Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (50): 7579-7585.doi: 10.3969/j.issn.2095-4344.2016.50.019
Previous Articles Next Articles
Research progress of stem cells in hair follicle regeneration
Ding Yu-wei, Xin Guo-hua, Zeng Yuan-lin
- Department of Burn Surgery, First Affiliated Hospital of Nanchang University, Nanchang 330000, Jiangxi Province, China
-
Revised:2016-09-04Online:2016-12-02Published:2016-12-02 -
Contact:Zeng Yuan-lin, Doctoral supervisor, Chief physician, Department of Burn Surgery, First Affiliated Hospital of Nanchang University, Nanchang 330000, Jiangxi Province, China -
About author:Ding Yu-wei, Studying for master’s degree, Department of Burn Surgery, First Affiliated Hospital of Nanchang University, Nanchang 330000, Jiangxi Province, China -
Supported by:the Scientific Plan of Jiangxi Educational Department, No. 12003628
CLC Number:
Cite this article
Ding Yu-wei, Xin Guo-hua, Zeng Yuan-lin. Research progress of stem cells in hair follicle regeneration[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(50): 7579-7585.
share this article
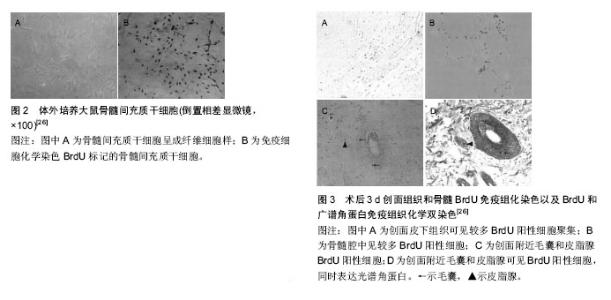
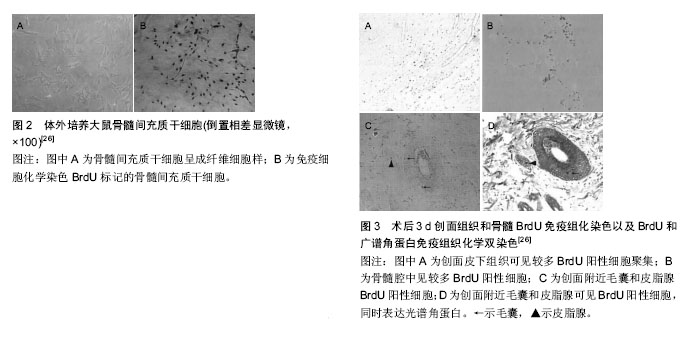
2.1 毛囊及毛囊的再生 2.1.1 毛囊的结构和特性 毛囊具有维持皮肤自身稳定及促进创伤愈合作用,毛囊由表皮层和真皮层两部分组成,毛囊表皮层包括紧密包绕毛囊的内根鞘和外根鞘,内根鞘在皮脂腺开口水平与内陷的表皮相连,起决定毛发形状的作用。外根鞘与抗原递呈细胞和黑色素细胞共同对毛发的调节发挥作用,由外根鞘形成明显突起的区域为毛囊隆突区,内含多种干细胞。干细胞与周围的上皮细胞、内根鞘、外根鞘、基底膜或者纤维细胞相互作用,这对于干细胞功能的维持起着重要作用;毛囊真皮层包括真皮乳头和真皮鞘,真皮乳头位于竖毛肌与毛囊接触部下面,主要通过控制毛母质细胞的数目而达到控制毛发密度和长度的效果[2]。毛囊主要呈周期性生长,一般经历生长期、退行期、休止期3个阶段,毛囊隆突以上部位(相当于毛囊上1/3)为恒定部,此部分一般不发生凋亡和再生,隆突以下为循环部(毛囊下2/3),此部分呈现以上3个阶段为主的周期性变化,为毛囊能够在被破坏后进行自身再生修复提供了基础条件[3]。 2.1.2 毛囊的再生 毛囊的再生与毛囊干细胞的增殖分化直接相关。毛囊隆突中存在2种干细胞:毛囊干细胞及毛囊黑素干细胞,其中毛囊干细胞作为皮肤来源细胞的前体细胞,具有与其他成体干细胞一样的多向分化潜能[4]。当真皮浅层受损致毛囊横断时,毛囊的再生是通过毛囊隆突细胞受到刺激后以部分存活的毛囊为模版进行增生而实现的。这一过程类似胚胎期毛囊发育过程的修复[5],首先毛囊上皮细胞被真皮基质诱导增厚形成毛囊基板,基板再逐渐内陷形成毛乳头芽,同时,其下的真皮基质干细胞分化形成毛乳头,从而实现毛囊再生。Biernaskie等[6]将已标记的来自真皮的皮肤前体干细胞植入小鼠背部缺损皮肤,发现已植入的细胞在三四周表达Ⅰ型胶原、波形蛋白、纤连蛋白等成纤维细胞的标记,还有部分植入的皮肤前体干细胞进入皮下组织并呈现出脂肪细胞近似形态,并且证明皮肤前体干细胞具有维持真皮结构稳态并诱导毛囊再生的功能[7]。Gho等[8]在轻中度创面上移植直径0.5-0.6 mm的部分毛囊,受区和枕部供区的毛发均生长良好,提示毛囊纵向分开移植后可能形成2个新的毛囊;孙锡金等[9]在小鼠皮内注射毛囊干细胞和其他类型细胞,结果表明,含毛囊干细胞的移植组可见新生毛囊形成,其他类型细胞移植组未见毛囊形成。