Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (14): 2170-2179.doi: 10.3969/j.issn.2095-4344.2017.14.007
Previous Articles Next Articles
Biological behaviors of articular chondrocytes on the polydimethylsiloxane withdifferential surface modifications and stiffnesses
Xiong Zhi-miao, Ye Zhao-yang, Zhou Yan, Tan Wen-song
- State Key Laboratory of Bioreactor Engineering, East China University of Science and Technology, Shanghai 200237, China
-
Received:2017-03-12Online:2017-05-18Published:2017-06-10 -
Contact:Ye Zhao-yang, M.D., Associate professor, State Key Laboratory of Bioreactor Engineering, East China University of Science and Technology, Shanghai 200237, China -
About author:Xiong Zhi-miao, Studying for master’s degree, State Key Laboratory of Bioreactor Engineering, East China University of Science and Technology, Shanghai 200237, China -
Supported by:the Natural Science Foundation of Shanghai, No. 16ZR1408700
CLC Number:
Cite this article
Xiong Zhi-miao, Ye Zhao-yang, Zhou Yan, Tan Wen-song. Biological behaviors of articular chondrocytes on the polydimethylsiloxane withdifferential surface modifications and stiffnesses[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(14): 2170-2179.
share this article
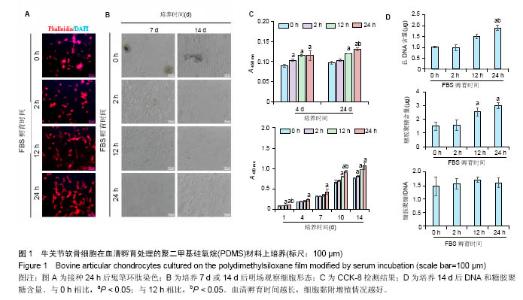
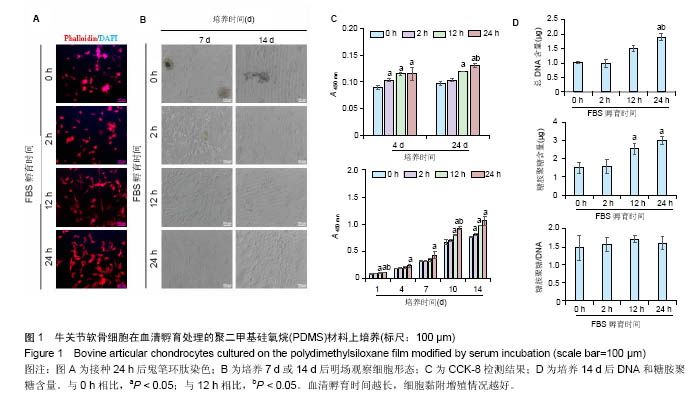
2.1 血清孵育处理PDMS薄膜对软骨细胞培养的影响PDMS薄膜材料(两组份质量比为 A∶B = 10∶1)经血清孵育处理不同时间(0,2,12和24 h)后,将牛关节软骨细胞以4×104/film的浓度接种在薄膜材料上培养以观察细胞在材料上24 h内的黏附情况,或者以2×104/film接种培养以观察其在材料上14 d内的生长情况。其中,血清孵育0 h(未经处理)的PDMS薄膜作为对照。如图1A所示,细胞培养 24 h后,经鬼笔环肽染色细胞骨架F肌动蛋白,可以发现细胞的铺展程度在不同材料表面有明显差别。血清孵育0 h的薄膜表面细胞多数呈圆形,而随血清孵育处理时间延长,细胞铺展程度加强,呈多角形态。图1B是将细胞培养7或 14 d,在显微镜明场下观察的照片。实验发现,血清孵育 0 h的薄膜上出现细胞聚集的现象,而在血清孵育12,24 h的材料上细胞呈单层生长,并且血清孵育处理过的比没有处理过的材料上的细胞数明显较多。通过CCK-8检测,对细胞数进行间接的定量比较,可见在接种4,24 h后,细胞贴附量随血清孵育处理时间的延长而显著增加,而在14 d的培养时间内,延长血清孵育处理时间可以进一步促进了细胞增殖,见图1C。培养14 d后,通过定量检测薄膜上的DNA和糖胺聚糖含量,发现相比于其他材料,血清孵育处理12,24 h的材料上,DNA含量和总糖胺聚糖含量都显著要高,而糖胺聚糖/DNA在各实验组间并没有显著差别,见图1D。"
