Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (36): 5897-5904.doi: 10.3969/j.issn.2095-4344.1000
Molecular mechanisms of bone morphogenetic protein, Wnt, fibroblast growth factor and sonic hedgehog signaling pathways in tooth development
Liu Zhe1, 2, Lian Wenwei 1, 2
- (1Department of Prosthodontics, Affiliated Stomatological Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China; 2Key Laboratory of Oral Biomedicine of Jiangxi Province, Nanchang 330006, Jiangxi Province, China)
-
Received:2018-08-15 -
Contact:Lian Wenwei, Master, Chief physician, Master’s supervisor, Department of Prosthodontics, Affiliated Stomatological Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China; Key Laboratory of Oral Biomedicine of Jiangxi Province, Nanchang 330006, Jiangxi Province, China -
About author:Liu Zhe, Master candidate, Department of Prosthodontics, Affiliated Stomatological Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China; Key Laboratory of Oral Biomedicine of Jiangxi Province, Nanchang 330006, Jiangxi Province, China
CLC Number:
Cite this article
Liu Zhe1, 2, Lian Wenwei 1, 2. Molecular mechanisms of bone morphogenetic protein, Wnt, fibroblast growth factor and sonic hedgehog signaling pathways in tooth development[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(36): 5897-5904.
share this article
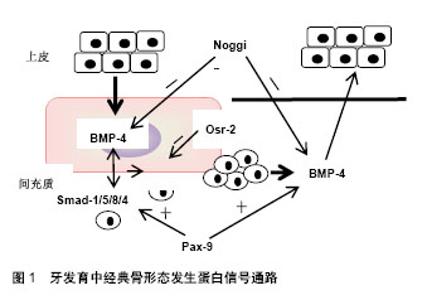
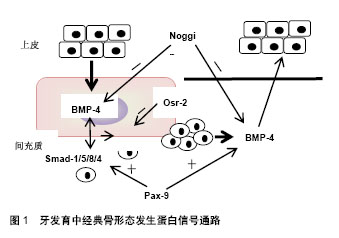
2.1 牙齿的发育过程 小鼠是研究牙齿发育过程中细胞生物学行为的理想模型。胚胎龄8.5(E8.5),来自于外胚层的具有多分化潜能的神经嵴细胞形成第一鳃弓,牙胚的发育开始。以后神经嵴细胞逐渐转变为间充质细胞。E10.5,特定位点的间充质细胞诱导局部口腔上皮细胞增殖,形成上皮板。此后,可将牙胚的发育过程概括为以下几个阶段:①蕾状期:E12.5-13.5,牙板末端膨大,形如花蕾,周围间充质细胞大量增殖,聚集在上皮下方。E13.5,在内层上皮中央形成簇状的细胞团,称为原发性釉结。原发性釉结为早期信号中心,可分泌多种细胞因子调节细胞增殖、分化和牙形态发生;②帽状期:始于E14.5左右,该期蕾状上皮向四周展开,呈帽状包绕间充质细胞。上皮层将来发育成釉质,分为3层细胞:内釉上皮层、外釉上皮层及内外上皮之间的星网状层。内釉上皮层原发性釉结发生退化,逐渐被继发性釉结取代。上皮下方包绕的间充质细胞突起形似乳头状,称为牙乳头,以后发育成牙本质和牙髓;③钟状期:E17.5- E19.5,牙乳头突起更明显,而两侧的内釉上皮与外釉上皮从颈环处向深层延伸,形似吊钟,牙胚发育逐渐成熟。从钟状期开始,细胞的命运已经决定,各区域的细胞表现为不同的形态和功能;④牙本质及釉质形成期:由上皮细胞分化而来成釉细胞与间充质细胞来源的成牙本质细胞各自产生基质并矿化。至此,牙齿发育基本完成[2]。 2.2 骨形态发生蛋白信号通路 骨形态发生蛋白是一类多功能糖蛋白,因能够诱导骨和软骨的形成而得名,目前共发现有20多种。骨形态发生蛋白作为配体蛋白,与细胞膜受体、胞外拮抗分子、Smad蛋白等因子共同构成了骨形态发生蛋白信号通路。研究发现,骨形态发生蛋白通路主要参与神经、骨骼、牙等外胚层器官发育,其发挥生物学效应可通过依赖Smad蛋白的经典途径和依赖丝裂原活化蛋白激酶(Mitogen-activated protein kinase,MAPK)非经典途径两种方式[3]。 经典通路中,骨形态发生蛋白与跨膜苏/丝氨酸激酶受体家族的骨形态发生蛋白受体(BMP Receptor,BMPR) -Ⅱ结合并使之磷酸化,活化的骨形态发生蛋白R-Ⅱ再招募骨形态发生蛋白R-Ⅰ,启动胞内信号传导。细胞质中,Smad-1/5/8最早发生磷酸化,并进一步与Smad-4 形成异源二聚体,转运至细胞核后调节靶基因的表达[4]。而MAPK介导的非经典途径路径由MAPK激酶激酶(MAP3K)-MAPK激酶(MAP2K)-MAPK组成,其中MAPK又包括ERK、p38、JNK和ERK5 4种亚型。