Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (30): 4841-4848.doi: 10.3969/j.issn.2095-4344.0965
Previous Articles Next Articles
An overview of patented technology for fully-degradable polylactic acid stents
Yang Jin-hui, Yu Qun, Zhao Jie
- Examining Division for Materials Engineering Inventions, State Intellectual Property Office of China, Beijing 100088, China
-
Received:2018-03-02Online:2018-10-28Published:2018-10-28 -
Contact:Yang Jin-hui, Examining Division for Materials Engineering Inventions, State Intellectual Property Office of China, Beijing 100088, China -
About author:Yang Jin-hui, Examining Division for Materials Engineering Inventions, State Intellectual Property Office of China, Beijing 100088, China
CLC Number:
Cite this article
Yang Jin-hui, Yu Qun, Zhao Jie. An overview of patented technology for fully-degradable polylactic acid stents[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(30): 4841-4848.
share this article
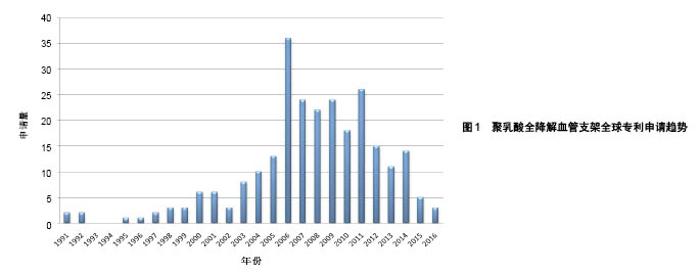
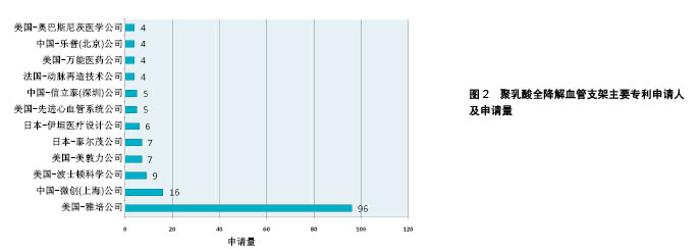
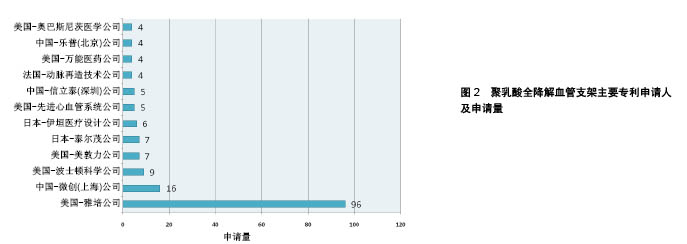
2.1 聚乳酸全降解血管支架领域专利申请态势 2.1.1 全球专利申请量年度趋势 截至2017年5月,聚乳酸全降解血管支架领域的全球专利申请共258项。20世纪90年代起,聚乳酸全降解血管支架领域的专利申请开始相继出现,图1反映了该领域全球专利申请趋势。从中可看出,1991年出现了有关聚乳酸全降解血管支架的专利申请,1991至1999年期间,该领域处于技术萌芽期,专利申请量维持在较低水平。在2000至2006年间,由于金属裸支架、药物洗脱支架在临床使用中逐渐凸显了其不足之处,研究人员开始寻求开发更理想的全降解血管支架,其专利申请量开始快速增长,至2006年,专利申请量出现爆发式增长。2007年至今,聚乳酸全降解血管支架技术进入平稳发展期,专利申请数量基本保持平稳。 2.1.2 主要申请人分布 图2显示了聚乳酸全降解血管支架领域主要专利申请人分布,美国雅培公司以96项专利申请占据了该领域37%的专利申请份额,中国微创公司以16项专利申请位列第二,排在之后的申请人分别是波士顿科学、美敦力、泰尔茂、伊恒医疗、先进心血管、信立泰等。在申请量排名前列的申请人中,美国申请人占据席位最多,其次是中国和日本。"
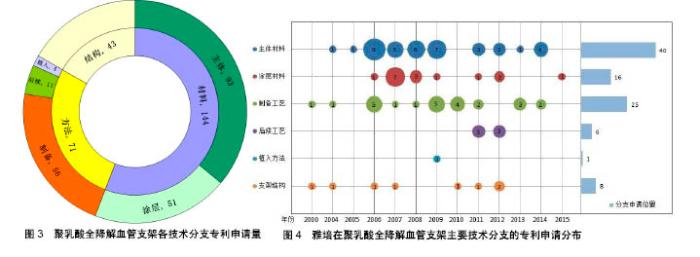
针对聚乳酸材料自身存在力学、降解和手术可操作性等技术问题,该领域专利申请的焦点主要集中在材料、工艺方法、结构3方面,各技术分支截至2017年5月的专利申请量如图3所示。其中,对材料的研究改进超过总申请的一半,也是该领域最早的专利申请方向,主要包括对主体材料和涂层材料的改进。此外,对工艺方法和结构设计的改进也是解决上述技术问题的渠道之一,但是限于聚乳酸材料自身的缺陷,单纯的工艺方法和结构设计并不能很好的解决上述技术问题,因此这些方向的申请量相比较材料方法占比较低。 2.1.4 重要申请人分析 在聚乳酸全降解血管支架领域,美国的雅培公司和中国的上海微创医疗器械(集团)有限公司在申请量上远超其他申请人,其分别是国际和国内血管支架领域的领先者,以下主要针对上述两个重要申请人的核心技术进行分析。 国外重要申请人——美国雅培公司:雅培公司在全球血管支架领域处于领先地位,目前全球唯一正式上市的完全可降解支架是雅培的聚乳酸支架Absorb。图4显示了雅培公司在聚乳酸全降解血管支架领域的几个主要技术分支上的专利申请分布,其在主体材料技术分支的申请量最大。其专利申请大部分都有较多同族专利,在世界各主要国家和地区都有专利布局,如美国、中国、欧洲、日本、中国香港、韩国等。 从图4可看出,雅培公司最早于2000年开始进行聚乳酸全降解血管支架领域的技术研发,初期专利申请主要分布在制备工艺、支架结构及主体材料研发方面。