Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (28): 4518-4525.doi: 10.3969/j.issn.2095-4344.2284
Previous Articles Next Articles
Properties of a novel photocrosslinked fish collagen peptide-hyaluronic acid hydrogel
Guo Enhui, Xu Zitong, Liang Yize, Zhou Liang, Lu Zhaoxiang, You Liang, Xia Yujun
Medical College, Qingdao University, Qingdao 266071, Shandong Province, China
-
Received:2019-10-21Revised:2019-10-25Accepted:2019-11-25Online:2020-10-08Published:2020-09-01 -
Contact:Xia Yujun, PhD, Professor, Doctoral supervisor, Medical College, Qingdao University, Qingdao 266071, Shandong Province, China -
About author:Guo Enhui, Medical College, Qingdao University, Qingdao 266071, Shandong Province, China -
Supported by:the Qingdao Livelihood Science and Technology Program, No. 15-9-2-76-nsh
CLC Number:
Cite this article
Guo Enhui, Xu Zitong, Liang Yize, Zhou Liang, Lu Zhaoxiang, You Liang, Xia Yujun. Properties of a novel photocrosslinked fish collagen peptide-hyaluronic acid hydrogel[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(28): 4518-4525.
share this article
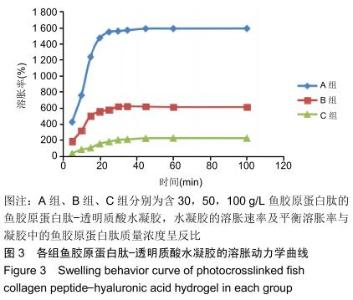
