Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (19): 3063-3071.doi: 10.3969/j.issn.2095-4344.2017.19.018
Previous Articles Next Articles
Research progress and challenge of the stem cell therapy for intervertebral disc regeneration
Liang Hang, Deng Xiang-yu, Shao Zeng-wu
- Orthopedic Hospital of Union Hospital, Tongji Medical College of Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China
-
Online:2017-07-08Published:2017-08-10 -
Contact:Shao Zeng-wu, M.D., Professor, Doctoral supervisor, Orthopedic Hospital of Union Hospital, Tongji Medical College of Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China -
About author:Liang Hang, Orthopedic Hospital of Union Hospital, Tongji Medical College of Huazhong University of Science and Technology, Wuhan 430022, Hubei Province, China -
Supported by:the National Key Research and Development Program of Ministry of Science and Technology, No. 2016YFC1100100
CLC Number:
Cite this article
Liang Hang, Deng Xiang-yu, Shao Zeng-wu. Research progress and challenge of the stem cell therapy for intervertebral disc regeneration [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(19): 3063-3071.
share this article

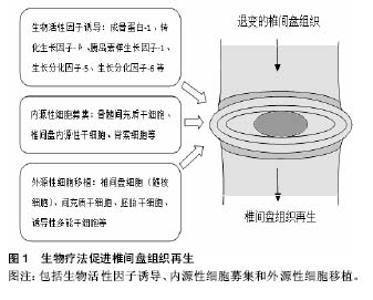
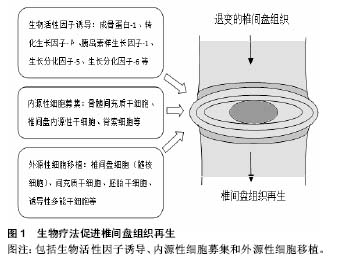
其中,生物活性因子在近年来的研究中被证实可在椎间盘的修复中发挥积极作用。生长因子(如成骨蛋白1、转化生长因子β、胰岛素样生长因子1、生长分化因子5和生长分化因子6)在体内实验中被证实具有促进细胞外基质合成的作用[7-11]。白细胞介素1受体拮抗剂被证实能够减少细胞因子及蛋白水解酶在髓核细胞中的产生,从而减少细胞外基质降解,提高细胞外基质质量,来维持椎间盘细胞生存的微环境[12-13]。同样,拮抗肿瘤坏死因子的疗法被证实具有类似效果,通过减少细胞外基质降解,促进细胞外基质的合成,对椎间盘细胞具有保护作用[14]。然而,生物制剂半衰期短暂、需重复操作等问题使该疗法的发展及应用受限,如何维持生物制剂的有效浓度、减少不必要的重复操作成为生物疗法进一步研究的核心问题[15]。另一方面,正常椎间盘组织细胞含量相对较低,而在退变的椎间盘组织中,因微环境的改变导致的椎间盘细胞凋亡将进一步减少正常细胞存活量,即便生物制剂浓度能够得到维持,缺乏充足有活力的正常椎间盘细胞也会在很大程度上影响治疗效果[16]。因此,将有活力的细胞代替生物制剂移植入退变的椎间盘内,利用其生存及增殖能力合成细胞外基质、调节椎间盘微环境成为进一步治疗退行性椎间盘疾病的研究热点。 组织再生过程中可从周围环境中通过募集作用来维持活细胞量及细胞功能,这样的稳态维持系统同样存在于椎间盘组织。Illien-Junger等[17-18]在体外对牛椎间盘组织行整块培养,发现外源性人间充质干细胞可通过软骨终板迁入,并能够对抗由饥饿、压力、创伤诱导的椎间盘退变。并且Pattappa等[19]证实椎间盘细胞在诱导退变的条件下可通过释放化学分子(如CCL5、CXCL6)来募集外源性细胞。然而,外源性细胞需通过脉管系统实现迁入募集,而椎间盘则是人体最大的无脉管系统的组织。Tzaan、Sakai等[20-21]在动物实验中证实在无血管的椎间盘组织中,很少有其他组织来源的细胞迁入,证实椎间盘组织在受损或退变的情况下,无法通过募集作用来维持正常细胞量及细胞功能而无法实现修复及再生。因此运用椎间盘内源性细胞的修复再生作用成为椎间盘组织再生研究的关键。 近几年有大量项研究证实移植自体椎间盘源性的细胞或关节软骨细胞能够有效延缓椎间盘退变进程、促进椎间盘组织再生[22-32]。Hohaus等[33]在犬的动物模型中证实自体髓核细胞移植能够延缓椎间盘退变;并在临床试验中证实与单纯椎间盘髓核摘除术相比,该方法能有效缓解患者腰痛、维持椎间隙高度。自体关节软骨细胞及椎间盘细胞能够分别从自身的关节软骨及椎间盘组织中提取并分离。然而,此种方法的应用同样存在诸多限制。一方面,关节软骨细胞虽然能分泌Ⅱ型胶原及蛋白聚糖,但其分泌蛋白聚糖与Ⅱ型胶原的比率明显低于髓核细胞,因此将其作为移植细胞来修复椎间盘组织并不合适[34-35];另一方面,椎间盘组织的在正常人体的量是相对较少的,侵入性的操作以及从自体进行大量的髓核细胞提取会导致严重并发症[36-37];而突出或者脱出的椎间盘组织中正常的椎间盘细胞量更低,从中获取的髓核细胞常表现为过早的衰老,难以在体外进行扩增,这将严重影响其修复及再生效率[35, 38-40]。因此,目前获取可移植细胞的最佳方式便是充分利用干细胞技术。 2.1.2 干细胞移植促进椎间盘再生 近年来,干细胞移植技术在椎间盘修复再生领域得到广泛关注与研究,基于干细胞的疗法能够有效的修复椎间盘功能、维持椎间盘结构稳定[41-45]。并动物实验中证实能够促进椎间盘再生、修复椎间隙高度、促进细胞外基质合成,从而改善椎体功能[43-44, 46-49]。目前,间充质干细胞针对椎间盘组织修复再生的作用在体外培养与体内移植方面的研究均取得显著成果。间充质干细胞与髓核细胞共培养能够显著增加髓核细胞生物活性。Watanabe等[50]在人髓核细胞与间充质干细胞共培养研究中发现髓核细胞增殖及DNA复制合成能力明显增强,证实间充质干细胞能够提高髓核细胞生存能力。Miyamoto等[51]发现将兔髓核细胞与人间充质干细胞共培养能改变髓核细胞基因表型,抑制降解酶及炎症因子的表达,从而减少细胞外基质降解,增加细胞外基质合成,维持椎间盘的微环境。动物实验中,Crevensten等[52]将同种异体的间充质干细胞移植入大鼠椎间盘组织中,发现4周后间充质干细胞得到显著扩增,椎间隙高度得到明显提高,证明蛋白聚糖在椎间盘组织中产量增加,因此作者认为移植的间充质干细胞对退变的椎间盘组织有明显的修复效果。Miyamoto等[51]进一步实验,将兔椎间盘部分髓核细胞抽离并移植入间充质干细胞,结果显示间充质干细胞能促进剩余的髓核细胞产生Ⅱ型胶原。间充质干细胞移植技术治疗椎间盘退变在大型动物模型中同样得到验证。Hiyama等[42]将间充质干细胞移植入椎间盘退变的犬模型中,结果显示间充质干细胞移植促进椎间盘再生的作用在影像学、组织学及生物化学分析方面均有明显体现。 运用于椎间盘再生的间充质干细胞可来源于骨 髓[42-44]、软骨终板[41]、脐带血[44]、滑膜组织[51, 53]、脂肪组织等[47, 54]。Chun等[47]在兔的椎间盘损伤模型中使用脂肪来源的间充质干细胞来促进椎间盘组织修复再生,发现移植的脂肪来源间充质干细胞能够有效增加细胞外基质,并且与对照组相比,损伤的软骨骨化水平显著降低。同样,Liang等[55]通过在兔椎间盘退变模型中移植脂肪来源间充质干细胞来观察其对退变的椎间盘是否有修复再生作用,结果发现在移植后24周,退变的椎间盘细胞外基质得到有效提高,椎间隙高度也得到维持与加强。Murrel等[56]将嗅神经源性的间充质干细胞移植入兔椎间盘中,发现其成功的向髓核样细胞分化。这些研究表明,来源不同的间充质干细胞都有治疗椎间盘退变的潜能。 Yim等[57]对24项针对不同的间充质干细胞类型、治疗策略、细胞剂量、多种椎间盘退变环境中的疗效的研究进行分析,涵盖了小鼠、大鼠、兔、犬、猪、羊等动物模型。作者指出骨髓间充质干细胞、脂肪间充质干细胞以及滑液间充质干细胞是相对安全并能有效阻止椎间盘退变的。并且,每个椎间盘实施106剂量的细胞移植既能显著修复椎间盘组织,又能最大程度的减少移植后的间充质干细胞凋亡。然而迄今没有确切证据证明某一特定的间充质干细胞在促进椎间盘再生作用中有明显优势。