Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (21): 3438-3444.doi: 10.3969/j.issn.2095-4344.0526
Role of SIRT1 in mesenchymal stem cell differentiation
Wang Gang-gang, Song Wei
- Beijing Advanced Innovation Center for Biomedical Engineering, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China
-
Revised:2018-03-14Online:2018-07-28Published:2018-07-28 -
Contact:Song Wei, Ph.D., Master’s supervisor, Lecturer, Beijing Advanced Innovation Center for Biomedical Engineering, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China -
About author:Wang Gang-gang, Master candidate, Beijing Advanced Innovation Center for Biomedical Engineering, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China -
Supported by:the National Natural Science Foundation of China, No. 31300769
CLC Number:
Cite this article
Wang Gang-gang, Song Wei. Role of SIRT1 in mesenchymal stem cell differentiation[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(21): 3438-3444.
share this article

2.1 间充质干细胞分化相关信号通路 2.1.1 Wnt信号通路 Wnts是富含半胱氨酸的高保守的分泌配体,目前已发现有19个家族成员,分别在细胞增殖、分化和凋亡等方面发挥作用[14]。Wnt信号能够激活多种信号通路,其中,需要β-catenin作为中间因子的被称为经典Wnt信号通路。在Wnt信号缺失时,细胞中的β-catenin在轴蛋白和腺瘤性息肉蛋白的协同作用下,被糖原合成激酶3β(glycogen synthase kinase-3β,GSK-3β)磷酸化,随后泛素化降解。而Wnt配体则可与跨膜受体Frizzled受体和脂蛋白受体相关蛋白(lipoprotein receptor related protein,LRP)辅助受体(LRP5/6)结合,激活胞质蛋白,抑制GSK-3β的活性,从而稳定并转运β-catenin入核,与核内的T细胞因子和淋巴增强因子结合,调节下游基因的转录[15]。β-catenin参与的经典Wnt通路可通过调节Nanog基因表达调控间充质干细胞的自我更新和干性维持[16]。间充质干细胞非经典的Wnt信号通路尚未完全清楚,但目前已发现许多相关信号通路,包括Wnt/Calcium、Wnt/PCP、Wnt/JNK、Wnt/Rho等[9]。无论经典Wnt信号通路还是非经典Wnt信号通路,都参与调节间充质干细胞的体外诱导分化过程。 Wnt信号通路参与调节骨和软骨的形成。Dong等[17]对鸡胸骨软骨细胞进行实验发现,过表达Wnt8c和Wnt9a可上调Runx2和Ⅹ型胶原蛋白的表达,下调Sox2和Ⅱ型胶原蛋白的表达,促进软骨细胞肥大成熟,过表达β-catenin有同样效果,说明激活经典Wnt信号通路可诱导软骨细胞肥大成熟。Wnt信号通路也可调节间充质干细胞向成骨细胞和软骨细胞分化。Etheridge等[15]在2004年发现间充质干细胞中表达部分Wnt信号(Wnt2、Wnt4、Wnt5a、Wnt11、Wnt16)、Frizzled受体及Wnt信号抑制因子Axin2。体外诱导间充质干细胞向成骨细胞分化,部分Wnt信号表达上调。Qnarto等[18]研究证明加入外源Wnt3a可促进间充质干细胞向成骨细胞分化,但抑制成骨细胞向成熟骨细胞分化。Wnt3a对于成骨方向分化的作用受细胞分化程度、细胞来源动物的年龄、添加Wnt3a的浓度等因素影响。在诱导间充质干细胞向软骨细胞分化的过程中,Wnt3a表达上调[19]。使用Wnt抑制剂会促进间充质干细胞早期向软骨细胞分化,但缺乏与转化生长因子β(transforming growth factor β,TGF-β)的长期协同作用,对后期软骨细胞的成熟没有促进作用;敲除Wnt抑制基因Axin2则会同时激活β-catenin和成纤维细胞因子受体1,促使间充质干细胞分化,导致小鼠头骨内发生软骨内骨化,颅缝过早闭合[19]。