Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (33): 5399-5407.doi: 10.3969/j.issn.2095-4344.0647
Previous Articles Next Articles
Treatment of complex anal fistula with adipose-derived stem cells: roles and mechanisms
Liu Yan-ni1, Ni Min2, Zhang Rui1, Huang Xiao-bo1, Zhou Chun-gen1, Jiang Bin2
- 1Nanjing University of Chinese Medicine, Nanjing 210000, Jiangsu Province, China; 2Nanjing Traditional Chinese Medicine Hospital, Nanjing 210000, Jiangsu Province, China
-
Revised:2018-07-22Online:2018-11-28Published:2018-11-28 -
Contact:Jiang Bin, MD, Chief physician, Nanjing Traditional Chinese Medicine Hospital, Nanjing 210000, Jiangsu Province, China -
About author:Liu Yan-ni, Master candidate, Nanjing University of Chinese Medicine, Nanjing 210000, Jiangsu Province, China -
Supported by:the Major Funded Projects of Nanjing Health Department, No. ZKX17034, ZKX15040
CLC Number:
Cite this article
Liu Yan-ni, Ni Min, Zhang Rui, Huang Xiao-bo, Zhou Chun-gen, Jiang Bin. Treatment of complex anal fistula with adipose-derived stem cells: roles and mechanisms[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(33): 5399-5407.
share this article
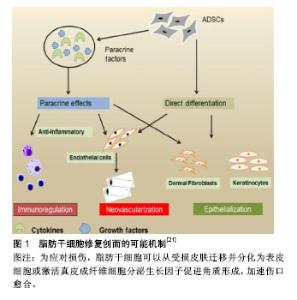
2.1 ASCs目前研究现状 间充质干细胞中应用最早的是骨髓干细胞,但由于骨髓干细胞提取有一定难度,组织含量较少,且随着年龄增长,骨髓干细胞数量会逐渐减少,临床使用中还存在伦理、法律及免疫排斥等方面的局限性[10-12]。随着ASCs的出现,克服了骨髓干细胞的缺陷,在许多疾病治疗中发挥着越来越重要的作用。起初,日本和欧洲学者利用ASCs构建工程化脂肪组织,用于软组织缺损修复、隆胸、面部填充、乳房再造等方面,获得了满意效果[13]。随后ASCs被用于组织修复与重建、自身免疫、体液免疫相关的各个系统疾病。在组织修复方面,ASCs已经被广泛应用于治疗骨科创伤、难愈性皮肤溃疡、糖尿病下肢病变等各类疾病[14-19]。在骨愈合临床试验中发现,ASCs与生物材料结合移植可增加骨折断端血管生成,促进骨质再生[14]。Jo等[15]进行关节腔注射自体ASCs治疗膝关节炎,未见严重不良反应。无论是低剂量组、中剂量组或高剂量组,关节腔注射ASCs后膝关节功能均在2年内得到明显改善,膝关节疼痛也得到明显减轻。Bura等[16]从自体脂肪中提纯出ASCs治疗下肢动脉闭塞导致的慢性溃疡,在创面边缘注射脂肪干细胞能有效减小创面的深度和范围,其中超过一半的患者创面达到了完全愈合。Rajashekhar等[17]将ASCs注射到糖尿病裸鼠视网膜功能障碍部位的血管网,发现局部注射ASCs可显著减少细胞凋亡和血管渗漏,增加血管生成,减轻神经退行性改变。 2.2 ASCs移植促进创面愈合的作用及机制 皮肤或组织创面愈合是涉及炎症、上皮形成、新生血管生成、增殖和胶原基质形成的复杂过程[20-21]。ASCs可通过多种机制来发挥这些作用,如诱导分化、调节炎症免疫、促进血管新生、激活成纤维细胞等作用,在创面愈合的各个环节中发挥重要的作用(图1)。Rodriguez等[22]在裸鼠皮肤伤口皮下注射ASCs,通过愈合后的皮肤免疫组织化学和血液灌注分析发现,与自发愈合组相比,ASCs治疗组创面愈合速度更快。Lee等[23]进行的一项初步研究中,采用肌肉内多次注射ASCs治疗严重肢体缺血患者,随访6个月发现 66.7%的患者临床症状、疼痛评分量表和跛行距离较治疗前均显着改善。总结文献发现, ASCs在创面愈合中的作用机制主要有以下几个方面。"
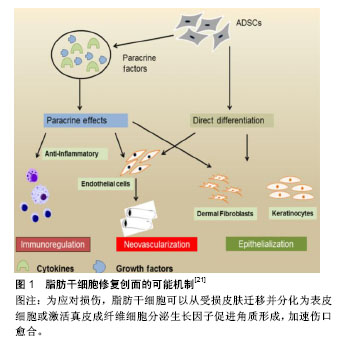
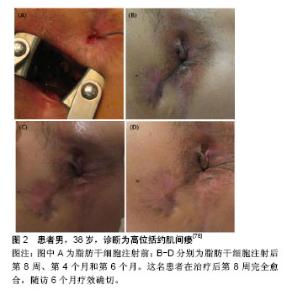
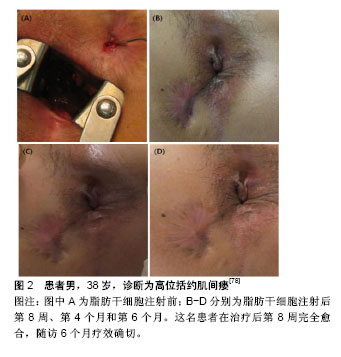
2.2.1 诱导分化作用 ASCs是一种多能细胞,具有较强的增殖能力,ASCs在不同诱导条件下可以分化为脂肪细胞、软骨细胞、成骨细胞、肌细胞、神经胶质细胞及神经细胞[24-26]。ASCs通过自分泌或者旁分泌方式分泌多种细胞因子,如成纤维生长因子2、血管内皮生长因子、表皮生长因子、肿瘤坏死因子α、血小板衍生生长因子和胰岛素样生长因子1等[27]。在缺氧情况下,ASCs还可刺激分泌多种细胞因子,加速创面愈合。Fischer等[28]将ASCs移植于创面局部,取愈合组织进行免疫荧光染色后发现,ASCs可表达血管内皮细胞标志物CD31,提示ASCs在体内可分化为血管内皮细胞和平滑肌细胞,参与创面的上皮重建[29]。ASCs的诱导分化作用也是创面愈合过程中组织细胞增长迅速的主要原因。 2.2.2 调节炎症反应 炎症反应是创面愈合的基础,但过度的炎症反应会导致局部组织愈合减慢,甚至出现愈合停止或组织坏死。ASCs通过诱导创面局部促炎因子分泌减少,抗炎因子增加,减轻创面的炎症反应[30-31]。从活化的外周血单核细胞中发现ASCs显著抑制促炎细胞因子如肿瘤坏死因子α的分泌。ASCs在治疗炎症性肠病方面的疗效显著,一个重要的原因可能由于MSCs调节机体炎症反应,减轻全身及局部炎症,促进创面愈合。 2.2.3 旁分泌及免疫调节作用 ASCs可以通过与细胞直接接触和旁分泌各种细胞因子等发挥免疫调节作用。几项研究表明,ASCs能够调节免疫系统中各种细胞的功能[32-33]。ASCs可与B淋巴细胞、T淋巴细胞、NK细胞、单核细胞衍生的树突细胞和嗜中性粒细胞相互作用,影响免疫细胞的分化和活化[34-38]。ASCs还可以释放各种与抑制免疫反应相关的细胞因子,如转化生长因子β、白细胞介素10、前列腺素E2、胰岛素样生长因子1和一氧化氮等[39-42]。ASCs通过调节T细胞诱导免疫系统的平衡,从而产生持续的治疗作用。意大利研究小组发现,直到随访1年后间充质干细胞治疗效果才能保持稳定。 2.2.4 促进血管新生和成纤维细胞激活 血管新生是创面愈合的关键环节,ASCs通过自分泌或者旁分泌方式分泌多种细胞因子,促进新生血管生成,建立和恢复局部血液供应,加速肉芽组织增生,促进难愈性创面愈合。在大鼠缺血后肢模型中,采用静脉内给药或肌肉内注射CD31阴性的ASCs,极大地改善了局部血管的血液供应,提高了缺血皮瓣的存活率[43-44]。Marino等[45]使用ASCs治疗外周动脉疾病患者的慢性皮肤溃疡,获得同样的结果。