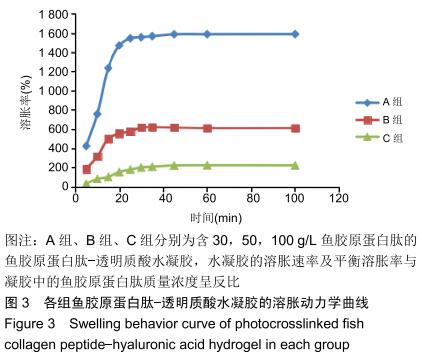
2.2 水凝胶的溶胀性 图3中显示了不同组分水凝胶材料在37 ℃去离子水中的溶胀曲线。 由图可见,在溶胀初期水凝胶的吸水溶胀速度较快,后期逐渐变缓至溶胀平衡,不同配比的水凝胶都可在100 min内达到溶胀平衡。对比各组,当鱼胶原蛋白肽质量浓度为 30 g/L时,水凝胶达溶胀平衡时的溶胀率为1 582%;当鱼胶原蛋白肽质量浓度为50 g/L时,水凝胶达溶胀平衡时的溶胀率为610%;当鱼胶原蛋白肽质量浓度为100 g/L时,水凝胶达溶胀平衡时的溶胀率仅为225%,可看出随着水凝胶中鱼胶原蛋白肽含量的增高,达溶胀平衡时的最大溶胀率迅速缩小,各组比较差异有显著性意义(P < 0.05)。尽管各组均具有较快的溶胀速率,但因配比不同,溶胀速率大小也呈A组>B组>C组的趋势,各组比较差异有显著性意义(P < 0.05)。 "
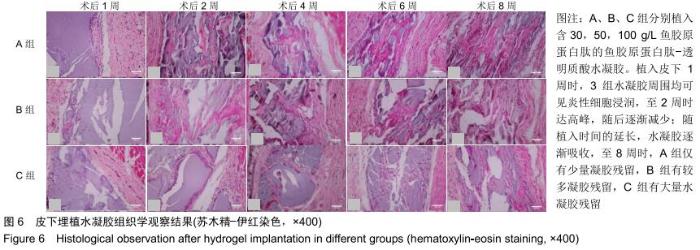
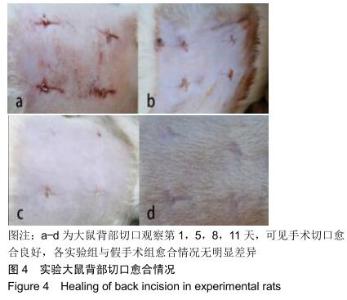
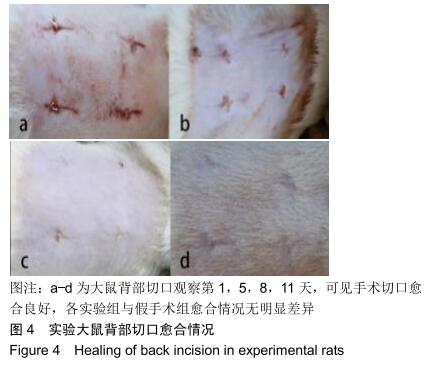
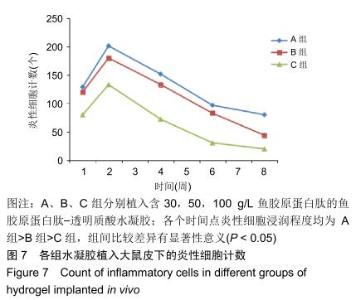
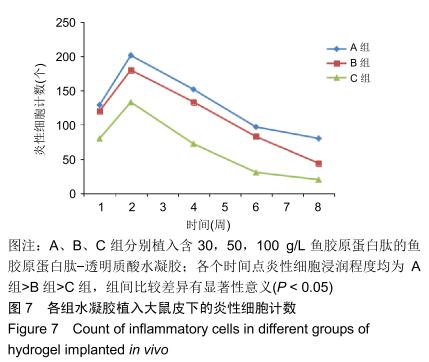
组织学观察显示,各组切片周围细胞排列规则,炎症均未波及皮肤及肌肉组织,未见组织变形坏死或异常增生;术后1周时,各组水凝胶周围均有少量炎性细胞及成纤维细胞浸润,炎性细胞以淋巴细胞为主,可见少量巨噬细胞,极少见中性粒细胞;术后2周时,周边炎性细胞浸润较第1周增多,可见淋巴细胞、成纤维细胞、新生血管长入凝胶;术后4周时,水凝胶周边炎性细胞浸润开始减少,纤维母细胞增生并分泌胶原纤维,水凝胶周围可见明显包裹的纤维薄膜,与周围组织界限清楚;术后6周时,周边炎性细胞浸润减少,组织细胞进一步长入凝胶;术后8周时,周边炎性浸润尚未完全消失,但已较少,凝胶内部大部分长入组织,见图6。各组炎性细胞计数见图7所示,术后1周各组水凝胶周围均有少量炎性细胞浸润,至术后2周时周边炎性细胞浸润明显增多,随后各个时间点炎性细胞浸润逐渐减少,各个时间点炎性细胞浸润程度均为A组>B组>C组,组间比较差异有显著性意义(P < 0.05)。 "
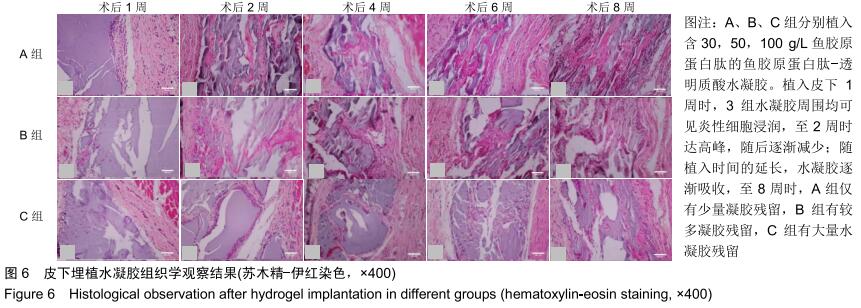
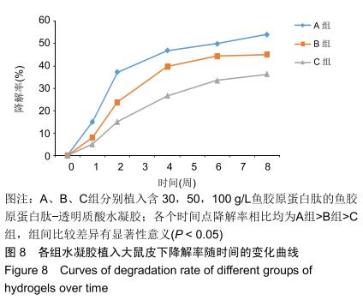
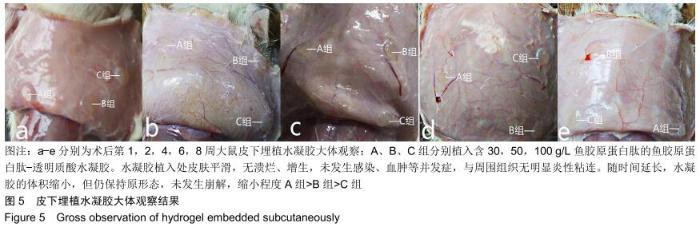