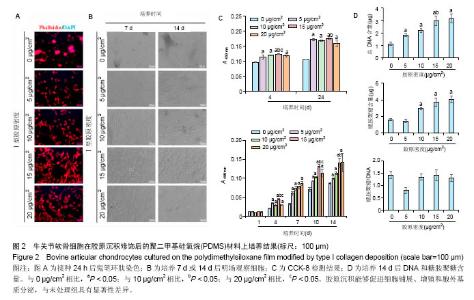
2.2 胶原沉积处理PDMS薄膜对软骨细胞培养的影响PDMS薄膜材料(两组份质量比为 A∶B = 10∶1)经孵育沉积不同密度(0,5,10,15和20 μg/cm2)的胶原处理后,将牛关节软骨细胞以4×104/film的密度接种在薄膜材料上培养以观察细胞在材料上24 h内的黏附情况,或者以2×104/film接种培养以观察其在材料上14 d内的生长情况。其中,以0 μg/cm2的胶原沉积处理的PDMS薄膜作为对照。如图2A所示,细胞培养24 h后,经鬼笔环肽染色细胞骨架,可以观察到在0-20 μg/cm2的范围内,细胞铺展程度随胶原沉积密度增加而加强,见图2B。在培养14 d的过程中,在显微镜明场下观察到在存在胶原沉积的材料表面,细胞能够均匀分布,单层贴壁生长,而且细胞量也随胶原沉积密度的增加有所增加。与此相对的是,在未经胶原沉积的材料上,细胞呈聚集分布,细胞量也显著要少。CCK-8检测进一步定量分析细胞量,也证实了无论是细胞贴附量还是细胞生长,在0-15 μg/cm2范围内都随胶原沉积密度的增加而增加,见图2C。需要指出的是,在胶原沉积密度过高的情况下(20 μg/cm2),出现了细胞黏附和生长相对于低胶原沉积密度并没有增加反而略有下降的趋势,这可能暗示了存在饱和胶原沉积修饰的情况。图2D所示为细胞培养14 d后,对DNA和糖胺聚糖含量的测定。可以看到,随胶原沉积密度的增加,DNA含量逐渐增加,而超过15 μg/cm2之后,DNA含量趋于稳定。糖胺聚糖含量在除了胶原沉积密度为5 μg/cm2时,也随胶原沉积密度增加而增加,而在胶原沉积密度高于15 μg/cm2时不再增加,糖胺聚糖/DNA数值在各组间并无差别。但是,在胶原沉积密度为5 μg/cm2时,总糖胺聚糖含量和糖胺聚糖/DNA数值比未经修饰时均有显著降低的情况。"
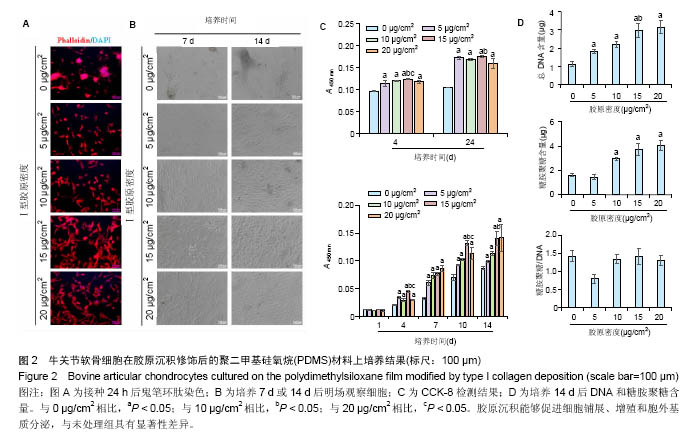
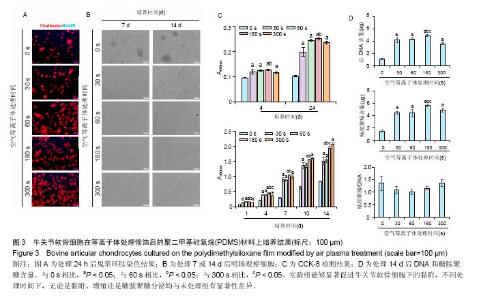
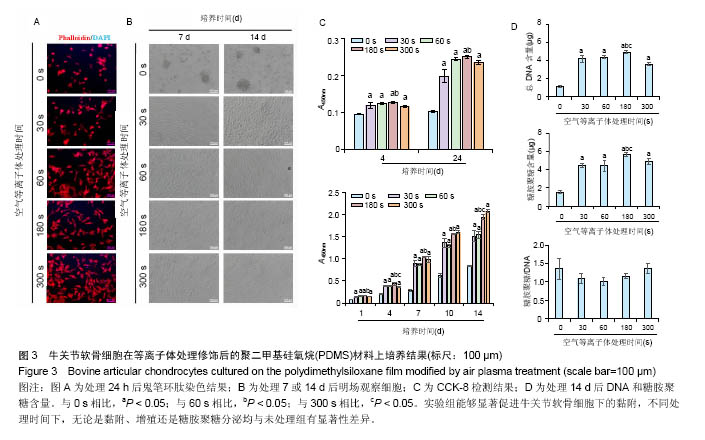
2.3 等离子体处理PDMS薄膜对软骨细胞培养的影响PDMS薄膜材料(两组份质量比为 A∶B = 10∶1)经等离子体处理不同时间(0,30,60,180和300 s)后,将牛关节软骨细胞以4×104/film的密度接种在薄膜材料上培养以观察细胞在材料上24 h内的黏附情况,或者以2×104/film接种培养以观察其在材料上14 d内的生长情况。其中,处理0 s的PDMS薄膜作为对照。如图3A所示为接种24 h后,对细胞骨架的染色。可以看到,随着等离子体处理时间的延长, 牛关节软骨细胞在PDMS薄膜上的铺展面积逐渐增加,细胞也从圆形变成多角形态。在细胞培养14 d内显微镜明场下观察到,未经处理的材料上细胞出现明显的聚集,而等离子体处理的材料上细胞均匀分布,而且随处理时间的增加细胞能更好的生长,但在处理时间达到60 s后,细胞基本呈现相似的形态和密度,见图3B。CCK-8检测对细胞量的分析也证实了此观察。在24 h内,在等离子体处理的材料上,细胞更容易贴附,且处理180 s时效果最好;在14 d内,细胞在修饰过的材料上的生长明显要好,且处理时间越长越有利,见图3C。通过对DNA和糖胺聚糖含量测定,也发现DNA含量在等离子体处理过的薄膜上显著增加,且随处理时间延长进一步提高,但至处理时间为300 s反而有所降低;总糖胺聚糖含量也是在处理过的薄膜上要高,其中在处理180 s的材料上最大,而糖胺聚糖/DNA数值在等离子体处理时间为30、60和180 s的材料上略低于未经处理和处理300 s的材料上的值,见图3D。"
