Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (8): 1261-1266.doi: 10.3969/j.issn.2095-4344.0146
Previous Articles Next Articles
Choke vessel growth in perforator flaps and the conception of angiosome
Li Wen-bo, Zhang Chao, Shi Jie, Huang Qiang, Jia Ding-ding, Gao Qiu-ming
- Department of Orthopaedic Trauma, Orthopedics Center of Lanzhou General Hospital of Military Region of Chinese PLA, Lanzhou 730050, Gansu Province, China
-
Received:2017-11-22Online:2018-03-18Published:2018-03-18 -
Contact:Gao Qiu-ming, Master, Chief physician, Master’s supervisor, Department of Orthopaedic Trauma, Orthopedics Center of Lanzhou General Hospital of Military Region of Chinese PLA, Lanzhou 730050, Gansu Province, China -
About author:Li Wen-bo, Master candidate, Department of Orthopaedic Trauma, Orthopedics Center of Lanzhou General Hospital of Military Region of Chinese PLA, Lanzhou 730050, Gansu Province, China -
Supported by:the National Science Foundation for Distinguished Young Scholars of China, No. 81600700; the Natural Science Foundation of Gansu Province, No. 1506RJZA298
CLC Number:
Cite this article
Li Wen-bo, Zhang Chao, Shi Jie, Huang Qiang, Jia Ding-ding, Gao Qiu-ming. Choke vessel growth in perforator flaps and the conception of angiosome[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1261-1266.
share this article

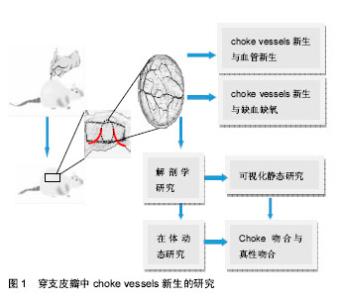
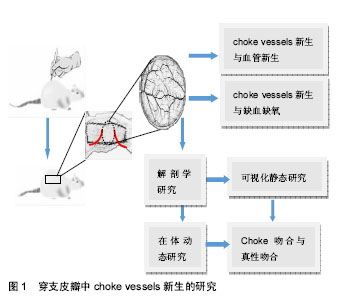
2.1 穿支皮瓣 以管径细小的皮肤穿支血管供血的轴型皮瓣,穿支皮瓣的特征是以穿支血管为蒂,穿支血管分为肌间隔穿支和肌皮穿支两种[1]。目前文献对穿支血管穿过深筋膜后的走形鲜有报道,而且可以肯定的是每一条穿支到达皮肤的走形是不一样的。 穿支是从深筋膜穿出供应其浅表,分支成星形状,单独供应一块特定的皮肤,即所谓的皮肤穿支体[2-3]。通常有两种类型:一种是典型的具有星形分支的垂直血管,以穿支为中心均匀向各个方向发散,一种是斜向上走形的血管,要么没有分支,要么会有两个相反的分支。有两个相反分支的血管体一般会斜行走向活动性好或者相对固定的皮肤,其走形一般离深筋膜有一定距离。而有3个或更多主要分支(星形分支)的穿支在临床皮瓣设计上更能达到理想的效果。 2.2 choke vessels choke vessels是连接相邻穿支血管的桥梁,是跨区穿支皮瓣血供来源的必由之路,对跨区皮瓣成活至关重要。Cormack等[4]通过分析相邻穿支体之间的关系,将皮肤的血管供区依次分为解剖学供区、血流动力学供区及潜在供区。解剖学供区是某一源动脉呈树形分布所到之处,是基本的血管供区;血流动力学供区是当一侧血管被切断或阻断时,其管内血压下降,解剖学供区血管内的血流就会跨越原来的吻合部位choke vessels区向压力低的一侧供血,这种紧邻原解剖学供区的扩张部位被称为血流动力学供区;潜在供区是在跨越动力学供区的基础上,再继续向远处供区延伸的区域。以这种血流跨越解剖学供区向血流动力学供区甚至向更远的潜在供区供血为理论基础,临床上可行跨区皮瓣切取来修复单血管体皮瓣无法覆盖的大面积皮肤缺损[5]。某一穿支的血流可通过choke血管的扩张安全到达相邻的穿支体区,但第三穿支体区的成活确实无法预测的。研究表明,皮瓣坏死经常发生在血流动力学区和潜在供区,即所谓的chokeⅠ区和chokeⅡ区[6]。带蒂皮瓣术后,至少相邻穿支体区可安全成活,但第三个血管体区往往面临坏死,而且chokeII区血管不会扩张。第三穿支体区的坏死与chokeⅡ区有关,chokeⅡ区是血流阻力的分水岭[5]。 2.3 穿支血管和choke vessels的可视化静态研究 起初对皮肤血管的研究是依靠血管造影技术来实现的。Taylor等[7]对大量标本进行皮肤血管造影,统计出口径在 0.5 mm以上的皮肤穿支374条,并对皮肤血供的来源进行了区分,他们根据皮肤内血管三维立体分布的特点提出了“血管体(angiosome)”的概念,指出每一皮肤血管除供应其分布范围内的皮肤和皮下组织外,还供应同一范围内的肌肉、肌腱、骨骼等多种组织在内的三维组织块。