此外,Vasiliadis、Minogue等[35, 58]通过实验表明与骨髓间充质干细胞相比,脂肪间充质干细胞能分化为跟髓核细胞更加相似的表型。Wang等[41]最近的研究表明软骨终板来源的干细胞与骨髓间充质干细胞比较,在MRI及组织样本中展现出更强的促进髓核组织再生作用。 虽然各项研究已证实间充质干细胞对治疗椎间盘退变有显著效果,但直接移植间充质干细胞进入靶向椎间盘组织仍缺乏可靠的方式。移植细胞在移植靶点的回漏以及其移植后的较低存活率对干细胞疗法的运用产生极大限制[57, 59-61]。并且,Vadala等[61]在对椎间盘退变的兔模型行间充质干细胞移植后发现移植靶点有骨赘形成。值得关注的是,近年来生物支架的运用能够有效避免移植细胞的回漏并提高移植细胞的生存及适应能力[31, 62-66]。Ruan等[67]分别在体外实验及体内实验内中证实搭载PLGA支架的髓核细胞有更强的增殖能力,在犬的实验模型中能够延缓椎间盘退变的进展。然而,生物支架的使用同样受到生物相容性、可降解性等诸多限制,寻求更高效、更安全的细胞移植方法有待进一步研究。 2.1.3 临床试验 近年来,诸多干细胞移植治疗椎间盘退行性疾病的研究进入临床试验阶段。Yoshikawa等[68]通过临床试验证实了移植的自体骨髓间充质干细胞能够帮助退变的椎间盘组织再生。该研究对象是2例有腰腿疼痛、麻木症状,且影像学提示有明显腰椎间盘退变的患者。研究人员从每位患者的髂骨取骨髓液,分离出骨髓间充质干细胞并用自体血清体外培养,在完成椎管减压术后,将包含自体骨髓间充质干细胞的明胶海绵移植入退变的椎间盘。术后2年,最初的症状得到有效缓解,X射线及CT随访显示椎间盘退变得到改善,脊柱稳定性得到提高,MRI中T2加权显示移植区为高信号(含水量增加)。 Orozco等[60]在一项针对10例伴有慢性腰痛的椎间盘退变的病例临床试验中,通过移植自体骨髓间充质干细胞进入退变的椎间盘组织来治疗其椎间盘退行性疾病。12个月后全部10例患者的背痛同样得到有效缓解,脊柱功能得到明显改善。并且与其他手术方式如脊柱融合术或椎体成形术相比具有明显优势,MRI中T2加权显示移植区椎间盘组织水含量升高,但是椎间隙高度并未恢复。因此,该临床试验的研究者认为干细胞疗法对椎间盘退行性疾病的治疗是有效且安全的,尽管椎间隙高度并未得到改善,但患者椎间盘组织水含量在术后12个月得到显著提高,干细胞疗法可作为部分伴有明显腰痛的椎间盘退行性疾病患者的另一选择。 Meisel等[69-70]通过分析112例椎间盘退行性疾病患者,探究与单纯髓核摘除术相比,在椎间盘髓核摘除术后移植自体椎间盘细胞的安全性及有效性,以及能否提高髓核细胞外基质水平。自体椎间盘细胞移植术在单节段髓核摘除术后3个月进行。术后2年的随访表明,与单纯行髓核摘除术相比,后续行自体椎间盘细胞移植的患者的腰痛得到更为明显的改善;并且术区的椎间隙高度得到维持,相邻节段的椎间盘含水量更为丰富,而单纯行椎间盘髓核摘除术的患者椎间隙高度出现下降。研究者认为,与单纯手术相比,术后采用自体椎间盘细胞移植技术能够安全、有效的缓解腰痛,促进椎间盘组织再生,干细胞移植技术在治疗椎间盘退行性疾病中有很大潜力。 Coric等[71]在一项前瞻性临床试验中,针对15个椎间盘退变导致腰痛的病例使用异体关节软骨细胞而非间充质干细胞或椎间盘细胞进行移植,病例均为L3至S1之间的单节段椎间盘退变,每个患者术中采用1.0-2.0 mL的细胞悬液(每毫升107个细胞)移植注射。术后12个月的随访中,均未出现不良反应,脊柱功能均得到一定恢复,其中10例在术后6个月的MRI显示椎间盘退变程度较前有明显改善,3例因腰痛持续而接受手术治疗。 总之,越来越多的临床试验已证实干细胞移植治疗椎间盘退变有确切疗效,不仅能够有效缓解腰痛,恢复脊柱盘功能,其安全性也得到越来越多的认可(表1),随着研究进一步深入,干细胞移植技术或许将成为治疗椎间盘退变的另一选择。"


2.2 干细胞移植治疗椎间盘退变面临的挑战 2.2.1 适应椎间盘微环境 尽管一系列体外、体内实验均证实干细胞移植能够促进椎间盘再生,但如何使移植的干细胞适应椎间盘特殊的微环境并适时的发挥作用成为研究难点。大多数器官、组织再生过程中包含了一系列细胞活动,如炎症的发生、细胞的死亡与去分化、细胞的增殖、外源性细胞的募集、合成因子的分泌、细胞外基质的合成等,但这一系列过程都发生在有脉管的组织中,而椎间盘是人体最大的无脉管组织,椎间盘细胞主要通过扩散作用利用相邻椎体血管内的氧气及营养物质[72]。因此椎间盘微环境与其他组织不同,表现为细胞量少、葡萄糖含量低、低氧、低pH值、缺乏营养物质等[73-77]。而当可供利用的营养物质不能满足椎间盘细胞的需求时,椎间盘细胞的活性及功能将会受到影响。近年来研究者利用生物制剂或细胞等生物疗法来促进退变的椎间盘修复再生,然而椎间盘独特的微环境将会对移植细胞的生存、代谢以及分化造成不利影响[76]。因此,如何使移植细胞适应椎间盘微环境并正确的分化是研究人员所面临的巨大挑战。间充质干细胞作为目前研究最多的移植供体细胞,其增殖和分化方向受多种因素调节,尤其是其生存的微环境及临近的细胞类型。体外培养中,使用特定的生长因子对间充质干细胞行预处理能够引导间充质干细胞向有特定表型的细胞方向分 化[78]。Steck等[79]将间充质干细胞在含有特定组织生长因子(转化生长因子β)的环境下培养,发现其分化的细胞表型与髓核细胞十分相似,并能够分泌Ⅱ型胶原和蛋白聚糖。Risbwd等[80]证实低氧条件下有利于髓核细胞表型的维持,并且低氧条件与转化生长因子β1协同可促进间充质干细胞向髓核细胞表型分化。鉴于此,研究者认为椎间盘组织的低氧、压力条件下或许对干细胞向椎间盘细胞分化产生有利影响,但是其高渗及酸性环境却不利于干细胞的生存[81-83]。炎症介质是导致椎间盘退变的主要因素之一,间充质干细胞必须改善退变椎间盘的炎症及营养物质缺乏的微环境才能发挥有效的修复作用[76]。而且,尽管研究表明低氧环境能加强椎间盘组织对间充质干细胞的保护作用,但是炎症因子(如白细胞介素1b和肿瘤坏死因子α)的积累将对移植的间充质干细胞的分化产生不利影响,抑制其向软骨类细胞分化而促进成骨类物质沉积,从而影响椎间盘的修复再生[76, 84-88]。因此,在针对退变的椎间盘组织行干细胞移植前,应确保移植的干细胞在独特的椎间盘微环境中能够向预先设计的方向分化。例如,有研究证实移植前对间充质干细胞预处理能诱导其正确分化。如移植前对间充质干细胞使用生长因子(转化生长因子β和骨形态发生蛋白)预处理能够刺激其向髓核细胞分化[89-91],或移植前将间充质干细胞与髓核细胞、脊索细胞或细胞外基质成分共培养来达到诱导其分化的效果[92-99]。此外,Le Visage等[100]取人体退变椎间盘中的髓核细胞以及纤维环细胞在体外与间充质干细胞共培养,发现纤维环细胞以及髓核细胞均发生了显著增殖,而蛋白多糖仅在纤维环细胞与间充质干细胞共培养时产量增加。因此,该研究者认为间充质干细胞在进行移植前或许无需促其分化为髓核样细胞或纤维环样细胞,移植的间充质干细胞可在椎间盘组织内向临近的细胞类型方向分化,促进椎间盘细胞增殖,帮助退变的椎间盘修复与再生。总之,在椎间盘独特、复杂、多变的微环境下,移植细胞的生存及效用面临极大挑战,虽然目前诸多研究已经进入临床试验阶段并取得一定疗效,但其长期的安全性以及作用的分子机制仍不明确,仍需进一步研究。 2.2.2 选择合适的干细胞 尽管椎间盘细胞移植治疗椎间盘退变的研究取得很好的前景,但是从自体获得可用的椎间盘细胞是非常困难的。一方面,选择健康的椎间盘提取细胞不仅会对其造成医源性损伤,而且在特殊的椎间盘微环境下,健康的椎间盘中有活力的细胞数量本身相对较少,如果从中获取细胞将会导致一系列并发症,加快健康椎间盘的退变。另一方面,选择突出或者脱出的退变的椎间盘提取细胞存在更大的限制,退变的椎间盘中健康的细胞数量进一步减少,从中提取的细胞常常表现为早衰,其细胞活性及增殖能力均下降,移植后的生存能力及功能性也会明显降低,不宜作为移植细胞的选择[35, 38-40]。因此,尽管自体椎间盘细胞移植在治疗椎间盘退变方面取得显著成果,但细胞来源的短缺或许将成为限制其发展的潜在因素。近年来,同种异体及异种来源的细胞被用于动物实验研究中,取得不错效果[25, 66]。但目前仍缺乏临床试验证据,同时受限于伦理道德问题,有待进一步深入研究。 在现有的临床试验中,间充质干细胞被认为是用于椎间盘再生的最佳供体细胞[101-102]。间充质干细胞是具有自我更新能力的多能成熟干细胞,具有向骨、软骨、脂肪、神经、肌肉、肌腱等所有中胚层细胞方向分化的能力[103]。间充质干细胞来源广泛,能够比较容易的从骨髓获取,同时也可在脂肪组织、胎盘组织、脐带血、结缔组织、皮肤、滑膜组织等获取[104]。使用间充质干细胞进行移植术前,可从自体骨髓提取、分离间充质干细胞进行体外培养并大量扩增,能够获得足够量的移植细胞。但目前仍缺乏检测手段来确认移植的间充质干细胞的分化方向,同样无法确保其准确的分化为椎间盘类软骨细胞而非成骨细胞。因此其不稳定的分化及潜在的致癌风险是不可忽视安全因素。 