例如,将聚合物和溶剂的组合物施用到可降解支架基底上,使温度等于或大于聚合物玻璃化转变温度,以除去溶剂在可降解支架表面形成涂层的制备工艺(US8632845B2[15]);在支架的壁上附接生物相容性材料层,材料孔径适合内皮细胞生长,并能防止尺寸超过其孔径大小的血栓通过,降低再狭窄和血栓形成的风险(EP2266502A1[16])。 2006年,雅培公司联合波士顿科学公司收购佳腾公司,继承了佳腾公司在聚乳酸血管支架方向的研究成果,在此基础上继续进行研发,生产出的可降解聚乳酸血管支架成为全球第一款上市的可降解支架产品。自2006年开始,雅培公司在聚乳酸全降解血管支架领域的研究投入进一步增加,专利申请量大幅度增加,在该领域的各主要技术分支均有涉及。例如主体材料方面,在基质中加入聚合物纳米颗粒,在支架成形过程中提供成核点形成基质聚合物结晶域,限制聚合物链的移动自由度,增强支架的韧性、强度和模量(WO2008002479A2[17]);分散相聚合物的部分链段进入连续相基质聚合物,增强两者之间的黏附,从而增强支架材料的力学性能,同时生物陶瓷颗粒分散在基质中,其降解产物能改变基质的降解速率(US2008081063A1[18]);在聚合物基质中添加可降解金属以提高支架的显影性(WO2008016696A2[19])等。涂层材料方面,设置多层涂层结构,包括促进细胞生长、抗增生的药物等,以实现支架的多功能性(US8709071B1[20]);等离子处理支架表面产生反应基团,与涂层化学键接,增加涂层和基体的黏附力,避免在卷曲或释放后导致的分离(US2009148591A1[21])等。制备工艺方面,使用溶剂处理部分左旋聚乳酸以诱导结晶,通过轴向和径向变性诱导聚合物达到或高于玻璃化转变温度,通过统一的变形处理,使得管材获得统一的诱导圆周定向,增加支架的机械稳定性(US2014265060A1[22]);控制退火温度,加入晶核形成剂,获得更小更均一的晶粒、更高的结晶密度,改善断裂韧性(US2014084515A1[23])等。 国内重要申请人——上海微创医疗器械(集团)有限公司:上海微创医疗器械(集团)有限公司是一家中国领先的医疗器械集团, 2016年,其自主研发的FiresorbTM生物可吸收雷帕霉素靶向洗脱冠脉支架系统通过了CFDA的创新医疗器械特别审批申请,意味着其加快了上市节奏。"
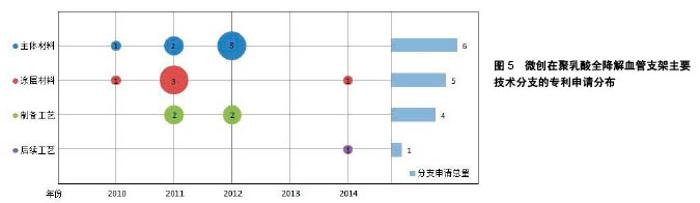
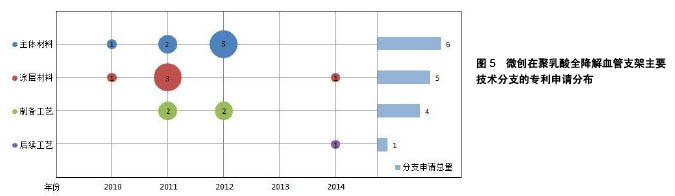
微创公司在聚乳酸全降解血管支架领域的主要技术分支上的专利申请分布如图5所示。从中可以看出,微创公司进入聚乳酸全降解血管支架领域的时间较晚,2010年才开始有相关专利申请。其专利有一半以上通过PCT途径申请,但只有少量申请进入欧洲、美国,其他大部分都只在中国进行了申请。 微创公司的专利申请主要集中在改进可降解支架的力学性能方面。例如,在生物可降解聚合物中加入磁性纳米粒子,通过外部的交变磁场使粒子升温发热,从而实现对支架的局部加热,使支架发生热塑性形变,待支架冷却后即可固定成形,可以有效降低支架扩张后的回缩率(CN102371006A[24]);通过控制组合聚合物的玻璃化转变温度,使支架不仅具有形状记忆性能,还具有足够的机械力学性能(CN102327652A[25]);通过共混可降解金属基材料,提高了支架的力学性能,并且实现了支架的显影 (CN103877624A[26]);使管材在切割成支架之前,在径向和轴向方向上实现高度取向,使材料在径向和轴向的强度和韧性得到大幅度提高(CN103876869A[27])等。 "
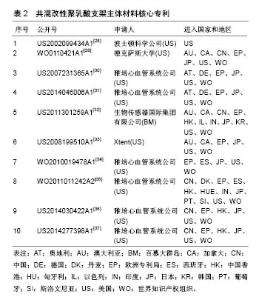
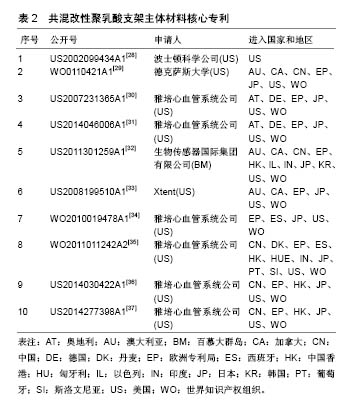
2.2 共混改性聚乳酸支架主体材料专利技术发展 在聚乳酸全降解血管支架领域,对支架主体材料的改进是各主要申请人研究的重点,主要包括共混改性聚乳酸材料、共聚改性聚乳酸材料和纯聚乳酸材料3方面,其中共混改性聚乳酸材料申请量最大,也能更好地改进支架性能,以下对共混改性聚乳酸支架主体材料的专利技术发展进行分析。 2.2.1 核心专利 核心专利体现了行业领域的研究重点,通过同族专利数目的多少和引用频次的高低等因素,筛选出排名前10的核心专利,如表2所示。从表2可以看出,共混改性聚乳酸支架主体材料核心专利申请国主要集中在美国,并且大部分都有大量同族专利,主要市场遍及中国、美国、日本、欧洲、中国香港、澳大利亚、加拿大等国家和地区。从核心专利的申请人来看,其基本为行业内排名前列的重要企业,如雅培、波士顿科学、Xtent。"
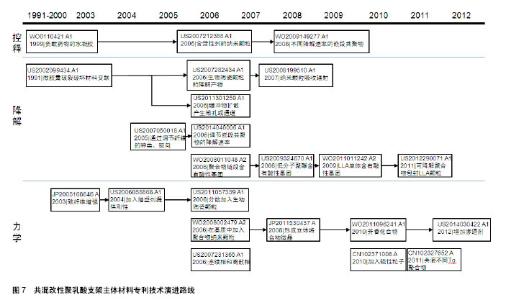
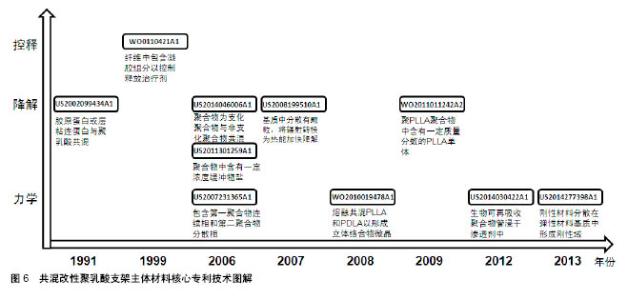
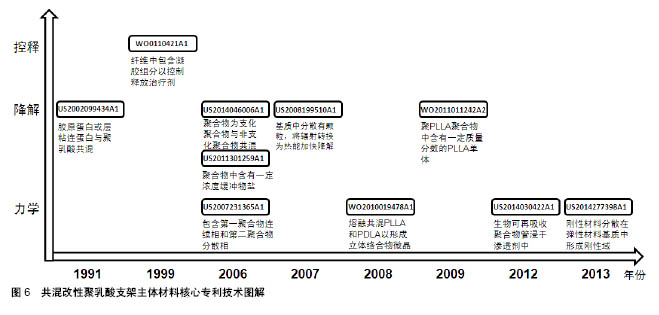
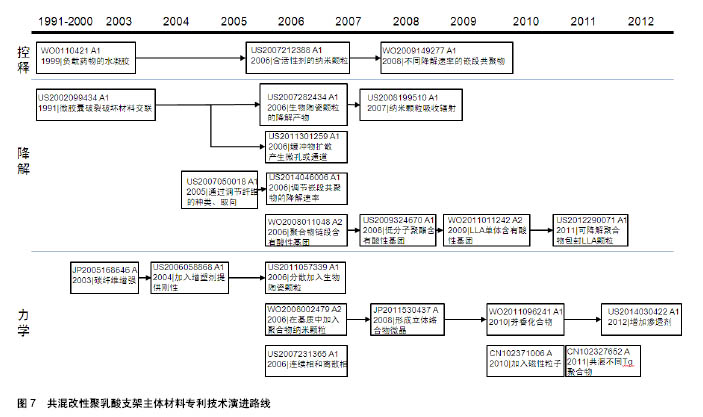
