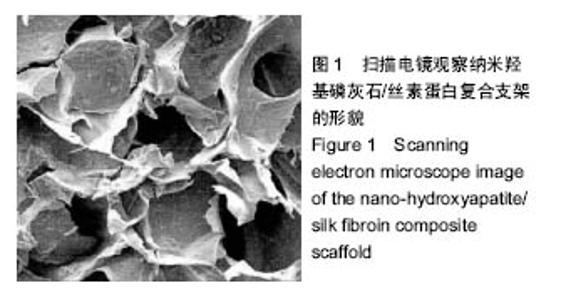
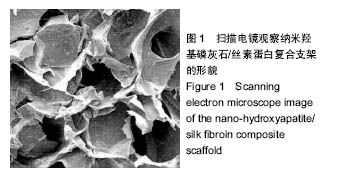
Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (18): 2802-2810.doi: 10.3969/j.issn.2095-4344.2017.18.003
Previous Articles Next Articles
Nano-hydroxyapatite/silk fibroin composite materials loaded with recombinant human bone morphogenetic protein 2 used for spinal fusion
- Shuqing Medical College, Zhengzhou 450064, Henan Province, China
-
Received:2017-02-11Online:2017-06-28Published:2017-07-07 -
About author:Chen Xiao-min, Master, Associate professor, Shuqing Medical College, Zhengzhou 450064, Henan Province, China
CLC Number:
Cite this article
Chen Xiao-min, Ma Xiao, Shao Nan-qi, Dai Yong-xia, Gao Qing, Fu Jin-fang . Nano-hydroxyapatite/silk fibroin composite materials loaded with recombinant human bone morphogenetic protein 2 used for spinal fusion[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(18): 2802-2810.
share this article
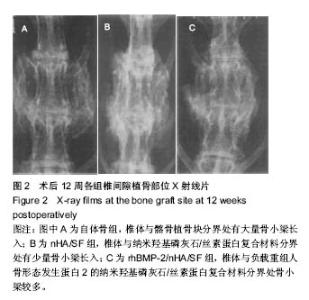
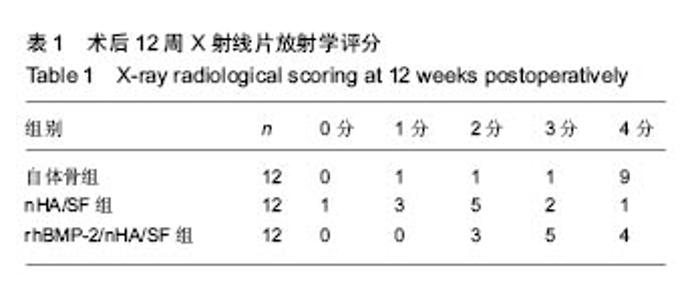
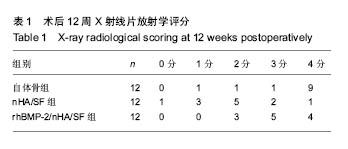
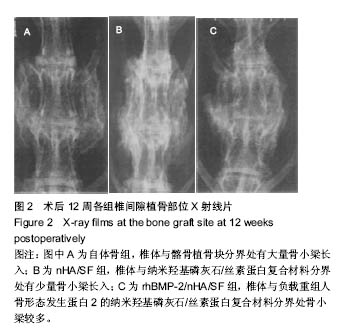
2.2 实验动物一般情况及标本大体观察结果 术后所有实验动物一般情况良好,未出现腹泻、伤口感染、移植物排斥等明显不良反应。自体骨组1只动物出现功能障碍,均于1周内恢复。nHA/SF组和rhBMP-2/nHA/SF组各有1只切口出现清水样渗出物,经处理后自愈。所有实验动物无一只死亡。术后12周处死实验动物后重点观察各组标本融合部位,质地均较硬,其中自体骨组大量骨痂形成,可见骨连接,融合迹象明显。nHA/SF组可见骨痂,但骨性连接不坚固,未见融合迹象。rhBMP-2/nHA/SF组材料与周围肌肉组织结合比较紧密,部分骨痂形成,已有骨连接。 2.3 放射学检查结果 见图2和表1。自体骨组:椎体与髂骨植骨块分界处有大量骨小梁长入,密度高,X射线评估有10只符合融合标准。nHA/SF组:双侧L4/5椎体与纳米羟基磷灰石/丝素蛋白复合材料分界处有少量骨小梁长入,X射线评估有3只符合融合标准。rhBMP-2/nHA/SF组:骨小梁较多,X射线评估有9只符合融合标准。"
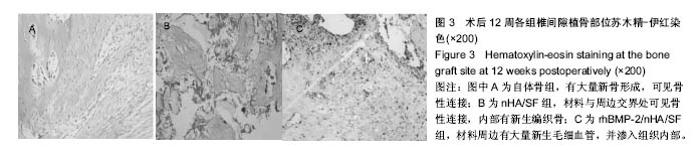
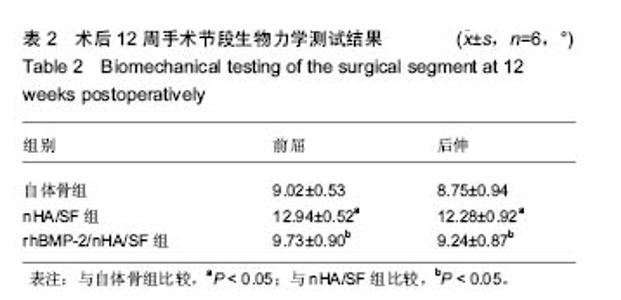
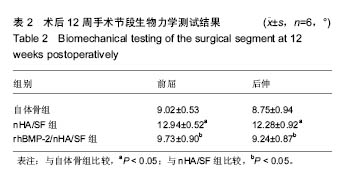
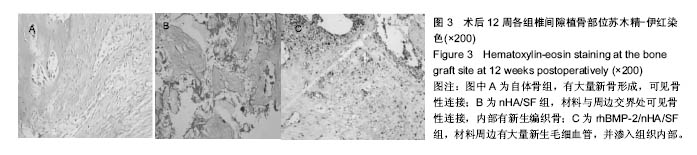
2.5 组织学观察及半定量分析结果 见图3。自体骨组:髂骨周边可见大量新骨形成,可见骨性连接。nHA/SF组:材料小部分降解,与周边交界处可见骨性连接,内部有新生编织骨,未见成熟骨组织。rhBMP-2/nHA/SF组:材料大部分降解,周边有大量新生毛细血管,并渗入组织内部,交界处未见纤维囊。经半定量分析后发现,自体骨组的新骨形成面积占(0.52±0.03)%,nHA/SF组的新骨形成面积占(0.41±0.02)%,rhBMP-2/nHA/SF组的新骨形成面积占(0.51±0.01)%。nHA/SF组比自体骨组低、rhBMP-2/ nHA/SF组比nHA/SF组高,差异均有显著性意义(P < 0.05)。"
| [1]Barz T, Melloh M, Lord SJ, et al. A conceptual model of compensation/decompensation in lumbar segmental instability. Med Hypotheses. 2014;83(3):312-316.[2]Souied EH, Cohen SY, de Pouvourville G, et al. Treatment of wet age-related macular degeneration by ranibizumab in "real life" in France: treatment behaviours and associated visual outcome. Acta Ophthalmol. 2015;93(2):e179-180.[3]Travaille KL, Salinas-De-León P, Bell JJ. Indication of visitor trampling impacts on intertidal seagrass beds in a New Zealand marine reserve. Ocean & Coastal Management. 