He等[20]在研究Toll样受体4(Toll-like receptor 4,TLR4)对间充质干细胞分化的调控时发现,TLR4促进骨髓来源间充质干细胞向成骨细胞分化过程中,Wnt3a和Wnt5a的mRNA和蛋白水平上调;沉默Wnt3a会抑制TLR4对间充质干细胞增殖的促进作用,沉默Wnt5a则抑制TLR4对间充质干细胞成骨分化的促进作用。Luo等[21]发现,在体外诱导人脂肪来源间充质干细胞向软骨细胞分化的过程中,Wnt1、β-catenin、GSK-3β的表达量在前12 d大幅上调,至第18天达到峰值,Sox2、Ⅱ型胶原、聚集蛋白多糖的表达量则在第12天达到峰值后逐渐下降;Wnt的拮抗剂Dkk处理会持续上调Sox2、Ⅱ型胶原、聚集蛋白多糖的表达,而加入外源Wnt1则下调这些软骨标志物的表达。这些研究证明Wnt通路可通过与成纤维细胞因子、转化生长因子β、TLR4等相互作用,调节间充质干细胞的自我更新、增殖及向成骨细胞和软骨细胞方向分化。 已有许多研究证实使用Wnt激动剂可以抑制3T3细胞、脂肪前体细胞向脂肪细胞分化,Wnt激动剂对间充质干细胞也有类似的抑制作用[2]。在诱导间充质干细胞向脂肪细胞方向分化过程中,抑制Wnt信号的分泌型卷曲相关蛋白4(secreted frizzled related protein 4, sFRP4)和Dkk1基因表达上调;而用BIO(GSK-3β抑制剂)处理或沉默sFRP4和Dkk1基因,都会导致诱导过程中间充质干细胞油脂积累减少,脂肪标志物表达量减少[2]。Bermeo等[22]研究发现核被膜蛋白MAN1调控间充质干细胞分化的过程与Wnt通路相关,低水平的MAN1显著上调核内β-catenin表达,并增加Smad和相关复合物的核内积累,从而抑制间充质干细胞向脂肪细胞分化。 经典和非经典Wnt信号通路都可促进间充质干细胞向肌细胞分化,每个Wnt配体(Wnt1、Wnt3、Wnt4、Wnt7a、Wnt7b、Wnt11)至少促进1个肌细胞标志物的表达[2]。Brunt等[23]用氯化锂激动经典Wnt信号,可增加β-catenin的生物利用率,同时提高来源于心脏病患者的间充质干细胞向心肌细胞分化的概率。He等[24]证实,非经典Wnt信号通路的配体Wnt11可上调间充质干细胞中心肌相关转录因子Gata-4表达,从而促进其向心肌细胞分化。Wnt11还可以促进生肌决定因子、肌形成蛋白等肌细胞标志物的表达[25]。 最近的研究表明,Wnt信号通路也可促进间充质干细胞向神经细胞分化。Yu等[26]证明Wnt3a可增加间充质干细胞中神经元特异性烯醇化酶、巢蛋白等神经标志物的表达,用siRNA干扰β-catenin表达则会抑制这些神经标志物的表达,sFRP4和Dkk1对此过程同样有抑制作用。 2.1.2 TGF-β信号通路 TGF-β超家族参与的信号通路也被证实与间充质干细胞的自我更新和分化有关。TGF-β超家族包括3种TGF-β(TGF-β1、TGF-β2和TGF-β3),还包括骨形态发生蛋白(bone morphogenetic protein,BMP)、激活素和生长分化因子[14]。TGF-β的生物活性需要通过TGF-β受体的信号传导发挥作用,其受体是一种跨膜蛋白,细胞外的TGF-β与Ⅱ型受体结合后,可被Ⅰ型受体识别,与其结合形成异源三聚体并被活化。活化的Ⅰ型受体激活细胞内的Smad受体激酶(RSmad),使其与通用型Smad(CoSmad)形成RSmad/CoSmad异聚复合物,向细胞核内移动,复合物上的发夹样结构与DNA上的Smad结合位点结合,从而调节靶基因转录[26-27]。BMP发挥作用的途径与TGF-β相类似[28]。 TGF-β信号通路可调节间充质干细胞增殖及成骨分化、脂肪分化、内皮分化。Ng等[29]通过转录组分析证实抑制TGF-β通路会减缓间充质干细胞增殖。Casado-Díaz等[30]用同样的方法发现在诱导间充质干细胞向脂肪细胞分化的过程中,TGF-β信号表达显著下调,过氧化物酶体增殖剂激活受体γ(peroxisome proliferators activated receptors,PPARγ)则表达上调。TGF-β信号通路对成骨分化的调控是一个极为复杂的过程。Grafe等[31]在其综述中,总结了已发现的TGF-β超家族信号在成骨分化中的作用方式和途径,BMPs与TGF-βs均可作用于Erk/MAPK、p38/MAPK、JNK等信号通路,影响下游成骨分化相关转录因子Runx2、Osterix的表达。TGF-β1/2/3、BMP-2/4/6/7/9可以在成骨分化早期,即间充质干细胞向前成骨细胞分化过程中发挥作用,其他的超家族信号则作用于后期成骨细胞成熟阶段[31](表1)。TGF-β1对骨的形成和重吸收、间充质干细胞增殖和成骨分化有重要意义,而TGF-β2则在诱导内皮分化过程中发挥作用[27]。"

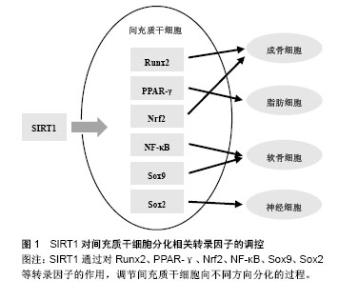
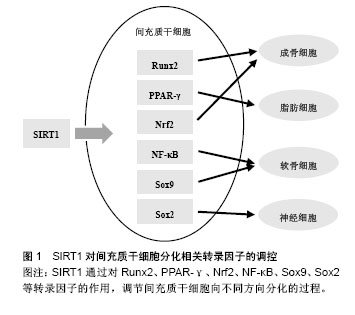