实际上,皮肤溃疡可能伴有严重的肢体缺血,即使周围组织成功重建后,仍然可能无法痊愈,灌注ASCs可以提高创面血液供应,增加组织血氧含量,加速创面愈合。成纤维细胞激活在血管新生中发挥重要作用。通过体内动物研究证实,在皮肤组织重塑过程中ASCs激活成纤维细胞,促进局部血管新生,从而加速创面边缘上皮化[46]。 2.3 ASCs治疗复杂性肛瘘的临床应用现状 肛瘘是肛管直肠周围间隙感染、损伤等因素形成的肛管直肠与肛周会阴皮肤相通的病理通道[47]。对于“复杂”肛瘘存在许多定义。1961年,Parks博士[48]将肛瘘分为括约肌间瘘、经括约肌瘘、括约肌上瘘和括约肌外瘘。2016年美国结直肠外科医师学会(ASCRS)更新的《肛周脓肿和肛瘘治疗指南》中[49-50],他们将“简单”瘘定义为括约肌间瘘或低位经括约肌瘘,侵犯外括约肌低于30%。“复杂”瘘定义中将术后易致肛门失禁的肛瘘均纳入复杂性肛瘘范畴,包括瘘管穿过肛门外括约肌30%以上的肛瘘(高位括约肌间、括约肌上和括约肌外)、女性前侧肛瘘以及复发性肛瘘或先前存在大便失禁的肛瘘,炎症性肠病或放射引起的肛瘘等。 2.3.1 目前国内外复杂性肛瘘手术治疗现状 复杂性肛瘘主要手术治疗方式有肛瘘切开挂线术和(或)分期肛瘘切开术、隧道式支管拖线术、纤维蛋白胶注射术、肛瘘栓填塞术、直肠黏膜推移皮瓣术、括约肌间瘘管结扎术(LIFT)以及括约肌间瘘管结扎术+肛瘘栓填塞术(LIFT Plug)等10余种手术方式[51-53]。以肛瘘切割挂线术为代表的传统手术,虽然手术治愈率较高,但术后创面大,疼痛剧烈,术后创面愈合时间长;手术损伤肛门括约肌,术后大便失禁发生率较高,给患者带来很大的心理及经济负担[54-56]。2015年的一项多中心回顾性研究报道537例患有低位瘘管(少于括约肌复合体的1/3)的患者,瘘管切开术后5年的愈合率为81%,26.8%的患者有严重失禁,只有26.3%的患者生活质量与正常人相当[57]。Ritchie等[58]报道切割挂线术后的平均失禁率为12%。随着瘘管内部开口的位置越移向近端,失禁率越高。非特异性肛腺感染性的肛瘘术后失禁率为18%。 2.3.2 微创手术治疗现状 随着外科技术的发展,微创观念的深入,外科手术治疗方式得到不断发展。肛瘘栓、纤维蛋白胶、直肠推移皮瓣术、括约肌间瘘管结扎术(LIFT)等为代表的微创手术逐渐开展。虽然早期研究数据显示肛瘘栓对低位肛瘘治疗成功率为70%-100%,但近期研究证明其愈合率低于50%[59-62]。一项多中心试验的肛瘘塞治疗复杂性肛瘘的荟萃分析显示,术后随访6个月和12个月,成功率分别为41%和49%[63]。在不同回顾性和前瞻性数据中显示,纤维蛋白胶治疗肛瘘成功率在14%-63%之间[64-65]。Lindsey等[64]进行的一项前瞻性试验中,纤维蛋白胶治疗6例单纯性肛瘘,3/6(50%)愈合,治疗复杂性肛瘘13例,9/13(69%)愈合。直肠推移皮瓣术是一种括约肌保留技术。研究报道显示肛腺感染性肛瘘采用直肠推移皮瓣治愈率为66%-87%[66-68],复发率较高。与修复治疗失败相关的因素包括既往放射线、潜伏期克罗恩病、活动性直肠炎、直肠阴道瘘、恶性肿瘤、肥胖以及先前尝试修复的次数有关[69-70]。尽管括约肌在瓣膜游离过程中并未分离,但内括约肌纤维可能部分会包绕在皮瓣中,报道中约35%的患者术后有轻度至中度失禁,术后测压报告静息压和收缩压下降[71]。括约肌间瘘管结扎术也是一种不损伤肛门括约肌的术式。一项251例括约肌间瘘管结扎术式的回顾性分析中发现,中位随访71个月的治愈率为87.65%。低位经括约肌瘘、括约肌间瘘、高位经括约肌瘘、半马蹄形瘘和马蹄形瘘的愈合率分别为92.1%,85.2%,60.0%,89.0%和40.0%[72]。而且,括约肌间瘘管结扎术式无法解决已有感染造成的损伤修复困难的问题。因此,各种微创手术也存在治愈率较低,愈合时间长,复发率较高等局限性。 2.3.3 ASCs治疗复杂性肛瘘 目前,文献报道ASCs用于肛腺感染性肛瘘、克罗恩肛瘘、直肠阴道瘘、炎症性肠病等相关的复杂性肛瘘均取得了较好的疗效。但由于目前ASCs处于早期临床应用阶段,不同实验室干细胞制备技术的差异,在不同文献中采用不同的实验剂量;各研究中心为探索手术适应证,将ASCs与各种肛瘘微创方式相结合;或者由于受试者自身条件或实验条件的限制,采用不同来源的ASCs治疗复杂性肛瘘。 (1)选择ASCs的剂量:由于各实验室对ASCs的培养、扩增技术不同,有1×107,2×107,3×107,4×107,9×106等不同剂量,也有ASCs结合纤维蛋白胶混悬液、ASCs结合基质血管成分混悬液等不同形式。2003年Garcia-Olmo等[73]学者报道了临床上首例使用ASCs治疗炎症性肠病相关的克罗恩肛瘘,该33岁女性患者,患有克罗恩病11年,因肛周脓肿术后形成直肠阴道瘘;使用肿瘤坏死因子α单抗和挂线引流术后症状没有改善,局部注射9×106自体ASCs治疗1周后,手术伤口完全闭合,仅留有轻微的炎症症状,无大便失禁或阴道出血,3个月内疾病未复发。2012年英国克利夫兰的北帝斯医院结直肠外科报道了3例自体ASCs治疗复杂性肛瘘患者,研究人员清理3例患者的瘘管,封闭内口后,将5 mL ASCs和20-30 mL脂肪组织中基质血管成分混悬液采用十字交叉格状注射到瘘管及周围组织内,达到最大的组织密度并填满相邻组织间隙,然后堵塞外口。经过两三年的随访,3例患者都能维持较好的治疗效果[74]。2013年,韩国报道了一项ASCs剂量递增Ⅰ期临床研究[75],10例克罗恩肛瘘患者分为3组,注射前彻底搔刮瘘管,用2-0 vicryl线缝合关闭内口,注射细胞体积与瘘道的大小成正比,将每组所需的ASCs均匀注入瘘管壁和瘘管内口周围黏膜组织中,再将凝血酶和纤维蛋白原的混合溶液灌注到瘘道中。前3例患者(组1)给予1×107/mL ASCs的第一次剂量注射,4周后,该剂量被认为是安全时,另外4例患者(组2)给予2×107/mL ASCs,其中1例患者中途退出。再过4周后,第2次剂量认为是安全的,第3次给最后一组的3例患者给予4×107/mL ASCs。注射8周后随访,第2组中2例患者显示完全愈合,第3组中1例患者显示完全愈合,其他患者均部分愈合,引流量较术前减少。对第8周显示完全愈合的3例患者又追踪随访6个月,以监测ASCs治疗的持续有效性和安全性。所有患者对治疗都表现出良好的耐受性,没有发生3级或4级严重不良事件,且没有发生与研究药物有关的不良事件。在跟踪治疗8个月后,仍保持疗效并无复发现象。因此,该研究显示了ASCs治疗克罗恩肛瘘的耐受性、安全性和潜在疗效,并为进一步的临床研究提供了基础支持。Lee等[76]进行了一项评估ASCs与纤维蛋白胶结合使用的Ⅱ期临床试验。首先打开瘘管,用2-0 vicryl缝合瘘管内口,ASCs注射在内口处黏膜下层及瘘管周围。瘘管用ASCs和纤维蛋白胶混合剂灌注。注射前用探针测量瘘管的直径和长度来确定瘘管的大小。当瘘管直径大约不超过1 cm时,每厘米瘘管长度注射3×107/mL ASCs,当瘘管直径在1.0-2.0 cm之间时,重复注射2次ASCs。注射8周后瘘管未愈合者,进行第2次注射。第2次注射ASCs的剂量为第1次注射所用细胞数量的1.5倍。注射第4,6,8周后拍摄病变部位,并与注射后第1天照片进行比较。根据修改后的方案分析,82%(27/33)的患者在第8周时表现出完全愈合,而在意向治疗分析中为64.3% (27/42)。在27例完全愈合患者中,26例完成1年随访,其中23例(88%)完全闭合。另一项研究对其长期结果进行了分析,在24个月时,经过修改后的方案分析中80.6% (21/26)患者完全愈合,在改进的意向性治疗分析中为75%(27/36)[77]。没有与ASCs治疗相关的不良事件发生。Choi等[78]比较1×107/mL ASCs与2×107/mL ASCs 2种不同剂量ASCs治疗与克罗恩不相关的复杂性肛瘘的临床安全性与有效性,将患者分为两组,组1使用1×107/mL ASCs,组2使用2×107/mL ASCs。注射后第8周评估疗效,第8周瘘管完全闭合者再随访6个月。未愈合者接受2倍的初始注射剂量。患者在第8周,第4个月和第6个月时分别拍照片记录肛周局部变化(图2)。注射ASCs的15例患者中13例完成了这项研究。在第8周时69.2%(9/13)患者瘘管完全闭合。第1组中5例患者中有3例,第2组中8例患者中有6例瘘管完全闭合,发现两组之间无明显差异。瘘管完全闭合的9例患者中有6例参与了随访,其中5例患者(83.3%)在第6个月时仍维持疗效。未观察到3级或4级不良事件。因此,对于传统手术无法治愈的复杂性肛瘘,ASCs移植可能是一个很好的选择方式。ASCs治疗肛瘘具有良好耐受性和安全性,但各实验室使用剂量不统一,需要大样本的临床试验寻找适合患者疾病特点的细胞剂量。"