上述研究说明毛囊干细胞具有良好的向毛囊分化增殖的能力。 近年来国内外关于毛囊再生的研究进展迅速,有研究者在体外进行毛囊重构以实现毛囊再生,并且在动物身上行毛囊再生的研究取得了不少的进展,如Qiao等[10]将胚鼠毛囊真皮和上皮细胞用悬滴法培养后形成细胞聚集体,将细胞聚集体进一步培养后发现能形成初始毛囊。还有研究者发现来源于鼠须的真皮乳头细胞也有利于毛囊的再生,Osada等[11]从鼠触须分离出真皮乳头细胞,经过体外培养并将传代次数少的真皮乳头细胞与表皮细胞一起注射到裸鼠皮下可生成毛发,而传代次数多的真皮乳头细胞不能生成毛发,但将真皮乳头细胞体外培养并聚集成微球后再与表皮细胞一起注射到裸鼠皮下,发现传代数多的真皮乳头细胞聚集成微球可以形成毛发,表明细胞间拥有一定紧密作用后可诱导毛囊的再生。Young等[12]将密度> 42×103个/cm2的真皮乳头细胞在聚乙烯-乙烯醇材料表面生长、聚集、自组装成与体内类似的球形微结构,然后与新生小鼠表皮细胞混合后注射于裸鼠皮下,成功诱导生成毛发结构,表明真皮乳头细胞在一定的密度下才能转化生成毛囊。Thangapazham等[13]和Higgins等[14]分别利用真皮乳头细胞诱导毛发再生功能,把真皮乳头细胞植入真皮-表皮复合物中和在三维球体培养环境下进行培养,达到毛囊重建的目的。 2.2 干细胞向毛囊的定向分化 毛囊的再生虽与毛囊干细胞等增殖分化密不可分,但当毛囊严重损伤甚至造成缺失时是难以靠自身重建再生的,为此研究者将目光转移至其他干细胞的应用上,并进行了相关研究,取得了不少有意义的成果。 2.2.1 胚胎干细胞向毛囊的分化 胚胎干细胞是一种高度未分化的细胞,是全能细胞系,能够大量繁殖并保持未分化状态,在一定条件下也能够分化成人体3个胚层的全部细胞,这也是胚胎干细胞相比其他干细胞的优势所在[15]。 目前国内外研究已证实体外可诱导胚胎干细胞分化为血管内皮细胞、平滑肌细胞、肌细胞、造血细胞、软骨细胞等,而且胚胎干细胞双向分化的潜能决定其既可以向表皮分化,也可向毛囊等皮肤附属器分化。Coraux等[16]将胚胎干细胞与人真皮成纤维细胞来源的无细胞饲养基质共培养,K-14阳性细胞占有1.1%,在骨形态发生蛋白4的作用下K-14阳性细胞升高至5.4%,其诱导作用限于胚胎干细胞培养的第4-8天,诱导后的阳性细胞转移到硝酸纤维素膜上,继续培养可见K-14阳性细胞能够形成复层上皮和毛囊等附件组织结构。还有研究表明,将胚胎干细胞和人羊膜进行共同培养,胚胎干细胞可诱导分化为具有表皮干细胞表型的干细胞,并将其移植于裸鼠腹腔、皮下及肾被囊中,均能分化为毛囊等皮肤附属器结构[17]。 2.2.2 表皮干细胞向毛囊的分化 人的表皮干细胞作为皮肤组织中特异性干细胞可以在体外进行有效培养并构成含附件的组织工程皮肤。Blanpain等[18]实验结果显示无论表皮干细胞标记物整合素α6是否高表达,标记CD34high的鼠隆突区细胞均可诱导出毛发来。有国外学者使用表皮干细胞另一种标记物Sox9作为基因标记物[19],发现Sox9阳性细胞具有分化为毛囊等皮肤附件结构的潜能[20]。Huang等[21]将含有表皮干细胞的胎猪皮肤前体组织移植到裸鼠背部创面,发现移植物可继续生长发育成为具有毛囊、皮脂腺等皮肤附件的完整皮肤组织。Lee等[22]用新生(出生24 h内)幼鼠的表皮细胞和成纤维细胞构建的组织工程皮肤移植到无胸腺小鼠背部,在无胸腺小鼠背部形成了毛囊及毛发,并且1年后无胸腺小鼠仍可以长时间表达毛囊增生,其认为损伤后毛囊可以再生和循环。有研究者在体外诱导胚胎干细胞分化为表皮干细胞,采用荧光染料Hoeehst 33342染色后,种植到有成纤维细胞的胶原-明胶海绵真皮替代物内,并移植到与胚胎干细胞同基因的129/J小鼠皮下,采用苏木精-伊红染色、免疫组化、Vann Gieson染色观察到细胞存活且呈现出毛囊样结构,也证实这些结构中含有CD29和细胞角蛋白18等表皮干细胞特异性标志物的表达[23],通过对表皮干细胞向毛囊分化的研究发现其具有用于临床治疗的潜能。 2.2.3 间充质干细胞向毛囊的分化 间充质干细胞是一种具有多向分化潜能的成体干细胞,通过建立不同培养条件后,可分化为表皮细胞、内皮细胞、心肌细胞、骨骼肌细胞等[24-25]。有研究者将BrdU标记的间充质干细胞经阴茎静脉注入大鼠体内,取3 d及7 d后的创面组织,行BrdU免疫组化单染色以及BrdU和广谱角蛋白免疫组化双染色,显示毛囊、皮脂腺内存在BrdU阳性细胞,同时表达广谱角蛋白,推断在缺损创面下间充质干细胞具有向上皮和毛囊转化能力[26-27],见图2,3。罗旭等[28]取SD大鼠股骨和胫骨,分离培养骨髓间充质干细胞群,传代至第3代后将其接种于激光微孔猪脱细胞真皮基质(LP-ADM)、无孔猪脱细胞真皮基质,构建骨髓间充质干细胞-微孔猪脱细胞真皮基质和骨髓间充质干细胞-无孔猪脱细胞真皮基质,分别移植于裸鼠背部全层皮肤缺损创面上,通过外观、苏木精-伊红染色、透射电镜发现创面出现毛囊新生现象,由此推断外源性骨髓间充质干细胞在一定诱导条件下可定向分化为毛囊等结构。 