2.2.2 专利技术发展演进 通过对共混改性聚乳酸支架主体材料各个时期的专利文献进行梳理和分析,可得到该领域主要技术方向的专利技术发展路线,如图7所示。 药物控释性能:聚乳酸材料作为聚合物其本身即可负载治疗性药物,但药物的释放速率往往不能得到很好的控制。德克萨斯大学在WO0110421A1中提出将负载治疗药物的水凝胶与聚合物基质共混,通过水凝胶来控制药物的释放速率。US2007212388A1将聚合物基质设计为多孔结构[38],负载有活性剂的聚合物纳米颗粒分散在基质的孔内,纳米颗粒以受控方式释放活性剂。波士顿科学公司的WO2009149277A1中[39],支架材料是两种嵌段共聚物的共混物,嵌段共聚物的不同嵌段具有不同的降解速率,将药物组分与嵌段共聚物共轭或离子连接,药物将随着嵌段共聚物的降解而释放,通过对嵌段种类和相对量的控制,达到所需的药物释放速率。 降解速率:全降解血管支架的优势即在于其后期可完全降解,但其降解速率需要得到有效控制,降解速率过快会导致其治疗效果不能得到充分的发挥,降解速率过慢又可能发生血管再狭窄,对血管自我修复和后续治疗也会有产生阻碍作用。 控制降解速率的一种方式是在基体材料中加入能影响其降解过程的物质。例如,US2002099434A1公开了支架材料中包含能够诱导其交联的微胶囊,通过能量的施加导致微胶囊破裂,破坏材料的交联,进而控制支架的降解速率;US2007282434A1中,共聚物基质中生物陶瓷颗粒的降解产物能改变共聚物的降解速率[40];US2011301259A1在聚合物基质中加入了盐类缓冲物质,支架与液体接触时,缓冲物从支架中快速地扩散出来,由此产生了微孔或通道,水分子可以通过这些微孔或通道渗入支架中从而加快支架的降解速率;US2008199510A1包含纳米颗粒[41],颗粒能吸收辐射转化为热量,加快主体结构的降解。 另一种方式是通过将具有不同降解速率的成分共混,来调节组合物的降解速率。例如,US2007050018A1提出,在聚合物基质中加入与其降解速率不同的纤维,通过调节纤维的种类以及纤维在聚合物基质中的取向,调节支架的降解速率[42]。US2014046006A1公开了将基质聚合物与星形嵌段共聚物共混,基质聚合物与嵌段共聚物的外部链段形成连续相,嵌段共聚物的内部链段分散在连续相中形成离散相,通过调节嵌段共聚物的降解速率来控制材料的降解速率 还有一种方式是改变支架所处环境的pH来调控支架的降解速率,通过在聚合物基质中加入含有酸性基团的低分子聚酯、左旋乳酸单体等物质,酸性基团在体内的水解释放会降低环境pH,从而增加支架降解速率,如WO2008011048A2[43]、US2009324670A1[44]、WO2011011242A2。更进一步地,US2012290071A1加入了可降解聚合物包封的左旋乳酸LLA颗粒[45],当支架暴露于水分环境时,表面可降解聚合物在一定时间内阻止左旋乳酸与水分接触,随后表面聚合物降解释放其中的左旋乳酸,加速支架降解。 力学性能:血管支架需要具备合适的力学性能,如径向强度、韧性、模量等,以支撑狭窄闭塞段血管,减少血管弹性回缩。早期专利多在聚合物基质中直接加入增强相以提高支架材料的强度,如JP2005168646A加入碳纤维增强材料强度[46],US2006058868A1加入增塑剂提供刚性[47],US2011057339A1加入生物陶瓷颗粒增加韧性和模量[48]。 2006年,雅培公司在其专利WO2008002479A2中提出,在基质中加入聚合物纳米颗粒,纳米颗粒在聚合物成形过程中提供成核点形成基质聚合物结晶域,限制聚合物链的移动自由度,增强聚合物的韧性、强度和模量。随后的专利申请中,也有部分专利申请沿袭了这个思路,通过影响聚合物的结晶过程,控制支架材料的力学性能。JP2011530437A将左旋聚乳酸和右旋聚乳酸的聚合物共混熔融,以在共混物中形成左旋聚乳酸/右旋聚乳酸立体络合物微晶[49];WO2011096241A1的支架中含有具有芳香环的芳香族化合物,由于芳香环的堆积作用,使脂肪族聚酯的分子链更规则地排列,支架的机械强度提高[50];US2014030422A1在聚合物基质中增加渗透剂以诱导结晶增加聚合物结晶度,提高机械性能。 除此以外,还有其他几种方式改善支架材料的力学性能。US2007231365A1包括第一聚合物的连续相和第二聚合物的离散相,离散相的部分链段进入连续相,增强了两者之间的黏附,从而增强材料的强度;CN102371006A的支架主体中分布有若干磁性粒子,通过外部的交变磁场使磁性纳米粒子升温发热,从而实现对支架的局部加热,使支架发生热塑性形变,待支架冷却后即可固定成形,可以有效降低扩张后的回缩率;CN102327652A通过将具有不同玻璃化转变温度的聚合物共混,使组合聚合物的玻璃化转变温度较高,在人体内支架处于玻璃态,有足够的机械强度支撑管腔。"
| [1] 世界卫生组织官方网站http://www.who.int/mediacentre/factsheets/fs317/zh[2] Lloyd-Jones D,Adams RJ,Brown TM,et al.Heart disease and stroke statistics-2009 update: a report from the American Heart Association statistics committee and stroke statistics subcommittee. Circulation. 2009;119:480-486.[3] 甄珍,奚廷斐,郑玉峰.全降解冠脉支架研究进展[J].新材料产业, 2011, 13(8):8-12.