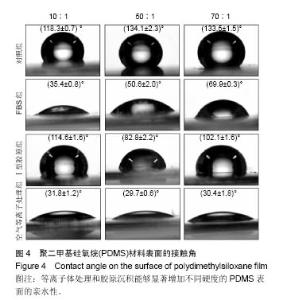
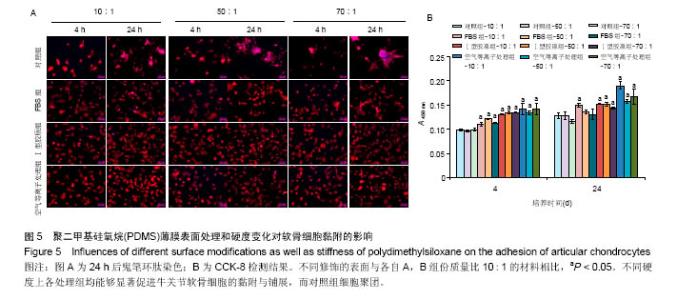
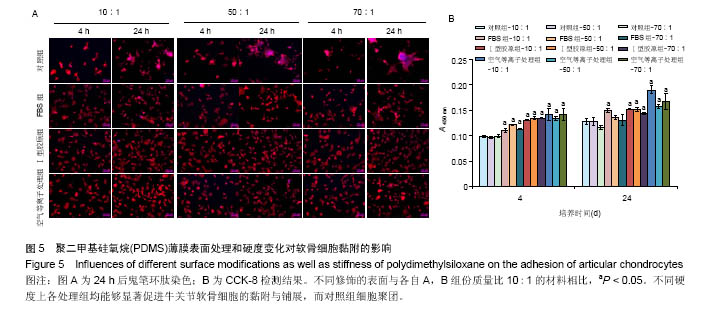
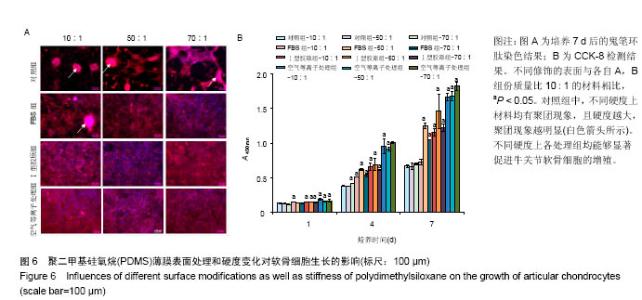
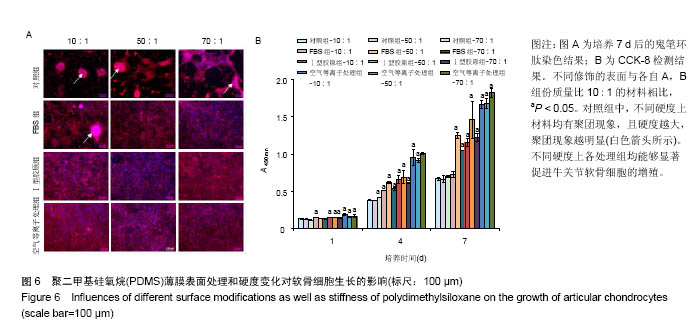
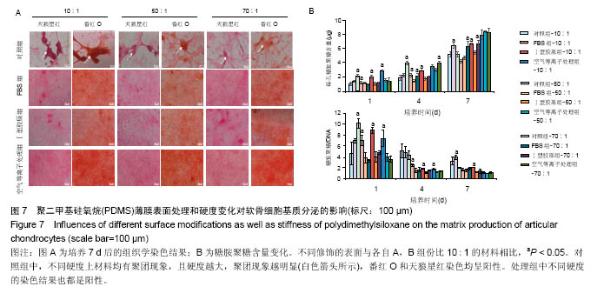
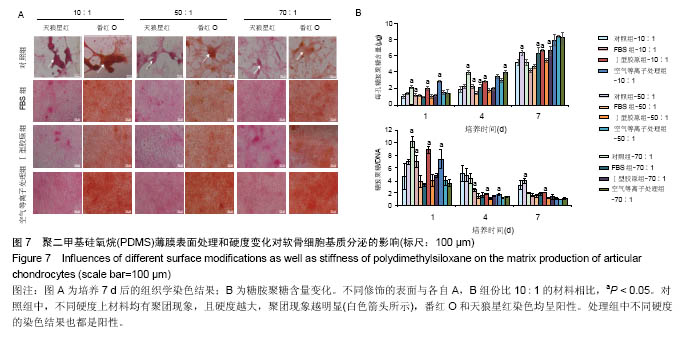
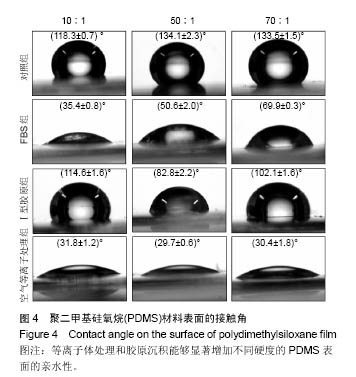
两组份A和B的质量比为10∶1(硬度最高)时表面接触角要比50∶1和70∶1时要低。除了组份质量比为10∶1结合胶原沉积的材料,所有表面修饰过的PDMS材料的接触角都要比未经修饰的低,而且等离子体处理对接触角降低的程度最大,血清孵育其次,而胶原沉积改变最小。对于血清孵育后的材料而言,随A,B组份质量比的增加,接触角越大;而胶原沉积处理和等离子体处理后PDMS材料的接触角在A,B组份质量比为50∶1时呈现出略低的接触角。 2.4 不同表面处理结合PDMS薄膜硬度变化对软骨细胞培养的影响 以两组份A和B的质量比分别为10∶1、50∶1和70∶1制备PDMS薄膜材料,然后通过血清孵育(处理时间24 h)、胶原沉积(胶原密度15 μg/cm2)以及等离子体处理(处理时间180 s)分别进行修饰。材料硬度随质量比从10∶1,50∶1到70∶1的增加而逐步降低。对所有材料进行接触角测定,结果如图4所示。对于未经任何修饰的薄膜而言,两 2.4 不同表面处理结合PDMS薄膜硬度变化对软骨细胞培养的影响 以两组份A和B的质量比分别为10∶1、50∶1和70∶1制备PDMS薄膜材料,然后通过血清孵育(处理时间24 h)、胶原沉积(胶原密度15 μg/cm2)以及等离子体处理(处理时间180 s)分别进行修饰。材料硬度随质量比从10∶1,50∶1到70∶1的增加而逐步降低。对所有材料进行接触角测定,结果如图4所示。对于未经任何修饰的薄膜而言, 图5A所示为将牛关节软骨细胞以4×104/film的密度接种在所有材料上,培养4,24 h后对细胞骨架的染色。可以看到,在未经修饰的PDMS材料表面,较硬的材料(A∶B= 10∶1)上,细胞铺展略好,而且细胞聚集的情况减弱。与未经修饰的对照材料相比,所有修饰过的材料表面上细胞铺展面积明显要大,而且细胞均能均匀分布。CCK-8测定显示血清孵育、胶原沉积和等离子体处理均可以显著提高细胞的黏附效率,并在等离子体处理的材料上最好;而且在24 h时,对不同修饰的材料而言,都是较硬的材料上细胞黏附量要略高,见图5B。 将牛关节软骨细胞以2×104/film接种在所有材料表面上培养7 d。图6A所示,通过对细胞骨架染色发现,未经处理的材料上细胞基本以聚集的形式存在,而在组份质量比70∶1的材料上铺展略好;而经过修饰的材料表面,只有血清孵育处理后在高硬度的材料上出现少量聚集,其余材料上细胞均能均匀分布于基材表面,且基本铺满整个表面。通过CCK-8检测,也发现经过修饰的材料更有利于细胞生长,且在血清孵育、胶原沉积和等离子体处理的表面上依次增加。结合材料硬度变化,发现血清孵育和胶原沉积处理后,中等硬度(A∶B=50∶1)的材料都呈现出更能促进细胞生长的趋势(图6B,培养4和7 d);对于等离子体处理的材料而言,较软的硬度(A∶B=70∶1)在7 d后材料表面的细胞量最高,见图6B。 培养7 d后,细胞在材料表面所分泌的胶原和糖胺聚糖通过天狼星红和番红O染色来分别表征,结果如图7A所示。