Chinese Journal of Tissue Engineering Research
Previous Articles Next Articles
Application and prospects of Nel-like molecular 1 in promoting bone regeneration and repair and treating osteoporosis
Cao Xi1, Cui Wei2, Chen Yueping2, Feng Yang2, Lu Tong1
- 1Department of Orthopedics, Changshu Hospital of Traditional Chinese Medicine, Changshu 215500, Jiangsu Province, China; 2Department of Orthopedic Trauma and Hand Surgery, Ruikang Hospital Affiliated to Guangxi University of Chinese Medicine, Nanning 530001, Guangxi Zhuang Autonomous Region, China
-
Online:2019-03-18Published:2019-03-18 -
Contact:Cui Wei, MD, Master’s supervisor, Chief physician, Department of Orthopedic Trauma and Hand Surgery, Ruikang Hospital Affiliated to Guangxi University of Chinese Medicine, Nanning 530001, Guangxi Zhuang Autonomous Region, China -
About author:Cao Xi, Master, Department of Orthopedics, Changshu Hospital of Traditional Chinese Medicine, Changshu 215500, Jiangsu Province, China -
Supported by:the Natural Science Foundation of Guangxi Zhuang Autonomous Region, No. 2015GXNSFAA139136; the Central Governmental Special Fund for Local University Development: Orthopedics in 2017, No. 04B2017082
CLC Number:
Cite this article
Cao Xi, Cui Wei, Chen Yueping, Feng Yang, Lu Tong. Application and prospects of Nel-like molecular 1 in promoting bone regeneration and repair and treating osteoporosis [J]. Chinese Journal of Tissue Engineering Research, doi: 10.3969/j.issn.2095-4344.0596.
share this article
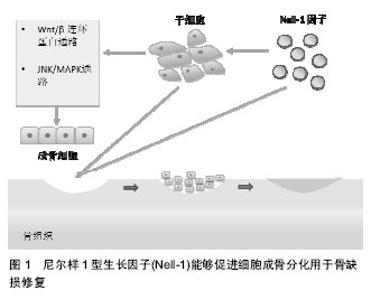
2.1 Nell-1促进成骨 Nell-1蛋白最早由洛杉矶大学加州分校(UCLA)牙医学院的Ting等[10]于1999年首次从单侧冠状缝早闭症患者的切除颅骨中分离出。Ting等[10]发现,Nell-1生长因子表达升高与膜内成骨及单侧冠状缝早闭症及成骨有密切联系,但其具体作用机制未知。随后,同样来自UCLA的研究人员Zhang等[11-24]对Nell-1因子展开了大量研究。 Zhang等[11]对Nell-1过表达转基因小鼠进行实验研究后发现,Nell-1因子减少会明显影响颅骨发育过程中的成骨细胞分化进程,而Nell-1过度表达与颅骨早闭症病理过程、成骨细胞分化及凋亡之间存在密切联 系[10-11]。Desai等[15]发现,除了与颅骨发育有关外,Nell-1严重缺乏还会导致小鼠出现颅腔增大伴随颅骨边缘变薄、椎间隙变窄和脊柱曲度改变以及胸廓畸形等异常表现,认为Nell-1除了能对髓内骨成骨发挥作用,在软骨内骨化和脊柱软骨正常发育方面也具有重要作用。2006年,Aghaloo等[16]发现MC3T3-E1型成骨细胞成骨分化过程中,Nell-1早期能够减少转录因子OSX和下调碱性磷酸酶(ALP)表达,而在分化晚期能增加成骨细胞分化成熟标志物——骨桥蛋白和骨钙蛋白的表达。国内的胡镜宙等[17]通过实时定量PCR技术也观察到了相类似的结果,说明Nell-1因子能够提高成骨细胞活性、促进细胞矿化和成骨细胞分化。 值得注意的是,Aghaloo认为Nell-1因子调节成骨和促进骨再生的能力与骨形态发生蛋白2相当,并表示需要进一步对Nell-1调控成骨相关的具体信号通路进行研究。但是,Cowan等[25]在随后的研究中发现,单独使用Nell-1转导成肌细胞C1C12无法诱导成肌细胞进行成骨分化,而联合使用Nell1+骨形态发生蛋白2转导对促进细胞成骨分化和促进成骨具有协同作用,且优于单独使用骨形态发生蛋白2。 