2015; 114(Pt 3):145-150.[4]闫伟,杨莉,凌梅,等.自体骨髓富集间质干细胞与羟基磷灰石磷酸三钙材料复合在脊柱融合中的应用[J].中国组织工程研究, 2016, 20(8):1075-1080.[5]郑扬,李危石,刘忠军. 骨组织3D 打印:骨再生的未来[J]. 北京大学学报:医学版, 2015, 47(2):203-206.[6]You R, Li X, Liu Y, et al. Response of filopodia and lamellipodia to surface topography on micropatterned silk fibroin films. J Biomed Mater Res A. 2014;102(12):4206-4212.[7]年争好,李晖,李瑞欣,等. 纳米羟基磷灰石/胶原蛋白/丝素蛋白复合骨组织工程支架材料的生物相容性[J]. 中国组织工程研究, 2015,19(8):1149-1154.[8]光善仪,柯福佑,沈玉华. 非织造多孔丝素蛋白/纳米羟基磷灰石复合支架材料的制备和表征[J].功能材料, 2015,46(22): 22149-22152.[9]Kim RY, Oh JH, Lee BS, et al. The effect of dose on rhBMP-2 signaling, delivered via collagen sponge, on osteoclast activation and in vivo bone resorption. Biomaterials. 2014; 35(6):1869-1881.[10]孟红亚,张长春,朱坤,等. rhBMP-2在脊柱融合术中应用安全性研究进展[J]. 国际骨科学杂志, 2015,36(1):26-29.[11]Zhou P, Xia Y, Cheng X, et al. Enhanced bone tissue regeneration by antibacterial and osteoinductive silica-HACC-zein composite scaffolds loaded with rhBMP-2. Biomaterials. 2014;35(38):10033-10045.[12]Byvaltsev VA, Kalinin AA, Belykh EG, et al. Optimization of segmental lumbar spine instability treatment using minimally invasive spinal fusion technique. Zh Vopr Neirokhir Im N N Burdenko. 2015;79(3):45-54.[13]Ploumis A, Christodoulou P, Kapoutsis D, et al. Surgical treatment of lumbar spinal stenosis with microdecompression and interspinous distraction device insertion. A case series. J Orthop Surg Res. 2012;7:35.[14]费琦,赵凡,杨雍,等.腰椎后路融合手术对失稳模型节段稳定性及相邻节段力学的影响[J].中华医学杂志, 2015,95(45):3681- 3686.[15]牛伟民,于德水,李名武,等.椎体间纤维性融合重建兔脊柱稳定性的初步探索[J].中国脊柱脊髓杂志,2012,22(12):1108-1112.[16]Takabatake K, Yamachika E, Tsujigiwa H, et al. Effect of geometry and microstructure of honeycomb TCP scaffolds on bone regeneration. J Biomed Mater Res A. 2014;102(9): 2952-2960.[17]Kaur G, Pandey OP, Singh K, et al. A review of bioactive glasses: Their structure, properties, fabrication and apatite formation. J Biomed Mater Res A. 2014;102(1):254-274.[18]Tomizawa Y. Late spontaneous nonanastomotic transgraft hemorrhage from biological material-impregnated fabric vascular graft may be due to autologous tissue detachment: a clinical hypothesis. Artif Organs. 2014;38(12):1058-1060.[19]Lai GJ, Shalumon KT, Chen SH, et al. Composite chitosan/silk fibroin nanofibers for modulation of osteogenic differentiation and proliferation of human mesenchymal stem cells. Carbohydr Polym. 2014;111:288-297.