Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (21): 3388-3393.doi: 10.3969/j.issn.2095-4344.2017.21.018
Previous Articles Next Articles
Isolation and cultivation of human placental chorionic-derived mesenchymal stem cells: optimization of the tissue explants method
Jin Yu-lin, Wu Jie-ying, Lu Yan, Chen Jin-song, Li Fa-tao, Tang Jie, Liu Dong, Liang Qi-hua, Li Yan, Tang Xue-wei, Xie Gui-e, Wu Shao-qing
- Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou 510623, Guangdong Province, China
-
Revised:2017-06-12Online:2017-07-28Published:2017-08-02 -
Contact:Wu Shao-qing, Master, Senior technologist, Master’s supervisor, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou 510623, Guangdong Province, China -
About author:Jin Yu-lin, Master, Associate chief physician, Guangzhou Women and Children’s Medical Center, Guangzhou Medical University, Guangzhou 510623, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81102248; the Medical Research Foundation of Guangdong Province, No. B2010274; the Science and Technology Project of Guangdong Province, No. 2014A020212027; the grant from Guangzhou Institute of Pediatrics/Guangzhou Women and Children’s Medical Center, No. YIP-2016-014
CLC Number:
Cite this article
Jin Yu-lin, Wu Jie-ying, Lu Yan, Chen Jin-song, Li Fa-tao, Tang Jie, Liu Dong, Liang Qi-hua, Li Yan, Tang Xue-wei, Xie Gui-e, Wu Shao-qing. Isolation and cultivation of human placental chorionic-derived mesenchymal stem cells: optimization of the tissue explants method[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(21): 3388-3393.
share this article
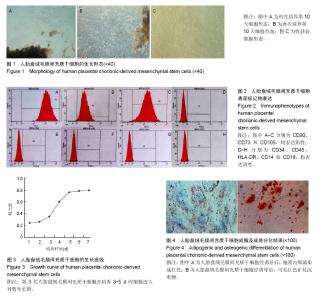
2.1 原代人胎盘绒毛膜间充质干细胞细胞形态、获得时间和细胞得率 镜下观察人胎盘绒毛膜间充质干细胞贴壁生长。初次培养的细胞,1周后,就能够看到极少数成纤维样细胞从组织块上爬出来,在第10天能够看到细胞呈旋涡状生长(图1A),形态均一,获得细胞的平均时间为(17.73± 1.14) d,每个Φ100 mm培养皿获得原代细胞为(6.97± 0.98)×105个。再次接种培养的细胞,在第10天能够看到组织块上爬出的细胞较多(图1B),获得细胞的平均时间为(10.03±1.30) d,每个培养皿获得原代细胞为(13.82± 1.44)×105个。结果表明,2种接种情况下,接种培养的细胞形态无明显差异,获得时间上,再次接种培养的细胞收获时间快,差异有显著性意义(P=0.000 < 0.05);获得细胞数量上,再次接种培养获得细胞约初次培养的2倍,差异有显著性意义(P=0.000 < 0.05)。达到90%融合度的细胞形态为长梭形成纤维细胞状,呈平行或漩涡状生长(图1C)。 2.2 人胎盘绒毛膜间充质干细胞免疫表型分析 流式细胞术检测第3代人胎盘绒毛膜间充质干细胞,表面标记抗体CD90、CD105、CD73的表达率分别(98.54±0.21)%,(99.62±0.12)%,100%,均高于95%,而CD34、CD45、CD14、CD19、HLA-DR检测为阴性,表达率分别为(0.82± 0.25)%,(1.22±0.27)%,(1.15±0.14)%,(0.11±0.09)%,(1.21±0.31)%,均低于2%,见图2。 2.3 人胎盘绒毛膜间充质干细胞增殖能力分析 CCK-8法绘制第3代人胎盘绒毛膜间充质干细胞的生长曲线,1-2 d间充质干细胞增殖不明显;3-5 d细胞进入对数生长期,细胞增殖加速;6-7 d细胞进入平台期,生长缓慢,见图3。 2.4 人胎盘绒毛膜间充质干细胞诱导分化潜能分析 第3代人胎盘绒毛膜间充质干细胞经成骨、成脂分化培养基诱导4周后,油红O染色可见胞浆内脂滴染成红色(图4A),而茜素红染色,可见红色矿化沉着物(图4B)。说明人胎盘绒毛膜间充质干细胞在体外特定环境下具有向成骨细胞或脂肪细胞分化的潜能。"

| [1] Trounson A, McDonald C. Stem Cell Therapies in Clinical Trials: Progress and Challenges. Cell Stem Cell. 2015;17(1): 11-22. [2] 赖平,陈懿建,罗耀玲,等.人胎盘源间充质干细胞的分离?培养及生物学鉴定[J].赣南医学院学报,2016,36(2):183-186.[3] 韩潇,白海,赵强,等.人脐带间充质干细胞体外分离培养及其生物学特性的研究[J].重庆医学,2016,45(7):876-879.[4] Jung N, Park S, Choi Y, et al. Tonsil-Derived Mesenchymal Stem Cells Differentiate into a Schwann Cell Phenotype and Promote Peripheral Nerve Regeneration. Int J Mol Sci. 2016; 17(11). pii: E1867.[5] Park JR, Kim E, Yang J, et al. Isolation of human dermis derived mesenchymal stem cells using explants culture method: expansion and phenotypical characterization. Cell Tissue Bank. 2015;16(2):209-218. [6] Patel J, Shafiee A, Wang W, et al. Novel isolation strategy to deliver pure fetal-origin and maternal-origin mesenchymal stem cell (MSC) populations from human term placenta. Placenta. 2014;35(11):969-971.[7] Zhang X, Huang F, Chen Y, et al. Progress and prospect of mesenchymal stem cell-based therapy in atherosclerosis. Am J Transl Res. 2016;8(10):4017-4024. [8] 余永春,刘小峰,高平,等.胎盘间充质干细胞牙向分化的体外研究[J]. 口腔医学,2016,36(5):394-398.[9] 杜莉莉,金玉楠,李昆,等.胎盘来源的间充质干细胞体外分离培养及向软骨细胞分化的研究[J].解剖科学进展,2008,14(1):83-86.