最近荷兰研究人员Fahmygarcia等[18]的一项研究也报道了类似的结果,他对NELL-1、骨形态发生蛋白2、高迁移率族蛋白以及结缔组织生长因子进行研究后发现,低剂量Nell-1因子能够促进间充质干细胞分化为前成骨细胞,但是相对于骨形态发生蛋白2,Nell-因子1的促血管化作用和促细胞迁移能力高于促进成骨能力。而联用Nell-1和骨形态发生蛋白2能够显著促进间充质干细胞向成骨细胞分化和提高成骨分化作用。Fahmy-garcia等的研究结论认为Nell-1因子与成骨过程有联系,可应用于骨缺损再生修复。 2.2 Nell-1成骨相关信号通路 2.2.1 Wnt信号 截至目前,Wnt信号通路能够促进成骨和抑制脂肪生成这一观点已经经过了研究论证,而Wnt/β-连环蛋白通路更是一条与促进成骨作用密切相关的典型信号通路[19]。整合素作为一种膜表面受体具有介导细胞黏附和整合包括细胞外生长因子及细胞激素信号、组织细胞骨架、调节基因表达等等作用[20]。近年研究表明,Nell-1成骨信号与Wnt典型信号通路及整合素之间存在联系。Shen等[21]发现,Nell-1的血小板反应蛋白-1样N-端结构域(N-terminal thrombospondin-1- like domain,TSPN)能够直接与膜表面受体整合素β1结合,从而促进细胞的黏附、增殖和成骨分化。 一项近期的研究表明,Nell-1因子的成骨作用需要通过诱导和激活Wnt/β连环蛋白典型信号通路完成。Shen等[21]通过免疫细胞化学染色β连环蛋白细胞核后发现,使用Nell-1能够增强β连环蛋白细胞核定位信号,而当Wnt信号被抑制时,Nell-1的成骨作用会受到干扰。Shen等[21]还使用Frizzled蛋白(FZD)受体替代低分子脂蛋白相关蛋白5/6共受体与Wnt蛋白配体结合,FZD与Wnt结合后拮抗Wnt信号通路同时影响了Nell-1的下游调节作用。 此外,Runx2也是Wnt通路中另一个重要蛋白,Runx2在成骨细胞的分化和成骨中扮演着特异性转录因子这一重要角色。Runx2能够上游调控Nell-1的表达同时也是Nell-1的重要启动子。Runx2与Nell-1的表达还具有偶联特点,Runx2蛋白缺乏会导致Nell-1水平降 低[12]。国内张弘等[22]近期的研究发现,使用Nell-1诱导骨髓间充质干细胞成骨分化时,Nell-1能够通过调控Runx2的P1启动子促进骨髓间充质干细胞向成骨细胞分化[24]。 2.2.2 丝裂原活化蛋白激酶(mitogen- activated protein kinases,MAPK)信号 MAPK信号通路是另一条与细胞生长及分化相关的重要通路。哺乳动物体内的MAPK通路可以分为以下几类:p38、ERK1/2和JNK等。Cowan等[25]发现Nell-1与骨形态发生蛋白2的协同效应与JNK/MAPK通路有关。Cowan等[25]使用重组腺病毒介导Nell-1和骨形态发生蛋白2转导成肌细胞进行观察后发现,Nell-1主要通过磷酸化激活JNK通路诱导骨桥蛋白表达同时使成肌细胞出现成骨细胞表型,而Nell-1信号在其余的ERK1/2和p38MAPK通路中并未出现类似作用。除此以外,Nell-1信号还能与Hedgehog信号协同促进细胞成骨分化和促进成骨,但其具体信号通路仍有待研究。 2.3 Nell-1因子优点 Nell-1因子调控成骨的优点主要有以下几点:①调控细胞种类广泛,包括间充质祖细胞(mesenchymal progenitor cells)[23-24]、周细胞(pericyte)[12]、前成骨细胞(pre-osteoblasts cells)[25]、成牙质细胞(odontoblast)和成软骨细胞(chondrocytes) 等[26-27];②抑制成脂分化[28],Nell-1能够通过氧化物酶体增殖因子活化受体γ(PPARGγ)通路和CCAAT/增强子结合蛋白α(CCAAT-enhancer binding protein alpha,C/EBPα)通路抑制细胞成脂肪分化;③具有成骨和成软骨特异性[22];④抑制炎症反应[29-30],Nell-1能够降低血清肿瘤坏死因子α和白细胞介素6水平从而抑制炎症反应[29];⑤无毒性作用,Nell-1因子在促进细胞增殖和成骨效应过程中不会诱导毒性反应[31]。 2.4 Nell-1因子骨再生修复应用研究 Nell-1因子是一种有效的成骨生长因子,因此。Nell-1因子在修复颌面软骨、关节软骨及长骨缺损和促进脊柱融合方面都表现出了极大的潜力[32],见图1。"
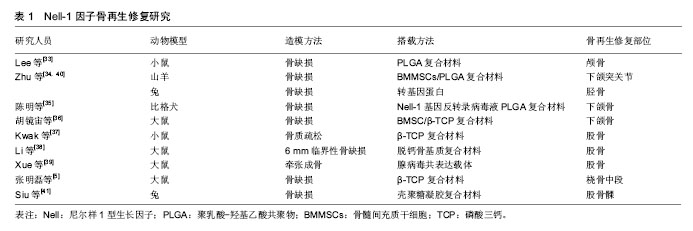
2.4.1 颅颌骨再生修复 Nell-1因子最初发现与颅骨发育有关,Nell-1促进成骨相关的研究最早也与颅骨成骨有关[15]。迄今为止,Nell-1因子参与颅颌面骨再生修复的研究报道数量较多。韩国抱川中文医科大学的Lee 等[33]对Nell-1的颅骨缺损再生修复效果进行了研究,他们使用CD-1型小鼠进行右侧颅骨临界缺损模型造模,全厚层颅骨缺损深度达4 mm,随后将小鼠分为4组,分别为空白对照组、细胞渗透性激动剂干预组(SAG干预组)、Nell-1蛋白干预组及使用激动剂联合Nell-1蛋白干预组(联合干预组),并采用微型CT和组织学分析评估成骨情况。治疗4周和8周,SAG干预组和Nell-1干预组微型CT显示骨缺损部位有新骨生成,但联合干预组的新生骨量明显多于其余几组,且组织学分析显示缺损部位有血管化加强迹象。 