2.2 间充质干细胞分化相关转录因子 在间充质干细胞分化过程中涉及的转录因子十分复杂繁多,Augello等[35]在2010年的综述中对间充质干细胞向成骨细胞和软骨细胞分化相关的转录因子做了总结:Runx2和Osterix是间充质干细胞向成骨细胞分化过程中最重要的两个转录因子,对骨发生和骨形成都十分必要;间充质干细胞向软骨细胞分化过程中则需要转录因子Sox9的参与。Grafe等[31]的综述中也有同样的结论,并指出在间充质干细胞向软骨细胞分化过程中,Nkx-2.3、Sox6、Sox5等也有重要作用,且Nkx-2.3与Sox9相互促进表达。此外,文中还总结了调控间充质干细胞向脂肪细胞和肌细胞分化过程的转录因子:C/EBPα、C/EBPβ、C/EBPδ、PPARγ等可调节间充质干细胞向脂肪分化的过程;Mrf4、Mrf5、肌分化因子及肌形成蛋白等则在间充质干细胞向肌细胞分化的过程中发挥作用[31]。Arminan等[36]通过与心肌细胞共培养体外诱导间充质干细胞分化,发现转录因子Nkx-2.5和Gata-4的入核转运在分化中有重要作用。 除上述分化相关转录因子外,与间充质干细胞干性维持相关的因子在分化过程中也有重要意义。核转录因子Nrf2可通过促进抗氧化酶基因表达驱动抗氧化反应,在人间充质干细胞体外传代培养和成骨分化过程中,其细胞内定位发生变化。用OTA阻断Nrf2的入核通道,会减缓早期的间充质干细胞自我更新,抑制成骨向分化,用t-BHQ加强Nrf2入核转运则会起到相反的作用[10]。Lin等[37]发现NF-κB通过与miR-34a的启动子区域结合促进miR-34a的转录,增强NF-κB活性会下调成年小鼠骨髓来源间充质干细胞向成骨细胞分化。Meng等[38]实验证明miR-34a与异质性核糖核蛋白A1相互作用,促进间充质干细胞向软骨细胞方向分化。此外,Sox2表达量微量变化即可触发胚胎干细胞向不同方向分化,抑制早期神经干细胞分化[39],减弱间充质干细胞的自我更新能力和分化潜能[40]。 2.3 SIRT1对间充质干细胞分化的影响 2.3.1 SIRT1对相关信号通路的调控 SIRT1是Wnt信号通路的重要调控因子,可通过去乙酰化β-catenin促进下游分化相关因子表达[41]。SIRT1对Wnt信号通路的作用受多种因素影响。Yoon等[42]用SIRT1的激动剂白藜芦醇处理间充质干细胞,可增加β-catenin的稳定性和核内积聚,且作用强度呈现时间和剂量依赖。但另有研究者报道,白藜芦醇对于间充质干细胞的影响与其在体外传代培养的次数相关:早期(第1-3代)的间充质干细胞,白藜芦醇处理可抑制β-catenin的活性,增强细胞自我更新和多向分化的能力;晚期(第9-10代)的间充质干细胞受同样处理,则激活β-catenin,促进细胞分化,加速细胞衰老[43]。这种现象可能与细胞中SIRT1表达量不同相关,早期间充质干细胞中SIRT1表达量高,而晚期间充质干细胞几乎不表达SIRT1。研究者用白藜芦醇处理早期但抑制SIRT1表达的间充质干细胞,得到的结果与晚期间充质干细胞一致,证实了上述猜想[43]。Zhou等[43]研究发现SIRT1可通过调节Wnt通路促进间充质干细胞向成肌细胞分化,抑制其向脂肪细胞分化。使用5-氮胞苷诱导间充质干细胞分化过程中,过表达SIRT1或加入白藜芦醇都会促进间充质干细胞成肌分化,而敲除SIRT1或使用NAM抑制SIRT1的活性则促进间充质干细胞成脂分化。此外,在对癌细胞的研究中,研究者发现SIRT1可通过去乙酰化Wnt信号的胞外拮抗物sFRPs基因启动子区域的组蛋白以及Wnt信号关键蛋白Dishevelled,介导这些基因的表观遗传学沉默[9]。 SIRT1可通过调节Smad蛋白实现对TGF-β信号通路的调控。有研究表明,SIRT1可与Smad3结合,使其去乙酰化,而白藜芦醇加强这种结合作用,同时降低Smad3的转录活性;敲除SIRT1基因会增加Smad3的乙酰化水平,并显著增强TGF-β1的转录活性[44]。Kume等[45]证实,SIRT1对Smad7也有相同的作用。过表达的SIRT1会使Smad7表达下调,而敲除SIRT1基因则使Smad7表达上调。 TGF-β也可反过来调控SIRT1的表达。Xu等[46]发现,培养间充质干细胞时加入外源TGF-β可显著下调SIRT1的mRNA和蛋白表达,而加入TGF-β的抑制剂吡非尼酮则会上调SIRT1表达。 2.3.2 SIRT1对相关转录因子的调控 SIRT1通过对Runx2、PPARγ等转录因子的作用,调节间充质干细胞向不同方向分化的过程(图1)。SIRT1可调节成骨、成脂相关转录因子。体外培养的间充质干细胞,激动SIRT1可改变其谱系,上调成骨分化和成肌分化,下调成脂分化[9,47]。白藜芦醇处理间充质干细胞,可增加成骨标志物的表达,阻断成脂分化,而SIRT1的抑制剂NAM处理则会得到相反的结果[47]。SIRT1可使骨硬化蛋白编码基因Sost的启动子在H3K9位点去乙酰化,也可去乙酰化Runx2、β-catenin、PPARγ等非组蛋白,从而促进间充质干细胞成骨分化,抑制间充质干细胞成脂分化[47]。Said等[48]在对大鼠卵巢功能的研究中发现,白藜芦醇可上调SIRT1和PPARγ的表达,侧面证实了SIRT1与脂肪分化相关转录因子PPARγ的关联。这种作用在生物体内也得到了证实:SIRT1的杂合子模型(SIRT1+/-)小鼠,与对照相比,骨形成减少,骨髓脂肪细胞增加。近年,研究者培育出的间充质干细胞特异性SIRT1敲除小鼠,也表现出骨小梁体积及骨密度降低,分离出的间充质干细胞体外培养成骨向分化减弱[41]。"
