Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (29): 7654-7662.doi: 10.12307/2026.225
Previous Articles Next Articles
Metabolic dysregulation in osteoarthritis: mechanisms and targeted therapeutic strategies
Guo Shanshan1, Ma Ding2, Dong Bingchen2
- 1Central Laboratory & Translational Medicine Center, Shaanxi Provincial People's Hospital, Xi'an 710068, Shaanxi Province, China; 2Department of Orthopedics, Xi’an No. 9 Hospital, Xi’an 710054, Shaanxi Province, China
-
Received:2025-09-02Revised:2025-11-20Online:2026-10-18Published:2026-03-06 -
Contact:Dong Bingchen, MS, Attending physician, Department of Orthopedics, Xi’an No. 9 Hospital, Xi’an 710054, Shaanxi Province, China -
About author:Guo Shanshan, PhD, Assistant Researcher, Central Laboratory & Translational Medicine Center, Shaanxi Provincial People’s Hospital, Xi’an 710068, Shaanxi Province, China -
Supported by:Youth Cultivation Project of Xi’an No. 9 Hospital, No. 2025qn02 (to DBC)
CLC Number:
Cite this article
Guo Shanshan, Ma Ding, Dong Bingchen. Metabolic dysregulation in osteoarthritis: mechanisms and targeted therapeutic strategies[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7654-7662.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
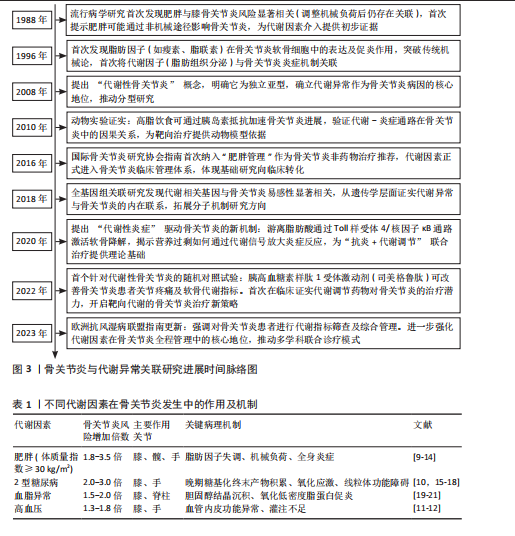
2.1 骨关节炎与代谢异常关联研究进展时间脉络图 见图3。 2.2 代谢异常与骨关节炎的流行病学关联 代谢综合征涵盖肥胖、高血压、血脂异常、高血糖及胰岛素抵抗等核心组分,这些因素不仅独立促进骨关节炎的病理进程,更通过协同效应显著提升心血管事件风险[9-12]。大型多中心研究证实,慢性肥胖人群的膝关节内侧软骨损伤风险增加3.12倍(95%CI:2.85-3.42),且体质量指数每升高5 kg/m2,膝骨关节炎发病风险相应增加35%(RR=1.35,95%CI:1.28-1.42)[13]。值得注意的是,肥胖患者非负重关节(如手部)的骨关节炎风险亦显著上升1.8倍(95%CI:1.5-2.1),这一现象佐证了代谢因子的独立致病机制,超越了传统机械负荷理论的解释范畴[14]。 糖脂代谢紊乱在骨关节炎发展中扮演关键角色[10]。慢性高血糖与胰岛素抵抗通过三重途径驱动关节退变[15]:①诱导促炎因子(肿瘤坏死因子α、白细胞介素1β)及基质金属蛋白酶的异常合成;②促进晚期糖基化终末产物积累,激活晚期糖基化终末产物受体信号通路;③引发线粒体功能障碍,导致氧化应激反应增强[16]。流行病学数据显示,2型糖尿病患者骨关节炎发病风险较非糖尿病人群升高2.3倍(95%CI:2.0-2.6),且该效应独立于体质量指数[17-18]。在脂代谢层面,高胆固醇血症与低密度脂蛋白水平升高可加速骨关节炎进展(HR=1.85,95%CI:1.62-2.11),而高密度脂蛋白则表现出保护效应——其浓度每增加0.26 mmol/L,骨关节炎风险相应降低15%(HR=0.85,95%CI:0.79-0.92)[19-21](表1)。 饮食模式与代谢状态存在显著交互作用。韩国横断面研究(n=8 342)发现,高血糖指数饮食使女性症状性膝骨关节炎风险增加40%(OR=1.40,95%CI:1.12-1.75)[10];而北美大型队列(n=12 591)证实,严格遵循地中海饮食(低高血糖指数特征)的个体膝骨关节炎风险降低30%(HR=0.70,95%CI:0.62-0.79)。这种代谢异常不仅增加骨关节炎易感性,更直接影响临床转归——加拿大关节置换登记研究(n=24 768)显示,代谢综合征患者术后功能恢复延迟(6 min步行距离下降18%,P < 0.01),再手术风险增加2.2倍(95%CI:1.8-2.7)[22]。 值得注意的是,代谢综合征各组分通过“肥胖→胰岛素抵抗→高血糖→血脂异常”的恶性循环形成正反馈网络:肥胖诱发胰岛素抵抗,导致高血糖及晚期糖基化终末产物积累;高血糖又进一步加剧血脂异常,共同促进关节组织退变[23]。这种复杂的协同效应使得针对单一代谢因素的干预策略效果有限,亟需建立综合代谢调控体系以改善骨关节炎的临床预后[24]。 2.3 代谢性炎症的核心机制 2.3.1 脂肪因子紊乱与关节组织损伤 脂肪组织不仅是能量储存器官,也是重要的内分泌器官。在肥胖状态下,脂肪组织功能失调导致多种脂肪因子分泌异常,形成慢性低度炎症状态,即“代谢性炎症”[25-26],这些脂肪因子通过血液循环到达关节组织,直接参与骨关节炎的病理过程。 瘦素是研究最为深入的脂肪因子之一。在骨关节炎患者关节液中,瘦素水平显著升高,且与软骨破坏程度呈正相关[25]。瘦素通过多种途径促进骨关节炎进展:①刺激软骨细胞产生基质金属蛋白酶,特别是基质金属蛋白酶13,加速细胞外基质降解[27];②促进促炎因子(白细胞介素6、白细胞介素1β和肿瘤坏死因子α)的产生;③诱导软骨细胞肥大分化,加速软骨钙化[28];④促进骨赘形成[26]。临床研究发现,老年人群血液瘦素水平与膝关节软骨容积呈负相关,提示瘦素具有作为骨关节炎生物标志物的潜力[29]。 脂联素的作用则与瘦素相反[29],这种具有抗炎作用的脂肪因子在肥胖患者和骨关节炎患者中显著降低[30]。脂联素可通过多种机制保护关节:①下调白细胞介素1诱导的基质金属蛋白酶13表达[31];②上调金属蛋白酶组织抑制"
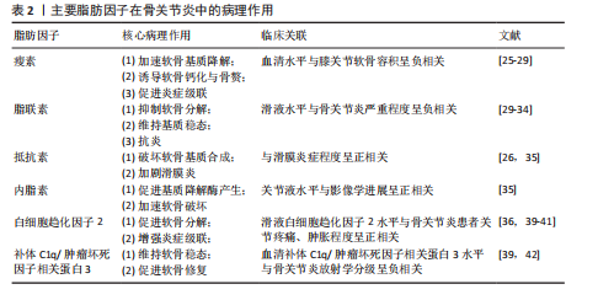
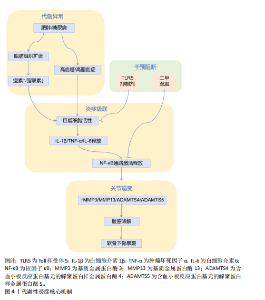
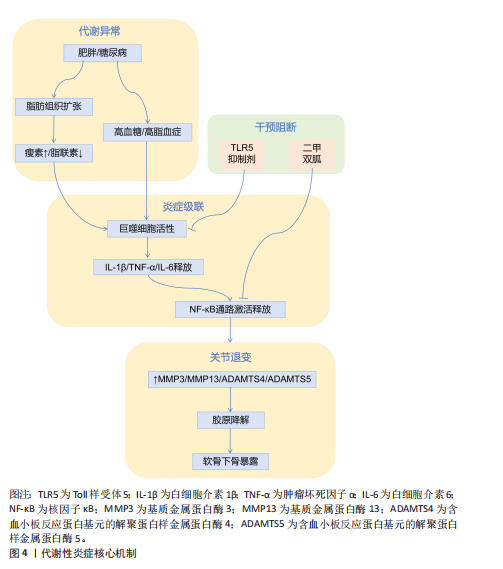
物2的表达[32];③抑制核因子κB信号通路[33]。临床研究证实,血浆和关节滑液中的脂联素水平与骨关节炎疾病严重程度呈负相关,表明脂联素具有保护作用[34]。 抵抗素和内脂素是另外两种促炎脂肪因子。抵抗素抑制软骨蛋白聚糖合成,并促进白细胞介素6、白细胞介素8和前列腺素E2释放[26]。内脂素又称B细胞前体克隆增强因子,在骨关节炎软骨中高表达,通过刺激基质金属蛋白酶3/基质金属蛋白酶9、前列腺素E2及含血小板结合蛋白基序的解聚素样金属蛋白酶5加剧软骨分解[35]。 新型脂肪因子白细胞趋化因子2(Leukocyte cell-derived chemotaxin 2,LECT2)和补体C1q/肿瘤坏死因子相关蛋白3(C1q/TNF-related protein 3,CTRP3 )也是连接脂代谢紊乱与骨关节炎的关键递质[36-38]。动物模型证实,白细胞趋化因子2通过胆固醇-25-羟化酶(CH25H)-细胞色素P450家族成员7B1(CYP7B1)-维A酸相关孤儿受体α(RORα)信号轴抑制胆固醇逆转运,导致软骨细胞内胆固醇蓄积[39]。过表达白细胞趋化因子2的小鼠出现严重软骨破坏、骨赘形成及软骨下骨板增厚[40]。临床研究进一步发现,高脂饮食后人体循环白细胞趋化因子2水平上升40%,且骨关节炎患者关节组织中白细胞趋化因子2表达显著高于健康对照,首次建立了脂代谢异常与骨关节炎的直接因果关联[41]。补体C1q/肿瘤坏死因子相关蛋白3通过激活过氧化物酶体增殖物激活受体α-酰基辅酶A硫脂酶12通路维持软骨稳态,干预实验显示重组补体C1q/肿瘤坏死因子相关蛋白3可使软骨降解标志物基质金属蛋白酶13表达降低52%[39,42](表2)。 2.3.2 炎症信号级联放大 代谢异常引发的炎症反应在骨关节炎进展中起核心作用[43-44]。脂肪组织产生的促炎因子与关节局部炎症形成恶性循环,不断放大炎症信号[45]。促炎细胞因子包括白细胞介素1β、肿瘤坏死因子α和白细胞介素6,主要由活化的巨噬细胞合成,在骨关节炎相关的炎症反应中起关键作用[21,46](图4)。流行病学研究已将糖尿病和肥胖确定为骨关节炎发展的促成因素,在人体内诱导局部或全身低度炎症状态[47]。