[4] Serruys PW,Luijten HE,Beatt KJ,et al.Incidence of restenosis after successful coronary angioplasty: A time-related phenomenon: A quantitative angiographic study in 342 consecutive patients at 1, 2, 3, and 4 months. Circulation. 1988;77:361-371.[5] Gruentzig AR,King SB,Schlumpf M,et al.Longterm follow-up after percutaneous transluminal coronary angioplasty: The early Zurich experience.N Engl J Med.1987;316:1127-1132.[6] Serruys PW,de Jaegere P,Kiemeneij F,et al.A comparison of balloon-expandable-stent implantation with balloon angioplasty in patients with coronary artery disease: Benestent Study Group.N Engl J Med.1994;331:489-495.[7] Martin DM,Boyle FJ.Drug-eluting stents for coronary artery disease: A review.Med Eng Phys. 2011;33(2):148-163.[8] Lemos PA,Serruys PW,van Domburg RT,et al.Unrestricted utilization of sirolimus-eluting stents compared with conventional bare stent implantation in the “real world”: The Rapamycin-Eluting stent Evaluated At Rotterdam Cardiology Hospital (RESEARCH) registry. Circulation.2004;109:190-195.[9] Di Mario C,Gil R,Camenzind E,et al.Quantitative assessment with intracoronary ultrasound of the mechanisms of restenosis after percutaneous transluminal coronary angioplasty and directional coronary atherectomy.Am J Cardiol.1995;75:772-777.[10] Yamawaki T,Shimokawa H,Kozai T,et al.Intramural delivery of a specific tyrosine kinase inhibitor with biodegradable stent suppresses the restenotic changes of the coronary artery in pigs in vivo.J Am Coll Cardiol.1998;32:780-786.[11] Eggebrecht H,Rodermann J,Hunold P,et al.Images in cardiovascular medicine: Novel magnetic resonance-compatible coronary stent: The absorbable magnesiumalloy stent. Circulation. 2005;112: e303-e304.[12] Tamai H,Igaki K,Kyo E,et al.Initial and 6-month results of biodegradable poly-l-lactic acid coronary stents in humans. Circulation. 2000;102:399-404.[13] Vogt F,Stein A,Rettemeier G,et al.Long-term assessment of a novel biodegradable paclitaxel-eluting coronary polylactide stent.Eur Heart J. 2004;25:1330-1340.[14] Ormiston JA,Webster MW,Armstrong G.First-in-human implantation of a fully bioabsorbable drug-eluting stent:the BVS poly-L-lactic acid everolimus-eluting coronary stent.Catheter Cardiovasc Interv. 