[10] Pop DM, Sori??u O, ?u?man S, et al. Potential of placental- derived human mesenchymal stem cells for osteogenesis and neurogenesis. Rom J Morphol Embryol. 2015;56(3):989-996.[11] Nitkin CR, Bonfield TL2. Concise Review: Mesenchymal Stem Cell Therapy for Pediatric Disease: Perspectives on Success and Potential Improvements. Stem Cells Transl Med. 2016. pii: sctm.2015-0427. [12] 刘芳,杨辉,汪兆艳,等.自体骨髓间充质干细胞移植治疗唐氏综合征临床观察[J].转化医学杂志,2016,5(5):288-291.[13] Matigian N, Brooke G, Zaibak F, et al. Multipotent human stromal cells isolated from cord blood, term placenta and adult bone marrow show distinct differences in gene expression pattern. Genom Data. 20143:70-74. [14] Li J, Wei Y, Yan L, et al. Multiplacenta derived stem cell/cytokine treatment increases survival time in a mouse model with radiation-induced bone marrow damage. Cytotechnology. 2016;68(6):2677-2686. [15] 高舒平,曲春辉,周瑞,等.干细胞与转化医学研究进展浅析[J].中国实验动物学报,2016,24(4):439-442.[16] 陆琰,吴洁莹,陈劲松,等.胎盘间充质干细胞脐血血浆培养体系的建立[J].广东医学,2015,36(15):2312-2315.[17] Park S, Park HR, Lee WD, et al. Establishment of a xeno-free culture system that preserves the characteristics of placenta mesenchymal stem cells. Cytotechnology. 2015;67(5):851- 860. [18] Yong KW, Choi JR, Wan Safwani WK. Biobanking of Human Mesenchymal Stem Cells: Future Strategy to Facilitate Clinical Applications. Adv Exp Med Biol. 2016;951:99-110.[19] Du WJ, Chi Y, Yang ZX, et al. Heterogeneity of proangiogenic features in mesenchymal stem cells derived from bone marrow, adipose tissue, umbilical cord, and placenta. Stem Cell Res Ther. 2016;7(1):163.[20] Heyes R, Iarocci A, Tchoukalova Y, et al. Immunomodulatory Role of Mesenchymal Stem Cell Therapy in Vascularized Composite Allotransplantation. J Transplant. 2016;2016: 6951693. [21] Saparov A, Ogay V, Nurgozhin T, et al. Preconditioning of Human Mesenchymal Stem Cells to Enhance Their Regulation of the Immune Response. Stem Cells Int. 2016; 2016:3924858. [22] Murphy N, Lynch K, Lohan P, et al. Mesenchymal stem cell therapy to promote corneal allograft survival: current status and pathway to clinical translation. Curr Opin Organ Transplant. 2016;21(6):559-567.[23] Meng YH, Zhu XH, Yan LY, et al. Bone mesenchymal stem cells improve pregnancy outcome by inducing maternal tolerance to the allogeneic fetus in abortion-prone matings in mouse. Placenta. 2016;47:29-36. [24] 韩之波,杨舟鑫,池颖,等.人脐带、胎盘绒毛膜来源间充质干细胞的生物学特性比较研究[J].中国实验血液学杂志,2012,20(3): 692-696.[25] Katsiani E, Garas A, Skentou C, et al. Chorionic villi derived mesenchymal like stem cells and expression of embryonic stem cells markers during long-term culturing. Cell Tissue Bank. 2016;17(3):517-529. [26] Qin SQ, Kusuma GD, Al-Sowayan B, et al. Establishment and characterization of fetal and maternal mesenchymal stem/stromal cell lines from the human term placenta. Placenta. 2016;39:134-146. [27] Ziadlou R, Shahhoseini M, Safari F, et al. Comparative analysis of neural differentiation potential in human mesenchymal stem cells derived from chorion and adult bone marrow. Cell Tissue Res. 2015;362(2):367-377. [28] Kil K, Choi MY, Kong JS, et al. Regenerative efficacy of mesenchymal stromal cells from human placenta in sensorineural hearing loss. Int J Pediatr Otorhinolaryngol. 2016;91:72-81. [29] Liu H, Murthi P, Qin S, et al. A novel combination of homeobox genes is expressed in mesenchymal chorionic stem/stromal cells in first trimester and term pregnancies. Reprod Sci. 2014;21(11):1382-1394. [30] Klein C, Strobel J, Zingsem J, et al. Ex vivo expansion of hematopoietic stem- and progenitor cells from cord blood in coculture with mesenchymal stroma cells from amnion, chorion, Wharton's jelly, amniotic fluid, cord blood, and bone marrow. Tissue Eng Part A. 2013;19(23-24):2577-2585.