路径中,骨形态发生蛋白在细胞膜位置与Ⅰ和Ⅱ型骨形态发生蛋白受体、肿瘤坏死因子受体相关因子6、转化生长因子激酶ab形成共聚体,泛素化TAK-1,再经MAP2K、MAPK传导转位至细胞核。骨形态发生蛋白信号通路是一庞大而又复杂的系统,受多重信号调控,使骨形态发生蛋白信号水平处于动态平衡的状态。Noggin、Osr-2、Usag-1、Dkk-2、Ectodin、卵泡抑制素(Folliculostatin)是骨形态发生蛋白胞外信号拮抗因子[5],一旦缺失,将严重影响器官的发育。 骨形态发生蛋白通路相关蛋白在牙发育中的表达和作用:骨形态发生蛋白,尤其是骨形态发生蛋白2,4,7对牙发育至关重要,不仅在牙胚发育的起始过程诱导间充质细胞的增殖,参与发育信号中心的构建,而且被认为是成釉细胞和成牙本质细胞最终分化的基础蛋白。 骨形态发生蛋白4是牙齿发育过程中最先表达的骨形态发生蛋白亚型,可介导上皮与间充质间信号传递,在蕾状期初始阶段诱导间充质细胞增殖、聚集和分化,至蕾状-帽状期过渡期,骨形态发生蛋白4诱导上皮原发性釉结的形成,参与牙胚形态发生。钟状期主要在牙源性细胞的前体细胞中表达,对决定细胞的命运和硬组织形成至关重要。具体作用机制表现为,蕾状早期上皮表达的骨形态发生蛋白4与间充质细胞表面骨形态发生蛋白受体Ib和骨形态发生蛋白受体-Ⅱ结合,经经典路径诱导核内Msx-1基因表达,进而上调间充质内骨形态发生蛋白4、淋巴增强因子(LEF)1等生长因子的水平,促进细胞的增殖和分化[6]。随着胚胎的发育,间充质内的骨形态发生蛋白4与上皮细胞膜表面的骨形态发生蛋白受体-Ia结合,刺激局部细胞增殖形成原发性釉结。在该过程中,Msx-1、Pax-9基因对骨形态发生蛋白引导的经典通路发挥正向调控作用,Noggin、Osr-2起负向作用,从而维持骨形态发生蛋白信号平衡。其中,同源框基因Msx-1、Pax-9牙板期接受上皮信号后,在间充质持续表达,促进骨形态发生蛋白、Smad蛋白的分泌[7]。Noggin可作用于骨形态发生蛋白4,抑制其与骨形态发生蛋白受体的结合,阻断信息传导。条件性敲除Noggin基因,上皮中的骨形态发生蛋白信号活跃,细胞大量增殖,导致部分细胞凋亡,破坏了牙胚的完整性,在小鼠模型中可表现出左右两侧上切牙牙胚的发育终止于蕾状期,并融合成一个牙胚[8]。Osr-2在蕾状期间充质中表达,抑制Msx-1基因功能,阻断骨形态发生蛋白信号传导,从而调控牙齿形态发生[9-10],见图1。"

骨形态发生蛋白2亦是牙发育关键的信号蛋白,在帽状期,釉结中大量存在,并持续表达至出生后。Laurikkala发现[11],釉结中,持续增加的骨形态发生蛋白2诱导骨形态发生蛋白信号拮抗剂Ectodin分泌,抑制Msx基因,从而调控骨形态发生蛋白信号强度。钟状期,骨形态发生蛋白2诱导上皮细胞和牙乳头细胞分化为成釉细胞及成牙本质细胞,参与牙硬组织形成、矿化和成熟。此外,骨形态发生蛋白2还具有成骨和成血管作用。牙成釉细胞相关蛋白是釉质发育必不可少一种蛋白分子。Lee等[12]发现成釉细胞分化期,上皮中的骨形态发生蛋白2与骨形态发生蛋白受体Ib结合,磷酸化胞内牙成釉细胞相关蛋白,活化的牙成釉细胞相关蛋白激活下游MAPK通路,参与成釉细胞的分化及釉质形成。牙本质发育中,骨形态发生蛋白2经p38 MAPK上调细胞内β-catenin含量,活化经典Wnt信号通路,诱导成牙本质细胞分化[13]。Rakian等[14]发现,骨形态发生蛋白2基因缺失,牙周组织中血管内皮生长因子A无表达,表现为微血管形成较少和骨发育缺陷。 骨形态发生蛋白7是牙胚发育重要的起始信号分子,主要表达下颌磨牙区。其作用机制与骨形态发生蛋白4类似,蕾状期由上皮细胞分泌后诱导间充质Msx-1、Msx-2的表达,参与牙胚的发生[15],在钟状期骨形态发生蛋白7可介导间充质细胞的分化[16]。干细胞再生实验中骨形态发生蛋白7和骨形态发生蛋白2彼此触发表达,协同诱导骨的形成[17]。Usag-1既是骨形态发生蛋白信号的拮抗因子,又是Wnt信号的拮抗因子。在牙胚发育中,Usag-1选择性结合骨形态发生蛋白7和Lef-1引起部分细胞的凋亡。实验显示,Usag-1基因表达缺失将导致发育个体出现多生牙[18]。 2.3 Wnt信号通路 Wnt信号通路是公认的器官和组织生长、发育中重要的分子信号级联,可参与决定细胞命运,并调控细胞的增殖、分化、迁移和极化。Wnt信号通路又进一步分为依赖β-连环素(β-catenin)经典型和非依赖β-catenin通路两种。经典通路中,Wnt-1,-2,-3,-8(a,b)和-10(a,b)等配体与卷曲蛋白(Frizzled ,FZD)和低密度脂蛋白相关蛋白(low density lipoprotein receptor related protein,LRP)-5/6构成复合体,抑制胞内β-catenin降解,随着β-catenin浓度升高激活核内LEF传递信息[19]。非经典通路包括Wnt/ca+通路,JNK通路和PKA通路,涉及的配体有Wnt-4,-5(a,b),-6,-7(a,b)和-11等。LRP-4可介导非经典通路信息负向传导[20]。 Dkk蛋白是重要的Wnt信号拮抗剂,可与LRP5/6结合,阻断典型通路信息传导[21]。Dkk家族有4种类型,即Dkk-1、Dkk-2、Dkk-3、Dkk-4。其中,Dkk-1和Ectodin、Sostdc1、Usag-1机制相类似,可结合LRP-6,阻断Wnt经典通路。而Dkk-2、Dkk-3与LRP-5/6或者膜表面的Kremen-2受体结合,表现出正向或者负向两种调节作 用[22]。Wnt信号胞外拮抗剂还包括分泌型的Frizzled相关蛋白(secreted frizzled-related protein,SFRP)、Wnt 抑制因子(WIF)-1、硬化蛋白。SFRP的机制为其与细胞膜上的FZD受体竞争性结合Wnt分子,屏蔽胞外Wnt信号,抑制信息传导[23]。