2007; 69:128-131[15] ABBOTT CARDIOVASCULAR SYSTEMS INC.Coating stent e.g. self-expandable stents, by applying composition including polymer component and solvent to stent substrate, and exposing polymer component to temperature equal to/greater than glass transition temperature of component:美国,US8632845B2[16] ABBOTT LAB VASCULAR ENTERPRISES LTD.Apparatus, useful for stenting, comprises stent (having proximal and distal ends and a lumen extending between them, and tubular body) and a biocompatible material layer attached to a portion of stent between the proximal and distal ends:美国,EP2266502A1[17] ABBOTT CARDIOVASCULAR SYSTEMS INC, Implantable medical device such as stent for treating diseases associated with lumen in mammals e.g. atherosclerotic stenosis, has structural element containing composite having polymer nanoparticles dispersed within polymer matrix:美国,WO2008002479A2[18] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent, e.g. for maintaining vascular patency and/or drug delivery, has structural unit including continuous phase containing matrix polymer, and discrete phases containing modifier polymer with discrete phase segments:美国,US2008081063A1[19] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent useful in the treatment of artherosclerosis comprises a body consisting of composite containing a polymer, and radiopaque metallic particles mixed or dispersed within the polymer:美国,WO2008016696A2[20] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent comprises stent body containing structural elements, each element contains luminal surface, abluminal surface and sidewall surface, prohealing coating, and antiproliferative coating, where the body is made of bioabsorbable material:美国,US8709071B1[21] ABBOTT CARDIOVASCULAR SYSTEMS INC.Polymeric stent for being implanted in bodily lumen for treating polymer surface, has functional group miscible or compatible with coating polymer selected as poly-L-lactide-co-glycolide, polypropylene glycol, or polyethylene glycol:美国,US2009148591A1[22] ABBOTT CARDIOVASCULAR SYSTEMS INC.Method for fabricating implantable medical device e.g. stent from deformed polymer tube, involves treating portion of tube with solvent, radially and axially deforming tube and fabricating stent from deformed and treated tube:美国,US2014265060A1[23] ABBOTT CARDIOVASCULAR SYSTEMS INC.Polymeric tubular implantable stent e.g. self-expandable stent for use during treatment of atherosclerotic stenosis in blood vessels, has tube annealed at temperature in temperature range of above glass transition temperature of polymer:美国,US2014084515A1[24] 上海微创医疗器械(集团)有限公司.