此外,胚胎干细胞及诱导性多能干细胞能分化为各种椎间盘细胞(如脊索细胞),同样可作为供体细胞用于椎间盘再生,且研究证实脊索细胞能产生髓核细胞并在移植术后更强的生存能力[16, 105-106]。Sheikh等[107]在兔的椎间盘退变模型中证实胚胎干细胞来源的软骨祖细胞能分化为脊索细胞。Liu等[106]研究证实使用髓核组织基质能使诱导性多能干细胞向脊索细胞分化。 然而,目前无论是移植细胞类型的选择上(椎间盘细胞、干细胞或者其他特定的细胞如软骨细胞等),还是细胞供体的选择上(自体、同种异体、异种),均缺乏临床研究的确切证据,不同移植细胞类型的长期的安全有效性以及具体的分子机制有待进一步的研究。 2.2.3 用于适合的患者 椎间盘退变受诸多因素影响,遗传、年龄、吸烟、肥胖等与退行性椎间盘疾病密切相关。尽管有许多干细胞移植治疗椎间盘退行性疾病的研究已进入临床试验阶段,但是针对不同的退变程度以及疼痛水平,究竟何种患者适合采取细胞移植疗法至今仍未得到明确[108]。 通过对现有临床试验成果的分析与总结,适用于干细胞移植疗法的椎间盘退行性疾病患者需满足以下3个条件。其一,针对有慢性腰痛症状的早期椎间盘退行性改变的患者,伴或不伴随影像学上明显的椎间盘退变表现,适合行干细胞移植术。因为早期的椎间盘退行性变并不适合采取手术方式来治疗,而传统的非手术方法仅以缓解症状为目的,并不能阻止椎间盘退变的进展,而持续进展的椎间盘退变将引起继发性的不可逆性退行性脊柱疾病。因此,在相应的继发性病变出现前,针对早期退变的椎间盘使用细胞移植疗法或许能够阻止甚至逆转其病理发展,帮助椎间盘组织修复再生,同时又能帮助患者缓解疼痛,改善脊柱功能。其二,影像学提示为早期椎间盘退行性疾病且为单个节段的病变的患者。目前大多数临床试验选择的患者均为单个节段的椎间盘退变,对于多个节段的椎间盘退变患者尚未进行针对性临床研究。单个节段的椎间盘退变一般处于病理发展的早期,退变的程度相对较轻,脊柱的功能及稳定性尚能维持,此时采取干细胞移植疗法较传统保守治疗及手术治疗有较大优势,或许能够在一定程度上逆转或延缓其椎间盘退变的发展,取得良好效果。其三,针对部分行手术治疗后的椎间盘退行性疾病患者,可采取干细胞疗法。一方面,部分患者在接受手术治疗后仍存在腰痛症状或腰痛症状很快出现反复,运用细胞移植技术或许可以有效缓解疼痛、促进病变椎间盘组织术后的修复重建,提高患者生活质量;另一方面,与单纯手术治疗(如单纯髓核摘除术)相比,在术后加用干细胞移植疗法能够能更好的缓解患者的疼痛症状,同时有利于维持病变椎间盘的椎间隙高度,增加相邻椎间盘组织的含水量,帮助维持脊柱的稳定性及功能。因此,针对术后的椎间盘退行性疾病患者,加用干细胞移植疗法作为辅助治疗或许能够提高手术疗效,减少术后并发症,减缓病情进展,协同手术方法进一步解除病痛,提高脊柱稳定性与功能。 然而,对于存在多个节段的椎间盘退变的患者或已引起继发性退行性病变的患者,细胞移植疗法也许很难达到预期疗效,因此针对椎间盘退行性疾病的早期诊断对于干细胞疗法的使用及疗效至关重要。然而目前的临床诊断方法手段对于早期的椎间盘退行性改变仍缺乏鉴别能力,病变早期的患者一般不伴随明显症状而不能及时就诊,早期有症状的患者以目前的影像学水平也很难鉴别,因此缺乏足够的证据来识别早期椎间盘退行性疾病的患者,目前临床上对于椎间盘退行性改变的诊断技术有待进一步提高。 "

| [1] Ivanova JI, Birnbaum HG, Schiller M, et al. Real-world practice patterns, health-care utilization, and costs in patients with low back pain: the long road to guideline-concordant care. Spine J. 2011;11(7): 622-632.[2] Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859): 2163-2196.[3] Kepler CK, Ponnappan RK, Tannoury CA, et al. The molecular basis of intervertebral disc degeneration. Spine J. 2013;13(3): 318-330.[4] Isgro M, Buraschi R, Barbieri C, et al. Conservative management of degenerative disorders of the spine. J Neurosurg Sci. 2014;58(2 Suppl 1): 73-76.[5] Miller SM. Low back pain: pharmacologic management. Prim Care. 2012;39(3): 499-510.[6] Wang MY, Vasudevan R, Mindea SA. Minimally invasive lateral interbody fusion for the treatment of rostral adjacent-segment lumbar degenerative stenosis without supplemental pedicle screw fixation. J Neurosurg Spine. 2014;21(6): 861-866.[7] Walsh AJ, Bradford DS, Lotz JC. In vivo growth factor treatment of degenerated intervertebral discs. Spine (Phila Pa 1976). 2004;29(2): 156-163.[8] Osada R, Ohshima H, Ishihara H, et al. Autocrine/paracrine mechanism of insulin-like growth factor-1 secretion, and the effect of insulin-like growth factor-1 on proteoglycan synthesis in bovine intervertebral discs. J Orthop Res. 1996;14(5): 690-699.[9] Imai Y, Okuma M, An HS, et al. Restoration of disc height loss by recombinant human osteogenic protein-1 injection into intervertebral discs undergoing degeneration induced by an intradiscal injection of chondroitinase ABC. Spine (Phila Pa 1976). 2007;32(11): 1197-1205.[10] Wei A, Williams LA, Bhargav D, et al. BMP13 prevents the effects of annular injury in an ovine model. Int J Biol Sci. 2009;5(5): 388-396.[11] An HS, Takegami K, Kamada H, et al. Intradiscal administration of osteogenic protein-1 increases intervertebral disc height and proteoglycan content in the nucleus pulposus in normal adolescent rabbits. Spine (Phila Pa 1976). 2005;30(1): 25-31; discussion 31-32.[12] Le Maitre CL, Freemont AJ, Hoyland JA. A preliminary in vitro study into the use of IL-1Ra gene therapy for the inhibition of intervertebral disc degeneration. Int J Exp Pathol. 2006;87(1): 17-28.[13] Le Maitre CL, Hoyland JA, Freemont AJ. Interleukin-1 receptor antagonist delivered directly and by gene therapy inhibits matrix degradation in the intact degenerate human intervertebral disc: an in situ zymographic and gene therapy study. Arthritis Res Ther. 2007; 9(4): R83.