高血糖环境和肥胖可增加促炎因子如白细胞介素1β、白细胞介素6和肿瘤坏死因子α的表达,进一步激活核因子κB信号通路,增加关节软骨细胞的分解代谢活性并促进细胞外基质降解,最终导致骨关节炎进展。在应激诱导的骨关节炎小鼠模型中,高脂饮食组血清肿瘤坏死因子α水平显著升高,软骨损伤更严重;而Toll样受体5缺陷(Tlr5 KO)小鼠组血清白细胞介素6水平显著低于其他组,表明肥胖通过增加促炎因子表达加剧骨关节炎进展[48]。另一研究证明,高脂饮食小鼠血清中白细胞介素1β显著升高,白细胞介素1β通过激活核因子κB信号通路在骨关节炎软骨细胞中诱导炎症反应[49]。值得注意的是,二甲双胍不仅降低肥胖个体的体质量指数,还降低骨关节炎患者关节置换手术比率[18]。在小鼠研究中,肿瘤坏死因子α和白细胞介素1β显著增加原代关节软骨细胞中基质金属蛋白酶3、基质金属蛋白酶13、含血小板结合蛋白基序的解聚素样金属蛋白酶4和含血小板结合蛋白基序的解聚素样金属蛋白酶5的mRNA表达水平,而二甲双胍有效抑制肿瘤坏死因子α和白细胞介素1β诱导的基质金属蛋白酶13和基质金属蛋白酶3表达,揭示二甲双胍通过减弱炎症细胞因子诱导的分解代谢反应保护关节软骨细胞[50]。因此,抑制促炎因子表达对骨关节炎治疗具有积极疗效[51]。 白细胞介素1β和肿瘤坏死因子α是骨关节炎中的关键促炎因子[47,52]。在肥胖引起的低炎症状态下,这些细胞因子水平升高,抑制软骨基质合成,促进基质金属蛋白酶表达,诱导一氧化氮合酶产生,增加氧化应激,促进软骨细胞凋亡[46]。临床研究表明,抑制白细胞介素1的药物可缓解骨关节炎症状,减缓疾病进展[49]。白细胞介素6家族细胞因子(包括白细胞介素6、制瘤素M和白血病抑制因子)在骨关节炎中同样发挥重要作用,15%-30%血液循环中的白细胞介素6来自脂肪组织[48]。白细胞介素6可与白细胞介素1协同作用,抑制软骨细胞合成蛋白多糖,诱导软骨细胞产生过氧化反应,导致胶原降解[53]。临床研究发现,老年人血液中白细胞介素6水平可预测2年后骨密度损失,且白细胞介素6水平与膝关节软骨损失呈正相关[54]。基质金属蛋白酶是细胞外基质降解的关键执行者,其中基质金属蛋白酶13是降解Ⅱ型胶原效率最高的酶。在代谢异常状态下,脂肪因子和炎症因子协同上调基质金属蛋白酶表达,同时降低金属蛋白酶组织抑制物表达,打破基质金属蛋白酶-金属蛋白酶组织抑制物平衡,导致细胞外基质过度降解,加速软骨破坏[50]。 2.3.3 细胞内代谢通路重编程 近年研究发现,骨关节炎软骨细胞经历显著的代谢重编程,主要表现为糖酵解增强、脂肪酸氧化障碍和线粒体功能障碍[55]。在骨关节炎的不良微环境(如缺氧、营养缺乏、炎症因子刺激)下,软骨细胞从相对静止状态转变为高度激活状态,导致具有致炎和促蛋白分解作用的中间代谢产物增加[56](图5)。 糖代谢异常是骨关节炎代谢重编程的核心特征,在缺氧和炎症条件下,软骨细胞糖酵解速率显著提高,产生过量乳酸,降低细胞外pH值,激活酸性敏感离子通道,导致疼痛[48,57]。同时,高血糖环境促进晚期糖基化终末产物积累,晚期糖基化终末产物通过与受体结合,激活核因子κB信号通路,促进炎症因子表达[48]。在2型糖尿病患者中,长期高血糖导致晚期糖基化终末产物积累,促进基质硬化[56]。此外,晚期糖基化终末产物与软骨细胞膜上的受体结合,激活丝裂原活化蛋白激酶信"

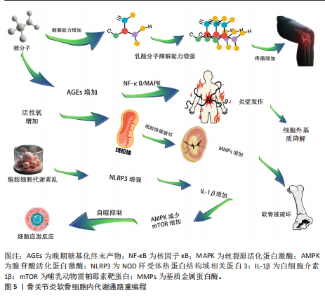
号通路,增强白细胞介素6和白细胞介素8表达,加剧炎症反应[58-59]。晚期糖基化终末产物还促使关节软骨基质分解代谢增加,阻碍细胞外基质更新[60]。研究报道骨关节炎患者软骨中晚期糖基化终末产物水平显著高于健康个体,晚期糖基化终末产物积累使胶原网络脆弱化,增加骨关节炎风险[59]。 活性氧是含有氧的高反应性分子,肥胖和糖尿病可诱导体内活性氧水平升高[61-62]。脂肪组织和高血糖水平营造促炎环境,导致M1型巨噬细胞和促炎细胞因子增加,进一步刺激活性氧产生[63-64]。在骨关节炎软骨细胞中,过量活性氧激活丝裂原活化蛋白激酶和核因子κB信号通路,破坏软骨分解代谢和合成代谢平衡[65]。研究表明,H2O2孵育软骨细胞会导致活性氧增加、软骨细胞死亡及细胞外基质降解[66];"
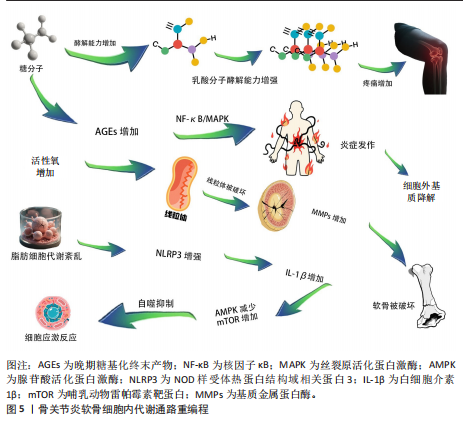
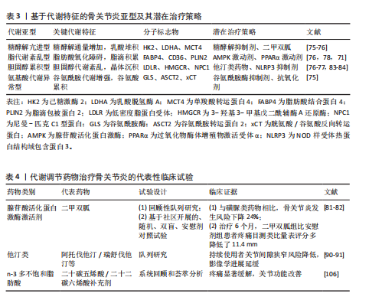
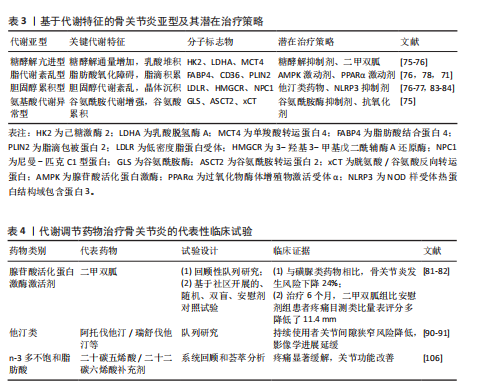
而活性氧抑制剂可降低膝关节软骨损伤严重程度,减少基质金属蛋白酶13和含血小板结合蛋白基序的解聚素样金属蛋白酶5表达,增加Ⅱ型胶原和聚集蛋白聚糖表达[67]。在白细胞介素1β诱导的骨关节炎模型中,氧化应激标志物8-羟基脱氧鸟苷水平升高4倍,添加抗氧化剂可降低基质金属蛋白酶13和含血小板结合蛋白基序的解聚素样金属蛋白酶5表达[68]。研究还发现骨关节炎软骨细胞中活性氧水平升高阻碍线粒体呼吸链,引起线粒体DNA突变,促使基质金属蛋白酶1和基质金属蛋白酶3表达增强[68-69]。 脂质代谢过程包括细胞内脂质的合成和降解,当稳态平衡破坏时易患骨关节炎[19]。研究显示代谢紊乱个体的骨关节炎患病率是对照组的2倍以上[19]。脂代谢紊乱参与骨关节炎进展:肥胖患者血液中升高的游离脂肪酸可进入软骨细胞,在缺氧条件下因β-氧化受阻而堆积,形成脂滴,诱发内质网应激和线粒体功能障碍;胆固醇结晶在关节组织沉积可激活NOD样受体热蛋白结构域相关蛋白3炎症小体,促进白细胞介素1β成熟和释放[19]。能量感应通路在骨关节炎代谢重编程中起调控作用,正常条件下腺苷酸活化蛋白激酶维持能量稳态[70-71];而在骨关节炎中,腺苷酸活化蛋白激酶活性降低,哺乳动物雷帕霉素靶蛋白信号增强,促进合成代谢和细胞应激[72]。实验研究表明,二甲双胍通过激活腺苷酸活化蛋白激酶可减轻骨关节炎动物模型的软骨破坏,医用臭氧通过腺苷酸活化蛋白激酶/哺乳动物雷帕霉素靶蛋白通路增强自噬,抑制白细胞介素1β诱导的基质金属蛋白酶13表达,藤茶黄酮则通过激活该通路改善脂质代谢并减少氧化损伤[50,71,73]。 2.4 骨关节炎代谢异质性分型与精准诊疗前景 2.4.1 基于代谢特征的骨关节炎分型研究 骨关节炎患者存在显著的代谢异质性,这为精准分型提供了理论基础。2024年发表在《Phenomics》的一项研究采用非靶向代谢组学技术分析了不同严重程度骨关节炎患者的关节软骨代谢特征[74]。研究发现,与Kellgren-lawrence(K-L)3级患者相比,K-L 4级患者的胆碱、2-丙基哌啶、鼠李糖和单甲基戊二酸等代谢物显著上调,而1-甲基组胺、鞘磷脂、玉米赤霉醇等代谢物显著下调。这些差异代谢物主要富集于甘油磷脂代谢、组氨酸代谢、赖氨酸降解和甘氨酸/丝氨酸/苏氨酸代谢等通路[74]。 基于代谢组学的分型研究为骨关节炎的精准治疗提供了新思路。通过整合转录组、代谢组等多组学数据,可将骨关节炎分为不同代谢亚型,如糖酵解主导型、脂肪酸氧化障碍型、胆固醇代谢紊乱型等[74]。不同亚型对治疗的反应可能不同,例如糖酵解主导型可能对糖酵解抑制剂更敏感,而胆固醇代谢紊乱型可能对他汀类药物反应更佳[74-78]。 2.4.2 生物信息学驱动的靶点挖掘 生物信息学技术在骨关节炎代谢靶点挖掘中发挥重要作用。2025年发表在《BMC Musculoskeletal Disorders》的一项研究通过整合骨关节炎和动脉粥样硬化的基因表达谱,发现2种疾病共享9个枢纽基因(CCR5、IFIT2、MMP1、CXCL9、RSAD2、IFIH1、TNF、IFIT3和TBX21),均富集于免疫炎症反应通路(如趋化因子信号、干扰素应答),提示慢性炎症是代谢性关节-血管共病的核心机制[8]。 多组学整合分析是挖掘关键代谢靶点的有力工具。通过结合转录组、代谢组和表观遗传组数据,可构建骨关节炎代谢调控网络,识别关键调控节点。例如,基于基因共表达网络分析,研究人员发现胆固醇调节元件结合蛋白1、过氧化物酶体增殖物激活受体γ和肝脏X受体等转录因子在骨关节炎脂代谢紊乱中起核心调控作用[76]。胆固醇调节元件结合蛋白1调控脂肪酸合成酶、硬脂酰辅酶A去饱和酶表达,促进软骨细胞脂质蓄积[76]。过氧化物酶体增殖物激活受体γ介导脂肪细胞分化和脂解过程,其失活导致脂联素分泌减少[76]。肝脏X受体维持胆固醇外流,抑制其活性加剧胆固醇结晶沉积[76-77]。 人工智能在骨关节炎代谢分型和靶点预测中的应用日益广泛。机器学习算法通过分析高维代谢组学数据,识别潜在生物标志物和药物靶点[74]。基于代谢组学数据构建分类器,对骨关节炎亚型的鉴别准确率> 95% (如通过血清胆碱水平区分K-L 4级患者)[74]。深度学习模型可预测患者对不同代谢调节药物的反应,指导个体化治疗。利用图神经网络预测药物反应,例如糖酵解亢进型对二甲双胍的敏感性是脂代谢紊乱型的3.2倍(曲线下面积=0.88)[78]。 2.5 靶向代谢通路的治疗策略 基于对骨关节炎代谢机制的深入理解,药物再定位成为开发骨关节炎新疗法的有效策略。已有多种代谢调节药物在临床前研究中显示出对骨关节炎的治疗潜力。 二甲双胍作为经典的腺苷酸活化蛋白激酶激活剂,可通过多种机制减轻骨关节炎进展:①激活腺苷酸活化蛋白激酶信号,抑制哺乳动物雷帕霉素靶蛋白C1信号,降低白细胞介素1β/肿瘤坏死因子α合成(临床前模型显示软骨降解减少60%)[70,72]。②改善线粒体功能,增强线粒体呼吸链复合物活性,降低活性氧水平(体外实验证实超氧化物歧化酶2表达上调2.1倍)[67]。