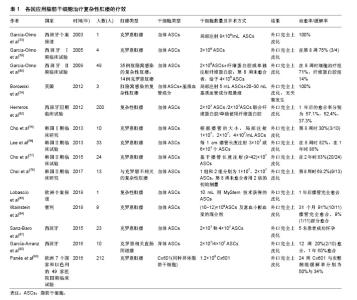
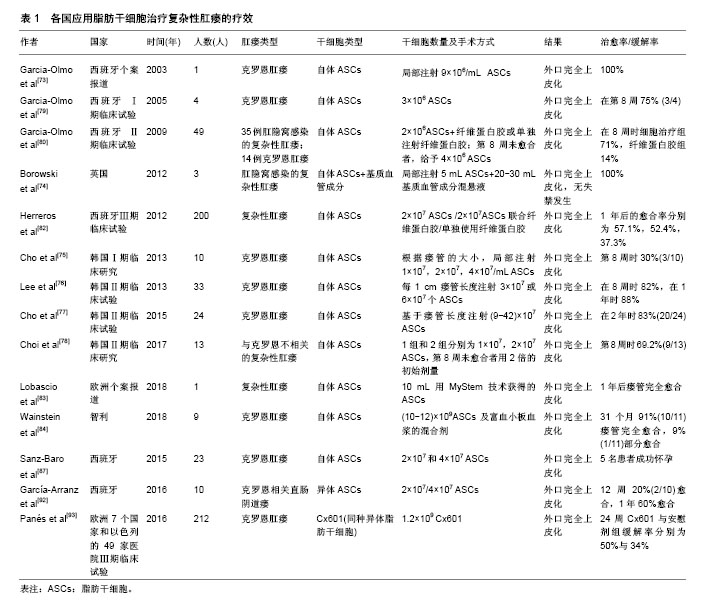
(2)ASCs与各种手术方式结合:目前ASCs治疗复杂性肛瘘的临床治疗方案不同,有单独使用ASCs、ASCs联合纤维蛋白胶或/和直肠内推移皮瓣等治疗。2005年,Garcia-Olmo等[79]Ⅰ期临床研究中,使用自体ASCs移植治疗4例克罗恩肛瘘,随访8周后治愈率为75%,证实了ASCs移植疗法治疗克罗恩肛瘘的可行性和安全性。因此,该团队在Ⅱ期多中心随机对照试验中[80],将49例复杂性肛瘘患者(肛腺感染性的普通肛瘘35例和克罗恩肛瘘14例),随机分配到纤维蛋白胶对照组和纤维蛋白胶联合2×106 ASCs治疗组,随访8周后,未治愈者,对照组采用第2种剂量纤维蛋白胶注射,治疗组用纤维蛋白胶联合4×106 ASCs治疗。在8周和1年时评估瘘管愈合情况和SF-12生活质量问卷,发现在治疗组的24例患者中17例(71%)瘘管愈合,而单独接受纤维蛋白胶的25例患者中,只有4例患者(16%)瘘管愈合。克罗恩肛瘘和普通肛瘘患者的愈合率相似。对于括约肌上肛瘘患者,治疗组比对照组更有效(P=0.001)。从而,在临床上评估了ASCs联合纤维蛋白胶注射治疗复杂性肛瘘的有效性和安全性。据报道,纤维蛋白胶可以提高细胞移植的存活率并增强移植细胞的生物学功能[81]。类似地,纤维素基质中的ASCs能在细胞凋亡条件下存活,并且在体外培养基中不补充营养素的情况下,可持续2周释放生长因子。此外,ASCs成功抑制T细胞增殖和活化促炎细胞因子。因此推测,ASCs中的纤维蛋白基质可能通过影响瘘管病变部位的微环境,从而对ASCs的治疗作用产生协同效果。2012年西班牙马德里报道的一项随机、双盲、多中心Ⅲ期临床研究中[82],19个临床中心的200例复杂型肛瘘患者随机被分为3组:A组64例患者接受2×107 ASCs治疗,B组60例患者接受2×107 ASCs联合纤维蛋白胶治疗,C组59例接受纤维蛋白胶治疗。如果第1次注射后,瘘管在12周时未愈合,则采用第2种注射剂量(A组和B组接受4×107 ASCs)。随访24-26周时,A,B,C 3组患者的愈合率分别为39.1%,43.3%,37.3% (P=0.79)。1年后的愈合率分别为57.1%,52.4%,37.3% (P=0.13)。这表明,2×107或4×107剂量的ASCs单独使用或联合纤维蛋白胶都是安全的,治疗半年后的愈合率可达到40%,1年后的愈合率可超过50%。近期ASCs治疗复杂性经括约肌肛瘘临床案例报道中[83],1例77岁的男性患者,在肛门左后侧有一长约6 cm,没有内口的盲瘘。患者曾经接受过脓肿切开引流术和肛瘘切开术。术前使用过氧化氢和氯化钠溶液进行每日瘘道灌洗。当瘘管停止排出脓液时进行外科手术。在腹部抽取脂肪组织,通过MYSTEM®EVO(一种非酶分离细胞技术)分离、浓缩后获得具有完整脂肪组织小叶的ASCs。用肛瘘刷搔刮瘘道,并用探针探查瘘管。从外口向假设的内口方向注射10 mL用MyStem技术获得的ASCs。外口使用21G皮下注射针注射10 mL ASCs在瘘管中。外口用3-0 Vicryl线缝合。患者术后第2天出院。术后第7,10,30和180天对患者进行随访。用肛门镜和经肛门超声检查证实瘘管是否愈合。术后1年时随访发现瘘管已完全愈合。Wainstein等[84]进行一项单中心、前瞻性观察性试验研究。该研究纳入了18岁以上的9例患者,均为临床缓解期或疾病稳定期,克罗恩疾病活动指数CDAI< 220,并且之前有过手术史和/或生物治疗史的难愈性克罗恩肛瘘患者。患者根据研究分2个阶段进行。第1阶段确定瘘管走行,清理瘘管,瘘管挂线引流,机械性吸脂获取约200 cm3皮下脂肪组织,并在实验室分离、培养和扩增获得ASCs。第2阶段抽取患者40-60 cm3的外周血,离心并提取获得富血小板血浆。去除挂线并对瘘管进一步清创,通过创建一个小的直肠黏膜推移皮瓣关闭瘘管内口,将(10-12)×109 ASCs及富血小板血浆的混合剂注入瘘管内口和瘘道。此外ASCs及富血小板血浆的混合溶液在填充瘘道之前用钙激活,从而形成生物塞,封堵瘘管。在治疗的11个瘘管,其中2个为回肠肛窝阴道瘘。中期随访31个月(21-37个月),发现91%(10/11)瘘管完全愈合,9%(1/11)部分愈合。所有患者的瘘管均没有复发,没有发生不良反应。手术后肛周疾病活动指数和炎症性肠病问卷评分均显著改善。因此,单独使用ASCs,ASCs联合纤维蛋白胶或/和直肠黏膜推移皮瓣治疗肛瘘都安全有效,临床治愈率较高[85-86]。 (3)ASCs的细胞来源:ASCs根据来源不同可分为自体、异体及同种异体。例如,2015年,西班牙进行了一项ASCs治疗克罗恩肛瘘相关的妇女妊娠临床回顾性观察研究[87]。23名肛瘘妇女每间隔三四个月依次接受2×107和4×107 2种剂量的自体ASCs局部灌注治疗。6名妇女在干细胞治疗缓解期成功怀孕,并且5名妇女足月妊娠,1名发生2次妊娠早期流产,流产率符合一般人群发生率(20%)[88],且明显低于文献报道与克罗恩疾病相关的流产率(40%)[89-91]。因此,ASCs治疗似乎并没有增加流产率,其认为接受ASCs治疗可以帮助克罗恩肛瘘患者跨越妊娠的心理障碍,减轻肠道炎症反应,增加怀孕成功率。最近一项Ⅰ-Ⅱ期临床试验研究异体ASCs治疗克罗恩相关直肠阴道瘘的安全性和可行性[92]。异体ASCs治疗10例直肠阴道瘘患者,如果外科医生认为有必要,则增加阴道或直肠推移皮瓣。治疗方案包括在阴道壁的黏膜下层和瘘道内注射2×107 ASCs。治疗12周后评估愈合情况,愈合定义为阴道和直肠两侧的再上皮化以及阴道没有引流物。治疗12周时2例患者愈合,8例患者接受4×107 ASCs注射。血液中细胞因子谱和免疫毒性检测,没有发现严重不良事件。随访1年,60%的患者达到完全愈合。这表明ASCs可作为不同类型的克罗恩病变的替代疗法之一。2016年,武田制药和TiGenix公司联合在欧洲7个国家和以色列的49家医院进行的一项随机、双盲、平行、安慰剂对照的Ⅲ期临床试验[93],总共212例克罗恩肛瘘患者纳入该试验。随机分为2组,治疗组107例采用1.2×109Cx601(同种异体脂肪干细胞),安慰剂组105例采用24 mL生理盐水。治疗24周后,Cx601治疗组与安慰剂组疾病缓解率分别为50%与34%(P=0.024);治疗相关的不良事件发生率分别为17%与29%;出现肛周脓肿分别为6例与9例,出现肛门痛分别为5例与9例。52周后,Cx601治疗组与安慰剂组缓解率分别为56.3%与38.6%(P=0.010),复发率分别为55.9%与75%(P=0.052)。在1年后随访发现CX601组与安慰剂组相比更加安全和有效。这一结果表明对传统治疗或生物治疗无效的克罗恩病肛瘘,同种异体脂肪干细胞移植是一种安全有效的治疗方法。由上可见,不同国家,不同临床中心采用了不同的细胞来源、剂量及手术方式(表1)。 研究间充质干细胞治疗复杂性肛瘘的安全性和有效性的临床试验日益增多。