Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (34): 5493-5501.doi: 10.3969/j.issn.2095-4344.2328
Previous Articles Next Articles
Potential of corn husk fibers reinforced chitosan-based hydrogels in cartilage tissue engineering scaffold
Fang Yulu, Yi Bingcheng, Shen Yanbing, Tang Han, Zhang Yanzhong
College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China
-
Received:2019-12-14Revised:2019-12-19Accepted:2020-02-12Online:2020-11-08Published:2020-09-11 -
Contact:Zhang Yanzhong, MD, Professor, Doctoral supervisor, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China -
About author:Fang Yulu, Master candidate, College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China -
Supported by:National Natural Science Foundation of China, No. 31771050; No.31570969
CLC Number:
Cite this article
Fang Yulu, Yi Bingcheng, Shen Yanbing, Tang Han, Zhang Yanzhong. Potential of corn husk fibers reinforced chitosan-based hydrogels in cartilage tissue engineering scaffold[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5493-5501.
share this article

2.1 CHFs/CC复合水凝胶的形貌及红外光谱表征 图2A是CC和CHFs/CC水凝胶样品的宏观形貌,直径均约为 14 mm,高约5 mm,且随着加入CHFs比例的增加,水凝胶由纯白色向淡黄色转变。从横截面观察,CC和CHFs/CC水凝胶均显示出三维互连多孔形貌和纤维成分的掺入使孔壁表面变得粗糙(图2B),这种互连多孔结构将有利于细胞的渗透、营养物质的运输和代谢废物的清除。进一步对孔径进行分析可知,CC水凝胶的孔径大小为(158.0±37.0) μm,但随着CHFs含量从1∶1增加到2∶1,CHFs/CC复合水凝胶的孔径则由(102.1±26.7) μm减小到(93.5±14.8) μm,使得孔结构变得有些致密。 图2C为CHFs、CC和CHFs/CC样品的红外光谱图。可知,CC水凝胶具有壳聚糖和柠檬酸的特征峰,如波数在 3 100-3 400 cm-1之间较宽的吸收带为壳聚糖分子内及分子间-OH键和-NH键的伸缩振动峰在同一处叠加的结果,波数在1 557,1 375,894 cm-1分别为壳聚糖的酰胺Ⅰ、酰胺Ⅲ谱带和β-构型糖苷键的特征峰[42];波数在1 716 cm-1处的吸收特征峰则为柠檬酸的C=O的伸缩振动峰;此外,CC水凝胶在1 557 cm-1左右产生一个新的特征吸收峰,这主要归结于壳聚糖的氨基与柠檬酸的羧基发生离子交联反应后形成了离子键[36]。CHFs的红外光谱与CC存在较高的相似性,但CHFs/CC复合水凝胶在波数2 876 cm-1处出现较尖的峰,而CC水凝胶内未出现,并且在波数约为3 330 cm-1处的峰强度有所增加,说明在CHFs的-OH基团和壳聚糖的-NH基团间存在氢键作用。 "

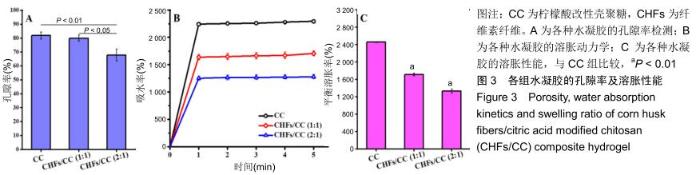
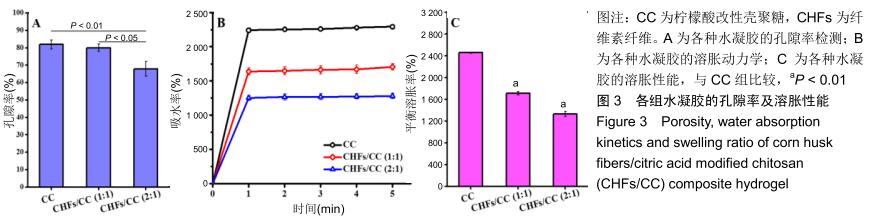
2.2 CHFs/CC复合水凝胶的孔隙率、溶胀性能 由图3A可知,CC水凝胶的孔隙率为81.92%,随着CHFs的添加,CHFs/CC(1∶1)复合水凝胶的孔隙率减少到79.98%,CHFs/CC(2∶1)复合水凝胶的孔隙率进一步降低到67.90%,这与图2B中扫描电镜观察到的结果基本一致。 图3B是CC、CHFs/CC(1∶1)和CHFs/CC(2∶1)水凝胶在PBS中的溶胀动力学。这些水凝胶均在1 min后基本达到最大吸水率,CHFs/CC复合水凝胶的吸水容量低于CC水凝胶,但CHFs/CC(1∶1)复合水凝胶仍表现出较好的吸水溶胀性能,4 h后达到溶胀平衡;由图3C可以发现与CC水凝胶相比,CHFs/CC复合水凝胶的平衡溶胀率下降(P < 0.01)。 "
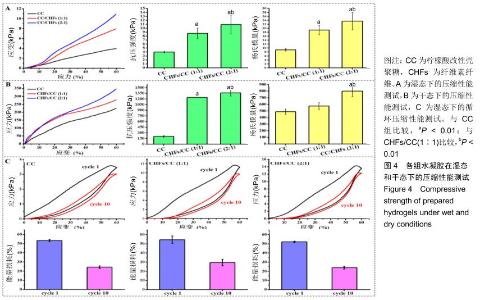