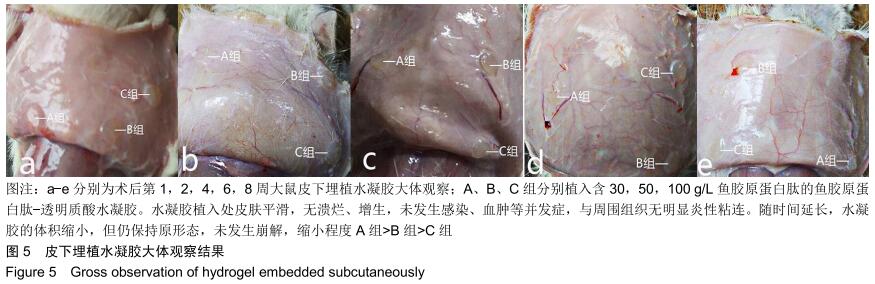
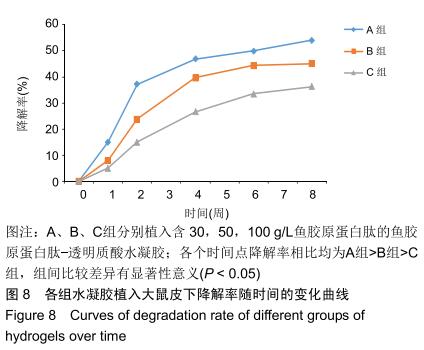
2.3.3 水凝胶的体内降解性 大体观察可见随时间延长水凝胶的体积不断缩小,降解程度A组>B组>C组,但至术后8周时都有凝胶残留,水凝胶仍保持原形态,未发生崩解,见图5。 组织学观察显示,术后1周时,各组仅在凝胶周边有少许组织细胞长入凝胶,凝胶内部完整,无组织细胞生长现象,A组组织细胞生长数目较B组多,C组仅少量细胞长入凝胶边缘。术后2周切时,组织细胞呈条索状长入各组凝胶,A组已有较多组织细胞长入凝胶内部,主要为成纤维细胞,淋巴细胞,极少见中性粒细胞,可见新生血管;B组也有组织细胞长入凝胶的情况,较A组为少;C组仅有少量组织细胞长入水凝胶。术后4周时,A组有更多组织细胞长入,成纤维细胞分泌胶原,新生血管进一步增多;B组亦有更多组织细胞长入,较A组为少;C组增多,但明显少于A、B两组。术后6周时,各组组织细胞进一步增多,凝胶逐渐吸收。术后8周时,凝胶内部大部分长入组织,A组凝胶残留已较少,凝胶降解吸收处组织形态已类似周围真皮组织;B组有较多凝胶残留;C组有大量水凝胶残留,水凝胶仍基本保持原来形态,尚未崩解,见图6。 各组水凝胶不同时间点的降解情况见图8,可见第2周降解速率较第1周明显加快,随后降解速率开始逐渐减慢,至第8周观察结束时,A组降解率达54%,B组降解率达45%,C组降解率达36%。各个时间点降解率相比均为A组>B组>C组,组间比较差异有显著性意义(P < 0.05)。 "
|
[1] 高春华,黄新友.组织工程与生物材料[J].上海生物医学工程, 2003,24(4):46-49.
[2] COSTA A, NARANJO JD, LONDONO R, et al. Biologic Scaffolds.Cold Spring Harb Perspect Med.2017;7(9).pii: a025676. doi:10.1101/cshperspect.a025676.
[3] 王馨,屈雷,窦忠英,等.组织工程生物支架材料[J].中国生物工程杂志,2003,23(10):15-18.
[4] BADYLAK SF, FREYTES DO, GILBERT TW. Reprint of: Extracellular matrix as a biological scaffold material: Structure and function.Acta Biomater.2015;23:S17-S26.