可以看出,所有材料表面均有天狼星红和番红O的阳性染色,但是等离子体处理的材料上着色明显比血清孵育和胶原沉积修饰的材料要强,而未经修饰的材料表面由于细胞发生聚集,在细胞聚集体中天狼猩红和番红O着色均较深。在未经处理的材料表面上,材料硬度对细胞外基质分泌略有影响,随着硬度提高(也即A,B组份质量比越低),天狼星红和番红O的染色有加深的趋势,这可能主要是因为不同硬度的材料上细胞聚集程度的差别引起的;而材料经过修饰后,不同硬度的表面上染色强度差别不明显,见图7A。通过对糖胺聚糖含量的定量测定发现,所有材料表面上总糖胺聚糖含量随培养时间延长逐步增加,见图7B。对于不同表面修饰而言,在培养1,4 d后,总糖胺聚糖量差别并不明显,而培养7 d后,等离子体处理的表面的最终的总糖胺聚糖量最高,而其他表面之间差别不显著。在不同修饰的表面上,材料硬度对总糖胺聚糖量影响不同。对于未经修饰的对照材料而言,在培养1,4 d后,糖胺聚糖量在较软的材料上更高,但在培养7 d后,中等硬度的材料却表现出较高的上糖胺聚糖分泌量最高。对于表面修饰过的材料来讲,在培养1 d后,都是在最软的材料上总糖胺聚糖量最高,而随着培养时间的延长,呈现了不同的变化规律。在培养4 d后,血清孵育修饰后总糖胺聚糖量在中间硬度的材料上最低;胶原沉积修饰后,总糖胺聚糖量在最软的材料上要显著高于较硬的材料上的值;而等离子体处理后,最硬的材料上的总糖胺聚糖值最高。在培养7 d后,血清孵育修饰后总糖胺聚糖量在最硬的材料上最高;胶原沉积修饰后,总糖胺聚糖量在中间硬度的材料上最低;而等离子体处理后,所有材料上的总糖胺聚糖值基本接近。对于糖胺聚糖/ DNA数值,在所有材料上,总体趋势是随培养时间的延长逐渐降低。在培养1 d后,不同修饰的材料上的糖胺聚糖/DNA差别不大,而在培养4,7 d后,所有修饰过的材料上的糖胺聚糖/DNA数值要低于未经修饰过的材料上的值。对于同一修饰条件下,在培养1 d后,未经修饰的材料上糖胺聚糖/DNA随材料硬度降低而增加,而三种修饰过的材料上,随硬度降低而降低,但随培养时间延长至4,7 d,除了7 d后未经修饰的材料中最软材料上糖胺聚糖/DNA值显著要低以外,硬度的影响逐渐较弱。"
| [1]Sia SK, Whitesides GM. Microfluidic devices fabricated in poly(dimethylsiloxane) for biological studies. Electrophoresis. 2003;24(21):3563-3576. [2]Kuncova-Kallio J, Kallio PJ. PDMS and its suitability for analytical microfluidic devices. Conference proceedings: annual International Conference of the IEEE Engineering in Medicine and Biology Society IEEE Engineering in Medicine and Biology Society Annual Conference. 2006;1:2486-2489. [3]Jang LW, Lee J, Razu ME, et al. Fabrication of PDMS Nanocomposite Materials and Nanostructures for Biomedical Nanosystems. IEEE Trans Nanobioscience. 