四川大学的Zhu等[34]对50只下颌髁突关节软骨缺损山羊模型进行了研究,骨软骨缺损大小为直径和深度分别为3 cm和5 cm,随后使用Nell-1蛋白修饰的骨髓间充质干细胞/聚乳酸-羟基乙酸共聚物(BMMSCs/PLGA)复合材料植入缺损部位后发现,相比未使用Nell-1蛋白修饰的两组对照组(BMMSCs/PLGA)复合材料组和单纯PLGA植入组),经Nell-1蛋白修饰的BMMSC/PLGA复合材料在植入6周时表现出了极大的软骨缺损修复能力,下颌髁突关节软骨缺损在植入后第24周时已被完全修复,且修复效果明显优于两对照组。陈明等[35]选用8只比格犬进行下颌骨缺损造模,下颌骨缺损大小为15 cm× 10 cm×5 mm,研究中使用了Nell-1和病毒反转录载体制作出了Nell-1基因反转录病毒液,随后将PLGA与Nell-1基因反转录病毒液制作成生物修复材料植入缺损部位,通过将实验组(Nell-1基因反转录病毒液PLGA复合材料植入组)与对照组(单纯PLGA植入组)进行比较后发现,实验组植入材料在术后第8周表面和内部都出现了成骨细胞移行,有部分区域出现了骨小梁和骨陷窝结构并伴随大量致密的不规则骨质;植入后第16周,骨缺损部位出现了骨性愈合,而对照组术后第16周仅在断端出现少量骨痂。 上海交通大学的胡镜宙[36]使用经Nell-1基因修饰后的骨髓间充质干细胞联合β-TCP复合材料修复大鼠下颌骨缺损,也获得了很好的修复效果。说明Nell-1因子能够用于颅颌面部位的骨缺损再生修复。 2.4.2 长骨再生修复 长骨结构及力学性能具有特殊性,长骨的骨组织再生修复过程较其他部位通常更为复杂,因此研究人员对Nell-1因子在长骨再生修复方面的作用进行了研究。UCLA的Kwak等[37]发现Nell-1因子能够有效增强骨质疏松小鼠长骨原位成骨,能够用于骨质疏松性骨折的再生修复。Li等[38]使用大鼠进行了股骨 6 mm临界性缺损模造模,随后将脱钙骨基质(DBX)搭载Nell-1蛋白植入缺损部位观察其作用,他们将大鼠模型分为3组,分别为:①1.5 g/L Nell-1蛋白及DBX组;②0.6 g/L Nell-1蛋白与DBX组以及,③磷酸盐缓冲溶液(PBS)与DBX(对照组),显微CT和组织学分析结果显示两组使用Nell-1蛋白的治疗组的大鼠再生骨量都明显多于PBS对照组,且再生骨量与Nell-1蛋白使用剂量成正比。 此外,来自解放军总医院的Xue等[39]以及四川大学的Zhu等[40]还对NELL-1因子对长骨缺损常用治疗方 法——牵张成骨术的影响进行了研究。Ad-GFP-NELL-1为能够同时表达绿色荧光蛋白(green Fluorescent Protein,GFP)报告基因和Nell-1的腺病毒共表达载体。Xue等[39]对Ad-GFP-NELL-1对大鼠股骨牵张成骨的影响进行了评估,他们将接受股骨牵张成骨术的大鼠分成d-GFP-NELL-1注射组、Ad-GFP注射组和生理盐水注射组3组。微型CT显示Ad-GFP-NELL-1组大鼠均获得骨性愈合且数量多于Ad-GFP组和生理盐水组。Ad-GFP-NELL-1组的大鼠生物力学测试也优于其余两组,与正常股骨的抗扭强度相当。祝颂松将兔胫骨牵张模型分为4组,并局部予生长因子治疗。4组分别为PBS治疗组、50 mg rhNell-1蛋白治疗组、50 mg重组人骨形态发生蛋白2蛋白治疗组和25 mg rhNell-1与25 mg重组人骨形态发生蛋白2联合治疗组。兔牵张模型达到骨性愈合评估标准4周后,通过组织学、影像学、双能X射线吸收测定法、微型CT和三点弯曲测试检查结果将4组进行对比,结果显示rhnell1联合重组人骨形态发生蛋白2治疗组成骨疗效优于其余分组。张明磊等[5]观察了含有NELL-1/骨形态发生蛋白2的β-磷酸钙复合物对大鼠桡骨中段骨折的影响,结果显示两组大鼠骨折后期的骨痂评分相当。这些研究说明Nell-1在长骨再生修复方面也有巨大的潜在应用价值。此外,Siu等[41]使用Nell-1修复兔股骨髁软骨缺损也取得了较好结果,Nell-1因子也有望用于退行性病变导致的关节软骨缺损再生修复。Nell-1因子骨再生修复研究见表1。 2.5 Nell-1因子的其他用途 除了能够用于促进骨缺损再生修复,Nell-1因子还能够用于促进椎间隙融合。苏州大学医学院的邹俊[42]发现,在使用Ad-NELL-1诱导大鼠脊柱后外侧融合时,大鼠脊柱融合率达到了80%。UCLA的James等[43]、Lu等[44]和Siu等[45]、韩国的Lee 等[46]、复旦大学的Yuan等[47]也都发现了Nell-1对脊柱融合具有良好的促进作用。解放军总医院的郭旭[48-49]还对Nell-1的骨溶解防治作用进行了研究,郭旭研究发现Nell-1能够平衡磨屑导致的骨溶解失衡,能够潜在应用于假体置换术以防止术后假体松动。此外,Nell-1还有望应用于治疗骨质疏松症[50]。"