[31] Yamahara K, Harada K, Ohshima M, et al. Comparison of angiogenic, cytoprotective, and immunosuppressive properties of human amnion- and chorion-derived mesenchymal stem cells. PLoS One. 2014;9(2):e88319.[32] Yan ZJ, Hu YQ, Zhang HT, Comparison of the neural differentiation potential of human mesenchymal stem cells from amniotic fluid and adult bone marrow. Cell Mol Neurobiol. 2013;33(4):465-475. [33] Bana N, Sanooghi D, Soleimani M, et al. A Comparative Study to Evaluate Myogenic Differentiation Potential of Human Chorion versus Umbilical Cord Blood-derived Mesenchymal Stem Cells. Tissue Cell. 2017. in press. [34] Faghihi F, Mirzaei E, Ai J, et al. Erratum to: Differentiation Potential of Human Chorion-Derived Mesenchymal Stem Cells into Motor Neuron-Like Cells in Two- and Three-Dimensional Culture Systems. Mol Neurobiol. 2016; 53(3):1873.[35] Manochantr S, Marupanthorn K, Tantrawatpan C, et al. The expression of neurogenic markers after neuronal induction of chorion-derived mesenchymal stromal cells. Neurol Res. 2015;37(6):545-552.[36] Faghihi F, Mirzaei E, Ai J, et al. Differentiation Potential of Human Chorion-Derived Mesenchymal Stem Cells into Motor Neuron-Like Cells in Two- and Three-Dimensional Culture Systems. Mol Neurobiol. 2016;53(3):1862-1872.[37] Brown PT, Squire MW, Li WJ. Characterization and evaluation of mesenchymal stem cells derived from human embryonic stem cells and bone marrow. Cell Tissue Res. 2014;358(1): 149-164. [38] Pop DM, Sori??u O, ?u?man S, et al. Potential of placental-derived human mesenchymal stem cells for osteogenesis and neurogenesis. Rom J Morphol Embryol. 2015;56(3):989-996. [39] Di Bernardo J, Maiden MM, Jiang G, et al. Paracrine regulation of fetal lung morphogenesis using human placenta-derived mesenchymal stromal cells. J Surg Res. 2014;190(1):255-263.[40] Oliveira MS, Barreto-Filho JB. Placental-derived stem cells: Culture, differentiation and challenges. World J Stem Cells. 2015;7(4):769-775.[41] 杜莉莉,金玉楠,李昆,等.胎盘来源的间充质干细胞体外分离培养及向软骨细胞分化的研究[J].解剖科学进展,2008,14(1):83-86.[42] Zhong ZN, Zhu SF, Yuan AD, et al. Potential of placenta-derived mesenchymal stem cells as seed cells for bone tissue engineering: preliminary study of osteoblastic differentiation and immunogenicity. Orthopedics. 2012;35(9): 779-788. [43] Bellavia M, Altomare R, Cacciabaudo F, et al. Towards an ideal source of mesenchymal stem cell isolation for possible therapeutic application in regenerative medicine. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158(3): 356-360. [44] Zhu Y, Song X, Han F, et al. Alteration of histone acetylation pattern during long-term serum-free culture conditions of human fetal placental mesenchymal stem cells. PLoS One. 2015;10(2):e0117068. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [3] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [4] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [5] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [6] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [7] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [8] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [9] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [10] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [11] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [12] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [13] | Huang Chuanjun, Zou Yu, Zhou Xiaoting, Zhu Yangqing, Qian Wei, Zhang Wei, Liu Xing. Transplantation of umbilical cord mesenchymal stem cells encapsulated in RADA16-BDNF hydrogel promotes neurological recovery in an intracerebral hemorrhage rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 510-515. |
| [14] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [15] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||