2015;14(8): 841-849. [4]Kim SJ, Lee JK, Kim JW, et al. Surface modification of polydimethylsiloxane (PDMS) induced proliferation and neural-like cells differentiation of umbilical cord blood-derived mesenchymal stem cells. Journal of materials science J Mater Sci Mater Med. 2008;19(8):2953-2962. [5]Kamei K, Mashimo Y, Koyama Y, et al. 3D printing of soft lithography mold for rapid production of polydimethylsiloxane -based microfluidic devices for cell stimulation with concentration gradients. Biomed Microdevices. 2015;17(2): 36. [6]Stanton MM, Parrillo A, Thomas GM, et al. Fibroblast extracellular matrix and adhesion on microtextured polydimethylsiloxane scaffolds. J Biomed Mater Res B Appl Biomater. 2015;103(4):861-869. [7]Kidambi S, Udpa N, Schroeder SA, et al. Cell adhesion on polyelectrolyte multilayer coated polydimethylsiloxane surfaces with varying topographies. Tissue Eng. 2007; 13(8):2105-2117. [8]Tsimbouri PM. Adult Stem Cell Responses to Nanostimuli. J Funct Biomater. 2015;6(3):598-622. [9]Makamba H, Kim JH, Lim K, et al. Surface modification of poly(dimethylsiloxane) microchannels. Electrophoresis. 2003; 24(21):3607-3619. [10]Zhou J, Ellis AV, Voelcker NH. Recent developments in PDMS surface modification for microfluidic devices. Electrophoresis. 2010;31(1):2-16.[11]Zhou J, Khodakov DA, Ellis AV, et al. Surface modification for PDMS-based microfluidic devices. Electrophoresis. 2012; 33(1):89-104. [12]Zuchowska A, Kwiatkowski P, Jastrzebska E, et al. Adhesion of MRC-5 and A549 cells on poly(dimethylsiloxane) surface modified by proteins. Electrophoresis. 2016;37(3):536-544. [13]Patrito N, McCague C, Norton PR, et al. Spatially controlled cell adhesion via micropatterned surface modification of poly(dimethylsiloxane). Langmuir. 2007;23(2):715-719. [14]Yang SC, Hou JL, Finn A, et al. Synthesis of multifunctional plasmonic nanopillar array using soft thermal nanoimprint lithography for highly sensitive refractive index sensing. Nanoscale. 