| [1] Egol KA,Nauth A,Lee M,et al.Bone Grafting: Sourcing, Timing, Strategies, and Alternatives.J Orthop Trauma. 2015;29 Suppl 12:S10-104. [2] Wang W,Yeung K W K. Bone grafts and biomaterials substitutes for bone defect repair: Areview. Bioactive Materials.2017;2(4): 224-247.[3] Arealis G,Nikolaou VS.Bone printing: new frontiers in the treatment of bone defects. Injury.2015;46 Suppl 8:S20-22.[4] Yu X,Tang X,Gohil SV, et al.Biomaterials for Bone Regenerative Engineering.Advanced Healthcare Materials.2015;4(9):1268.[5] 张明磊,宋显吉,尹若峰,等.神经源性表皮生长因子样分子-1局部植入促进大鼠长骨干骨折愈合的研究[J]. 中华实验外科杂志, 2015, 32(9):2211-2213.[6] 周海华,李祖兵. Nel样1型分子对成骨细胞分化的分子调控机制[J].国际口腔医学杂志,2010, 37(2):166-169.[7] Watanabe TK.Cloning and characterization of two novel human cDNAs (NELL1 and NELL2) encoding proteins with six EGF-like repeats.Genomics.1996;38(3):273-276.[8] 万林子,金晗,吴熙凤,等.脂质体介导NELL1基因对脂肪干细胞影响的体外研究[J].现代口腔医学杂志, 2015(3):161-165.[9] Smith B, Goldstein T, Ekstein C.Biologic adjuvants and bone: current use in orthopedic surgery. Curr Rev Musculoskelet Med. 2015 ;8(2):193-199.[10] Ting K,Vastardis H,Mulliken JB,et al.Human NELL-1 expressed in unilateral coronal synostosis. J Bone Miner Res.1999;14(1): 80-89.[11] Zhang X,Kuroda S,Carpenter D,et al.Craniosynostosis in transgenic mice overexpressing Nell-1. J Clin Invest. 2002; 110(6):861-870.[12] Zhang X,Carpenter D,Bokui N,et al.Overexpression of Nell-1, a craniosynostosis. J Bone Miner Res. 2003;18(12):2126-2134.[13] Zhang X, Kang T, Bessette C M, et al. Nell-1, a key Functional Mediator of Runx2, Partially Rescues Calvarial Defects inRunx2+/−Mice.J Bone Miner Res.2011 ;26(4):777-791.[14] Zhang X,Péault B,Chen W,et al.The Nell-1 growth factor stimulates bone formation by purified human perivascular cells. Tissue Engineering Part A.2011;17(19-20):2497.[15] Desai J,Shannon ME,Johnson MD,et al.Nell1-deficient mice have reduced expression of extracellular matrix proteins causing cranial and vertebral defects. Human Molecular Genetics.2006; 15(8):1329.[16] Aghaloo T,Cowan CM,Chou YF,et al.Nell-1-induced bone regeneration in calvarial defects.Am J Pathol. 2006;169(3): 903-915.[17] 胡镜宙,蒋欣泉,张志愿,等.实时定量PCR检测Nel样I型分子基因对大鼠骨髓基质细胞成骨相关基因表达的影响[J].中国口腔颌面外科杂志,2008, 6(1):48-53.[18] Fahmygarcia S,Van DM,Wittebuoma J,et al.Nell-1, HMGB1 and CCN2 enhance migration and vasculogenesis, but not osteogenic differentiation compared to BMP2. Tissue Eng Part A. 2018; 24(3-4):207-218[19] Li G,Liu J,Wang Y,et al.LNGFR targets the Wnt/β-catenin pathway and promotes the osteogenic differentiation in rat ectomesenchymal stem cells.Sci Rep.2017;7(1):11021.[20] Shen J,James AW,Chung J,et al.NELL-1 promotes cell adhesion and differentiation via Integrinβ1. J Cell Biochem. 2012;113(12): 3620-3608.