[5] 崔曼,张欣,马丽桃,等.新型三维复合仿生网络的构建及其组织相容性评价[J].天津医药, 2015,43(3):233-236.
[6] CHEUNG HK, HAN TT, MARECAK DM, et al. Composite hydrogel scaffolds incorporating decellularized adipose tissue for soft tissue engineering with adipose-derived stem cells. Biomaterials.2014;35(6):1914-1923.
[7] AHMED EM. Hydrogel: Preparation, characterization, and applications: A review.J Adv Res. 2015;6(2):105-121.
[8] ZHAO X, IRVINE SA, AGRAWAL A, et al. 3D patterned substrates for bioartificial blood vessels- The effect of hydrogels on aligned cells on a biomaterial surface.Acta Biomater.2015;26:159-168.
[9] DA SILVA DOMINGUES JF, ROEST S, WANG Y, et al. Macrophage phagocytic activity toward adhering staphylococci on cationic and patterned hydrogel coatings versus common biomaterials.Acta Biomater.2015;18:1-8.
[10] GOUJON LJ, SANTOSH H, BAHAREH S, et al. Tunable hydrogel thin films from reactive synthetic polymers as potential two-dimensional cell scaffolds.Langmuir. 2015; 31(20):5623.
[11] HOFFMAN AS. Hydrogels for biomedical applications. Ann N Y Acad Sci.2012;64(1):18-23.
[12] QIU Y, PARK K. Environment-sensitive hydrogels for drug delivery. Adv Drug Deliv Rev.2012;64(3):49-60.
[13] MALINEN MM, KANNINEN LK, CORLU A, et al. Differentiation of liver progenitor cell line to functional organotypic cultures in 3D nanofibrillar cellulose and hyaluronan-gelatin hydrogels. Biomaterials.2014;35(19): 5110-5121.
[14] PARHI R. Cross-Linked Hydrogel for Pharmaceutical Applications: A Review.Adv Pharm Bull. 2017;7(4):515-530.
[15] 李静静,郭璇,解军,等.琼脂糖/明胶/透明质酸/细胞外基质水凝胶的制备与性能表征[J].中国组织工程研究,2019,23(18): 2900-2908.
[16] NAAHIDI S, JAFARI M, LOGAN M, et al. Biocompatibility of hydrogel-based scaffolds for tissue engineering applications. Biotechnol Adv.2017;35(5):530-544.
[17] SPANG MT, CHRISTMAN KL. Extracellular matrix hydrogel therapies: In vivo applications and development.Acta Biomater. 2018;68:1-14.
[18] SALDIN LT, CRAMER MC, VELANKAR SS, et al. Extracellular Matrix Hydrogels from Decellularized Tissues: Structure and Function.Acta Biomater.2016;49:1-15.
[19] XIE B, JIN L, LUO Z, et al. An injectable thermosensitive polymeric hydrogel for sustained release of Avastin® to treat posterior segment disease.Int J Pharm.2015;490(1): 375-383.
[20] PATI F, DATTA P, ADHIKARI B, et al. Collagen scaffolds derived from fresh water fish origin and their biocompatibility.J Biomed Mater Res A.2012;100A(4):1068-1079.
[21] NAGAI N, NAKAYAMA Y, ZHOU YM, et al. Development of salmon collagen vascular graft: mechanical and biological properties and preliminary implantation study.J Biomed Mater Res B Appl Biomater.2010;87B(2):432-439.
[22] YAMADA S, YAMAMOTO K, IKEDA T, et al. Potency of Fish Collagen as a Scaffold for Regenerative Medicine.Biomed Res Int.2014;2014(4976):302932.
[23] XU Y, WANG X. Application of 3D biomimetic models in drug delivery and regenerative medicine. Curr Pharm Des. 2015; 21(12):1618-1626.
[24] FERNÁNDEZ-GARCÍA L, MARÍ-BUYÉ N, BARIOS JA, et al. Safety and tolerability of silk fibroin hydrogels implanted into the mouse brain.Acta Biomater.2016;45:262-275.
[25] GUZIEWICZ N, BEST A, PEREZ-RAMIREZ B, et al. Lyophilized silk fibroin hydrogels for the sustained local delivery of therapeutic monoclonal antibodies. Biomaterials. 2011;32(10):2642-2650.