[14] Tobinick EL, Britschgi-Davoodifar S. Perispinal TNF-alpha inhibition for discogenic pain. Swiss Med Wkly. 2003;133 (11-12): 170-177.[15] Larson JW, Levicoff EA, Gilbertson LG, et al. Biologic modification of animal models of intervertebral disc degeneration. J Bone Joint Surg Am. 2006;88 Suppl 2: 83-87.[16] Acosta FL Jr, Lotz J, Ames CP. The potential role of mesenchymal stem cell therapy for intervertebral disc degeneration: a critical overview. Neurosurg Focus. 2005; 19(3): E4.[17] Illien-Junger S, Gantenbein-Ritter B, Grad S, et al. The combined effects of limited nutrition and high-frequency loading on intervertebral discs with endplates. Spine (Phila Pa 1976). 2010; 35(19): 1744-1752.[18] Illien-Junger S, Pattappa G, Peroglio M, et al. Homing of mesenchymal stem cells in induced degenerative intervertebral discs in a whole organ culture system. Spine (Phila Pa 1976). 2012;37(22): 1865-1873.[19] Pattappa G, Eroglio M, Sakai D, et al. CCL5/RANTES is a key chemoattractant released by degenerative intervertebral discs in organ culture. Eur Cell Mater. 2014;27: 124-136.[20] Tzaan WC, Chen HC. Investigating the possibility of intervertebral disc regeneration induced by granulocyte colony stimulating factor-stimulated stem cells in rats. Adv Orthop. 2011;2011: 602089.[21] Sakai D, Nishimura K, Tanaka M, et al. Migration of bone marrow-derived cells for endogenous repair in a new tail-looping disc degeneration model in the mouse: a pilot study. Spine J. 2015;15(6): 1356-1365.[22] Nishimura K, Mochida J. Percutaneous reinsertion of the nucleus pulposus. An experimental study. Spine (Phila Pa 1976). 1998;23(14): 1531-1538; discussion 1539.[23] Gruber HE, Johnson TL, Leslie K, et al. Autologous intervertebral disc cell implantation: a model using Psammomys obesus, the sand rat. Spine (Phila Pa 1976). 2002;27(15): 1626-1633.[24] Okuma M, Mochida J, Nishimura K, et al. Reinsertion of stimulated nucleus pulposus cells retards intervertebral disc degeneration: an in vitro and in vivo experimental study. J Orthop Res. 2000;18(6): 988-997.[25] Nomura T, Mochida J, Okuma M, et al. Nucleus pulposus allograft retards intervertebral disc degeneration. Clin Orthop Relat Res. 2001;(389): 94-101.[26] Sato M, Asazuma T, Ishihara M, et al. An experimental study of the regeneration of the intervertebral disc with an allograft of cultured annulus fibrosus cells using a tissue-engineering method. Spine (Phila Pa 1976). 2003; 28(6): 548-553.[27] Watanabe K, Mochida J, Nomura T, et al. Effect of reinsertion of activated nucleus pulposus on disc degeneration: an experimental study on various types of collagen in degenerative discs. Connect Tissue Res. 2003;44(2): 104-108.[28] Bertram H, Kroeber M, Wang H, et al. Matrix-assisted cell transfer for intervertebral disc cell therapy. Biochem Biophys Res Commun. 2005;331(4): 1185-1192.[29] Iwashina T, Mochida J, Sakai D, et al. Feasibility of using a human nucleus pulposus cell line as a cell source in cell transplantation therapy for intervertebral disc degeneration. Spine (Phila Pa 1976). 2006; 31(11): 1177-1186.[30] Huang B, Zhuang Y, Li CQ, et al. Regeneration of the intervertebral disc with nucleus pulposus cell-seeded collagen II/hyaluronan/chondroitin-6-sulfate tri-copolymer constructs in a rabbit disc degeneration model. Spine (Phila Pa 1976). 2011;36(26): 2252-2259.[31] Ganey T, Libera J, Moos V, et al. Disc chondrocyte transplantation in a canine model: a treatment for degenerated or damaged intervertebral disc. Spine (Phila Pa 1976). 2003;28(23): 2609-2620.[32] Ruan DK, Xin H, Zhang C, et al. Experimental intervertebral disc regeneration with tissue-engineered composite in a canine model. Tissue Eng Part A. 2010;16(7): 2381-2389.[33] Hohaus C, Ganey TM, Minkus Y, et al. Cell transplantation in lumbar spine disc degeneration disease. Eur Spine J. 2008; 17 Suppl 4: 492-503.