一种生物可降解支架:中国, CN102371006A[P].2012-03-14.[25] 上海微创医疗器械(集团)有限公司.一种生物可降解支架及其制备工艺:中国,CN102327652A[P].2012-01-25.[26] 上海微创医疗器械(集团)有限公司.一种可降解聚酯支架及其制备工艺:中国,CN103877624A[P].2014-06-25.[27] 上海微创医疗器械(集团)有限公司.一种生物可降解聚合物支架的制备工艺:中国,CN103876869A[P].2014-06-25.[28] SCIMED LIFE SYSTEMS INC IN.Stent, useful for inserting into body lumen such as blood vessel, has tubular main body having biodegradable matrix containing collagen IV and laminin and biodegradable strengthening material:美国,US2002099434 A1[29] UNIV TEXAS.Biodegradable polymer fiber scaffold implants with controlled release of therapeutic agents, promote cell migration use for drug delivery, stents, arteriogenesis, bone fractures, muscle grafts, wound dressings, and optic disorders:美国,WO0110421 A1[30] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device, e.g. stent, comprises structural element including continuous phase comprised of first polymer, and discrete phase comprised of second polymer that has discrete phase segments and anchor segments:美国,US2007231365 A1[31] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device e.g. stent fabricated from polymer blend comprises biodegradable matrix polymer blended with biodegradable star-block copolymers having three arms, where the arms include inner segments and outer segments:美国,US2014046006 A1[32] BIOSENSORS INT GROUP.Biodegradable polymer stent such as bile duct stent useful in blood vessels e.g. aorta, comprises an aliphatic polyester polymer, and a phosphate salt including phosphate buffer salt, citrate buffer salt and sodium chloride:百慕大,US2011301259 A1[33] XTENT INC.Implant, useful e.g. in tissue, comprises structure (having material property at normal body temperature) adapted for implantation; and particles (which are adapted to convert incident radiation into heat energy) dispersed in structure:美国,US2008199510 A1[34] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent making method by forming tube from melt processed blend of (L-lactide) and