[21] Shen J,James AW,Zhang X,et al.Novel Wnt Regulator NEL-Like Molecule-1 Antagonizes Adipogenesis and Augments Osteogenesis Induced by Bone Morphogenetic Protein 2. Am J Pathol. 2016;186(2):419-434.[22] 张弘,姚宇,孙佳栋,等. NELL-1调控RUNX2的P1启动子诱导成骨分化[J].中华口腔医学研究杂志(电子版), 2017, 11(4):197-203.[23] Li CS,Zhang X,Péault B,et al.Accelerated Chondrogenic Differentiation of Human Perivascular Stem Cells with NELL-1. Tissue Eng Part A.2016;22(3-4):272-285[24] Mishra R,Sefcik R,Bishop T,et al.Proliferation and Osteogenic Differentiation of Human Mesenchymal Stem Cells on Poly(propylene fumarate) Tissue Engineering Scaffolds[C]// Osuwmc Trainee Research Day. 2015.[25] Cowan CM,Zhang X,James AW,et al.NELL-1 increases pre-osteoblast mineralization using both phosphate transporter Pit1 and Pit2. Biochem Biophys Res Commun.2012;422(3): 351-357.[26] Liu M,Wang Q,Tang R,et al.Nel-like Molecule 1 Contributes to the Odontoblastic Differentiation of Human Dental Pulp Cells. J Endod.2016;42(1):95-100.[27] Lee M,Siu RK,Ting K,et al.Effect of Nell-1 delivery on chondrocyte proliferation and cartilaginous extracellular matrix deposition.Tissue Eng Part A.2010;16(5):1791-1800.[28] James AW,Pan A,Chiang M,et al.A new function of Nell-1 protein in repressing adipogenic differentiation. Biochem Biophys Res Commun.2011;411(1):126-131.[29] 秦雪嫣,赵华翔,张倩,等. NELL-1:高效特异的新型生长因子[J]. 北京大学学报(医学版),2016,48(2):380-383.[30] Shen J,James AW,Zara JN,et al.BMP2-Induced Inflammation Can Be Suppressed by the Osteoinductive Growth Factor NELL-1.Tissue Engineering Part A.2013;19(21-22):2390.[31] 郭旭,彭江,王玉,等.腺病毒介导的NELL1基因对小鼠成骨细胞增殖分化的影响[J].解放军医学院学报,2012,33(5):509-511.[32] 陈微微. NELL-1蛋白对小鼠ATDC5细胞软骨分化的调控机制和对人血管周细胞成骨分化作用的研究[D].杭州:浙江大学, 2011.[33] Lee S, Wang C, Pan HC,et al.Combining Smoothened Agonist (SAG) and NEL-like protein-1 (NELL-1) Enhances Bone Healing. Plast Reconstr Surg.2017;139(6):1385-1396[34] Zhu S,Zhang B,Man C,et al.NEL-like molecule-1-modified bone marrow mesenchymal stem cells/poly lactic-co-glycolic acid composite improves repair of large osteochondral defects in mandibular condyle. Osteoarthritis Cartilage.2011;19(6):743-750.[35] 陈明,张宇,倪龙兴. nell-1基因直接体内转染修复犬下颌骨的缺损[J]. 医学争鸣,2007,28(13):1211-1213.[36] 胡镜宙. Nell-1基因修饰的组织工程化骨修复颌骨缺损的实验研究[D]. 上海:上海交通大学, 2007.[37] Kwak J,Zara JN,Chiang M,et al.NELL-1 Injection Maintains Long-Bone Quantity and Quality in an Ovariectomy-Induced Osteoporotic Senile Rat Model. Tissue Eng Part A. 2013;19(3-4): 426-436.