[34] Mwale F, Roughley P, Antoniou J. Distinction between the extracellular matrix of the nucleus pulposus and hyaline cartilage: a requisite for tissue engineering of intervertebral disc. Eur Cell Mater. 2004;8: 58-63; discussion 63-64.[35] Vasiliadis ES, Pneumaticos SG, Evangelopoulos DS, et al. Biologic treatment of mild and moderate intervertebral disc degeneration. Mol Med. 2014;20: 400-409.[36] Carragee EJ, Don AS, Hurwitz EL, et al. 2009 ISSLS Prize Winner: Does discography cause accelerated progression of degeneration changes in the lumbar disc: a ten-year matched cohort study. Spine (Phila Pa 1976). 2009; 34(21): 2338-2345.[37] Nassr A, Lee JY, Bashir RS, et al. Does incorrect level needle localization during anterior cervical discectomy and fusion lead to accelerated disc degeneration? Spine (Phila Pa 1976). 2009;34(2): 189-192.[38] Chadderdon RC, Shimer AL, Gilbertson LG, et al. Advances in gene therapy for intervertebral disc degeneration. Spine J. 2004; 4(6 Suppl): 341S-347S.[39] Gruber HE, Ingram JA, Norton HJ, et al. Senescence in cells of the aging and degenerating intervertebral disc: immunolocalization of senescence-associated beta-galactosidase in human and sand rat discs. Spine (Phila Pa 1976). 2007;32(3): 321-327.[40] Roberts S, Evans EH, Kletsas D, et al. Senescence in human intervertebral discs. Eur Spine J. 2006;15 Suppl 3: S312-316.[41] Wang H, Zhou Y, Huang B, et al. Utilization of stem cells in alginate for nucleus pulposus tissue engineering. Tissue Eng Part A. 2014;20(5-6): 908-920.[42] Hiyama A, Mochida J, Iwashina T, et al. Transplantation of mesenchymal stem cells in a canine disc degeneration model. J Orthop Res. 2008;26(5): 589-600.[43] Feng G, Zhao X, Liu H, et al. Transplantation of mesenchymal stem cells and nucleus pulposus cells in a degenerative disc model in rabbits: a comparison of 2 cell types as potential candidates for disc regeneration. J Neurosurg Spine. 2011;14(3): 322-329.[44] Serigano K, Sakai D, Hiyama A, et al. Effect of cell number on mesenchymal stem cell transplantation in a canine disc degeneration model. J Orthop Res. 2010;28(10): 1267-1275.[45] Jin ES, Min J, Jeon SR, et al. Analysis of molecular expression in adipose tissue-derived mesenchymal stem cells : prospects for use in the treatment of intervertebral disc degeneration. J Korean Neurosurg Soc. 2013;53(4): 207-212.[46] Anderson DG, Markova D, An HS, et al. Human umbilical cord blood-derived mesenchymal stem cells in the cultured rabbit intervertebral disc: a novel cell source for disc repair. Am J Phys Med Rehabil. 2013;92(5): 420-429.[47] Chun HJ, Kim YS, Kim BK, et al. Transplantation of human adipose-derived stem cells in a rabbit model of traumatic degeneration of lumbar discs. World Neurosurg. 2012;78(3-4): 364-371.[48] Ganey T, Hutton WC, Moseley T, et al. Intervertebral disc repair using adipose tissue-derived stem and regenerative cells: experiments in a canine model. Spine (Phila Pa 1976). 2009;34(21): 2297-2304.[49] Allon AA, Aurouer N, Yoo BB, et al. Structured coculture of stem cells and disc cells prevent disc degeneration in a rat model. Spine J. 2010;10(12): 1089-1097.[50] Watanabe T, Sakai D, Yamamoto Y, et al. Human nucleus pulposus cells significantly enhanced biological properties in a coculture system with direct cell-to-cell contact with autologous mesenchymal stem cells. J Orthop Res. 2010; 28(5): 623-630.[51] Miyamoto T, Muneta T, Tabuchi T, et al. Intradiscal transplantation of synovial mesenchymal stem cells prevents intervertebral disc degeneration through suppression of matrix metalloproteinase-related genes in nucleus pulposus cells in rabbits. Arthritis Res Ther. 2010;12(6): R206.[52] Crevensten G, Walsh AJ, Ananthakrishnan D, et al. Intervertebral disc cell therapy for regeneration: mesenchymal stem cell implantation in rat intervertebral discs. Ann Biomed Eng. 2004;32(3): 430-434.