Geddes、楼新法等[8-9]对明胶-氧化铅血管灌注造影技术进行了改良,应用“一次性整体血管标识技术”、“交互式医学影像控制系统”对全身口径大于0.5 mm的血管进行了全面系统的观察,绘制出了人体皮肤穿支分布图[10],统计出口径0.5 mm以上的皮肤穿支442条。这种利用明胶-氧化铅灌注后进行二维X射线摄片虽然能够提供清晰的血管区域影像,但其缺点是影像叠加,解剖层次不清,无法剔除不需要的解剖结构的干扰[11]。CT 血管造影(computed tomographic angiography,CTA)较X射线平片相比具有明显的优势:它能够屏蔽掉不需要的重叠的解剖结构,具有内在的高分辨率,能够进行三维重建。Saint等[12]运用CT血管造影技术对对多个皮肤单穿支进行了3D可视化研究,发现相邻穿支之间通过直接链接和间接链接两种方式吻合;位于跨区供血位置,首先对同一源动脉发出的邻近穿支进行供血,其次对其他源动脉的邻近穿支进行供血。同时提出了穿支体区(Perforasome) 概念。但CT血管造影主要是在新鲜标本上进行的,不能真实反映活体的choke vessels的生理特性。而且,X射线摄片和CT血管造影通常局限于choke 动脉,而不能同时观察choke静脉。 2.4 choke vessels在体动态研究 庄跃宏等[13-14]设计了一种研究 Choke vessels新的动物模型——大鼠背部皮窗。通过在大鼠背部安装皮窗对choke vessels生理状态下血流动力学的变化进行了动态观察。戴开宇等[15]运用上述大鼠背部皮窗模型观察大鼠背部跨区皮瓣的choke 静脉变化规律,发现在皮瓣切取后的早期,静脉血主要以“迷宫”式回流为主,而术后晚期则通过“迷宫式”及“瓣膜失效”两种方式进行回流,以“瓣膜失效”为主。胡斯旺等[16]对皮窗进行了改进,制成“新型大鼠皮肤血管实时观察窗套件”,对大鼠背部穿支体区间choke vessels变化规律进行了探索,研究发现,跨区皮瓣术后,穿支体间choke vessels动脉有两个扩增高峰,分别是术后4-6 d和10-16 d,这对临床上切取跨区皮瓣、延迟皮瓣有一定的指导意义。大鼠背部皮窗模型的优缺点见表1。 2.5 choke吻合与真性吻合 研究发现相邻穿支体间存在3种吻合:真性吻合、choke吻合与生理条件下不开放的潜在性吻合[17]。真性吻合是血管体之间大的管腔跟管腔之间的吻合,管径一般恒定不会逐渐变小。choke吻合位于血管体周围多个血管分支处,管径是逐渐减小的。真性吻合血管的血流量要比choke吻合区血流量大得多,因为血管阻力与管半径的四次方成反比。 一般认为,穿支皮瓣可以有效灌注其解剖学区域和相邻穿支体区,而第三穿支体区域或者距离血管蒂有两个穿支体区的区域有可能出现缺血缺氧甚至皮瓣坏 死[2]。但也有文献报道即使皮瓣内包含了3个甚至4个穿支体,皮瓣仍然全部成活[18-20]。穿支体间不同的吻合形式很好地解释了为什么有些案例中能成功切取第三穿支体区域甚至更大范围的皮瓣,反之在其他案例中则有可能造成皮瓣坏死。在真性吻合的穿支体间,血流容易通过真性吻合血管使皮瓣得到充足的血供以保证皮瓣的成活。而在choke吻合的穿支体间,血流通常只可流经一个choke区供给邻近的穿支体区域,却不足以流经第2个choke区向第3个或更远的穿支体区供血[17]。 2.6 choke vessels新生与血管新生 对延迟机制的研究加深了对皮瓣的认识[6,21-24],外科延迟术可通过choke vessels促进穿支皮瓣的扩展[25]。延迟通常是在术前1周左右,切割已设计好的皮瓣边缘,以阻断皮瓣内一些血管的血供以达到刺激choke vessels的目的,使choke vessels扩张增生满足末梢血供,从而改变临床预后。choke vessels的扩张是一种永久性的扩张[24]。choke vessels的生长是一个主动的过程,伴随着血管内皮细胞的增生和血管壁的扩张。皮瓣术后最初会有短暂的血管痉挛,紧接着在前72 h内血管由于血流中断会出现快速扩张。72 h后,血管在持续扩张的同时,还会伴有血管内皮细胞的增生。术后第7天,会观察到明显的血管径扩张,细胞体积也会增大,choke vessels呈现迂曲状分布。目前对choke vessels新生的机制存在好多种说法,包括血流对choke血管的生理影响。血流量的增加可能会对血管施加一种延伸的力量,造成最初内膜的破坏,从而引起血管细胞增生和肥大以及平滑肌的延伸。研究发现炎症在延迟过程中的血管变化和保护作用方面起到关键的作用[26],这种延迟过程中的保护作用和血管改变都受到炎症和修复过程的调控[27]。 血管新生中侧支循环的建立是由机械力引发的[28]。血管新生是内皮细胞在原有血管的基础上增殖和迁移,以出芽的方式形成新的血管。出芽的方向是由促进血管生成的因子趋化的,新生的血管芽与邻近的血管芽或已存在的血管吻合,形成新的血管网[29-30]。当一支主要的传导动脉被血栓或由于损伤发生堵塞时,压力差会改变侧支小动脉的血流方向。侧支血管血流量的增加会使血管壁切应力增加,从而激活血管内皮[31-32]。血管内皮细胞释放细胞活性因子,比如单核细胞趋化因子,启动炎性级联反应。循环中的中性粒细胞和单核细胞聚集并穿过血管壁向周围组织转移。在堵塞后3 d左右巨噬细胞堆集达到峰值[33]。随着侧支血管的继续生长,这些血管呈现出特有的螺旋形状。在动脉堵塞近72 h左右,侧支血管壁近一半的细胞呈现出有分裂的迹象。这与Taylor对choke vessels的研究结果相一致,他通过Brdu摄取和染色检测到了细胞分裂[24]。血管新生的另一种方式是套迭式血管形成,只是血管的重新组织,不涉及血管内皮细胞的增殖,此过程可能与切应力和血流速度有 关[34]。内皮细胞可感知切应力并进行信号转换,上调转化生长因子β、一氧化氮合酶和黏附分子的表达[35]。血"