AXIN2基因是Wnt信号的胞内拮抗物,其蛋白产物通过降解β-catenin负向调控Wnt信号通路[24]。 Wnt通路相关蛋白在牙发育中的表达和作用:Wnt信号通路不仅参与牙胚起始,介导上皮和间充质的信息传导,促进细胞的增殖、分化,而且调控牙胚形态发生和牙齿数量,对牙硬组织的形成亦有一定的诱导作 用[25]。其中,Wnt-10(a、b)和Wnt-5(a、b)是两种代表性Wnt蛋白亚型,作用于牙胚发育的起始期-成熟期整个阶段。 牙板期,Wnt-10b表达于上皮的局部增厚区,介导细胞的有丝分裂。蕾状早期(E13),上皮及间充质区均可见Wnt-10b的表达。帽状早期(E14-E15),Wnt-10b在继发性釉结区表达减少,可能与该区细胞有丝分裂减弱有关,而继发性釉结区以外的上皮及间充质Wnt-10b表达量持续增加。帽状后期(E15-E17),成釉器,包括釉上皮、中间层和星网状层中Wnt-10b表达减少,而颈环部位表达明显增加。钟状期(E19),在成牙本质细胞前体细胞中检测到Wnt-10b的表达[26]。提示Wnt-10b主要承担上皮与间充质信息交换,通过经典通路参与细胞的增殖、牙冠及牙根形态发生和牙本质形成。Wnt-10a主要表达于原发性釉结和继发性釉结中,可诱导间充质Wnt-10a表达,引发信息传导,参与细胞分裂和牙尖的形成。进一步研究发现,Wnt-10a是牙本质涎磷蛋白(DSPP)的上游调控蛋白,在牙本质发育中发挥不可替代的作用[27]。 Wnt5a是Wnt非经典通路的重要蛋白,可激活p42/44 MAPK、p38 MAPK、JNK和AKT信号通路刺激细胞增殖,对牙齿形态发生及牙本质生成亦具有促进作用[28]。牙板期,Wnt5a和Wnt的激动剂/抑制剂Mfrzb1和 Mfrp2在间充质区微弱表达,至帽状期,牙乳头区和成釉器的各个结构,尤其是继发性釉结中表达量的明显增加。钟状期,Wnt5a的表达集中于釉上皮、成牙本质细胞层和牙乳头的尖端。实验发现,Wnt5a能够增强Wnt/β-catenin通路信号活性,Wnt5a缺失,小鼠牙齿表现为形态小、牙尖圆钝及牙本质发育缺陷[29]。 Wnt通路的其他相关蛋白对牙齿的发育亦起着重要作用。Wnt3、Wnt4、Wnt6和Wnt受体MFz6在牙胚发育的早期阶段上皮上及周围即有表达。对于牙胚细胞的增殖具有重要作用。帽状期,Wnt蛋白主要表达于釉结中,参与调控牙胚形态发生。Wnt7b是牙胚初始阶段的重要因子,在此阶段邻接牙胚的上皮因Wnt-7b抑制Shh表达未增厚,由此而产生的口腔和牙胚间的分界使牙齿得以在既定的区域形成[30]。此外,Wnt/β-catenin还参与控制牙齿的数量。多项资料表明,牙胚发生初期超活化Wnt/β-catenin信号将引起上皮中Shh、特异性蛋白6 (Sp6,又称 Epfn)、成纤维细胞生长因子8表达增加诱发多生牙产生[31]。Dkk、LRP-4等负向调控信号的存在对于小鼠牙数的控制以及牙间隙的维持亦具有重要意义。 Wnt蛋白与其他通路的对话机制是牙胚发育重要的调控手段,对于牙胚形态正确形成和硬组织发育至关重要。蕾状期,间充质中Wnt/β-catenin诱导成纤维细胞生长因子3在该区的表达,并间接促进了原发性釉结的形成[25]。帽状期上皮中,Wnt/β-catenin通路中淋巴增强因子能够诱导釉结成纤维细胞生长因子4的表达[32],并与间充质中成纤维细胞生长因子10控制釉结中Shh表达量[31]。在间充质区Wnt/ β-catenin通路诱导Pax-9、Msx-1基因的表达上调骨形态发生蛋白水平。钟状期,正是由于间充质区Wnt/β-catenin通路的支持,保证了足够的骨形态发生蛋白的表达,使得牙源性细胞的分化和硬组织形成顺利进行[33]。研究发现。Wnt-5a及其受体Ror2诱发的非经典通路信号对骨形态发生蛋白、成纤维细胞生长因子、Shh通路信号传导亦有促进作用[28]。 2.4 成纤维细胞生长因子信号通路 成纤维细胞生长因子是一类分泌型肽类分子,因能够促进大脑垂体成纤维细胞生长而得名。至今在人和鼠中共鉴定出23种成纤维细胞生长因子。研究表明,成纤维细胞生长因子可与成纤维细胞生长因子受体(FGF receptor),一种特定的酪氨酸激酶跨膜受体(Tyrosine Kinase Receptors,TKR),结合激活不同传导路径,主要为Ras/MAPK通路发挥生物学效应,调控器官的形态发生、细胞的增殖、分化和迁移。这种多样化的活动需要通过诱导不同的反馈抑制剂,如Sproutys、成纤维细胞生长因子基因相似表达子(Similar expression to FGF genes,Sef)和MAPK磷酸酶3,来严格控制传导信号,限制成纤维细胞生长因子在时间和空间中的活动[34-35]。 成纤维细胞生长因子通路相关蛋白在牙发育中的表达和作用:成纤维细胞生长因子3,4,7,8,9,10,23是牙齿发育的关键因子,其中,成纤维细胞生长因子4,8,9,23存在于上皮中,成纤维细胞生长因子3,7,10主要在间充质中表达。成纤维细胞生长因子作为一类分裂素,能够促进DNA合成和细胞有丝分裂,参与牙胚细胞的增殖、分化。此外,成纤维细胞生长因子还可引起细胞非有丝分裂效应,对细胞迁移以及牙胚形态发生具有重要作用。 成纤维细胞生长因子4,8,9,23作为上皮信号,可在不同阶段介导组织间的相互作用,调控牙齿形态的发生及硬组织形成。此外,成纤维细胞生长因子4,8,9还可促进上皮中骨形态发生蛋白通路、Wnt通路信号传导,协同调控牙胚发育[36]。成纤维细胞生长因子8是牙胚发育重要的起始信号之一,主要作用于牙胚发育早期,后期牙胚中未检测到成纤维细胞生长因子8 mRNA的表达。牙板前期,成纤维细胞生长因子8维持了神经嵴细胞多元分化、自我更新等干细胞特征并促使其向间充质细胞转化。牙板期(E10),成纤维细胞生长因子8集中表达于上皮中,促进细胞增殖并延长了细胞存活时间,而且参与决定牙胚发生的位置[37]。 