[53] Pei M, Shoukry M, Li J, et al. Modulation of in vitro microenvironment facilitates synovium-derived stem cell-based nucleus pulposus tissue regeneration. Spine (Phila Pa 1976). 2012; 37(18): 1538-1547.[54] Longo UG, Papapietro N, Petrillo S, et al. Mesenchymal stem cell for prevention and management of intervertebral disc degeneration. Stem Cells Int. 2012;2012: 921053.[55] Liang CZ, Li H, Tao YQ, et al. Dual release of dexamethasone and TGF-beta3 from polymeric microspheres for stem cell matrix accumulation in a rat disc degeneration model. Acta Biomater. 2013;9(12): 9423-9433.[56] Murrell W, Sanford E, Anderberg L, et al. Olfactory stem cells can be induced to express chondrogenic phenotype in a rat intervertebral disc injury model. Spine J. 2009;9(7): 585-594.[57] Yim RL, Lee JT, Bow CH, et al. A systematic review of the safety and efficacy of mesenchymal stem cells for disc degeneration: insights and future directions for regenerative therapeutics. Stem Cells Dev. 2014;23(21): 2553-2567.[58] Minogue BM, Richardson SM, Zeef LA, et al. Characterization of the human nucleus pulposus cell phenotype and evaluation of novel marker gene expression to define adult stem cell differentiation. Arthritis Rheum. 2010;62(12): 3695-3705.[59] Jeong JH, Lee JH, Jin ES, et al. Regeneration of intervertebral discs in a rat disc degeneration model by implanted adipose-tissue-derived stromal cells. Acta Neurochir (Wien). 2010;152(10): 1771-1777.[60] Orozco L, Soler R, Morera C, et al. Intervertebral disc repair by autologous mesenchymal bone marrow cells: a pilot study. Transplantation. 2011;92(7): 822-828.[61] Vadala G, Sowa G, Hubert M, et al. Mesenchymal stem cells injection in degenerated intervertebral disc: cell leakage may induce osteophyte formation. J Tissue Eng Regen Med. 2012; 6(5): 348-355.[62] Sakai D, Mochida J, Yamamoto Y, et al. Transplantation of mesenchymal stem cells embedded in Atelocollagen gel to the intervertebral disc: a potential therapeutic model for disc degeneration. Biomaterials. 2003;24(20):3531-3541.[63] Yang H, Wu J, Liu J, et al. Transplanted mesenchymal stem cells with pure fibrinous gelatin-transforming growth factor-beta1 decrease rabbit intervertebral disc degeneration. Spine J. 2010;10(9): 802-810.[64] Leckie SK, Sowa GA, Bechara BP, et al. Injection of human umbilical tissue-derived cells into the nucleus pulposus alters the course of intervertebral disc degeneration in vivo. Spine J. 2013; 13(3): 263-272.[65] Subhan RA, Puvanan K, Murali MR, et al. Fluoroscopy assisted minimally invasive transplantation of allogenic mesenchymal stromal cells embedded in HyStem reduces the progression of nucleus pulposus degeneration in the damaged ntervertebral [corrected] disc: a preliminary study in rabbits. Scientific World J. 2014;2014: 818502.[66] Henriksson HB, Hagman M, Horn M, et al. Investigation of different cell types and gel carriers for cell-based intervertebral disc therapy, in vitro and in vivo studies. J Tissue Eng Regen Med. 2012;6(9): 738-747.[67] Ruan D, Xin H, Zhang C, et al. Experimental intervertebral disc regeneration with tissue-engineered composite in a canine model. Tissue Eng Part A. 2010;16(7): 2381-2389.[68] Yoshikawa T, Ueda Y, Miyazaki K, et al. Disc regeneration therapy using marrow mesenchymal cell transplantation: a report of two case studies. Spine (Phila Pa 1976). 2010; 35(11): E475-480.[69] Meisel HJ, Ganey T, Hutton WC, et al. Clinical experience in cell-based therapeutics: intervention and outcome. Eur Spine J. 2006;15 Suppl 3: S397-405.[70] Meisel HJ, Siodla V, Ganey T, et al. Clinical experience in cell-based therapeutics: disc chondrocyte transplantation A treatment for degenerated or damaged intervertebral disc. Biomol Eng. 2007;24(1): 5-21.[71] Coric D, Pettine K, Sumich A, et al. Prospective study of disc repair with allogeneic chondrocytes presented at the 2012 Joint Spine Section Meeting. J Neurosurg Spine. 