③阻断核因子κB核转位,减少基质金属蛋白酶13/含血小板结合蛋白基序的解聚素样金属蛋白酶5释放(动物实验显示国际骨关节炎研究学会评分下降40%)[68,79]。动物实验表明,二甲双胍可显著减轻前交叉韧带横断诱导的骨关节炎模型的软骨破坏[80]。一项回顾性队列研究结果显示,与磺脲类药物治疗相比,二甲双胍治疗的糖尿病患者骨关节炎的发生风险降低了24%[81]。2025年发表的一项报告也表明在肥胖和超重膝骨关节炎患者中,相比于安慰剂,持续使用二甲双胍6个月,患者疼痛显著减轻[82]。 他汀类药物因降脂和抗炎双重作用而被研究用于骨关节炎治疗[83-84]。他汀类药物作为3-羟基-3-甲基戊二酰辅酶A还原酶的选择性竞争抑制剂,通过阻断胆固醇内源性合成途径,可显著降低血清胆固醇水平,现已成为治疗高胆固醇血症的核心药物[85-86]。目前临床常用种类包括阿托伐他汀、氟伐他汀、洛伐他汀、匹伐他汀、普伐他汀、瑞舒伐他汀及辛伐他汀[84,86]。基础研究证实,他汀类在骨关节炎治疗中具有多重保护效应:①在培养的骨关节炎软骨细胞模型中,他汀可有效延缓软骨退变进程[83,87];②在多种骨关节炎动物模型(如前交叉韧带离断模型、自发性骨关节炎模型)中显示他汀抑制疾病进展的作用[88-89]。一项为期6.5年的随访研究发现,持续使用他汀类药物可显著延缓膝骨关节炎患者的整体病情进展(影像学与症状评分)[90];针对膝骨关节炎患者的对照研究显示,他汀使用者关节间隙狭窄风险较非使用者显著降低[91]。 过氧化物酶体增殖物激活受体γ在软骨组织中高表达,其激动剂在骨关节炎动物模型中表现出显著的抗炎与软骨保护效应[71,92]。研究发现,在STR/Ort小鼠骨关节炎进展过程中关节软骨过氧化物酶体增殖物激活受体γ表达下调,且在炎症微环境下该现象进一步加剧,导致膝关节软骨损伤及骨赘形成[93]。机制研究表明,过氧化物酶体增殖物激活受体γ可通过介导胆固醇外流途径促进兔骨关节炎软骨细胞的细胞外基质合成。最新证据显示,小泛素样修饰蛋白修饰的过氧化物酶体增殖物激活受体γ能有效改善软骨细胞脂代谢紊乱[94]。这些发现共同表明,激活过氧化物酶体增殖物激活受体γ对骨关节炎具有潜在治疗价值,但调控脂代谢延缓骨关节炎进展的具体分子机制仍需深入探索。与具有软骨保护效应的过氧化物酶体增殖物激活受体α/γ不同,过氧化物酶体增殖物激活受体δ激活可能加速骨关节炎进展[95]。当使用过氧化物酶体增殖物激活受体δ激动剂(GW501516)处理小鼠软骨细胞时,基质降解酶表达显著上调(基质金属蛋白酶2、基质金属蛋白酶3、含血小板结合蛋白基序的解聚素样金属蛋白酶2、含血小板结合蛋白基序的解聚素样金属蛋白酶5 mRNA水平升高),并增强软骨细胞脂肪酸氧化程度[96]。基因敲除研究进一步证实:软骨细胞特异性缺失过氧化物酶体增殖物激活受体δ基因可缓解小鼠内侧半月板切除手术诱导的骨关节炎症状,表明过氧化物酶体增殖物激活受体δ缺失对骨关节炎发展具有抑制作用[96]。过氧化物酶体增殖物激活受体δ加剧骨关节炎的潜在机制可能涉及:促进软骨细胞脂肪酸氧化诱导活性氧大量产生,进而加速软骨退变进程[96]。 前蛋白转化酶枯草溶菌素9(Proprotein convertase subtilisin/kexin type 9,PCSK9)抑制剂通过阻止低密度脂蛋白受体降解,可显著降低低密度脂蛋白胆固醇水平(降幅达50%-70%)。然而,这种临床降脂方案通常需与他汀类药物联用,而非单独使用。值得注意的是,在APOE*3Leiden. CETP小鼠高脂饮食诱导的膝骨关节炎模型中,阿托伐他汀与前蛋白转化酶枯草溶菌素9抑制剂联用虽有效降低血脂,却未能延缓小鼠软骨退变进程[97]。这表明尽管前蛋白转化酶枯草溶菌素9抑制剂对脂代谢紊乱治疗价值明确,但在代谢性骨关节炎中的应用仍存争议。 n-3多不饱和脂肪酸(Polyunsaturated fatty acid,PUFA)作为人体无法合成的必需脂肪酸,主要包括α-亚麻酸、二十碳五烯酸和二十二碳六烯酸,具有显著降低三酰甘油的作用。在白细胞介素1β诱导的软骨细胞及滑膜细胞炎症模型中,n-3多不饱和脂肪酸可降低炎症标志物表达[98-99]。犬软骨细胞实验显示n-3多不饱和脂肪酸减少诱导型一氧化氮合酶表达及一氧化氮生成[100],在人软骨细胞中降低基质金属蛋白酶13与前列腺素E2水平[101]。缓释二十碳五烯酸注射显著延缓手术诱导的小鼠膝骨关节炎进展[102]。但临床研究结果存在矛盾,补充磷虾油改善轻中度膝骨关节炎患者的疼痛、僵硬及关节功能[103],另一项随机对照试验未观察到n-3多不饱和脂肪酸对膝关节症状的显著改善[104]。此外,一项观察性研究表明,全膝关节置换术后血清n-3多不饱和脂肪酸水平与股四头肌肌力恢复呈正相关(r=0.356,P=0.004)[105]。研究提示n-3多不饱和脂肪酸可能通过减轻低度炎症延缓软骨降解[56]。一项荟萃分析结果表明,补充n-3多不饱和脂肪酸可以有效缓解骨关节炎患者疼痛和改善关节功能,而不增加治疗相关的不良事件风险。这些发现支持使用n-3多不饱和脂肪酸补充剂作为骨关节炎的替代治疗方法[106](表3,4)。 2.6 新型靶向治疗策略 多种新型靶向治疗策略正处于临床前或临床研究阶段。脂肪因子靶向治疗是重要研究方向。针对瘦素的单克隆抗体或受体拮抗剂在动物模型中显示出软骨保护作用,中和抗体LepAb在肥胖骨关节炎小鼠中使软骨缺损面积减少45%[76]。增加脂联素水平的策略(如脂联素受体激动剂AdipoRon)在骨关节炎动物模型中也取得了积极效果,AdipoRon通过腺苷酸活化蛋白激酶/过氧化物酶体增殖物激活受体α通路抑制炎症,恢复软骨合成代谢(Ⅱ型胶原表达增加1.8倍)。炎症小体抑制剂是另一重要方向。针对NOD 样受体热蛋白结构域包含蛋白 3(NOD-like receptor pyrin domain-containing protein 3,NLRP3)炎症小体的抑制剂可抑制白细胞介素1β成熟和释放,减轻关节炎症和软骨破坏[77]。代谢酶抑制剂也受到广泛关注。例如,靶向糖酵解关键酶己糖激酶2或乳酸脱氢酶A抑制剂可抑制软骨细胞过度糖酵解,减轻软骨破坏。同样,抑制谷氨酰胺代谢的关键酶谷氨酰胺酶可减少骨关节炎软骨细胞的氧化应激和炎症反应[75]。 2.7 非药物干预策略 生活方式干预是代谢相关骨关节炎的基础治疗策略。研究表明,体质量减轻10%可使膝骨关节炎患者的疼痛减轻50%(西部安大略省和麦克马斯特大学骨关节炎指数),并显著改善关节功能。减重的获益不仅源于机械负荷减轻,还与代谢炎症改善密切相关[76]。营养干预在调节代谢炎症中发挥重要作用,富含ω-3多不饱和脂肪酸的饮食可减少促炎脂肪因子的产生,降低炎症水平[78]。同样,热量限制和间歇性禁食可通过激活自噬、改善线粒体功能等机制减轻骨关节炎进展[78]。运动干预对代谢相关骨关节炎具有双重益处:一方面,运动可减轻体质量,降低关节机械负荷[107];另一方面,运动可改善胰岛素敏感性,增加肌肉力量,改善关节稳定性,并具有抗炎作用[107]。研究显示,水中运动对肥胖骨关节炎患者尤其有益,可在减轻关节负荷的同时提供有效运动刺激[108]。"
| [1] HU Y, CHEN X, WANG S, et al. Subchondral bone microenvironment in osteoarthritis and pain. Bone Res. 2021;9(1):20. [2] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182):1745-1759. [3] LOESER RF, GOLDRING SR, SCANZELLO CR, et al. Osteoarthritis: a disease of the joint as an organ. Arthritis Rheum. 2012;64(6):1697-1707. [4] TONG L, YU H, HUANG X, et al. Current understanding of osteoarthritis pathogenesis and relevant new approaches. Bone Res. 2022; 10(1):60. [5] PLOTZ B, BOMFIM F, SOHAIL MA, et al. Current Epidemiology and Risk Factors for the Development of Hand Osteoarthritis. Curr Rheumatol Rep. 2021;23(8):61. [6] YASUDA E, NAKAMURA R, MATSUGI R, et al. Association between the severity of symptomatic knee osteoarthritis and cumulative metabolic factors. Aging Clin Exp Res. 2018;30(5):481-488. [7] WEI G, LU K, UMAR M, et al. Risk of metabolic abnormalities in osteoarthritis: a new perspective to understand its pathological mechanisms. Bone Res. 2023;11(1):63. [8] JIN Y, ZHANG H. Comprehensive bioinformatics analysis uncover molecular pathways shared between osteoarthritis and atherosclerosis. BMC Musculoskelet Disord. 