近来研究发现早期上皮细胞分泌成纤维细胞生长因子8和信号素3f(Sema3f)分别聚集和分散间充质细 胞[38],最终使细胞浓缩在一小范围内。感受到压力变化的细胞在RhoA基因调控下的表达Pax-9、Msx-1和骨形态发生蛋白4,开启牙胚发育的下一个阶段。成纤维细胞生长因子9亦参与牙胚发育的起始阶段,并且还调控牙胚形态发生和牙源性细胞分化。从E10开始,成纤维细胞生长因子9由牙胚发生部位的上皮细胞分泌,参与间充质细胞的增殖和聚集[39],研究发现间充质来源的Spry-4可与上皮成纤维细胞生长因子R-1结合,阻断成纤维细胞生长因子9通路信号的传导[40]。随着发育进行,成纤维细胞生长因子9和成纤维细胞生长因子4在原发性釉结、继发性釉结中共表达,参与牙齿形状调控和牙尖的形成[41]。该阶段,Notch信号通路利用成纤维细胞生长因子与骨形态发生蛋白建立起一套平衡系统,调节釉结信号传导。系统中,Notch受体与jagged-2编码的配体蛋白结合,成纤维细胞生长因子可上调jagged的表达,间充质内骨形态发生蛋白则抑制基因的表达。Jagged基因敲除模型中,釉结结构被破坏,导致磨牙牙尖数目异常增多,切牙则表现为釉质矿化不良[42]。钟状期开始(E18),成纤维细胞生长因子9分子由继发性釉结向内釉上皮扩散,刺激细胞表达釉质蛋白基因同时促进釉质蛋白分泌,至E19成釉细胞退缩时成纤维细胞生长因子9表达量减少。文献报道,成纤维细胞生长因子4,9能够诱导Msx-1基因及其同源框基因Msx-2、Lef的表达增强骨形态发生蛋白、Wnt通路信号活性[43]。 成纤维细胞生长因子23可调节血磷浓度,被认为广泛存在于各种组织中。近年来学者主张成纤维细胞生长因子23主要表达于成骨细胞及与成骨细胞特征类似的成牙骨质细胞、成牙本质细胞中[44]。在釉质及牙本质形成时,成纤维细胞生长因子23表达量明显增加。有研究显示,成纤维细胞生长因子23的分泌受phex基因的调 控[45-49],Tbx-1、Isl-1 基因也可能作用此期,促进釉质的发育。 成纤维细胞生长因子3,10主要存在于间充区,对于牙冠形态发生和上皮细胞分化、成熟的调控至关重 要[48]。蕾状后期间充质细胞分泌的成纤维细胞生长因子3后与上皮细胞膜上成纤维细胞生长因子受体1b和成纤维细胞生长因子受体2b结合刺激细胞增殖,并参与形成原发性釉结。另外,成纤维细胞生长因子3还诱导釉上皮向上凹陷及两侧向根方迁移。小鼠切牙牙胚发育过程中,唇侧内、外釉上皮连接处部位活化素(Activin)上调成纤维细胞生长因子3分子水平,使得细胞增殖并向根向延伸,形成正常釉质覆盖唇面。舌侧Folliculostatin限制成纤维细胞生长因子3信息传递,导致该区发育停滞,釉质缺如[40,49]。钟状后期,成纤维细胞生长因子3可诱导完成有丝分裂的乳头细胞分化为成牙本质细胞及邻近的内釉上皮细胞分化为成釉细胞。研究发现,成纤维细胞生长因子4,8是成纤维细胞生长因子3上游信号分子,能够刺激成纤维细胞生长因子3在间充质中的表 达[32]。成纤维细胞生长因子10与成纤维细胞生长因子3同时共表达于间充质中,参与上皮细胞的增殖及和细胞命运的决定。文献报道,上皮细胞分泌Spry-4与间充质成纤维细胞生长因子受体2结合[40],阻断间充质成纤维细胞生长因子3、成纤维细胞生长因子10信息传导通路。成纤维细胞生长因子7在磨牙中表达于牙胚周围的骨形成区,而切牙胚中成纤维细胞生长因子7主要存在于颈环周围的间充质中[4,8]。 2.5 Shh信号通路 Shh是一种分泌型多肽,其主导的通路在在细胞数量控制方面起到至关重要的作用。Shh通路主要由Shh、跨膜蛋白受体Ptch和Smo以及下游转录因子Gli蛋白组成,其中,Ptch和Gli既是Shh通路的组成部分,亦是通路作用的靶向因子[50]。 Shh通路相关蛋白在牙发育中的表达和作用:在发育的牙胚中,Shh被限制表达于上皮部分,在起始阶段和接下来的牙冠、牙根形态发生中发挥重要作用[51]。牙板期,Shh作为最重要的信号级联诱导上皮细胞增殖、聚集、迁移及内陷进入蕾状发育阶段[52]。蕾状期,Shh局限上皮中央,诱导原发性釉结的形成,而其周围Shh表达即减少。Ohazama等[53]发现,牙胚区域浓度逐渐增高的Ptch-1 和 Gli-1,扩散至邻近的牙间隙区,诱导Shh拮抗因子Gas-1的在间充质的表达,抑制Shh通路信息传导,从而避免了牙胚细胞的过度增殖,牙冠的形态和数量由此决定。Cobourne等[54]在小鼠磨牙胚中转载外胚层发育重要信号通路外异蛋白(EDA)/EDAR延长Shh的表达能够产生额外牙胚。上述研究均表明Shh通路对于牙齿数量的控制至关重要。帽状期,Shh在继发性釉结中表达增加,至钟状期,Shh在继发性釉结持续高表达,正向调节内釉上皮的碱性磷酸酶活性及釉质蛋白的表达[55]。此外,Shh在颈环上皮中亦有强烈表达,诱导颈环处上皮增殖并向根方延伸形成上皮根鞘,与此同时,颈环周围间充质部分Ptch-1和Gli-1表达量亦增加,细胞分化开始。 在4条通路中Shh通路通常作为其他3条通路的下游通路参与调控活动,其中,Shh通路重要组成部分Pitx-2基因起到重要的介导作用[56]。牙胚发育早期,Shh的表达需要骨形态发生蛋白、成纤维细胞生长因子8信号通路的支持。研究显示,牙板期,成纤维细胞生长因子8 能够上调 Pitx-2基因表达水平,刺激Shh通路的活化。蕾状期(E12.5和E13.5),利用noggin基因减弱间充质骨形态发生蛋白4信号可抑制上皮Shh的表达[57]。而且上皮中骨形态发生蛋白/Smad可调控颈环部位Shh-Gli-1信息传导,进而刺激颈环周围间充质中核心因子ic(Nuclear factor ic,Nfic)的表达,促进牙根的发育[58]。在受到上皮生长因子调控时,Shh的表达还强烈依赖于间充质中Wnt5a[59]、成纤维细胞生长因子3、成纤维细胞生长因子10等因子。"