李森娟等[94]对干细胞移植治疗复杂性肛瘘的Meta分析中,实验组(干细胞移植组)与对照组(安慰剂组)比较,实验组能显著提高复杂性肛瘘的治愈率,是一种治疗复杂性肛瘘的有效手段。2018年一篇系统文献回顾中,间充质干细胞治疗难治性疾病的安全性和有效性研究[95]。间充质干细胞治疗的不良反应没有明显增加(OR=1.07,95%CI=0.61-1.89,P=0.81),严重不良反应也没有明显增加(OR=0.53,95% CI=0.28-0.98,P=0.04)。现有资料证明间充质干细胞治疗肛周克罗恩病较对照组能够提高疗效且没有增加不良反应。ASCs移植治疗复杂性肛瘘具有创伤小、不损伤肛门括约肌、可重复使用、治愈率高于其他生物制剂等优点。对那些使用常规外科手术或多次手术未能达到完全愈合的复杂性肛瘘患者,或合并有直肠阴道瘘以及有妊娠需要的克罗恩肛瘘患者,都是有效的治疗选择之一。 应用ASCs治疗复杂性肛瘘,是一项具有巨大发展潜力的医疗技术,可为一系列难治性结直肠外科疾病提供新的治疗方向,引起了各国临床医生、科研工作者的关注。国外文献研究也证实了ASCs治疗复杂性肛瘘疾病的安全性和有效性。但目前此研究仍处于初级阶段,可能存在一些问题需要进一步改进:①虽然ASCs来源丰富、取材简便,但干细胞不同于普通药物治疗,不同研究单位其培养方法、移植剂量和质量检测都有差异,因此确定ASCs最佳来源,建立ASCs标准化的培养体系,治疗流程非常重要;②临床研究大多是单中心、小样本回顾性病例报告,观察时间较短,样本量较少,缺乏有力的对照数据,未来需要开展多中心、大样本、前瞻性的临床随机对照研究;③目前虽然没有报道临床干细胞治疗增加致瘤风险,但建议在临床治疗前,需对ASCs进行体内外的致瘤分析。开展自体、异体及同种异体ASCs之间的实验研究及临床对照研究,明确各自的优缺点及适应证;④各临床中心的治疗评估体系不尽相同,可以采用统一的临床瘘管愈合评估标准以及实验室、影像学评估标准,增强结果的可信度;⑤自体ASCs移植需要进行2次手术,一次用于提取脂肪组织,另一次用于注射ASCs,总的治疗费用也高于一般手术,也给患者造成了一定经济压力。因此,加强对干细胞技术和人员的培养,降低干细胞提取过程的中间投资,减少治疗成本,从而更好的应用于临床。"
| [1] Minteer DM, Marra KG, Rubin JP. Adipose stem cells: biology, safety, regulation, and regenerative potential. Clin Plast Surg. 2015;42(2): 169-179.[2] Bajek A, Gurtowska N, Olkowska J, et al. Adipose-Derived Stem Cells as a Tool in Cell-Based Therapies. Arch Immunol Ther Exp (Warsz). 2016;64(6):443-454.[3] Ratajczak MZ. Stem cells and mechanisms regulating their trafficking - a new and challenging area of investigation in modern psychiatry. Prog Neuropsychopharmacol Biol Psychiatry. 2018;80(Pt A):1-2.[4] Bourin P, Bunnell BA, Casteilla L, et al. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy. 2013; 15(6):641-648.[5] Münch DP. Breast augmentation with autologous fat - experience of 96 procedures with the BEAULI-technique. Handchir Mikrochir Plast Chir. 2013;45(2):80-92.[6] Strioga M, Viswanathan S, Darinskas A, et al. Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells Dev. 2012;21(14): 2724-2752.[7] Gir P, Oni G, Brown SA, et al. Human adipose stem cells: current clinical applications. Plast Reconstr Surg. 2012;129(6):1277-1290.[8] Cadeddu F, Salis F, Lisi G, et al. Complex anal fistula remains a challenge for colorectal surgeon. Int J Colorectal Dis. 2015;30(5): 595-603.[9] Limura E, Giordano P. Modern management of anal fistula. World J Gastroenterol. 2015;21(1):12-20.[10] Rasmussen JG, Frøbert O, Holst-Hansen C, et al. Comparison of human adipose-derived stem cells and bone marrow-derived stem cells in a myocardial infarction model. Cell Transplant. 2014;23(2): 195-206.[11] Mimeault M, Hauke R, Batra SK. Stem cells: a revolution in therapeutics-recent advances in stem cell biology and their therapeutic applications in regenerative medicine and cancer therapies. Clin Pharmacol Ther. 2007;82(3):252-264.[12] Zhou Z, Chen Y, Zhang H, et al. Comparison of mesenchymal stromal cells from human bone marrow and adipose tissue for the treatment of spinal cord injury. Cytotherapy. 2013;15(4):434-448.[13] Yoshimura K, Sato K, Aoi N, et al. Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthetic Plast Surg. 2008;32(1):48-55.[14] Bhattacharya I, Ghayor C, Weber FE. The Use of Adipose Tissue-Derived Progenitors in Bone Tissue Engineering - a Review. Transfus Med Hemother. 2016;43(5):336-343.[15] Jo CH, Lee YG, Shin WH, et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells. 2014;32(5): 1254-1266.