2025;26(1):449. [9] SELLAM J, BERENBAUM F. Is osteoarthritis a metabolic disease? Joint Bone Spine. 2013;80(6): 568-573. [10] BASAK S, HRIDAYANKA KSN, DUTTAROY AK. Bioactives and their roles in bone metabolism of osteoarthritis: evidence and mechanisms on gut-bone axis. Front Immunol. 2024;14:1323233. [11] HUANG S, CHEN J, ZHANG H, et al. Inflammatory mechanisms underlying metabolic syndrome-associated and potential treatments. Osteoarthr Cartil Open. 2025;7(2):100614. [12] CRUZ CJ, PATTERSON FM, GAIRE J, et al. Comorbid hypertension and osteoarthritis exacerbates joint remodeling and gait compensations in female rats with milder effects observed in males. Osteoarthr Cartil Open. 2025;7(3):100649. [13] SAMPATH SJP, VENKATESAN V, GHOSH S, et al. Obesity, Metabolic Syndrome, and Osteoarthritis-An Updated Review. Curr Obes Rep. 2023;12(3): 308-331. [14] REYES C, LEYLAND KM, PEAT G, et al. Association Between Overweight and Obesity and Risk of Clinically Diagnosed Knee, Hip, and Hand Osteoarthritis: A Population-Based Cohort Study. Arthritis Rheumatol. 2016;68(8):1869-1875. [15] BERENBAUM F, WALLACE IJ, LIEBERMAN DE, et al. Modern-day environmental factors in the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2018;14(11):674-681. [16] LEPETSOS P, PAPAVASSILIOU AG. ROS/oxidative stress signaling in osteoarthritis. Biochim Biophys Acta. 2016;1862(4):576-591. [17] DE SILVA VA, SURAWEERA C, RATNATUNGA SS, et al. Metformin in prevention and treatment of antipsychotic induced weight gain: a systematic review and meta-analysis. BMC Psychiatry. 2016; 16(1):341. [18] LU CH, CHUNG CH, LEE CH, et al. Combination COX-2 inhibitor and metformin attenuate rate of joint replacement in osteoarthritis with diabetes: A nationwide, retrospective, matched-cohort study in Taiwan. PLoS One. 2018;13(1):e0191242. [19] YANG Y, WEI J, LI J, et al. Lipid metabolism in cartilage and its diseases: a concise review of the research progress. Acta Biochim Biophys Sin (Shanghai). 2021;53(5):517-527. [20] COURTIES A, SELLAM J, BERENBAUM F. Metabolic syndrome-associated osteoarthritis. Curr Opin Rheumatol. 2017;29(2):214-222. [21] THIJSSEN E, VAN CAAM A, VAN DER KRAAN PM. Obesity and osteoarthritis, more than just wear and tear: pivotal roles for inflamed adipose tissue and dyslipidaemia in obesity-induced osteoarthritis. Rheumatology (Oxford). 2015;54(4):588-600. [22] VERONESE N, STUBBS B, NOALE M, et al. Adherence to a Mediterranean diet is associated with lower prevalence of osteoarthritis: Data from the osteoarthritis initiative. Clin Nutr. 2017; 36(6):1609-1614. [23] WEN W, FAN H, ZHANG S, et al. Associations between metabolic dysfunction-associated fatty liver disease and atherosclerotic cardiovascular disease. Am J Med Sci. 2024;368(6):557-568. [24] BI X, SUN L, YEO MTY, et al. Integration of metabolomics and machine learning for precise management and prevention of cardiometabolic risk in Asians. Clin Nutr. 2025;50:146-153. [25] ZHONG W, MA M, XIE J, et al. Adipose-specific deletion of the cation channel TRPM7 inhibits TAK1 kinase-dependent inflammation and obesity in male mice. Nat Commun. 2023;14(1):491. [26] ARNETH B. Interactions among nutrition, metabolism and the immune system in the context of starvation and nutrition-stimulated obesity. Nutr Diabetes. 2025;15(1):26. [27] FRIEDMAN JM. Leptin and the endocrine control of energy balance. Nat Metab. 2019; 1(8):754-764. [28] ABELLA V, SCOTECE M, CONDE J, et al. Leptin in the interplay of inflammation, metabolism and immune system disorders. Nat Rev Rheumatol. 2017;13(2):100-109. [29] STANNUS OP, CAO Y, ANTONY B, et al. Cross-sectional and longitudinal associations between circulating leptin and knee cartilage thickness in older adults. Ann Rheum Dis. 2015;74(1):82-88. [30] KANG EH, LEE YJ, KIM TK, et al. Adiponectin is a potential catabolic mediator in osteoarthritis cartilage. Arthritis Res Ther. 2010;12(6):R231. [31] YAMAUCHI T, KAMON J, ITO Y, et al. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature. 2003;423(6941):762-769. [32] YAMAUCHI T, NIO Y, MAKI T, et al. Targeted disruption of AdipoR1 and AdipoR2 causes abrogation of adiponectin binding and metabolic actions. Nat Med. 2007;13(3):332-339. [33] KOSKINEN A, JUSLIN S, NIEMINEN R, et al. Adiponectin associates with markers of cartilage degradation in osteoarthritis and induces production of proinflammatory and catabolic factors through mitogen-activated protein kinase pathways. Arthritis Res Ther. 2011;13(6):R184. [34] HAO D, LI M, WU Z, et al. Synovial fluid level of adiponectin correlated with levels of aggrecan degradation markers in osteoarthritis. Rheumatol Int. 2011;31(11):1433-1437. [35] FUKUHARA A, MATSUDA M, NISHIZAWA M, et al. Visfatin: a protein secreted by visceral fat that mimics the effects of insulin. Science. 2005;307(5708):426-430. [36] ZHU S, BENNETT S, LI Y, et al. The molecular structure and role of LECT2 or CHM-II in arthritis, cancer, and other diseases. J Cell Physiol. 2022; 237(1):480-488. [37] WOLF RM, LEI X, YANG ZC, et al. CTRP3 deficiency reduces liver size and alters IL-6 and TGFβ levels in obese mice. Am J Physiol Endocrinol Metab. 2016;310(5):E332-345. [38] MAGHBOOLI Z, HOSSEIN-NEZHAD A, KHOSHECHIN G, et al. Possible association between circulating CTRP3 and knee osteoarthritis in postmenopausal women. Aging Clin Exp Res. 2019;31(7):927-934. [39] WILLIS SA, SARGEANT JA, YATES T, et al. Acute Hyperenergetic, High-Fat Feeding Increases Circulating FGF21, LECT2, and Fetuin-A in Healthy Men. J Nutr. 2020;150(5):1076-1085. [40] IKEDA D, AGETA H, TSUCHIDA K, et al. iTRAQ-based proteomics reveals novel biomarkers of osteoarthritis. Biomarkers. 2013;18(7):565-572. [41] CHOI WS, LEE G, SONG WH, et al. The CH25H-CYP7B1-RORα axis of cholesterol metabolism regulates osteoarthritis. Nature. 2019;566(7743):254-258. [42] HUANG Y, WAN G, TAO J. C1q/TNF-related protein-3 exerts the chondroprotective effects in IL-1β-treated SW1353 cells by regulating the FGFR1 signaling. Biomed Pharmacother. 2017; 85:41-46. [43] CHANG J, LIAO Z, LU M, et al. Systemic and local adipose tissue in knee osteoarthritis. Osteoarthritis Cartilage. 2018;26(7):864-871. [44] URBAN H, LITTLE CB. The role of fat and inflammation in the pathogenesis and management of osteoarthritis. Rheumatology (Oxford). 2018;57(suppl_4):iv10-iv21. [45] CAO C, SHI Y, ZHANG X, et al. Cholesterol-induced LRP3 downregulation promotes cartilage degeneration in osteoarthritis by targeting Syndecan-4. Nat Commun. 2022; 13(1):7139. [46] WANG T, HE C. Pro-inflammatory cytokines: The link between obesity and osteoarthritis. Cytokine Growth Factor Rev. 2018;44:38-50. [47] GALLO J, RASKA M, KRIEGOVA E, et al. Inflammation and its resolution and the musculoskeletal system. J Orthop Translat. 2017; 10:52-67. [48] GUSS JD, ZIEMIAN SN, LUNA M, et al. The effects of metabolic syndrome, obesity, and the gut microbiome on load-induced osteoarthritis. Osteoarthritis Cartilage. 2019;27(1):129-139. [49] JIANG M, LI X, YU X, et al. Oral Administration of Resveratrol Alleviates Osteoarthritis Pathology in C57BL/6J Mice Model Induced by a High-Fat Diet. Mediators Inflamm. 2017;2017:7659023. [50] LI J, ZHANG B, LIU WX, et al. Metformin limits osteoarthritis development and progression through activation of AMPK signalling. Ann Rheum Dis. 2020;79(5):635-645. [51] CONROZIER T. How to Treat Osteoarthritis in Obese Patients? Curr Rheumatol Rev. 2020;16(2):99-104. [52] JRAD AIS, TRAD M, BZEIH W, et al. Role of pro-inflammatory interleukins in osteoarthritis: a narrative review. Connect Tissue Res. 2023;64(3): 238-247. [53] ROGERO MM, CALDER PC. Obesity, Inflammation, Toll-Like Receptor 4 and Fatty Acids. Nutrients. 2018;10(4):432. [54] FROMMER KW, SCHÄFFLER A, REHART S, et al. Free fatty acids: potential proinflammatory mediators in rheumatic diseases. Ann Rheum Dis. 2015;74(1):303-310. [55] WANG C, YANG F, ZENG W, et al. Vine tea total flavonoids activate the AMPK/mTOR pathway to amelioration hepatic steatosis in mice fed a high-fat diet. J Food Sci. 2024;89(5):3019-3036. [56] ZHUO Q, YANG W, CHEN J, et al. Metabolic syndrome meets osteoarthritis. Nat Rev Rheumatol. 