[38] Li W,Zara JN,Siu RK,et al.Nell-1 enhances bone regeneration in a rat critical-sized femoral segmental defect model. Plast Reconstr Surg. 2011;127(2):580-587.[39] Xue J,Peng J,Yuan M,et al.NELL1 promotes high-quality bone regeneration in rat femoral distraction osteogenesis model.Bone. 2011; 48(3):485.[40] Zhu S,Song D,Jiang X,et al.Combined effects of recombinant human BMP-2 and Nell-1 on bone regeneration in rapid distraction osteogenesis of rabbit tibia. Injury. 2011;42(12): 1467-1473.[41] Siu RK,Zara JN,Hou Y,et al.NELL-1 Promotes Cartilage Regeneration in an In Vivo Rabbit Model.Tissue Engineering Part A.2012;18(3-4):252.[42] 邹俊. Nell-1基因诱导大鼠脊柱后外侧融合的实验研究[D]. 苏州:苏州大学, 2007.[43] James AW,Shen J,Tsuei R,et al.NELL-1 induces Sca-1+ mesenchymal progenitor cell expansion in models of bone maintenance and repair. JCI Insight.2017;2(12).pii: 92573.[44] Lu SS,Zhang X,Soo C,et al.The osteoinductive properties of Nell-1 in a rat spinal fusion model. 2007;7(1):50-60.[45] Siu RK,Lu SS,Li W,et al.Nell-1 protein promotes bone formation in a sheep spinal fusion model. Tissue Engineering Part A.2011; 17(7-8):1123.[46] Lee S,Zhang X,Shen J,et al.hPSCs and NELL-1 Synergistically Enhance Spinal Fusion in Osteoporotic Rats.Stem Cells.2015; 33(10):3158.[47] Yuan W,James AW,Asatrian G,et al.NELL-1 based demineralized bone graft promotes rat spine fusion as compared to commercially available BMP-2 product. J Orthop Sci. 2013;18(4):646-657.[48] 郭旭.Ad-GFP-NELL1对聚乙烯颗粒致颅骨骨溶解的防治作用[D]. 北京:军医进修学院解放军总医院,2012.[49] 郭旭,彭江,王玉,等.腺病毒介导NELL1促进骨溶解后的高质量骨再生[J].中国矫形外科杂志,2012,20(9):834-837.[50] James AW,Shen J,Zhang X,et al.NELL-1 in the treatment of osteoporotic bone loss.2015;6(Suppl 6):7362. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [5] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| [13] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [14] | Wei Congcong, Yao Mengxuan, Yang Meng, Li Huijie. Mechanism and treatment of osteolysis around artificial joint prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(27): 4401-4407. |
| [15] | Zheng Feng, Zhang Fucai, Xu Zhe. MicroRNA-98-5p promotes osteoblast proliferation and differentiation: possibilities and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4112-4117. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||