| [1] Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147.[2] Visweswaran M, Pohl S, Arfuso F, et al. Multi-lineage differentiation of mesenchymal stem cells - To Wnt, or not Wnt. Int J Biochem Cell Biol. 2015;68:139-147.[3] Cho DI, Kang WS, Hong MH, et al. The optimization of cell therapy by combinational application with apicidin-treated mesenchymal stem cells after myocardial infarction. Oncotarget. 2017;8(27):44281-44294.[4] Zang L, Hao H, Liu J, et al. Mesenchymal stem cell therapy in type 2 diabetes mellitus. Diabetol Metab Syndr. 2017;9:36.[5] Satoh A, Brace CS, Ben-Josef G, et al. SIRT1 promotes the central adaptive response to diet restriction through activation of the dorsomedial and lateral nuclei of the hypothalamus. J Neurosci. 2010;30(30):10220-10232.[6] Correia M, Perestrelo T, Rodrigues AS, et al. Sirtuins in metabolism, stemness and differentiation. Biochim Biophys Acta. 2017;1861(1 Pt A):3444-3455.[7] Rahman S, Islam R. Mammalian Sirt1: insights on its biological functions. Cell Commun Signal. 2011;9:11.[8] Yuan HF, Zhai C, Yan XL, et al. SIRT1 is required for long-term growth of human mesenchymal stem cells. J Mol Med (Berl). 2012;90(4):389-400.[9] Chen H, Liu X, Chen H, et al. Role of SIRT1 and AMPK in mesenchymal stem cells differentiation. Ageing Res Rev. 2014;13:55-64.[10] Yoon DS, Choi Y, Lee JW. Cellular localization of NRF2 determines the self-renewal and osteogenic differentiation potential of human MSCs via the P53-SIRT1 axis. Cell Death Dis. 2016;7:e2093.[11] Buhrmann C, Busch F, Shayan P, et al. Sirtuin-1 (SIRT1) is required for promoting chondrogenic differentiation of mesenchymal stem cells. J Biol Chem. 2014;289(32): 22048-22062.[12] Joe IS, Jeong SG, Cho GW. Resveratrol-induced SIRT1 activation promotes neuronal differentiation of human bone marrow mesenchymal stem cells. Neurosci Lett. 2015;584: 97-102.[13] Agathocleous M, Harris WA. Metabolism in physiological cell proliferation and differentiation. Trends Cell Biol. 2013; 23(10):484-492.[14] Bhaskar B, Mekala NK, Baadhe RR, et al. Role of signaling pathways in mesenchymal stem cell differentiation. Curr Stem Cell Res Ther. 2014;9(6):508-12.[15] Etheridge SL, Spencer GJ, Heath DJ, et al. Expression profiling and functional analysis of wnt signaling mechanisms in mesenchymal stem cells. Stem Cells. 