[20]Larsen RW, Wojtas L, Whittington C. Biomimetic Light Harvesting in Nanoporous Metal Organic Materials. Biophysical Journal. 2014;106(2):421.[21]Sharma MK, Khursheed A, Kazmi AA. Modified septic tank-anaerobic filter unit as a two-stage onsite domestic wastewater treatment system. Environmental Technology. 2014;35(17):2183-2193.[22]Pa?cu EI, Stokes J, McGuinness GB. Electrospun composites of PHBV, silk fibroin and nano-hydroxyapatite for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2013;33(8):4905-4916.[23]Thorfve A, Lindahl C, Xia W, et al. Hydroxyapatite coating affects the Wnt signaling pathway during peri-implant healing in vivo. Acta Biomater. 2014;10(3):1451-1462.[24]Zhi W, Zhang C, Duan K, et al. A novel porous bioceramics scaffold by accumulating hydroxyapatite spherulites for large bone tissue engineering in vivo. II. Construct large volume of bone grafts. J Biomed Mater Res A. 2014;102(8):2491-2501.[25]Vozzi G, Corallo C, Carta S, et al. Collagen-gelatin- genipin-hydroxyapatite composite scaffolds colonized by human primary osteoblasts are suitable for bone tissue engineering applications: in vitro evidences. J Biomed Mater Res A. 2014;102(5):1415-1421.[26]Venkatesan J, Kim SK. Nano-hydroxyapatite composite biomaterials for bone tissue engineering--a review. J Biomed Nanotechnol. 2014;10(10):3124-3140.[27]帅亚俊, 张璨, 邓连霞,等. 矿化柞蚕丝胶膜表面粗糙度的调控及其对骨髓间充质干细胞生长行为的影响[J]. 复合材料学报, 2015, 32(5):1527-1535.[28]Hofmann S, Stok KS, Kohler T, et al. Effect of sterilization on structural and material properties of 3-D silk fibroin scaffolds. Acta Biomater. 2014;10(1):308-317.[29]Lai GJ, Shalumon KT, Chen SH, et al. Composite chitosan/silk fibroin nanofibers for modulation of osteogenic differentiation and proliferation of human mesenchymal stem cells. Carbohydr Polym. 2014;111:288-297.[30]Zhu M, Wang K, Mei J, et al. Fabrication of highly interconnected porous silk fibroin scaffolds for potential use as vascular grafts. Acta Biomater. 2014;10(5):2014-2023.[31]Das S, Pati F, Choi YJ, et al. Bioprintable, cell-laden silk fibroin-gelatin hydrogel supporting multilineage differentiation of stem cells for fabrication of three-dimensional tissue constructs. Acta Biomater. 2015;11:233-246.[32]孙庆治. 纳米羟基磷灰石/丝素蛋白人工骨修复骨缺损[J]. 中国组织工程研究, 2015,19(8):1190-1194. [33]Tayton E, Purcell M, Smith JO, et al. The scale-up of a tissue engineered porous hydroxyapatite polymer composite scaffold for use in bone repair: an ovine femoral condyle defect study. J Biomed Mater Res A. 2015;103(4):1346-1356.