2012;8(12):729-737. [57] CENTERS FOR DISEASE CONTROL AND PREVENTION (CDC). State prevalence of self-reported doctor-diagnosed arthritis and arthritis-attributable activity limitation--United States, 2003. MMWR Morb Mortal Wkly Rep. 2006;55(17):477-481. [58] MOBASHERI A, RAYMAN MP, GUALILLO O, et al. The role of metabolism in the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2017;13(5): 302-311. [59] RASHEED Z, AKHTAR N, HAQQI TM. Advanced glycation end products induce the expression of interleukin-6 and interleukin-8 by receptor for advanced glycation end product-mediated activation of mitogen-activated protein kinases and nuclear factor-κB in human osteoarthritis chondrocytes. Rheumatology (Oxford). 2011; 50(5):838-851. [60] STEENVOORDEN MM, HUIZINGA TW, VERZIJL N, et al. Activation of receptor for advanced glycation end products in osteoarthritis leads to increased stimulation of chondrocytes and synoviocytes. Arthritis Rheum. 2006; 54(1):253-263. [61] MATSUDA M, SHIMOMURA I. Increased oxidative stress in obesity: implications for metabolic syndrome, diabetes, hypertension, dyslipidemia, atherosclerosis, and cancer. Obes Res Clin Pract. 2013;7(5):e330-e341. [62] NIEMANN B, ROHRBACH S, MILLER MR, et al. Oxidative Stress and Cardiovascular Risk: Obesity, Diabetes, Smoking, and Pollution: Part 3 of a 3-Part Series. J Am Coll Cardiol. 2017;70(2):230-251. [63] AHMED B, SULTANA R, GREENE MW. Adipose tissue and insulin resistance in obese. Biomed Pharmacother. 2021;137:111315. [64] PANENI F, BECKMAN JA, CREAGER MA, et al. Diabetes and vascular disease: pathophysiology, clinical consequences, and medical therapy: part I. Eur Heart J. 2013;34(31):2436-2443. [65] RENDRA E, RIABOV V, MOSSEL DM, et al. Reactive oxygen species (ROS) in macrophage activation and function in diabetes. Immunobiology. 2019; 224(2):242-253. [66] ZHOU F, MEI J, YUAN K, et al. Isorhamnetin attenuates osteoarthritis by inhibiting osteoclastogenesis and protecting chondrocytes through modulating reactive oxygen species homeostasis. J Cell Mol Med. 2019;23(6):4395-4407. [67] HAN J, PARK D, PARK JY, et al. Inhibition of NADPH Oxidases Prevents the Development of Osteoarthritis. Antioxidants (Basel). 2022;11(12):2346. [68] GUI T, LUO L, CHHAY B, et al. Superoxide dismutase-loaded porous polymersomes as highly efficient antioxidant nanoparticles targeting synovium for osteoarthritis therapy. Biomaterials. 2022;283:121437. [69] REED KN, WILSON G, PEARSALL A, et al. The role of mitochondrial reactive oxygen species in cartilage matrix destruction. Mol Cell Biochem. 2014;397(1-2):195-201. [70] XING H, LIANG C, WANG C, et al. Metformin mitigates cholesterol accumulation via the AMPK/SIRT1 pathway to protect osteoarthritis chondrocytes. Biochem Biophys Res Commun. 2022;632:113-121. [71] VASHEGHANI F, ZHANG Y, LI YH, et al. PPARγ deficiency results in severe, accelerated osteoarthritis associated with aberrant mTOR signalling in the articular cartilage. Ann Rheum Dis. 2015;74(3):569-578. [72] LIN XL, LIU MH, HU HJ, et al. Curcumin enhanced cholesterol efflux by upregulating ABCA1 expression through AMPK-SIRT1-LXRα signaling in THP-1 macrophage-derived foam cells. DNA Cell Biol. 2015;34(9):561-572. [73] LI J, WANG Y, CHEN D, et al. Oral administration of berberine limits post-traumatic osteoarthritis development and associated pain via AMP-activated protein kinase (AMPK) in mice. Osteoarthritis Cartilage. 2022;30(1):160-171. [74] SUN S, CHEN M, ZHANG T, et al. Identification of Key Factors in Cartilage Tissue During the Progression of Osteoarthritis Using a Non-targeted Metabolomics Strategy. Phenomics. 2024;4(3):227-233. [75] JAISWAL S, MISHRA V, MAJUMDER S, et al. Metabolomic profiling reveals grade-specific niacinamide accumulation and its therapeutic potential via SIRT1-CD38-EMT axis modulation in cervical cancer progression. Biochim Biophys Acta Mol Cell Res. 2025;1872(7):119994. [76] ZHAO C, KONG K, LIU P, et al. Regulating obesity-induced osteoarthritis by targeting p53-FOXO3, osteoclast ferroptosis, and mesenchymal stem cell adipogenesis. Nat Commun. 