2.2.4 诱导多功能干细胞向毛囊的分化 近年来诱导多功能干细胞成为国内外研究者的研究热点,其具有胚胎干细胞生物特性和分化潜能,单个细胞可向内、中、外胚层所有的细胞进行分化,进而形成各组织、器官[29]。Takahashi等[30]利用Oct3/4、Sox2、c-Myc和Klf4这4个因子以反转录病毒为载体将其导入小鼠胚胎成纤维细胞,使体细胞重新获得全能特性,重编程为诱导多能干细胞。近年Takahashi等[31]进一步把人皮肤成纤维细胞重编程诱导成诱导多功能干细胞,这已为研究诱导多功能干细胞定向分化为毛囊等皮肤附件奠定了基础。Bilousova等[32]利用维A酸和骨形态发生蛋白4对小鼠诱导多功能干细胞进行处理,可见其在体外分化为功能性的角质形成细胞,再将这些细胞置于已包被Ⅳ胶原的培养板上培养,体内研究证明它们能再生分化为毛囊、皮脂腺、汗腺等,基因和蛋白表达模式均与正常角质形成细胞相似。"
| [1] Sugaya K, Ishihara Y, Inoue S, et al. The effects of gamma rays on the regeneration of hair follicles are carried over to later hair cycles.Int J Radiat Biol. 2015; 91(12):957-963.[2] Balañá ME, Charreau HE, Leirós GJ. Epidermal stem cells and skin tissue engineering in hair follicle regeneration. World J Stem Cells. 2015;7(4):711-727.[3] Fuchs E. Cell biology: More than skin deep. J Cell Biol. 2015;209(5):629-631.[4] Zhang H, Zhang S, Zhao H, et al. Ovine Hair Follicle Stem Cells Derived from Single Vibrissae Reconstitute Haired Skin. Int J Mol Sci. 2015;16(8):17779-17797.[5] Su YS, Miao Y, Jiang JD, et al. A simple and rapid model for hair-follicle regeneration in the nude mouse. Clin Exp Dermatol. 2015;40(6):653-658.[6] Biernaskie J, Paris M, Morozova O, et al. SKPs derive from hair follicle precursors and exhibit properties of adult dermal stem cells. Cell Stem Cell. 2009; 5(6): 610-623.[7] Shu B, Xie JL, Xu YB, et al. Effects of skin-derived precursors on wound healing of denervated skin in a nude mouse model.Int J Clin Exp Pathol. 2015;8(3): 2660-2669.[8] Gho CG, Neumann HA. Improved hair restoration method for burns. Burns. 2011;37(3):427-433.[9] 孙锡金,胡志奇,苗勇.小鼠毛囊细胞注射移植实验研究[J].中华整形外科杂志,2012,28(1):44-49.[10] Qiao J, Turetsky A, Kemp P, et al. Hair morphogenesis in vitro: formation of hair structures suitable for implantation. Regen Med. 2008;3(5):683-692.[11] Osada A, Iwabuchi T, Kishimoto J, et al. Long-term culture of mouse vibrissal dermal papilla cells and de novo hair follicle induction. Tissue Eng. 2007;13(5):975-982.