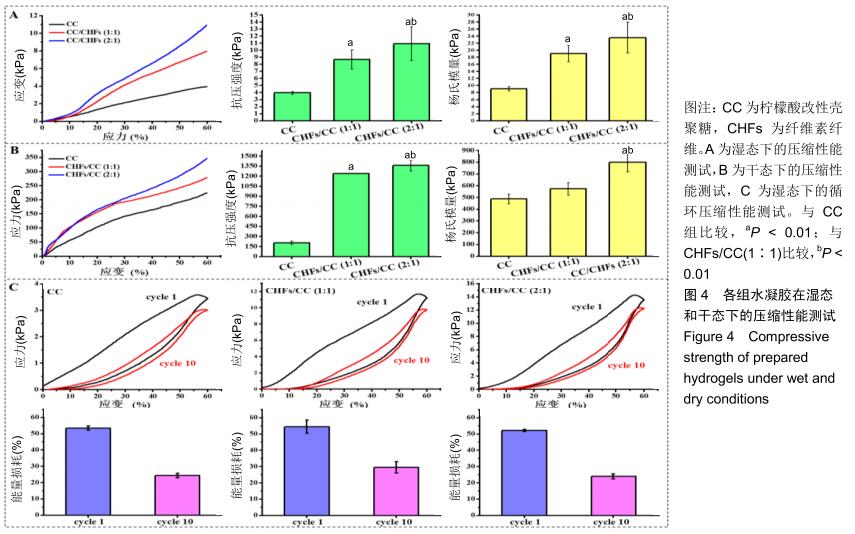
2.3 CHFs/CC复合水凝胶的压缩力学性能 湿态和干态下各水凝胶的抗压强度和杨氏模量,见图4A,B。如图所示,CC水凝胶在湿态和干态下的抗压强度分别为3.97,9.08 kPa,添加CHFs之后的两种复合水凝胶均表现出更高的抗压强度,湿态下达到CC水凝胶的两三倍,干态下则达到六七倍,且抗压强度随着CHFs复合量的增加而增加(P < 0.01)。CHFs/CC复合水凝胶的杨氏模量也表现出类似的增强效果,与CC水凝胶相比,CHFs/CC(1∶1)复合水凝胶湿态下的杨氏模量增加1倍(P < 0.01);当CHFs在CHFs/CC复合水凝胶中的添加比例从1∶1增加到2∶1时,湿态下的杨氏模量从19.04 kPa增加至23.54 kPa,干态下的杨氏模量从573.06 kPa增加至798.56 kPa(P < 0.01)。 在恒定应变(60%)下进行循环压缩-回复实验,以了解CHFs/CC复合水凝胶的能量耗散状况。图4C显示了CC、CHFs/CC(1∶1)和CHFs/CC(2∶1)复合水凝胶的第1次和第10次压缩-回复曲线。滞后回线显示压缩-回复测试期间的能量耗散状况,所有水凝胶都显著地耗散能量,但各组水凝胶的抗压强度均未发生显著下降。通过计算应力-应变曲线下的面积来分析支架的韧性[33],结果表明CHFs的添加明显增强了CC水凝胶的韧性。经过一个循环,CC、CHFs/ CC(1∶1)和CHFs/CC(2∶1)复合水凝胶的能量损耗分别达到了54.50%,53.47%和52.22%;第10次循环压缩后,能量损耗则分别下降到了29.45%,24.28%,24.02%,这种下降是由于水凝胶在第10次压缩进行前水凝胶已有一定程度的变形。 "
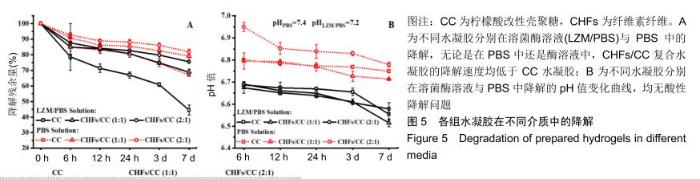
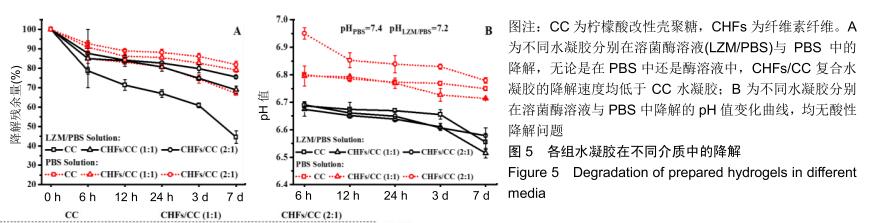
2.4 CHFs/CC复合水凝胶的体外降解性能 溶菌酶可破坏壳聚糖中的β-1,4糖苷键并破坏水凝胶的网络结构[24,43],通过测量含有105 U/mL溶菌酶的PBS中水凝胶的质量变化来评估CHFs/CC复合水凝胶的降解性质,同时比较各组水凝胶的酶促降解和在PBS中的降解。结果显示,各组水凝胶在6 h内都发生了较大幅度的质量损失,但随着时间增加降解速度明显变缓,并且在含溶菌酶的溶液中的降解速度大于在PBS中的降解速度,见图5A。无论是在PBS中还是酶溶液中,CHFs/CC复合水凝胶的降解速度均低于CC水凝胶。通过测量降解液的pH值,由图5B可知,在整个降解过程中降解液的pH均保持在6.0-7.0,无酸性降解问题。 "