[26] KOGAN G, ŠOLTÉS L, STERN R, et al. Hyaluronic acid: a natural biopolymer with a broad range of biomedical and industrial applications.Biotechnol Lett.2007;29(1):17-25.
[27] HSIEH MF, SHEU CL, YANG HW. Micro-Lithographic Fabrication of Collagen and Hyaluronic Acid Hydrogel Scaffolds. Adv Mater Res.2013;647:170-175. [28] 曹璐娟.透明质酸超分子凝胶及纳米凝胶的制备及生物医学应用研究[D].无锡:江南大学,2014.
[29] VALMIKINATHAN CM, MUKHATYAR VJ, JAIN A, et al. Photocrosslinkable chitosan based hydrogels for neural tissue engineering.Soft Matter.2012;8(6):1964-1976.
[30] CHIEN HW, CHENG PH, CHEN SY, et al. Low-fouling and functional poly(carboxybetaine) coating via a photo-crosslinking process.Biomater Sci.2017;5(3):523-531.
[31] CREDI C, DE MC, MOLENA E, et al. Direct photo-patterning of hyaluronic acid baits onto a fouling-release perfluoropolyether surface for selective cancer cell capture and immobilization. Mater Sci Eng C Mater Biol Appl.2016;62: 414-422.
[32] KIM S, LEE S, LEE J, et al. Tissue adhesive, rapid forming, and sprayable ECM hydrogel via recombinant tyrosinase crosslinking.Biomaterials.2018;178:401-412.
[33] GHUMAN H, GERWIG M, NICHOLLS FJ, et al. Long-term retention of ECM hydrogel after implantation into a sub-acute stroke cavity reduces lesion volume.Acta Biomater. 2017;63: 50-63.
[34] AHEARNE M. Development of an ECM hydrogel for corneal tissue engineering.Acta Ophthalmol.2014;92(s253).DOI: 10.1111/j.1755-3768.2014.1726.x.
[35] LIU C. Development of an ECM-mimetic, Electrospun Hydrogel Scaffold for Soft Tissue Repair Application. Dissertations & Theses-Gradworks,2014.
[36] HIROAKI S, SHUNJI Y, EIJI K, et al. In vivo biological responses and bioresorption of tilapia scale collagen as a potential biomaterial.J Biomater Sci Polym Ed.2009;20(10): 1353-1368.
[37] 魏志君,施春英,程连强,等.鱼胶原蛋白肽-透明质酸水凝胶制备及其生物相容性[J].青岛大学医学院学报,2017,53(5):600-603.
[38] BRIGHAM MD, ALEXANDER B, EDWARD L, et al. Mechanically robust and bioadhesive collagen and photocrosslinkable hyaluronic acid semi-interpenetrating networks.Tissue Eng Part A.2009;15(7):1645.
[39] RINAUDO M. Chitin and chitosan: Properties and applications. Cheminform.2007;31(7):603-632.
[40] 刘捷,王芳,马盈盈,等.热处理对聚乙烯醇/胶原水解物复合膜性能的影响[J].中国皮革, 2010,39(15):13-15.
[41] 周慧通,方大为,王波,等.冷冻干燥法制备CS/AMPS/PEGDA光交联多孔水凝胶及其性能[J].材料科学与工程学报, 2015,33(3): 432-437.
[42] 赵江,蒋国强,孙佳丽,等.释药用壳聚糖温敏凝胶体内外形态与溶蚀降解研究[J].高校化学工程学报, 2010,24(3):456-461.
[43] KAMATH KR, PARK K. Biodegradable hydrogels in drug delivery.Adv Drug Deliv Rev.1993;11(1):59-84.
[44] JIN S, BIAN F, LIU M, et al. Swelling mechanism of porous P(VP-co-MAA)/PNIPAM semi-IPN hydrogels with various pore sizes prepared by a freeze treatment.Polym Int. 2010; 58(2):142-148.
[45] KIM SW, YOU HB, OKANO T. Hydrogels: Swelling, Drug Loading, and Release.Pharm Res.1992;9(3):283-290. [46] LIM YH, KIM D, LEE DS. Drug releasing characteristics of thermo- and pH-sensitive interpenetrating polymer networks based on poly (N-isopropylacrylamide).J Appl Polym Sci. 1997; 64(13):2647-2655.