| [1] Taylor GI,Rozen WM,Whitaker IS.Establishing a perforator flap nomenclature based on anatomical principles.Plast Reconstr Surg.2012;129(5):877e-879e.[2] Taylor GI,Corlett RJ,Dhar SC,et al.The anatomical(angiosome) and clinical territories of cutaneous perforating arteries: Development of the concept and designing safe flaps. Plast Reconstr Surg.2011;127(4):1447-1459.[3] Rozen WM,Ashton MW,Le Roux CM,et al.The perforator angiosome: A new concept in the design of deep inferior epigastric artery perforator flaps for breast reconstruction. Microsurgery. 2010;30(1):1-7.[4] Cormack GC,Lamberty BG.A classification of fascio-cutaneous flaps according to their patterns of vascularisation.Br J Plast Surg. 1984;37(1):80-87.[5] Miyamoto S, Minabe T, Harii K. Effect of recipient arterial blood inflow on free flap survival area.Plast Reconstr Surg. 2008;121(2): 505-513.[6] 李浙峰,高伟阳,陈时益,等.吡格列酮药物延迟对大鼠跨区穿支皮瓣成活的影响[J].中国修复重建外科杂志, 2014,28(6):701-706.[7] Taylor GI,Palmer JH.The vascular territories (angiosomes) of the body experimental study and clinical applications.Br J Plast Surg. 1987;40(2):113-141.[8] Tang M,Geddes CR,Yang D,et al.Modified lead oxide-gelatin injection technique for vascular studies.Chin Clin Anat. 2002;1(1):73-78.[9] 楼新法, 梅劲, Geddes C.R.,等.明胶-氧化铅血管造影术的优化[J].中国临床解剖学杂志, 2006,24(3):259-262.[10] 杨大平,唐茂林,R Christopher,等.皮肤穿支血管的解剖学研究[J].中国临床解剖学杂志, 2006,24(3):232-235.[11] Bergeron L,Tang M,Morris SF.A review of vascular injection techniques for the study of perforator flaps.Plast Reconstr Surg. 2006;117:2050-2057.[12] Saint CM,Wong C,Schaverien MV,et al.The perforasome theory: vascular anatomy and clinical implications.Plast Reconstr Surg. 2009;124(5):1529-1544.[13] Zhuang Y,Hu S,Wu D,et al.A novel in vivo technique for observation of choke vessels in a rat skin flap model.Plast Reconstr Surg.2012;130(2):308-317.[14] 庄跃宏,胡斯旺,吴东方,等.一种研究 Choke 血管新的动物模型-大鼠背部皮窗[J].中国临床解剖学杂志, 2011,29(6):609-613.[15] 戴开宇,胡斯旺,庄跃宏,等.大鼠背部跨区皮瓣的choke静脉的变化规律[J].解剖学报, 2013,44(2):238-244.[16] 胡斯旺,庄跃宏,崔怀瑞, 等.大鼠背部穿支体区间choke血管变化规律初探[J].中国临床解剖学杂志, 2014,32(5):580-584.[17] Chubb DP, Taylor GI, Ashton MW. True and "Choke" Anastomoses between Perforator Angiosomes: Part II. Dynamic Thermographic Identifcation.Plast Reconstr Surg.2013;132(6): 1457-1464.[18] Aoki S,Tanuma K,Iwakiri I,et al.Clinical and vascular anatomical study of distally based sural ?ap.Ann Plast Surg.2008;61(1): 73-78.[19] Prasad V, Morris S F. Propeller DICAP ?ap for a large defect on the back: Case report and review of the literature.Microsurgery. 