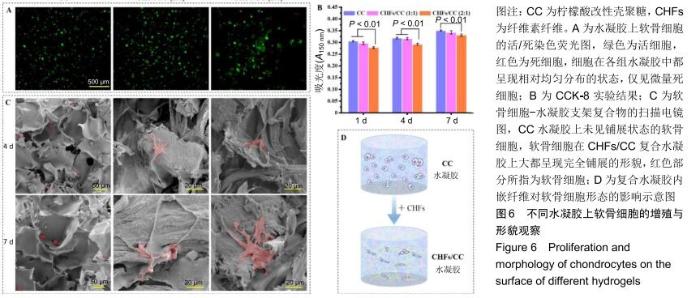
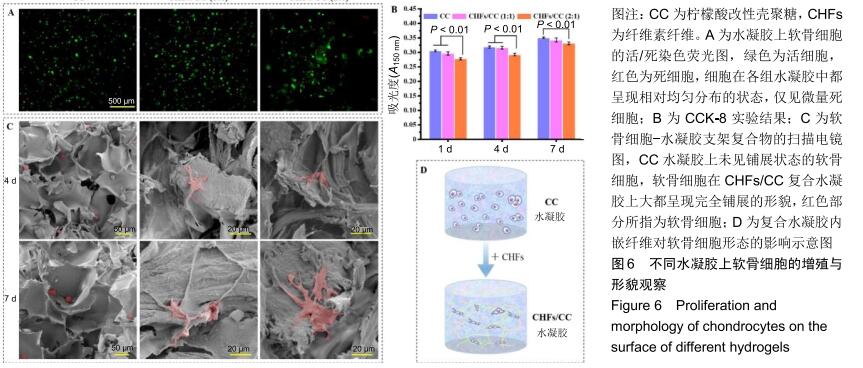
2.5 CHFs/CC复合水凝胶的细胞相容性评价 图6A为软骨细胞在CC、CHFs/CC(1∶1)和CHFs/CC(2∶1)水凝胶支架中培养7 d后活(绿色)、死(红色)细胞的荧光图像。可知,细胞在各组水凝胶中都呈现相对均匀分布的状态,并且仅观察到微量死细胞。增殖实验表明,所有支架上细胞数量均随着培养时间的延长而增加,但CHFs/CC复合水凝胶上的细胞增殖低于CC水凝胶(P < 0.01),见图6B。图6C为软骨细胞-水凝胶支架复合物在第4,7天的扫描电镜形貌图,CC水凝胶上的软骨细胞保持椭圆形,并且为单个、稀疏地分布在水凝胶片层的表面,视野内几乎没有观察到铺展状态的细胞;相反,软骨细胞在CHFs/CC复合水凝胶上大都呈现完全铺展的形貌,因此猜想CHFs的掺入使水凝胶孔结构的表面有纤维组分裸露,使支架表面特性发生改变,引起软骨细胞由球状向完全铺展的形貌转变(图6D)。此外,经过几天的细胞培养,水凝胶表面的多孔结构开始模糊,孔与孔之间难以发现明显的界线。 "
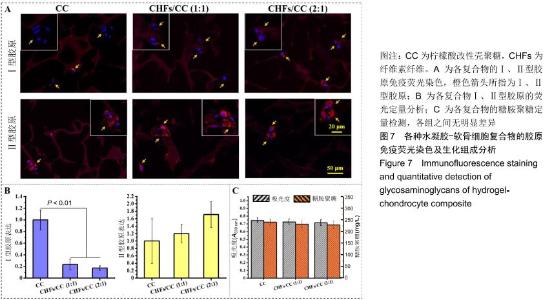
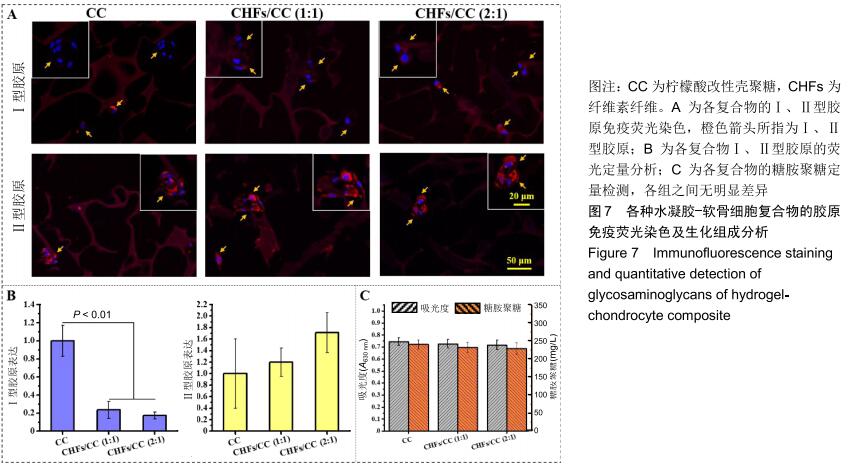
2.6 胶原免疫荧光染色及生化组成分析 软骨细胞可表达多种胶原蛋白,其中透明软骨细胞主要表达Ⅱ型胶原。对于Ⅰ、Ⅱ型胶原的免疫荧光染色,细胞核发出蓝色荧光,Ⅰ、Ⅱ型胶原蛋白发出红色荧光。经过28 d的培养,各组水凝胶上的软骨细胞均呈现多个细胞聚集团簇的形态,胶原主要集中表达于细胞膜、细胞质和核膜,见图7A。从荧光强度上进行比较,Ⅱ型胶原的荧光强度大于Ⅰ型胶原,即细胞在各水凝胶上表达的Ⅱ型胶原更多,并且随着CHFs添加量的增加而上升,见图7B。除此之外,对糖胺聚糖进行定量检测,以测定软骨细胞外基质的分泌情况。图7C为糖胺聚糖的定量测定结果,CC水凝胶中含量最高,为240.2 mg/L,CHFs/CC (1∶1)复合水凝胶次之,为232.4 mg/L,CHFs/CC(2∶1)复合水凝胶最少,为229.1 mg/L,但各组之间均无明显差异。 "
|
[1] FU J, YANG F, GUO Z. The chitosan hydrogels: from structure to function.New J Chem. 2018;42(21):17162-17180.