2004; 22(5):849-860. [16] Yu SJ, Kim HJ, Lee ES, et al. β-Catenin Accumulation Is Associated With Increased Expression of Nanog Protein and Predicts Maintenance of MSC Self-Renewal. Cell Transplant. 2017;26(2):365-377.[17] Dong YF, Soung do Y, Schwarz EM, et al. Wnt induction of chondrocyte hypertrophy through the Runx2 transcription factor. J Cell Physiol. 2006;208(1):77-86.[18] Quarto N, Behr B, Longaker MT. Opposite spectrum of activity of canonical Wnt signaling in the osteogenic context of undifferentiated and differentiated mesenchymal cells: implications for tissue engineering. Tissue Eng Part A. 2010; 16(10):3185-3197.[19] Maruyama T, Mirando AJ, Deng CX, et al. The balance of WNT and FGF signaling influences mesenchymal stem cell fate during skeletal development. Sci Signal. 2010;3(123): ra40.[20] He X, Wang H, Jin T, et al. TLR4 Activation Promotes Bone Marrow MSC Proliferation and Osteogenic Differentiation via Wnt3a and Wnt5a Signaling. PLoS One. 2016;11(3): e0149876.[21] Luo S, Shi Q, Zha Z, et al. Inactivation of Wnt/β-catenin signaling in human adipose-derived stem cells is necessary for chondrogenic differentiation and maintenance. Biomed Pharmacother. 2013;67(8):819-824.[22] Bermeo S, Al-Saedi A, Kassem M, et al. The Role of the Nuclear Envelope Protein MAN1 in Mesenchymal Stem Cell Differentiation. J Cell Biochem. 2017;118(12):4425-4435.[23] Brunt KR, Zhang Y, Mihic A, et al. Role of WNT/β-catenin signaling in rejuvenating myogenic differentiation of aged mesenchymal stem cells from cardiac patients. Am J Pathol. 2012;181(6):2067-2078.[24] He Z, Li H, Zuo S, et al. Transduction of Wnt11 promotes mesenchymal stem cell transdifferentiation into cardiac phenotypes. Stem Cells Dev. 2011;20(10):1771-1778.[25] Belema Bedada F, Technau A, Ebelt H, et al. Activation of myogenic differentiation pathways in adult bone marrow-derived stem cells. Mol Cell Biol. 2005;25(21): 9509-9519.[26] Yu Q, Liu L, Duan Y, et al. Wnt/β-catenin signaling regulates neuronal differentiation of mesenchymal stem cells. Biochem Biophys Res Commun. 2013;439(2):297-302.[27] Watabe T, Miyazono K. Roles of TGF-beta family signaling in stem cell renewal and differentiation. Cell Res. 2009;19(1): 103-115.