| [1] Cai J, Zhang Y, Liu P, et al. Generation of tooth-like structures from inte gration-free human urine induced pluripotent stem cells. Cell Regen (Lond). 2013;2(1):6 [2] Obara N, Suzuki Y, Irie K, et al.Expression of planar cell polarity genes during mouse tooth development. Arch Oral Biol. 2017;83: 85-91.[3] Mu Y,Gudey SK,Landström M.Non-Smad signaling pathways.Cell Tissue Res.2012; 7: 11-20.[4] Kohei Miyazono, Shingo Maeda, TakeshiI mamura.BMP receptor signaling: Transcriptional targ ets, regulation of signals, and signaling cross-talk.2005; 6(3):251-263.[5] Wang XP,Suomalainen M,Jorgez CJ,et al.Follistatin regulates enamel pattern in mouse inci-sior by asymmetrically inhibiting BMP signaling and ameloblast differentiation.Dev Cell. 2004; 7(5):719-30.[6] Guobin Yang, uohua Yuan, Wenduo Ye et al. An Atypical Canonical Bone Morphogenetic Protein (BMP) Signaling Pathway Regulates Msh Homeobox 1(Msx1) Expression during Odontogenesis. J Biol Chem. 2014; 289(45):31492-31502. [7] Kriangkrai R,Iseki S,Eto K,et al.Dual odontogenic origins develop at the early stage of rat maxillary incisor development. Anat Embryol. 2006; 211(2):101-108. [8] Hu X,Wang Y,He F, et al. Noggin Is Required for Early Development of Murine Upper Incisors.J dent Res. 2012; 9(4): 394-400.[9] Jia S,Kwon HE,Lan Y,et al.BMP4-Msx1 signaling and Osr2 control tooth organo-genesis through antagonistic regulation ofsecreted Wnt antagonists.Dev Biol.2016;420(1):110-119.[10] Kwon HJ,Park EK,Jia S,et al.Deletion of Osr2 Partially Rescues Tooth Development i- n Runx2 Mutant Mice.J Dent Res.2015; 94(8):1113-1119.[11] Laurikkala J,Kassai Y,Pakkasjärvi L,et al.Identification of a secreted BMP antagonist, ectodin, integrating BMP, FGF, and SHH signals from the tooth enamel knot. Dev Biol.2004;264(1): 91-105.[12] Lee HK,Park JT,Cho YS,et al.Odontogenic ameloblasts- associated protein (ODAM), via phosphorylation by bonemorphogenetic protein receptor type IB (BMPR-IB), is implicated in ameloblast differentiation.J Cell Biochem.2012; 113(5):1754-1765.[13] Yang J,Hui TQ,Yang DM,et al.Bone morphogeneic protein 2-induced human dental pulp cell differentiation involves p38 mitogen activated protein kinase actiwated canonical wnt pathway. Int Journal Of Oral Science.2015; 7:95-102.[14] Rakian A, Yang WC, Gluhak-Heinrich J, et al.Bone morphogenetic protein-2 gene controls tooth root development in coordination with formation of the periodont- ium.Int J Oral Sci.2013;5(2): 75-84.