[16] Bura A, Planat-Benard V, Bourin P, et al. Phase I trial: the use of autologous cultured adipose-derived stroma/stem cells to treat patients with non-revascularizable critical limb ischemia. Cytotherapy. 2014;16(2):245-257.[17] Rajashekhar G, Ramadan A, Abburi C, et al. Regenerative therapeutic potential of adipose stromal cells in early stage diabetic retinopathy. PLoS One. 2014;9(1):e84671.[18] Bielefeld KA, Amini-Nik S, Alman BA. Cutaneous wound healing: recruiting developmental pathways for regeneration. Cell Mol Life Sci. 2013;70(12):2059-2081.[19] Gould L, Abadir P, Brem H, et al. Chronic wound repair and healing in older adults: current status and future research. J Am Geriatr Soc. 2015;63(3):427-438.[20] Juniantito V, Izawa T, Yamamoto E, et al. Heterogeneity of macrophage populations and expression of galectin-3 in cutaneous wound healing in rats. J Comp Pathol. 2011;145(4):378-389.[21] Hassan WU, Greiser U, Wang W. Role of adipose-derived stem cells in wound healing. Wound Repair Regen. 2014;22(3):313-325.[22] Rodriguez J, Boucher F, Lequeux C, et al. Intradermal injection of human adipose-derived stem cells accelerates skin wound healing in nude mice. Stem Cell Res Ther. 2015;6:241.[23] Lee HC, An SG, Lee HW, et al. Safety and effect of adipose tissue-derived stem cell implantation in patients with critical limb ischemia: a pilot study. Circ J. 2012;76(7):1750-1760.[24] Desai VD, Hsia HC, Schwarzbauer JE. Reversible modulation of myofibroblast differentiation in adipose-derived mesenchymal stem cells. PLoS One. 2014;9(1):e86865.[25] 黄宏,朱方强,孙宏振,等.脂肪来源干细胞体外成骨诱导和成脂诱导分化[J].第三军医大学学报,2008,30(13):1219-1222.[26] Park A, Hogan MV, Kesturu GS, et al. Adipose-derived mesenchymal stem cells treated with growth differentiation factor-5 express tendon-specific markers. Tissue Eng Part A. 2010;16(9):2941-2951.[27] Dimarino AM, Caplan AI, Bonfield TL. Mesenchymal stem cells in tissue repair. Front Immunol. 2013;4:201.[28] Fischer LJ, McIlhenny S, Tulenko T, et al. Endothelial differentiation of adipose-derived stem cells: effects of endothelial cell growth supplement and shear force. J Surg Res. 2009;152(1):157-166.[29] Steinberg JP, Hong SJ, Geringer MR, et al. Equivalent effects of topically-delivered adipose-derived stem cells and dermal fibroblasts in the ischemic rabbit ear model for chronic wounds. Aesthet Surg J. 2012;32(4):504-519.[30] García-Gómez I, Elvira G, Zapata AG, et al. Mesenchymal stem cells: biological properties and clinical applications. Expert Opin Biol Ther. 2010;10(10):1453-1468.[31] Raffaghello L, Bianchi G, Bertolotto M, et al. Human mesenchymal stem cells inhibit neutrophil apoptosis: a model for neutrophil preservation in the bone marrow niche. Stem Cells. 2008;26(1): 151-162.[32] Yoo KH, Jang IK, Lee MW, et al. Comparison of immunomodulatory properties of mesenchymal stem cells derived from adult human tissues. Cell Immunol. 2009;259(2):150-156.[33] 卫传元,顾建英.脂肪来源干细胞在慢性创面愈合中作用的研究进展[J].中国临床医学,2017,24(4):650-655.[34] Bruno S, Deregibus MC, Camussi G. The secretome of mesenchymal stromal cells: Role of extracellular vesicles in immunomodulation. Immunol Lett. 2015;168(2):154-158.[35] Krampera M, Glennie S, Dyson J, et al. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood. 2003;101(9):3722-3729.[36] Ren G, Zhang L, Zhao X, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2(2):141-150.