2015;7(13):5760-5766.[15]Efimenko K, Wallace WE, Genzer J. Surface modification of Sylgard-184 poly(dimethyl siloxane) networks by ultraviolet and ultraviolet/ozone treatment. J Colloid Interface Sci. 2002;254(2):306-315. [16]Lee D, Yang S. Surface modification of PDMS by atmospheric-pressure plasma-enhanced chemical vapor deposition and analysis of long-lasting surface hydrophilicity. Sens Actuators B Chem. 2012;162(1):425-434.[17]Kuddannaya S, Bao J, Zhang Y. Enhanced in vitro biocompatibility of chemically modified poly(dimethylsiloxane) surfaces for stable adhesion and long-term investigation of brain cerebral cortex cells. ACS Appl Mater Interfaces. 2015;7(45):25529-25538. [18]Sun Y, Yong KM, Villa-Diaz LG, et al. Hippo/YAP-mediated rigidity-dependent motor neuron differentiation of human pluripotent stem cells. Nat Mater. 2014;13(6):599-604. [19]Hayman EG, Pierschbacher MD, Suzuki S, et al. Vitronectin--a major cell attachment-promoting protein in fetal bovine serum. Exp Cell Res. 1985;160(2):245-258. [20]Chastain SR, Kundu AK, Dhar S, et al. Adhesion of mesenchymal stem cells to polymer scaffolds occurs via distinct ECM ligands and controls their osteogenic differentiation. J Biomed Mater Res A. 2006;78(1):73-85. [21]Mata A, Fleischman AJ, Roy S. Characterization of polydimethylsiloxane (PDMS) properties for biomedical micro/nanosystems. Biomed Microdevices. 2005;7(4):281-93. [22]何懿,栾杰.PDMS微孔阵列细胞培养实验平台的初步构建与评测[J].组织工程与重建外科杂志,2011,7(3):121-128. [23]Hou S, Yang K, Qin M, et al. Patterning of cells on functionalized poly(dimethylsiloxane) surface prepared by hydrophobin and collagen modification. Biosens Bioelectron. 