[12] Young TH, Lee CY, Chiu HC, et al. Self-assembly of dermal papilla cells into inductive spheroidal microtissues on poly(ethylene-co-vinyl alcohol) membranes for hair follicle regeneration. Biomaterials. 2008;29(26):3521-3530.[13] Thangapazham RL, Klover P, Wang JA, et al. Dissociated human dermal papilla cells induce hair follicle neogenesis in grafted dermal-epidermal composites. J Invest Dermatol. 2014;134(2):538-540.[14] Higgins CA, Chen JC, Cerise JE, et al. Microenvironmental reprogramming by three-dimensional culture enables dermal papilla cells to induce de novo human hair-follicle growth. Proc Natl Acad Sci U S A. 2013;110(49):19679-19688.[15] Suh HN, Han HJ. Sonic hedgehog increases the skin wound-healing ability of mouse embryonic stem cells through the microRNA 200 family. Br J Pharmacol. 2015;172(3):815-828.[16] Coraux C, Hilmi C, Rouleau M, et al. Reconstituted skin from murine embryonic stem cells. Curr Biol. 2003; 13(10):849-853.[17] 杨晓龙,巴特,刘玲英.间充质干细胞促进慢性创面血管新生的机制及应用[J].中华损伤与修复杂志:电子版,2015, 10(1):76-79.[18] Blanpain C, Lowry WE, Geoghegan A, et al. Self-renewal, multipotency, and the existence of two cell populations within an epithelial stem cell niche. Cell. 2004;118(5):635-648.[19] Biernaskie J, Paris M, Morozova O, et al. SKPs derive from hair follicle precursors and exhibit properties of adult dermal stem cells. Cell Stem Cell. 2009;5(6): 610-623.[20] Alcolea MP, Jones PH. Lineage analysis of epidermal stem cells. Cold Spring Harb Perspect Med. 2014;4(1): a015206.[21] Huang Z, Yang J, Luo G, et al. Embryonic porcine skin precursors can successfully develop into integrated skin without teratoma formation posttransplantation in nude mouse model. PLoS One. 2010;5(1):e8717.[22] Lee LF, Jiang TX, Garner W, et al. A simplified procedure to reconstitute hair-producing skin. Tissue Eng Part C Methods. 2011;17(4):391-400.[23] 林羿,李海标,黄锦桃.含小鼠表皮干细胞的真皮替代物体内移植追踪实验[J].中华整形外科杂志,2015,21(6): 452-456.[24] Zahorec P, Koller J, Danisovic L, et al. Mesenchymal stem cells for chronic wounds therapy. Cell Tissue Bank. 2015;16(1):19-26.