[37] Prigione I, Benvenuto F, Bocca P, et al. Reciprocal interactions between human mesenchymal stem cells and gammadelta T cells or invariant natural killer T cells. Stem Cells. 2009;27(3):693-702.[38] Corcione A, Benvenuto F, Ferretti E, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107(1):367-372. [39] Groh ME, Maitra B, Szekely E, et al. Human mesenchymal stem cells require monocyte-mediated activation to suppress alloreactive T cells. Exp Hematol. 2005;33(8):928-934.[40] Batten P, Sarathchandra P, Antoniw JW, et al. Human mesenchymal stem cells induce T cell anergy and downregulate T cell allo-responses via the TH2 pathway: relevance to tissue engineering human heart valves. Tissue Eng. 2006;12(8):2263-2273.[41] Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005;105(4):1815-1822.[42] Sato K, Ozaki K, Oh I, et al. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood. 2007;109(1):228-234.[43] Li GY, Zhou F, Gong YQ, et al. Activation of VEGF and ERK1/2 and improvement of urethral function by adipose-derived stem cells in a rat stress urinary incontinence model. Urology. 2012;80(4):953.e1-8.[44] Cai A, Qiu R, Li L, et al. Atorvastatin treatment of rats with ischemia-reperfusion injury improves adipose-derived mesenchymal stem cell migration and survival via the SDF-1α/CXCR-4 axis. PLoS One. 2013;8(12):e79100.[45] Marino G, Moraci M, Armenia E, et al. Therapy with autologous adipose-derived regenerative cells for the care of chronic ulcer of lower limbs in patients with peripheral arterial disease. J Surg Res. 2013;185(1):36-44.[46] Kim EK, Li G, Lee TJ, et al. The effect of human adipose-derived stem cells on healing of ischemic wounds in a diabetic nude mouse model. Plast Reconstr Surg. 2011;128(2):387-394.[47] 汪建平.中华结直肠肛门外科学[M].北京:人民卫生出版社,2014: 776.[48] Parks AG. Pathogenesis and treatment of fistuila-in-ano. Br Med J. 1961;1(5224):463-469.[49] 王猛,王贵玉.2016年版美国结直肠外科医师学会《肛周脓肿、肛瘘和直肠阴道瘘治疗指南》解读[J].中国实用外科杂志, 2017,37(2):162-165.[50] Vogel JD, Johnson EK, Morris AM, et al. Clinical Practice Guideline for the Management of Anorectal Abscess, Fistula-in-Ano, and Rectovaginal Fistula. Dis Colon Rectum. 2016;59(12):1117-1133.[51] Bubbers EJ, Cologne KG. Management of Complex Anal Fistulas. Clin Colon Rectal Surg. 2016;29(1):43-49.[52] 蒋晓雪,郭修田.高位复杂性肛瘘手术治疗进展[J].甘肃中医药大学学报, 2017,34(3):90-93.[53] Marzo M, Felice C, Pugliese D, et al. Management of perianal fistulas in Crohn's disease: an up-to-date review[C]//APS Meeting. APS Meeting Abstracts, 2015:147-150. [54] 竺平,谷云飞,杨柏霖.复杂性肛瘘手术治疗的现存问题及对策[J].中西医结合学报,2009,7(12):1101-1103.[55] 吕长遥,伍静,何洪波.肛瘘微创手术治疗进展[J].现代中西医结合杂志, 2018,27(1):106-111.[56] Dudukgian H, Abcarian H. Why do we have so much trouble treating anal fistula. World J Gastroenterol. 2011;17(28):3292-3296.[57] Göttgens KW, Janssen PT, Heemskerk J, et al. Long-term outcome of low perianal fistulas treated by fistulotomy: a multicenter study. Int J Colorectal Dis. 2015;30(2):213-219.[58] Ritchie RD, Sackier JM, Hodde JP. Incontinence rates after cutting seton treatment for anal fistula. Colorectal Dis. 2009;11(6):564-571.[59] Adamina M, Hoch JS, Burnstein MJ. To plug or not to plug: a cost-effectiveness analysis for complex anal fistula. Surgery. 2010; 147(1):72-78.[60] El-Gazzaz G, Zutshi M, Hull T. A retrospective review of chronic anal fistulae treated by anal fistulae plug. Colorectal Dis. 2010;12(5): 442-447.