[2] GUPTA N, CRUZ MA, NASSER P, et al. Fibrin-genipin hydrogel for cartilage tissue engineering in nasal reconstruction.Ann Otol Rhinol Laryngol.2019;128(7):640-646.
[3] ASADI N, ALIZADEH E, SALEHI R, et al. Nanocomposite hydrogels for cartilage tissue engineering: a review.Artif Cell Nanomed Biotechnol. 2018;46(3):465-471.
[4] LI J, CHEN G, XU X, et al. Advances of injectable hydrogel-based scaffolds for cartilage regeneration.Regen Biomater. 2019;6(3):129-140.
[5] CAMARERO-ESPINOSA S, ROTHEN-RUTISHAUSER B, FOSTER EJ, et al. Articular cartilage: from formation to tissue engineering.Biomater Sci. 2016;4(5):734-767.
[6] CHUNG C, BURDICK JA. Engineering cartilage tissue.Adv Drug Deliv Rev.2008;60(2):243-262.
[7] ARMIENTO AR, STODDART MJ, ALINI M, et al. Biomaterials for articular cartilage tissue engineering: Learning from biology.Acta Biomater.2018;65:1-20.
[8] STECK E, BERTRAM H, WALTHER A, et al. Enhanced biochemical and biomechanical properties of scaffolds generated by flock technology for cartilage tissue engineering.Tissue Eng. Part A. 2010;16(12):3697-3707.
[9] LIU J, YANG B, LI M, et al. Enhanced dual network hydrogels consisting of thiolated chitosan and silk fibroin for cartilage tissue engineering. Carbohydr Polym.2020;227(1):115335.
[10] ZHAO Z, FANG R, RONG Q, et al. Bioinspired nanocomposite hydrogels with highly ordered structures. Adv Mater.2017;29(45):1703045.
[11] MCCULLEN SD, HASLAUER CM, LOBOA EG. Fiber-reinforced scaffolds for tissue engineering and regenerative medicine: use of traditional textile substrates to nanofibrous arrays.J Mater Chem. 2010;20(40):8776-8788.
[12] XU J, ZHANG J, GAO W, et al. Preparation of chitosan/PLA blend micro/nanofibers by electrospinning. Mater Lett. 2009;63(8):658-660.
[13] DONG W, ZENG Q, YIN X, et al. Preparation and blood compatibility of electrospun nanofibrous CTS/PLA mats from chitosan nanopowders and poly(lactic acid).Polym Compos.2018;39:416-425.
[14] RAD ZP, MOKHTARI J, ABBASI M. Calendula officinalis extract/PCL/ Zein/Gum arabic nanofibrous bio-composite scaffolds via suspension, two-nozzle and multilayer electrospinning for skin tissue engineering. Int J Biol Macromol.2019;135(19):530-543.
[15] GHOSAL K, MANAKHOV A, ZAJICKOVA L, et al. Structural and Surface Compatibility Study of Modified Electrospun Poly(epsilon-caprolactone) (PCL) Composites for Skin Tissue Engineering.AAPS PharmSciTech. 2017;18(1):72-81.
[16] YEKRANG J, SEMNANI D, SEYGHALANI AZ, et al. A novel biodegradable micro-nano tubular knitted structure of PGA braided yarns and PCL nanofibres applicable as esophagus prosthesis.Indian J Fibre Text Tes.2017;42(3):264-270.
[17] YOU Y, YOUK JH, LEE SW, et al. Preparation of porous ultrafine PGA fibers via selective dissolution of electrospun PGA / PLA blend fibers. Mater Lett.2006;62(6):757-760.
[18] BUTCHER AL, OFFEDDU GS, OYEN ML. Nanofibrous hydrogel composites as mechanically robust tissue engineering scaffolds. Trends Biotechnol.2014;32(11):564-570.
[19] VENUGOPAL E, SAHANAND KS, BHATTACHARYYA A, et al. Electrospun PCL nanofibers blended with Wattakaka volubilis active phytochemicals for bone and cartilage tissue engineering. Nanomedicine. 2019;21:102044.