poly(D-lactic acid) comprising stereocomplex crystallites by quenching to temperature below glass transition temperature of poly (L-lactide):美国,WO2010019478 A1[35] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent for treating diseased section of blood vessel comprises specific amount of L-lactide monomer mixed/dispersed/dissolved within polymeric scaffolding having poly(L-lactide) with specific crystallinity and number average molecular weight:美国,WO2011011242 A2[36] ABBOTT CARDIOVASCULAR SYSTEMS INC.Fabricating bioresorbable stent scaffold i.e. endoprosthesis stent, to treat atherosclerotic stenosis in blood vessels or heart valve, by providing a tube made of a bioresorbable polymer, exposing into solvent, and fabricating scaffold:美国,US2014030422 A1[37] ABBOTT CARDIOVASCULAR SYSTEMS INC.Medical device implantable within peripheral vessel of body to deliver anti-restenosis drug to treat peripheral vascular disease, comprises cylindrical radially expandable body made of polymer formulation and anti-restenosis drug:美国,US2014277398 A1[38] SAHAJANAND MEDICAL TECHNOLOGIES PVT LTD IN.Composition, useful for treating e.g. restenosis, comprises first polymer having pores; nanoparticles (comprising second polymer and active agent) dispersed within pores; and heparin covalently bonded to the polymer:印度, US2007212388A1[39] BOSTON SCI SCIMED INC.Block or graft copolymer used in coating of implantable medical device, e.g. stent, comprises first block including poly(lactide-glycolide) copolymer and second block comprising, e.g. polylactide homopolymer:美国,WO2009149277A1[40] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device useful as e.g. self-expandable stents, balloon-expandable stents, stent-grafts, and implantable cardiac pacemakers, comprises structural element including a bioceramic/copolymer composite:美国,US2007282434A1[41] XTENT INC.Implant, useful e.g. in tissue, comprises structure (having material property at normal body temperature) adapted for implantation; and particles (which are adapted to convert incident radiation into heat energy) dispersed in structure:美国, US2008199510A1[42] ARTERIAL REMODELING TECHNOLOGIES INC.Reduction of sharp surface and/or irregularity of polymeric stent, involves immersing polymeric stent into