[15] Ta?l? PN, Ayd?n S, Yalvaç ME, et al.BMP 2 and BMP 7 Induce Odonto- And Osteogenesis of Human Tooth Germ Stem Cells. Appl Biochem Biotechnol.2014; 172:3016-3025.[16] Gao B, Zhou X, Zhou X, et al.BMP7 and EREG Contribute to the Inductive Pot-ential of Dental Mesenchyme. Science report.2015; 5: 9903[17] 徐冉,张宇,邢舰誉, 等.BMP7对三维条件下小鼠iPS细胞成骨能力的影响[J].口腔医学研究,2016,32(4) : 322-325.[18] Murashima-Suginami A,Takahashi K,Sakata T,et al.Enhanced BMP signa-ling results in supernumerary tooth formation in USAG-1 deficient mouse.Biochem Biophys Res Commun.2008;369: 1012-1016.[19] Duan P, Bonewald LF. The role of the wnt/β-catenin signaling pathway in formation and m-aintenance of bone and teeth.Int J Biochem Cell Biol.2016; 77:23-29. [20] Gordon MD,Nusse R.Wnt signaling: multiple pathways, multiple receptors, and multip-le transcription factors.J Biol Chem.2006; 281(32) : 22429-22433.[21] Peng L, Dong G, Xu P, et al.Expression of Wnt5a in tooth germs and the related signal tra- nsduction analysis. Arch Oral Biol. 2010; 55(2):108-114.[22] Ahn Y, Sims C, Murray MJ, et al.MurrayMultiple modes of Lrp4 function in modu- lation of Wnt/β-catenin signaling during tooth development. Development.2017; 144: 2824-2836.[23] Bovolenta P, Esteve P, Ruiz JM, et al. Beyond Wnt inhibition: new functions of secreted Frizzled-related proteins in development and disease.J Cell Sci.2008;121(6):737-746. [24] Alldredge A, Fuhrmann S. Loss of Axin2 Causes Ocular Defects During Mouse Eye Devel-opment. Invest Ophthalmol Vis Sci.2016; 57(13):5253-5262.[25] Chen J, Lan Y, Baek JA, et al.Wnt/beta-catenin signaling plays an es- sential role in activation of odontogenic mesenchyme during early tooth development.Dev Biol.2009;334(1) :174-185.[26] Nadiri A, Kuchler-Bopp S, Haikel Y.Immunolocalization of BMP-2/-4, 成FGF-4, and WN-T10b in the developing mouse first lower molar.J Histochem Cytochem.2004;52(1):103-112.[27] Yamashiro T,Zheng L,Shitaku Y,et al.Wnt10a regulates dentin sialophosphoprotein m-RNA expression and possibly links odontoblastdifferentiation and tooth morphogenesis. Differentiation.2007;75(5):452-462. [28] Peng L, Dong G, Xu P, et al. Expression of Wnt5a in tooth germs and the related signal tra- nsduction analysis. Archives of oral biology.2010; 55 : 108-114.[29] Lin M,Li L,Liu C,et al.Wnt5a regulates growth, patterning, and odontoblast differ-entiation of developing mouse tooth.Dev Dyn. 2011; 240(2):432-440.[30] Jingjing Li, Lemonia Chatzeli, Eleni Panousopoulou et al. Epithelial stratification and pla- code invagination are separable functions in early morphogenesis of the molar tooth. Development. 