[20] SMYTH M, GARCIA A, RADER C, et al. Extraction and process analysis of high aspect ratio cellulose nanocrystals from corn (Zea mays) agricultural residue.Ind Crop Prod.2017;108:257-266.
[21] 徐传会,张洪亭.玉米皮纤维制取与性能研究分析[J].山东纺织经济, 2012(3):56-58.
[22] WANG X, CHENG L, LIU L. Structure and performance of corn bracts and its fiber.J Textile Res. 2016;37(7):7-12.
[23] XU Q, JI Y, SUN Q, et al. Fabrication of cellulose nanocrystal/chitosan hydrogel for controlled drug release.Nanomaterials.2019;9(2):53.
[24] CHEN Z, ZHAO M, LIU K, et al. Novel chitosan hydrogel formed by ethylene glycol chitosan, 1,6-diisocyanatohexan and polyethylene glycol-400 for tissue engineering scaffold: in vitro and in vivo evaluation. J Mater Sci-Mater Med.2014;25(8):1903-1913.
[25] KIM CH, PARK S, YANG DH, et al. Chitosan for Tissue Engineering.In book: Novel Biomaterials for Regenerative Medicine,2018:475-485.
[26] LI H, HU C, YU H, et al. Chitosan composite scaffolds for articular cartilage defect repair: a review.RSC Adv.2018;8(7):3736-3749.
[27] NETTLES DL, ELDER SH, GILBERT JA. Potential use of chitosan as a cell scaffold material for cartilage tissue engineering.Tissue Eng. 2002; 8(6):1009-1016.
[28] CHUAH YJ, PECK Y, LAU JEJ, et al.Hydrogel based cartilaginous tissue regeneration: recent insights and technologies.Biomater Sci. 2017;5(4):613-631.
[29] ENGKAGUL V, SEREEMASPUN A, CHIRACHANCHAI S. One pot preparation of chitosan/hyaluronic acid-based triple network hydrogel via in situ click reaction, metal coordination and polyion complexation in water.Carbohydr Polym.2018;200:616-623.
[30] BILLIET T, VANDENHAUTE M, SCHELFHOUT J, et al. A review of trends and limitations in hydrogel-rapid prototyping for tissue engineering. Biomaterials.2012;33(26):6020-6041.
[31] CHEN Y, SONG G, YU J, et al. Mechanically strong dual responsive nanocomposite double network hydrogel for controlled drug release of asprin.J Mech Behav Biomed Mater.2018;82:61-69.
[32] FELFEL RM, GIDEON-ADENIYI MJ, HOSSAIN KMZ, et al. Structural, mechanical and swelling characteristics of 3D scaffolds from chitosan-agarose blends.Carbohydr Polym.2019;204:59-67.
[33] HUANG S, ZHAO Z, FENG C, et al. Nanocellulose reinforced P(AAm-co-AAc) hydrogels with improved mechanical properties and biocompatibility.Compos Pt A-Appl Sci Manuf.2018;112:395-404.
[34] RADHAKRISHNAN J, SUBRAMANIAN A, SETHURAMAN S. Injectable glycosaminoglycan-protein nano-complex in semi-interpenetrating networks: A biphasic hydrogel for hyaline cartilage regeneration. Carbohydr Polym.2017;175:63-74.
[35] ZHANG YB, DING JX, XU WG, et al. Biodegradable thermogel as culture matrix of bone marrow mesenchymal stem cells for potential cartilage tissue engineering.Chin J Polym Sci. 2014;32(12):1590-1601.
[36] 杨上莹,袁卉华,易兵成,等.柠檬酸改性壳聚糖水凝胶的制备与性能[J].功能高分子学报,2018,31(3): 232-240.
[37] 王晓婷.玉米苞叶纤维素提取及纳米晶须制备[D].上海:东华大学,2016.
[38] KIM S, CUI ZK, KOO B, et al. Chitosan Lysozyme Conjugates for Enzyme-Triggered Hydrogel Degradation in Tissue Engineering Applications.ACS Appl Mater Interfaces.2018;10(48):41138-41145.