2013;18(1): 85-95.[72] Brodin H. Paths of nutrition in articular cartilage and intervertebral discs. Acta Orthop Scand. 1955;24(3): 177-183.[73] Grunhagen T, Shirazi-Adl A, Fairbank JC, et al. Intervertebral disk nutrition: a review of factors influencing concentrations of nutrients and metabolites. Orthop Clin North Am. 2011;42(4): 465-477, vii.[74] Holm S, Maroudas A, Urban JP, et al. Nutrition of the intervertebral disc: solute transport and metabolism. Connect Tissue Res. 1981;8(2): 101-119.[75] Risbud MV, Shapiro IM. Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol. 2014; 10(1): 44-56.[76] Krock E, Rosenzweig DH, Haglund L. The Inflammatory Milieu of the Degenerate Disc: Is Mesenchymal Stem Cell-based Therapy for Intervertebral Disc Repair a Feasible Approach? Curr Stem Cell Res Ther. 2015;10(4): 317-328.[77] Wuertz K, Haglund L. Inflammatory mediators in intervertebral disk degeneration and discogenic pain. Global Spine J. 2013;3(3): 175-184.[78] Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411): 143-147.[79] Steck E, Bertram H, Abel R, et al. Induction of intervertebral disc-like cells from adult mesenchymal stem cells. Stem Cells. 2005;23(3): 403-411.[80] Risbud MV, Guttapalli A, Albert TJ, et al. Hypoxia activates MAPK activity in rat nucleus pulposus cells: regulation of integrin expression and cell survival. Spine (Phila Pa 1976). 2005; 30(22): 2503-2509.[81] Stoyanov JV, Gantenbein-Ritter B, Bertolo A, et al. Role of hypoxia and growth and differentiation factor-5 on differentiation of human mesenchymal stem cells towards intervertebral nucleus pulposus-like cells. Eur Cell Mater. 2011;21: 533-547.[82] Liang C, Li H, Tao Y, et al. Responses of human adipose-derived mesenchymal stem cells to chemical microenvironment of the intervertebral disc. J Transl Med. 2012;10: 49.[83] Wuertz K, Godburn K, Neidlinger-Wilke C, et al. Behavior of mesenchymal stem cells in the chemical microenvironment of the intervertebral disc. Spine (Phila Pa 1976). 2008;33(17): 1843-1849.[84] Orth P, Rey-Rico A, Venkatesan JK, et al. Current perspectives in stem cell research for knee cartilage repair. Stem Cells Cloning. 2014; 7: 1-17.[85] Matta C, Mobasheri A, Gergely P, et al. Ser/Thr-phosphoprotein phosphatases in chondrogenesis: neglected components of a two-player game. Cell Signal. 2014;26(10): 2175-2185.[86] Mennan C, Wright K, Bhattacharjee A, et al. Isolation and characterisation of mesenchymal stem cells from different regions of the human umbilical cord. Biomed Res Int. 2013; 2013: 916136.[87] Liu S, Hou KD, Yuan M, et al. Characteristics of mesenchymal stem cells derived from Wharton's jelly of human umbilical cord and for fabrication of non-scaffold tissue-engineered cartilage. J Biosci Bioeng. 2014;117(2): 229-235.[88] Patel AA, Spiker WR, Daubs M, et al. Evidence for an inherited predisposition to lumbar disc disease. J Bone Joint Surg Am. 2011;93(3): 225-229.[89] Fong CY, Chak LL, Biswas A, et al. Human Wharton's jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Rev. 2011;7(1):1-16.[90] Harkness EF, Macfarlane GJ, Silman AJ, et al. Is musculoskeletal pain more common now than 40 years ago?: Two population-based cross-sectional studies. Rheumatology (Oxford). 2005;44(7):890-895.[91] La Rocca G, Lo Iacono M, Corsello T, et al. Human Wharton's jelly mesenchymal stem cells maintain the expression of key immunomodulatory molecules when subjected to osteogenic, adipogenic and chondrogenic differentiation in vitro: new perspectives for cellular therapy. Curr Stem Cell Res Ther. 2013;8(1):100-113.[92] Cornejo MC, Cho SK, Giannarelli C, et al. Soluble factors from the notochordal-rich intervertebral disc inhibit endothelial cell invasion and vessel formation in the presence and absence of pro-inflammatory cytokines. Osteoarthritis Cartilage. 