2016; 143: 670-681.[31] Aurrekoetxea M, Irastorza I, García-Gallastegui P, et al. Wnt/β- Catenin Re-gulates the Activity of Epiprofin/Sp6, SHH, FGF, and BMP to Coordinate the Stages of odontogenesis. Front Cell Dev Biol. 2016; 4:25.[32] Kratochwil K, Galceran J, Tontsch S, et al.BMP4, a direct target of LEF1 and Wnt signaling, can rescue the arrest of tooth organogenesis in Lef1−/− mice. Genes Dev. 2002;16(24):3173-3185.[33] Fujimori S, Novak H,Weissenböck M,et al.Wnt/β-catenin signaling in the dental mesenc-hyme regulates incisor development by regulating BMP4.Dev Biol.2010; 348(1):97-106.[34] Thisse B, Thisse C.Functions and regulations of fibroblast growth factor sig-naling during embryonic development.Developmental Biology.2005;287(2):390-402.[35] 欧明明,黄晓峰,韩培彦.成纤维细胞生长因子与器官发育[J] .中国组织工程研究, 2011,15(15):488-492.[36] Kriangkrai R,Iseki S,Eto K,et al.Dual odontogenic origins develop at the early stage of rat maxillary incisor developmen.Anat Embryol (Berl).2006;211(2):101-108.[37] Shao M, Liu C, Song Y, et al.FGF8 signaling sustains progenitor status and multipotency of cranial neural crest-derived mesenchymal cells in vivo and in vitro. J Mol Cell Biol. 2015;7(5): 441-454.[38] Mammoto T, Mammoto A, Torisawa YS,et al. Mechanochemical control of mesenchymal condensation and embryonic tooth organ formation.Dev Cell.2011;21(4) : 758-769.[39] Tai YY, Chen RS, Lin Y, et al.FGF-9 accelerates epithelial invagination for ectodermal organogenesis in real time bioengineered organ manipulation.Cell Commun Signal.2012;10 : 34.[40] Klein OD, Lyons DB, Balooch G, et al.An FGF signaling loop sustains the generation of differentiated progeny from stem cells in mouse incisors. Development.2008; 135(2) : 377-385.[41] Nadiri A, Kuchler-Bopp S, Haikel Y,et al.Immunolocalization of BMP-2/-4, FGF-4, and WNT10b in the Developing Mouse First Lower Molar. J Histochem Cytochem. 2004;52(1):103-112.[42] Mitsiadis TA, Graf D, Luder H, et al.BMPs and FGFs target Notch sig-nalling via jagged 2 to regulate tooth morphogenesis and cytodifferentiation. Develo pment.2010; 137(18) : 3025-3035.[43] Laurikkala J,Kassai Y,Pakkasjärvi L,et al.Identification of a secreted BMP antagonist,ecto-din, integrating BMP, FGF,and SHH signals from the tooth enamel knot.Dev Biol.2003;264(1): 91-105.[44] Yoshiko Y, Wang H, Minamizaki T, et al. Mineralized tissue cells are a principal source of FGF23 . Bone.2007;40(6): 1565-1573.