[39] SHEN ZS, CUI X, HOU RX, et al. Tough biodegradable chitosan-gelatin hydrogels via in situ precipitation for potential cartilage tissue engineering.RSC Adv.2015;5(69):55640-55647.
[40] THOMAS LV, RAHUL VG, NAIR PD. Effect of stiffness of chitosan- hyaluronic acid dialdehyde hydrogels on the viability and growth of encapsulated chondrocytes.Int J Biol Macromol.2017;104:1925-1935.
[41] SINGH YP, ADHIKARY M, BHARDWAJ N, et al. Silk fiber reinforcement modulates in vitro chondrogenesis in 3D composite scaffolds.Biomed Mater.2017;12(4):045012.
[42] 刘名金.纤维素纳米晶体增强壳聚糖纤维性能研究[D].上海:东华大学, 2016.
[43] QIANYING D, LIHUA LI, YIQUN LI, et al. Depolymerization of chitosan with the aid of lysozyme and hydrogen peroxide.Chem World. 2005; 46(6):338-340.
[44] 胡星,严涛海.NaOH溶液制备玉米苞叶纤维的初步研究[J].轻纺工业与技术,2012,41(6):12-13.
[45] 黄志成.菠萝叶纳米纤维素/壳聚糖复合膜的制备及其性能研究[D].湛江:广东海洋大学,2014.
[46] LIU XJ, ZHANG YM, LI XS. Tough biopolymer IPN hydrogel fibers by bienzymatic crosslinking approach. Chin J Polym Sci. 2015;33(12): 1741-1749.
[47] ZHANG YB, LIU XC, ZENG LD, et al. Polymer fiber scaffolds for bone and cartilage tissue engineering. Adv Funct Mater. 2019;29(36): 1903279.
[48] ZHANG YB, YU JK, REN KX, et al. Thermosensitive hydrogels as scaffolds for cartilage tissue engineering. Biomacromolecules. 2019; 20(4):1478-1492.
[49] WANG CY, FENG NB, CHANG F, et al. Injectable cholesterol-enhanced stereocomplex polylactide thermogel loading chondrocytes for optimized cartilage regeneration.Adv Healthc Mater. 2019;8(14):1900312.
[50] SANDELL LJ, SUGAI JV, TRIPPEL SB. Expression of collagens I, II, X, and XI and aggrecan mRNAs by bovine growth plate chondrocytes in situ.J Orthop Res.1994;12(1):1-14. |
| [1] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [2] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [3] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [4] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Intravenous, topical tranexamic acid alone or their combination in total knee arthroplasty: a meta-analysis of randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 948-956. |
| [5] | Zhan Fangbiao, Cheng Jun, Zou Xinsen, Long Jie, Xie Lizhong, Deng Qianrong. Intraoperative intravenous application of tranexamic acid reduces perioperative bleeding in multilevel posterior spinal surgery: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 977-984. |
| [6] | Yang Xin, Jin Zhe, Feng Xu, Lu Bing. The current situation of knowledge and attitudes towards organ, eye tissue, body donation of residents in Shenyang [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 779-784. |
| [7] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [8] | Zhang Guomei, Zhu Jun, Hu Yang, Jiao Hongwei. Stress of three-dimensional finite element models of E-MAX porcelain inlay [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 537-541. |
| [9] | Cheng Jun, Tan Jun, Zhao Yun, Cheng Fangdong, Shi Guojia. Effect of thrombin concentration on the prevention of postoperative cerebrospinal leakage by fibrin glue [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 570-575. |
| [10] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [11] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [12] | Ye Haimin, Ding Linghua, Kong Weihao, Huang Zutai, Xiong Long. Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 621-625. |
| [13] | Yu Langbo, Qing Mingsong, Zhao Chuntao, Peng Jiachen. Hot issues in clinical application of dynamic contrast-enhanced magnetic resonance imaging in orthopedics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 449-455. |
| [14] | Li Yanle, Yue Xiaohua, Wang Pei, Nie Weizhi, Zhang Junwei, Tan Yonghai, Jiang Hongjiang. Intramedullary nail fixation versus plate fixation in the treatment of displaced midshaft clavicular fractures in adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 471-476. |
| [15] | Wang Xinting, Xu Dandi, Zhang Junxia, Su Hailong Wang Qi. Stability of load-bearing cross barrier of different arch structures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3838-3843. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||