bath containing solvent and biodegradable polymer:美国,US2007050018A1[43] ABBOTT CARDIOVASCULAR SYSTEMS INC.Fabricating stent useful for bodily lumen involves forming a biodegradable polymer with a polymerization initiated with an acid containing group, and fabricating the stent from the formed polymer which includes acid group from the initiator:美国,WO2008011048A2[44] ABBOTT CARDIOVASCULAR SYSTEMS INC.Implantable medical device e.g. vascular stent comprises blend of high molecular weight polyester and low molecular weight polyester containing the blend and acidic moiety:美国,US2009324670A1[45] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent useful for delivering drug or therapeutic agent for treating atherosclerosis, comprises scaffolding made of poly(L-lactide) and several particles dispersed throughout scaffolding:美国,US2012290071A1[46] TERUMO CORP.Stent for detaining in constricted/closed tubular cavities e.g. blood vessels and useful in treating various diseases, is formed using fiber composition containing biodegradable polymer and carbon fiber having preset length:日本,JP2005168646A[47] ADVANCED CARDIOVASCULAR SYSTEM.Medical article for use as medical implant comprises implant comprising polymeric material that comprises plasticizing agent capable of increasing strain-to-failure in the polymeric material while placing the implant in a subject:美国, US2006058868A1[48] ADVANCED CARDIOVASCULAR SYSTEM.Fabrication of implantable medical device by forming suspension solution including fluid, matrix polymer, and bioceramic particles, forming composite mixture, and mixing composite mixture with modifier polymer and additional matrix polymer:美国,US2011057339A1[49] ABBOTT CARDIOVASCULAR SYSTEMS INC.Stent making method by forming tube from melt processed blend of (L- lactide) and poly(D-lactic acid) comprising stereocomplex crystallites by quenching to temperature below glass transition temperature of poly (L-lactide):美国,JP2011530437A[50] TERUMO CORP.In-vivo absorptive stent useful for preventing reocclusion of lesion site in e.g. blood vessel and trachea, contains mixture of in-vivo absorptive aliphatic polyester and aromatic compound having 1 or more aromatic rings:日本,WO2011096241A1 |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||