[47] 杨兵,侯萍,张会,等.光聚合型pH敏感水凝胶的制备及药物缓释性能[J].高分子材料科学与工程, 2019,35(4):19-25.
[48] ABUREESH MA, OLADIPO AA, GAZI M. Facile synthesis of glucose-sensitive chitosan-poly(vinyl alcohol) hydrogel: Drug release optimization and swelling properties.Int J Biol Macromol. 2016;90:75-80.
[49] 胡杨,蒋远干,但卫华,等.胃蛋白酶提取猪皮胶原的研究[J].中国皮革,2010,39(23):11-16.
[50] ZHANG W, WANG X, MA J, et al. Preparation of chitosan/pumpkin polysaccharide hydrogel for potential application in drug delivery and tissue engineering.J Porous Mater. 2017;24(2):497-506.
[51] 宋福来,刘万顺,韩宝芹.壳聚糖即型水凝胶的理化性质、止血功能和生物相容性研究[J].功能材料, 2014,45(9):9065-9069.
[52] 郭清奎,吕志前,张袆,等.新型可完全降解材料聚外消旋乳酸-三亚甲基碳酸酯聚合物体内降解行为和组织相容性[J].中国组织工程研究与临床康复,2011,15(34):6368-6374.
[53] HUANG KF, HSU WC, HSIAO JK, et al. Collagen-glycosaminoglycan matrix implantation promotes angiogenesis following surgical brain trauma.Biomed Res Int. 2014;2014(3):672409.
[54] LIM SM, SONG DK, OH SH, et al. In vitro and in vivo degradation behavior of acetylated chitosan porous beads.J Biomater Sci Polym Ed.2012;19(4):453-466.
[55] 符锋,秦喆,李晓红,等.胶原/壳聚糖复合支架植入大鼠不同部位降解速率的变化[J].中国组织工程研究,2017,21(6):864-870.
[56] GORHAM SD, LIGHT ND, DIAMOND AM, et al. Effect of chemical modifications on the susceptibility of collagen to proteolysis. II. Dehydrothermal crosslinking.Int J Biol Macromol.1992;14(3):129-138.
[57] HUTMACHER DW. Scaffolds in tissue engineering bone and cartilage.Biomaterials. 2000;21(24):2529-2543.
[58] LUO J, MENG Y, ZHENG L, et al. Fabrication and characterization of Chinese giant salamander skin composite collagen sponge as a high-strength rapid hemostatic material. J Biomater Sci Polym Ed.2019;30(4):247-262.
[59] 杨中民.明胶—透明质酸/纳米生物玻璃复合材料的制备及性能研究[D].湘潭:湖南科技大学,2013.
[60] WEI Y, CHANG YH, LIU CJ, et al. Integrated Oxidized-Hyaluronic Acid/Collagen Hydrogel with beta-TCP Using Proanthocyanidins as a Crosslinker for Drug Delivery. Pharmaceutics. 2018;10(2):37.
[61] 吴炜,毛天球,封兴华,等.胶原-透明质酸支架的制备及其与软骨细胞复合培养的实验研究[J].中国修复重建外科杂志, 2007, 21(4):401-405.
[62] 陈琳,吕洋,管利东,等.生物支架材料胶原膜交联前后的特性分析[J].中国修复重建外科杂志, 2008,22(2):183-187.
[63] SHAHBAN S, BROWN R, CHEEMA U, et al. Enhancing the mechanical properties of collagen by photo-chemical cross-linking. Int J Surg.2012;10(8):S54.
[64] 杜国辉.交联透明质酸的制备、性质及治疗骨关节炎的疗效研究[D].济南:山东大学,2010.
[65] 顾华,何黎.透明质酸在表皮重建中生物学意义的实验研究[C].中华医学会第16次全国皮肤性病学术年会,中国广东广州,2010. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Liu Fang, Shan Zhengming, Tang Yulei, Wu Xiaomin, Tian Weiqun. Effects of hemostasis and promoting wound healing of ozone sustained-release hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3445-3449. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||