2008;24(4):912-916.[24]Koo S, Muhammad R, Peh GS, et al. Micro- and nanotopography with extracellular matrix coating modulate human corneal endothelial cell behavior. Acta Biomater. 2014;10(5):1975-1984. [25]Fallas JA, O'Leary LE, Hartgerink JD. Synthetic collagen mimics: self-assembly of homotrimers, heterotrimers and higher order structures. Chem Soc Rev. 2010;39(9):3510-3527. [26]Markov DA, Lillie EM, Garbett SP, et al. Variation in diffusion of gases through PDMS due to plasma surface treatment and storage conditions. Biomedical microdevices. 2014;16(1):91-96. [27]李艳琼,王升高,程莉莉,等.低温空气等离子体改性PDMS的研究[J].真空与低温,2008,14(2):86-90.[28]Yildirim ED, Ayan H, Vasilets VN, et al. Effect of dielectric barrier discharge plasma on the attachment and proliferation of osteoblasts cultured over poly(?-caprolactone) scaffolds. Plasma Process Polym. 2008;5(1):58-66.[29]Pinto S, Alves P, Matos CM, et al. Poly(dimethyl siloxane) surface modification by low pressure plasma to improve its characteristics towards biomedical applications. Colloids Surf B Biointerfaces. 2010;81(1):20-26.[30]Zhao Y, Tan K, Zhou Y, et al. A combinatorial variation in surface chemistry and pore size of three-dimensional porous poly(epsilon-caprolactone) scaffolds modulates the behaviors of mesenchymal stem cells. Mater Sci Eng C Mater Biol Appl. 2016;59:193-202. [31]Schnabel M, Marlovits S, Eckhoff G, et al. Dedifferentiation- associated changes in morphology and gene expression in primary human articular chondrocytes in cell culture. Osteoarthritis Cartilage. 2002;10(1):62-70. [32]Chen M, Xu L, Zhou Y, et al. Poly(epsilon-caprolactone)- based substrates bearing pendant small chemical groups as a platform for systemic investigation of chondrogenesis. Cell proliferation. 