2012;32(8):617-621.[20] Teo TC.The propeller ?ap concept.Clin Plast Surg. 2010;37(4): 615-626.[21] Ceylan R, Kaya B, Çaydere M, et al. Comparison of ischaemic preconditioning with surgical delay technique to increase the viability of single pedicle island venous flaps: An experimental study. J Plast Surg Hand Surg.2014;48(6):368-374.[22] Do?an F,Özyazgan ?.Flap Preconditioning by Electrical Stimulation as an Alternative to Surgical Delay: Experimental Study.Ann Plast Surg.2015;75(5):560-564.[23] Temiz G,Ye?ilo?lu N,?irino?lu H, et al.Increasing the survival of transverse rectus abdominis musculocutaneous flaps with a Botulinum toxin-A injection:A comparison of surgical and chemical flap delay methods.J Plast Reconstr Aesthet Surg.2016;69(7): 944-951.[24] Dhar SC,Taylor GI.The delay phenomenon:the story unfolds.Plast Reconstr Surg. 1999;104(7):2079-2091.[25] Acart Ürk TO,Dincyürek H,Dagholug K.Delay by staged elevation of flaps and importance of inclusion of the perforator artery.J Hand Microsurg.2015;7(1):1-5.[26] Williams BA,Currie RW,Morris SF.Impact of Arteriogenesis in Plastic Surgery: Choke Vessel Growth Proceeds via Arteriogenic Mechanisms in the Rat Dorsal Island Skin Flap.Microcirculation. 2009;16(3):235-250.[27] Kleintjes WG. Delay phenomenon: can it be explained on the basis of inflammation and repair?. Plast Reconstr Surg.2001; 108(4):1090-1091.[28] Hänze J,Weissmann N,Grimminger F,et al.Cellular and molecular mechanisms of hypoxia-inducible factor driven vascular remodeling.Thromb Haemost.2007;97(5):774-787.[29] Hansen-Smith FM. Capillary network patterning during angiogenesis. Clin Exp Pharmacol Physiol. 2000;27(10):830-835.[30] Shweiki D,Itin A,Soffer D,et al.Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature.1992;359(6398):843-845.[31] Faber JE,Chilian WM, Deindl E,et al.A brief etymology of the collateral circulation. 2014;34(9):1854-1859.[32] Unthank JL,Sheridan KM,Dalsing MC.Collateral growth in the peripheral circulation: a review. Vasc Endovascular Surg.2004; 38(4):291-313.[33] Khmelewski E,Becker A,Meinertz T,et al.Tissue resident cells play a dominant role in arteriogenesis and concomitant macrophage accumulation.Circ Res.2004;95(6):E56-E64.[34] Kurz H,Burri PH,Djonov VG.Angiogenesis and vascular remodeling by intussusception:from form to function.News physiol Sci.2003;18(2):65-70.[35] Djonov VG,Galli AB,Burri PH.Intussusceptive arborization contributes to vasculartree formation in the chick chorio—allantoic membrane.Anat Embryol.2000;202(5):347-357.