[28] Alfaro MP, Vincent A, Saraswati S, et al. sFRP2 suppression of bone morphogenic protein (BMP) and Wnt signaling mediates mesenchymal stem cell (MSC) self-renewal promoting engraftment and myocardial repair.J Biol Chem. 2010;285(46):35645-35653.[29] Ng F, Boucher S, Koh S, et al. PDGF, TGF-beta, and FGF signaling is important for differentiation and growth of mesenchymal stem cells (MSCs): transcriptional profiling can identify markers and signaling pathways important in differentiation of MSCs into adipogenic, chondrogenic, and osteogenic lineages. Blood. 2008;112(2):295-307.[30] Casado-Díaz A, Anter J, Müller S, et al. Transcriptomic Analyses of Adipocyte Differentiation From Human Mesenchymal Stromal-Cells (MSC). J Cell Physiol. 2017; 232(4):771-784.[31] Grafe I, Alexander S, Peterson JR, et al. TGF-β Family Signaling in Mesenchymal Differentiation. Cold Spring Harb Perspect Biol. 2017 May 15. pii: a022202. doi: 10.1101/cshperspect.a022202. [Epub ahead of print][32] Tseng YH, Kokkotou E, Schulz TJ, et al. New role of bone morphogenetic protein 7 in brown adipogenesis and energy expenditure. Nature. 2008;454(7207):1000-1004.[33] Liao J, Wei Q, Zou Y, et al. Notch Signaling Augments BMP9-Induced Bone Formation by Promoting the Osteogenesis-Angiogenesis Coupling Process in Mesenchymal Stem Cells (MSCs). Cell Physiol Biochem. 2017;41(5):1905-1923.[34] Song BQ, Chi Y, Li X, et al. Inhibition of Notch Signaling Promotes the Adipogenic Differentiation of Mesenchymal Stem Cells Through Autophagy Activation and PTEN-PI3K/AKT/mTOR Pathway. Cell Physiol Biochem. 2015;36(5):1991-2002.[35] Augello A, De Bari C. The regulation of differentiation in mesenchymal stem cells. Hum Gene Ther. 2010;21(10): 1226-1238. [36] Armiñán A, Gandía C, Bartual M, et al. Cardiac differentiation is driven by NKX2.5 and GATA4 nuclear translocation in tissue-specific mesenchymal stem cells. Stem Cells Dev. 2009;18(6):907-918. [37] Lin TH, Gibon E, Loi F, et al. Decreased osteogenesis in mesenchymal stem cells derived from the aged mouse is associated with enhanced NF-κB activity. J Orthop Res. 2017; 35(2):281-288.[38] Meng X, Cui J, Wang Y, et al. Heterogeneous nuclear ribonucleoprotein A1 interacts with microRNA-34a to promote chondrogenic differentiation of mesenchymal stem cells. Am J Transl Res. 2017;9(4):1774-1782.