2016;49(4):512-522.[33]刘振宁,韩长旭,赵敏.生长分化因子5诱导脂肪干细胞复合Ⅰ型胶原支架成软骨细胞的分化[J].中国组织工程研究, 2015,19(19):2999- 3004. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [5] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [6] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [7] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [8] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [9] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [10] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| [11] | Gao Cangjian, Yang Zhen, Liu Shuyun, Li Hao, Fu Liwei, Zhao Tianyuan, Chen Wei, Liao Zhiyao, Li Pinxue, Sui Xiang, Guo Quanyi. Electrospinning for rotator cuff repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 637-642. |
| [12] | Guan Jian, Jia Yanfei, Zhang Baoxin , Zhao Guozhong. Application of 4D bioprinting in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 446-455. |
| [13] | Wang Kun, He Benxiang. Asperosaponin VI therapy for Achilles tendinopathy in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 211-217. |
| [14] | Liu Jiali, Suo Hairui, Yang Han, Wang Ling, Xu Mingen. Influence of lay-down angles on mechanical properties of three-dimensional printed polycaprolactone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2612-2617. |
| [15] | Huang Bo, Chen Mingxue, Peng Liqing, Luo Xujiang, Li Huo, Wang Hao, Tian Qinyu, Lu Xiaobo, Liu Shuyun, Guo Quanyi . Fabrication and biocompatibility of injectable gelatin-methacryloyl/cartilage-derived matrix particles composite hydrogel scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 10(16): 2600-2606. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||