[25] Jin SZ, Liu BR, Xu J, et al. Ex vivo-expanded bone marrow stem cells home to the liver and ameliorate functional recovery in a mouse model of acute hepatic injury. Hepatobiliary Pancreat Dis Int. 2012;11(1): 66-73.[26] 李海红,付小兵,王君,等.骨髓间充质干细胞分化为皮肤附属器细胞的初步实验研究[J]. 中国修复重建外科杂志, 2006,20(6):675-678.[27] 郭明锋,马杰,蔡卫林,等.皮肤附属结构构建与再生研究进展[J].中华创伤杂志,2014,30(1):85-88.[28] 罗旭,辛国华,曾逃方,等.微孔化猪脱细胞真皮基质与大鼠骨髓间充质细胞对裸鼠皮肤附件细胞再生的作用[J].中华烧伤杂志,2013,29(6):541-547.[29] Shimojo D, Onodera K, Doi-Torii Y, et al. Rapid, efficient, and simple motor neuron differentiation from human pluripotent stem cells. Mol Brain. 2015;8(1):79.[30] Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676.[31] Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotency in human somatic cells via a transient state resembling primitive streak-like mesendoderm. Nat Commun. 2014;5:3678.[32] Bilousova G, Chen J, Roop DR. Differentiation of mouse induced pluripotent stem cells into a multipotent keratinocyte lineage. J Invest Dermatol. 2011;131(4): 857-864.[33] 弓家弘,陆树良.干细胞治疗糖尿病难愈创面研究进展[J].中华烧伤杂志,2014,30(6):518-521.[34] Moore PJ. Stem cells as a novel therapy for amyotrophic lateral sclerosis. Br J Biomed Sci. 2015; 72(2):74-75. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Li Jing, Xie Jianshan, Cui Huilin, Cao Ximei, Yang Yanping, Li Hairong. Expression and localization of diacylglycerol kinase zeta and protein kinase C beta II in mouse back skin with different coat colors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1196-1200. |
| [5] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [6] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [7] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [8] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [9] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [10] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [11] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [12] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [13] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [14] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [15] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||