[45] Onishi T, Umemura S, Shintani S, et al. Phex mutation causes overexpression of FGF23 in teeth. Arch Oral Biol.2008;53(2): 99-104.[46] Mitsiadis TA, Tucker AS, De Bari C, et al. A regulatory relation- ship between Tbx1 and FGF signaling during tooth morphogenesis and ameloblast lineage determination. Dev Biol. 2008;320(1):39-48.[47] Naveau A, Zhang B, Meng B, et al.Isl1 Controls Patterning and Mineralization of Enamel in the Continuously Renewing Mouse Incisor. J Bone Miner Res. 2017;32(11):2219-2231.[48] Kettunen P, Laurikkala J, Itäranta P,et al. Associations of FGF-3 and FGF-10 with signaling networks regulating tooth morphogenesis. Dev Dyn.2000; 219(3):322-32.[49] Wang XP, Suomalainen M, Felszeghy S, et al.An Integrated Gene Reg-ulatory Network Controls Stem Cell Proliferation in Teeth. PLoS Biol.2007; 5(6) : e159.[50] 王苏平,吴晓君,闫旭,等. Sonic Hedgehog信号通路在胚胎发育及神经修复中的现状与进展[J].中国组织工程研究.2015,19(46): 7523-7528.[51] Khan M, Seppala M, Zoupa M, et al.Hedgehog pathway gene expre- ssion during early development of the molar tooth root in the mouse. Gene Expr Patterns. 2007;7(3):239-243.[52] Li J, Chatzeli L, Panousopoulou E, et al.Epithelial stratification and pla-code invagination are separable functions in early morphogenesis of the molar tooth.Development.2016;143: 670-681.[53] Ohazama A, Haycraft CJ, Seppala M, et al. Primary cilia regulate Shh ac- tivity in the control of molar tooth number. Development. 2009;136(6):897-903.[54] Cobourne MT, Sharpe PT. Making up the numbers: The molecular control of Mammalian dental formula. Semin Cell Dev Biol.2010; 21(3):314-324.[55] Takahashi S, Kawashima N, Sakamoto K, et al.Differentiation of an amelo-blast-lineage cell line (ALC) is induced by Sonic hedgehog signaling. Biochem Biophys Res Commun.2007; 353(2):405-411.[56] Fraser GJ,Graham A,Smith MM.Conserved deployment of genes during odontogenesis across osteichthyans.Proc Biol Sci.2004; 271(1555):2311-2317.[57] Li J, Feng J, Liu Y et al. BMP-SHH signaling network controls epithelial stem cell fate via regulation of its niche in the developing tooth.Dev Cell.2015;33(2):125-135.[58] Huang X, Xu X, Bringas P Jr,et al.Smad4-Shh-Nfic Signaling Cascade -Medi-ated Epithelial-Mesenchymal Interaction Is Crucial in Regulating Tooth Root Development.J Bone Miner Res.2010;25(5) : 1167-1178.[59] Cai J, Mutoh N, Shin JO, et al.Wnt5a plays a crucial role in determining tooth size during murine tooth development.Cell Tissue Res.2011;345(3):367-377. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||