[61] Kleif J, Hagen K, Wille-Jørgensen P. Acceptable results using plug for the treatment of complex anal fistulas. Dan Med Bull. 2011;58(3): A4254.[62] Safar B, Jobanputra S, Sands D, et al. Anal fistula plug: initial experience and outcomes. Dis Colon Rectum. 2009;52(2):248-252.[63] Stamos MJ, Snyder M, Robb BW, et al. Prospective multicenter study of a synthetic bioabsorbable anal fistula plug to treat cryptoglandular transsphincteric anal fistulas. Dis Colon Rectum. 2015;58(3):344-351.[64] Lindsey I, Smilgin-Humphreys MM, Cunningham C, et al. A randomized, controlled trial of fibrin glue vs. conventional treatment for anal fistula. Dis Colon Rectum. 2002;45(12):1608-1615.[65] Altomare DF, Greco VJ, Tricomi N, et al. Seton or glue for trans-sphincteric anal fistulae: a prospective randomized crossover clinical trial. Colorectal Dis. 2011;13(1):82-86.[66] Mitalas LE, Dwarkasing RS, Verhaaren R, et al. Is the outcome of transanal advancement flap repair affected by the complexity of high transsphincteric fistulas. Dis Colon Rectum. 2011;54(7):857-862.[67] Madbouly KM, El Shazly W, Abbas KS, et al. Ligation of intersphincteric fistula tract versus mucosal advancement flap in patients with high transsphincteric fistula-in-ano: a prospective randomized trial. Dis Colon Rectum. 2014;57(10):1202-1208.[68] Soltani A, Kaiser AM. Endorectal advancement flap for cryptoglandular or Crohn's fistula-in-ano. Dis Colon Rectum. 2010;53(4):486-495.[69] Goos M, Manegold P, Grüneberger M, et al. Long-term results after endoanal advancement flap repair for fistulas-in-ano. How important is the aetiology. Int J Colorectal Dis. 2015;30(3):413-419.[70] Schwandner O. Obesity is a negative predictor of success after surgery for complex anal fistula. BMC Gastroenterol. 2011;11:61.[71] Uribe N, Balciscueta Z, Mínguez M, et al. "Core out" or "curettage" in rectal advancement flap for cryptoglandular anal fistula. Int J Colorectal Dis. 2015;30(5):613-619.[72] Malakorn S, Sammour T, Khomvilai S, et al. Ligation of Intersphincteric Fistula Tract for Fistula in Ano: Lessons Learned From a Decade of Experience. Dis Colon Rectum. 2017;60(10):1065-1070.[73] García-Olmo D, García-Arranz M, García LG, et al. Autologous stem cell transplantation for treatment of rectovaginal fistula in perianal Crohn's disease: a new cell-based therapy. Int J Colorectal Dis. 2003; 18(5):451-454. [74] Borowski DW, Gill TS, Agarwal AK, et al. Autologous adipose-tissue derived regenerative cells for the treatment of complex cryptoglandular fistula-in-ano: a report of three cases. BMJ Case Rep. 2012;2012.[75] Cho YB, Lee WY, Park KJ, et al. Autologous adipose tissue-derived stem cells for the treatment of Crohn's fistula: a phase I clinical study. Cell Transplant. 2013;22(2):279-285.[76] Lee WY, Park KJ, Cho YB, et al. Autologous adipose tissue-derived stem cells treatment demonstrated favorable and sustainable therapeutic effect for Crohn's fistula. Stem Cells. 2013;31(11): 2575-2581.[77] Cho YB, Park KJ, Yoon SN, et al. Long-term results of adipose-derived stem cell therapy for the treatment of Crohn's fistula. Stem Cells Transl Med. 2015;4(5):532-537.[78] Choi S, Ryoo SB, Park KJ, et al. Autologous adipose tissue-derived stem cells for the treatment of complex perianal fistulas not associated with Crohn's disease: a phase II clinical trial for safety and efficacy. Tech Coloproctol. 