2015;23(3):487-496.[93] de Vries SA, Potier E, van Doeselaar M, et al. Conditioned medium derived from notochordal cell-rich nucleus pulposus tissue stimulates matrix production by canine nucleus pulposus cells and bone marrow-derived stromal cells. Tissue Eng Part A. 2015;21(5-6): 1077-1084.[94] Hiyama A, Mochida J, Sakai D. Stem cell applications in intervertebral disc repair. Cell Mol Biol (Noisy-le-grand). 2008; 54(1): 24-32.[95] Paesold G, Nerlich AG, Boos N. Biological treatment strategies for disc degeneration: potentials and shortcomings. Eur Spine J. 2007;16(4): 447-468.[96] Purmessur D, Schek RM, Abbott RD, et al. Notochordal conditioned media from tissue increases proteoglycan accumulation and promotes a healthy nucleus pulposus phenotype in human mesenchymal stem cells. Arthritis Res Ther. 2011; 13(3): p. R81.[97] Stemper BD, Baisden JL, Yoganandan N, et al. Mechanical yield of the lumbar annulus: a possible contributor to instability: Laboratory investigation. J Neurosurg Spine. 2014; 21(4): 608-613.[98] Strassburg S, Richardson SM, Freemont AJ, et al. Co-culture induces mesenchymal stem cell differentiation and modulation of the degenerate human nucleus pulposus cell phenotype. Regen Med. 2010; 5(5): 701-711.[99] Yuan M, Yeung CW, Li YY, et al. Effects of nucleus pulposus cell-derived acellular matrix on the differentiation of mesenchymal stem cells. Biomaterials. 2013;34(16): 3948-3961.[100] Le Visage C, Kim SW, Tateno K, et al. Interaction of human mesenchymal stem cells with disc cells: changes in extracellular matrix biosynthesis. Spine (Phila Pa 1976). 2006;31(18): 2036-2042.[101] Sivakamasundari V, Lufkin T. Stemming the Degeneration: IVD Stem Cells and Stem Cell Regenerative Therapy for Degenerative Disc Disease. Adv Stem Cells. 2013;2013. pii: 724547.[102] Benneker LM, Andersson G, Iatridis JC, et al. Cell therapy for intervertebral disc repair: advancing cell therapy from bench to clinics. Eur Cell Mater. 2014;27: 5-11.[103] Sethe S, Scutt A, Stolzing A. Aging of mesenchymal stem cells. Ageing Res Rev. 2006; 5(1): 91-116.[104] McLain RF, Fleming JE, Boehm CA, et al. Aspiration of osteoprogenitor cells for augmenting spinal fusion: comparison of progenitor cell concentrations from the vertebral body and iliac crest. J Bone Joint Surg Am. 2005; 87(12): 2655-2661.[105] Gorensek M, Jaksimovic C, Kregar-Velikonja N, et al. Nucleus pulposus repair with cultured autologous elastic cartilage derived chondrocytes. Cell Mol Biol Lett. 2004;9(2): 363-373.[106] Liu Y, Rahaman MN, Bal BS. Modulating notochordal differentiation of human induced pluripotent stem cells using natural nucleus pulposus tissue matrix. PLoS One. 2014;9(7): e100885.[107] Sheikh H, Zakharian K, De La Torre RP, et al. In vivo intervertebral disc regeneration using stem cell-derived chondroprogenitors. J Neurosurg Spine. 2009;10(3): 265-272.[108] Malik KM, Cohen SP, Walega DR, et al. Diagnostic criteria and treatment of discogenic pain: a systematic review of recent clinical literature. Spine J. 2013;13(11): 1675-1689. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [3] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [4] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [5] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [6] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [7] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [8] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1012-1019. |
| [9] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [10] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [11] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1032-1037. |
| [12] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [13] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [14] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1051-1056. |
| [15] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||