2025;16(1):4532. [77] WU Y, SHEN S, CHEN J, et al. Metabolite asymmetric dimethylarginine (ADMA) functions as a destabilization enhancer of SOX9 mediated by DDAH1 in osteoarthritis. Sci Adv. 2023;9(6):eade5584. [78] LIANG F, ZHENG Y, ZHAO C, et al. Microalgae-Derived Extracellular Vesicles Synergize with Herbal Hydrogel for Energy Homeostasis in Osteoarthritis Treatment. ACS Nano. 2025;19(8): 8040-8057. [79] BANNURU RR, OSANI MC, VAYSBROT EE, et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthritis Cartilage. 2019;27(11):1578-1589. [80] LU H, WEI J, LIU K, et al. Radical-Scavenging and Subchondral Bone-Regenerating Nanomedicine for Osteoarthritis Treatment. ACS Nano. 2023; 17(6):6131-6146. [81] BAKER MC, SHETH K, LIU Y, et al. Development of Osteoarthritis in Adults With Type 2 Diabetes Treated With Metformin vs a Sulfonylurea. JAMA Netw Open. 2023;6(3):e233646. [82] PAN F, WANG Y, LIM YZ, et al. Metformin for Knee Osteoarthritis in Patients With Overweight or Obesity: A Randomized Clinical Trial. JAMA. 2025;333(20):1804-1812. [83] OESTERLE A, LAUFS U, LIAO JK. Pleiotropic Effects of Statins on the Cardiovascular System. Circ Res. 2017;120(1):229-243. [84] SABERIANPOUR S, ABOLBASHARI S, MODAGHEGH MHS, et al. Therapeutic effects of statins on osteoarthritis: A review. J Cell Biochem. 2022; 123(8):1285-1297. [85] GAI Z, WANG T, VISENTIN M, et al. Lipid Accumulation and Chronic Kidney Disease. Nutrients. 2019;11(4):722. [86] ISTVAN ES, DEISENHOFER J. Structural mechanism for statin inhibition of HMG-CoA reductase. Science. 2001;292(5519):1160-1164. [87] DU J, JIANG Q, MEI L, et al. Effect of high fat diet and excessive compressive mechanical force on pathologic changes of temporomandibular joint. Sci Rep. 2020;10(1):17457. [88] TANAKA T, MATSUSHITA T, NISHIDA K, et al. Attenuation of osteoarthritis progression in mice following intra-articular administration of simvastatin-conjugated gelatin hydrogel. J Tissue Eng Regen Med. 2019;13(3):423-432. [89] ZHOU B, CHEN D, XU H, et al. Proliferation of rabbit chondrocyte and inhibition of IL-1β-induced apoptosis through MEK/ERK signaling by statins. In Vitro Cell Dev Biol Anim. 2017;53(2):124-131. [90] CLOCKAERTS S, VAN OSCH GJ, BASTIAANSEN-JENNISKENS YM, et al. Statin use is associated with reduced incidence and progression of knee osteoarthritis in the Rotterdam study. Ann Rheum Dis. 2012;71(5):642-647. [91] HAJ-MIRZAIAN A, MOHAJER B, GUERMAZI A, et al. Statin Use and Knee Osteoarthritis Outcome Measures according to the Presence of Heberden Nodes: Results from the Osteoarthritis Initiative. Radiology. 2019;293(2):396-404. [92] SABATINI M, BARDIOT A, LESUR C, et al. Effects of agonists of peroxisome proliferator-activated receptor gamma on proteoglycan degradation and matrix metalloproteinase production in rat cartilage in vitro. Osteoarthritis Cartilage. 2002;10(9):673-679. [93] WATTERS JW, CHENG C, PICKARSKI M, et al. Inverse relationship between matrix remodeling and lipid metabolism during osteoarthritis progression in the STR/Ort mouse. Arthritis Rheum. 2007;56(9):2999-3009. [94] WANG X, WANG D, XIA P, et al. Ultrasound-targeted simvastatin-loaded microbubble destruction promotes OA cartilage repair by modulating the cholesterol efflux pathway mediated by PPARγ in rabbits. Bone Joint Res. 2021;10(10):693-703. [95] WANG H, SHI Y, HE F, et al. GDF11 inhibits abnormal adipogenesis of condylar chondrocytes in temporomandibular joint osteoarthritis. Bone Joint Res. 2022;11(7):453-464. [96] RATNESWARAN A, LEBLANC EA, WALSER E, et al. Peroxisome proliferator-activated receptor δ promotes the progression of posttraumatic osteoarthritis in a mouse model. Arthritis Rheumatol. 2015;67(2):454-464. [97] VAN GEMERT Y, KOZIJN AE, POUWER MG, et al. Novel high-intensive cholesterol-lowering therapies do not ameliorate knee OA development in humanized dyslipidemic mice. Osteoarthritis Cartilage. 