2017;21(5):345-353.[79] García-Olmo D, García-Arranz M, Herreros D, et al. A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum. 2005;48(7):1416-1423.[80] Garcia-Olmo D, Herreros D, Pascual I, et al. Expanded adipose-derived stem cells for the treatment of complex perianal fistula: a phase II clinical trial. Dis Colon Rectum. 2009;52(1):79-86.[81] Christman KL, Vardanian AJ, Fang Q, et al. Injectable fibrin scaffold improves cell transplant survival, reduces infarct expansion, and induces neovasculature formation in ischemic myocardium. J Am Coll Cardiol. 2004;44(3):654-660.[82] Herreros MD, Garcia-Arranz M, Guadalajara H, et al. Autologous expanded adipose-derived stem cells for the treatment of complex cryptoglandular perianal fistulas: a phase III randomized clinical trial (FATT 1: fistula Advanced Therapy Trial 1) and long-term evaluation. Dis Colon Rectum. 2012;55(7):762-772.[83] Lobascio P, Balducci G, Minafra M, et al. Adipose-derived stem cells (MYSTEM® EVO Technology) as a treatment for complex transsphincteric anal fistula. Tech Coloproctol. 2018;22(5):373-377.[84] Wainstein C, Quera R, Fluxá D, et al. Stem Cell Therapy in Refractory Perineal Crohn's Disease: Long-term Follow-up. Colorectal Dis. 2018 Jan 6. [Epub ahead of print][85] Solomon MJ, McLeod RS, O’Connor BI, et al. Combination of ciprofloxacin and metronidazole in severe perianal Crohn’s disease. Can J Gastroenterol. 1993;7:571-573.[86] Mizrahi N, Wexner SD, Zmora O, et al. Endorectal advancement flap: are there predictors of failure. Dis Colon Rectum. 2002;45(12): 1616-1621.[87] Sanz-Baro R, García-Arranz M, Guadalajara H, et al. First-in-Human Case Study: Pregnancy in Women With Crohn's Perianal Fistula Treated With Adipose-Derived Stem Cells: A Safety Study. Stem Cells Transl Med. 2015;4(6):598-602.[88] Mountifield R, Bampton P, Prosser R, et al. Fear and fertility in inflammatory bowel disease: a mismatch of perception and reality affects family planning decisions. Inflamm Bowel Dis. 2009;15(5): 720-725.[89] Mañosa M, Navarro-Llavat M, Marín L, et al. Fecundity, pregnancy outcomes, and breastfeeding in patients with inflammatory bowel disease: a large cohort survey. Scand J Gastroenterol. 2013;48(4): 427-432.[90] Hatch Q, Champagne BJ, Maykel JA, et al. Crohn's disease and pregnancy: the impact of perianal disease on delivery methods and complications. Dis Colon Rectum. 2014;57(2):174-178.[91] Cheng AG, Oxford EC, Sauk J, et al. Impact of mode of delivery on outcomes in patients with perianal Crohn's disease. Inflamm Bowel Dis. 2014;20(8):1391-1398.[92] García-Arranz M, Herreros MD, González-Gómez C, et al. Treatment of Crohn's-Related Rectovaginal Fistula With Allogeneic Expanded-Adipose Derived Stem Cells: A Phase I-IIa Clinical Trial. Stem Cells Transl Med. 2016;5(11):1441-1446.[93] Panés J, García-Olmo D, Van Assche G, et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn's disease: a phase 3 randomised, double-blind controlled trial. Lancet. 2016;388(10051):1281-1290.[94] 李森娟,沈桂鑫,邱敏,等.干细胞移植治疗复杂性肛瘘疗效的Meta分析[J].浙江医学,2018,40(3): 262-265.[95] Lightner AL, Wang Z, Zubair AC, et al. A Systematic Review and Meta-analysis of Mesenchymal Stem Cell Injections for the Treatment of Perianal Crohn's Disease: Progress Made and Future Directions. Dis Colon Rectum. 2018;61(5):629-640. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||