[34]Haversath M, Hülsen T, Böge C, et al. Osteogenic differentiation and proliferation of bone marrow-derived mesenchymal stromal cells on PDLLA + BMP-2-coated titanium alloy surfaces. J Biomed Mater Res A. 2016;104(1): 145-154.[35]Kolk A, Tischer T, Koch C, et al. A novel nonviral gene delivery tool of BMP-2 for the reconstitution of critical-size bone defects in rats. J Biomed Mater Res A. 2016;104(10): 2441-2455.[36]Kowalczewski CJ, Saul JM. Surface-mediated delivery of siRNA from fibrin hydrogels for knockdown of the BMP-2 binding antagonist noggin. Acta Biomater. 2015;25:109-120.[37]Rodríguez-Evora M, Reyes R, Alvarez-Lorenzo C, et al. Bone regeneration induced by an in situ gel-forming poloxamine, bone morphogenetic protein-2 system. J Biomed Nanotechnol. 2014;10(6):959-969.[38]Samorezov JE, Headley EB, Everett CR, et al. Sustained presentation of BMP-2 enhances osteogenic differentiation of human adipose-derived stem cells in gelatin hydrogels. J Biomed Mater Res A. 2016;104(6):1387-1397. [39]李练兵,张其清,侯志伟,等. 复合人工骨材料对骨形成影响的实验研究[J]. 北京生物医学工程, 2014,33(3):240-246.[40]Takeoka Y, Hayashi M, Sugiyama N, et al. In situ preparation of poly(l-lactic acid-co-glycolic acid)|[sol]|hydroxyapatite composites as artificial bone materials. Polymer J. 2014; 47(2): 164-170. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [3] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [4] | Yang Jun, Yang Qun, Zhang Rui, Jiang Chang. A novel slidable pedicle screw-rod system for lumbar tuberculosis: promoting bone graft fusion by producing stress stimulation to fused segment [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 914-918. |
| [5] | Li Jian, Bao Zhengqi, Zhou Pinghui, Zhu Ruizhi, Li Zhixiang, Wang Jinzi. Effects of posterior single open-door laminoplasty and anterior cervical corpectomy fusion on cervical sagittal balance parameters in the treatment of multilevel cervical spondylotic myelopathy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 949-953. |
| [6] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [9] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [10] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [11] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [12] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [13] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [14] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [15] | Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2612-2617. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||