[39] Boyer LA, Lee TI, Cole MF, et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell. 2005; 122(6):947-956.[40] Yoon DS, Choi Y, Jang Y, et al. SIRT1 directly regulates SOX2 to maintain self-renewal and multipotency in bone marrow-derived mesenchymal stem cells. Stem Cells. 2014; 32(12):3219-3231.[41] Simic P, Zainabadi K, Bell E, et al. SIRT1 regulates differentiation of mesenchymal stem cells by deacetylating β-catenin. EMBO Mol Med. 2013;5(3):430-440.[42] Yoon DS, Choi Y, Choi SM, et al. Different effects of resveratrol on early and late passage mesenchymal stem cells through β-catenin regulation. Biochem Biophys Res Commun. 2015;467(4):1026-1032.[43] Zhou H, Shang L, Li X, et al. Resveratrol augments the canonical Wnt signaling pathway in promoting osteoblastic differentiation of multipotent mesenchymal cells. Exp Cell Res. 2009;315(17):2953-2962.[44] Huang XZ, Wen D, Zhang M, et al. Sirt1 activation ameliorates renal fibrosis by inhibiting the TGF-β/Smad3 pathway. J Cell Biochem. 2014;115(5):996-1005.[45] Kume S, Haneda M, Kanasaki K, et al. SIRT1 inhibits transforming growth factor beta-induced apoptosis in glomerular mesangial cells via Smad7 deacetylation. J Biol Chem. 2007;282(1):151-158.[46] Xu Y, Wang S, Tang C, et al. Upregulation of long non-coding RNA HIF 1α-anti-sense 1 induced by transforming growth factor-β-mediated targeting of sirtuin 1 promotes osteoblastic differentiation of human bone marrow stromal cells. Mol Med Rep. 2015;12(5):7233-7238.[47] Bäckesjö CM, Li Y, Lindgren U, et al. Activation of Sirt1 decreases adipocyte formation during osteoblast differentiation of mesenchymal stem cells. J Bone Miner Res. 2006;21(7):993-1002.[48] Said RS, El-Demerdash E, Nada AS, et al. Resveratrol inhibits inflammatory signaling implicated in ionizing radiation-induced premature ovarian failure through antagonistic crosstalk between silencing information regulator 1 (SIRT1) and poly(ADP-ribose) polymerase 1 (PARP-1). Biochem Pharmacol. 2016;103:140-150. [49] Kauppinen A, Suuronen T, Ojala J, et al. Antagonistic crosstalk between NF-κB and SIRT1 in the regulation of inflammation and metabolic disorders. Cell Signal. 2013;25 (10):1939-1948. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [14] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||