[36] Deindl E,Buschmann I,Hoefer IE,et al.Role of ischemia and of hypoxiainducible genes in arteriogenesis after femoral artery occlusion in the rabbit.Circ Res.2001;89(9):779-786.[37] 于文渊,余道江,赵天兰,等.狭长窄蒂皮瓣成活过程中低氧诱导因子-1α的表达及作用[J].中华实验外科杂志,2016,33(1):256.[38] Ramjaun AR,Hodivala-Dilke K.The role of cell adhesion pathways in angiogenesis.Int J Biochem Cell Biol.2009;41(3):521-530.[39] 徐子寒.地拉罗司模拟缺氧环境诱导EMT促进窄蒂皮瓣微血管生成的实验研究[D].苏州:苏州大学, 2012.[40] Kolbenschlag J, Sogorski A, Kapalschinski N, et al. Remote Ischemic Conditioning Improves Blood Flow and Oxygen Saturation in Pedicled and Free Surgical Flaps.Plast Reconstr Surg. 2016;138(5):1089-1097.[41] Kneser U,Beier JP, Schmitz M, et al.Zonal perfusion patterns in pedicled free-style perforator flaps.J Plast Reconstr Aesthet Surg. 2014;67(1):e9-e17.[42] Zhang Y,Xu H,Wang T,et al.Remote limb ischemic post-conditioning attenuates ischemia-reperfusion injury in rat skin ?apby limiting oxidative stress.Acta Cir Bras. 2016;31(1):15-21.[43] Heusch G, Bøtker HE, Przyklenk K, et al.Remote Ischemic Conditioning. J Am Coll Cardiol. 2015;65(2):177-195.[44] Linden MD, Whittaker P, Frelinger AL 3rd, et al. Preconditioning ischemia attenuates molecular indices of platelet activation-aggregation.J Thromb Haemost.2006;4(12):2670-2677.[45] Xu Y,Huo Y,Toufektsian MC,et al.Activated platelets contribute importantly to myocardial reperfusion injury.Am J Physiol Heart Circ Physiol.2006;290(2):H692-H696.[46] Burdess A,Nimmo AF,Campbell N,et al.Perioperative platelet and monocyte activation in patients with critical limb ischemia.J Vasc Surg.2010;52(3):697-703.[47] Shimizu M,Saxena P, Konstantinov I E, et al. Remote ischemic preconditioning decreases adhesion and selectively modifies functional responses of human neutrophils.J Surg Res. 2010; 158(1):155-161.[48] Konstantinov IE,Arab S,Kharbanda RK,et al.The remote ischemic preconditioning stimulus modifies inflammatory gene expression in humans.Physiol Genomics.2004;19(1):143-150.[49] Küntscher MV,Hartmann B,Germann G.Remote ischemic preconditioning of flaps: a review. Microsurgery.2005;25(4): 346-352.[50] Gao ZM,Lin DM,Wang Y,et al.Role of the NO/cGMP Pathway in Postoperative Vasodilation in Perforator Flaps.J Reconstr Microsurg. 2015;31(2):107-121.[51] McDonald WS,Lo TP Jr,Thurmond M,et al.Role of nitric oxide in skin flap delay. Plast Reconstr Surg.2004;113(3):927-931. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||