2021;29(9):1314-1323. [98] LOEF M, SCHOONES JW, KLOPPENBURG M, et al. Fatty acids and osteoarthritis: different types, different effects. Joint Bone Spine. 2019; 86(4):451-458. [99] WU CL, JAIN D, MCNEILL JN, et al. Dietary fatty acid content regulates wound repair and the pathogenesis of osteoarthritis following joint injury. Ann Rheum Dis. 2015;74(11):2076-2083. [100] ADLER N, SCHOENIGER A, FUHRMANN H. Polyunsaturated fatty acids influence inflammatory markers in a cellular model for canine osteoarthritis. J Anim Physiol Anim Nutr (Berl). 2018;102(2):e623-e632. [101] YU H, LI Y, MA L, et al. A low ratio of n-6/n-3 polyunsaturated fatty acids suppresses matrix metalloproteinase 13 expression and reduces adjuvant-induced arthritis in rats. Nutr Res. 2015;35(12):1113-1121. [102] TSUBOSAKA M, KIHARA S, HAYASHI S, et al. Gelatin hydrogels with eicosapentaenoic acid can prevent osteoarthritis progression in vivo in a mouse model. J Orthop Res. 2020;38(10):2157-2169. [103] STONEHOUSE W, BENASSI-EVANS B, BEDNARZ J, et al. Krill oil improved osteoarthritic knee pain in adults with mild to moderate knee osteoarthritis: a 6-month multicenter, randomized, double-blind, placebo-controlled trial. Am J Clin Nutr. 2022;116(3):672-685. [104] MACFARLANE LA, COOK NR, KIM E, et al. The Effects of Vitamin D and Marine Omega-3 Fatty Acid Supplementation on Chronic Knee Pain in Older US Adults: Results From a Randomized Trial. Arthritis Rheumatol. 2020;72(11):1836-1844. [105] 黄泽晓,杨妹,林诗炜,等.血清n-3多不饱和脂肪酸水平与全膝关节置换早期股四头肌肌力变化的相关性[J].中国组织工程研究, 2021,25(9):1375-1380. [106] DENG W, YI Z, YIN E, et al. Effect of omega-3 polyunsaturated fatty acids supplementation for patients with osteoarthritis: a meta-analysis. J Orthop Surg Res. 2023;18(1):381. [107] HOCHBERG MC, ALTMAN RD, APRIL KT, et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res (Hoboken). 2012;64(4):465-474. [108] 位长强,于洪建,刘宁宁,等.下肢力线调整区域对胫骨高位截骨后膝关节功能及运动学的影响[J].中国组织工程研究,2025,29(27):5743-5749. |
| [1] | Lyu Guoqing, Aizimaitijiang·Rouzi, Xiong Daohai. Irisin inhibits ferroptosis in human articular chondrocytes: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1359-1367. |
| [2] | Liu Huan, Zeng Shaopeng, Chen Jun, He Linqian, Yang Ying, Zhang Jing. Aging-related dysregulation of glucose metabolism: crossroads of cancer and neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1527-1538. |
| [3] | Zhang Qian, Huang Dongfeng. Weighted gene co-expression network analysis combined with machine learning to screen and validate biomarkers for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1096-1105. |
| [4] | Bu Yangyang, Ning Xinli, Zhao Chen. Intra-articular injections for the treatment of osteoarthritis of the temporomandibular joint: different drugs with multiple combined treatment options [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1215-1224. |
| [5] | Gao Zengjie, , Pu Xiang, Li Lailai, Chai Yihui, Huang Hua, Qin Yu. Increased risk of osteoporotic pathological fractures associated with sterol esters: evidence from IEU-GWAS and FinnGen databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1302-1310. |
| [6] | Wei Bingqi, Zhang Xinyue, Ren Xingyue, Sun Jiahui, Chen Liu, Li Yijing, Qi Yifan, Wang Shangzeng. Zinc finger DHHC-type containing 2 emerges as a novel therapeutic target in osteoarthritis pathogenesis: genome-wide data analysis in European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7715-7723. |
| [7] | Li Zhengpeng, Shao Weigang, Zeng Hao, Xiang Kelin, Zhang Botao, Zou Shunyi, Chen Sheng, Qi Wen. Osteoarthritis characteristic genes and prediction of targeted food-medicine homology traditional Chinese medicine: bioinformatics analysis and kinetic simulation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7739-7748. |
| [8] | Sun Zhiyuan, Xu Kai, Tian Xuewen, Shang Qinghui. Metabolic dysfunction-related fatty liver disease: pathological mechanisms mediated by common and heterogeneous pathways [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7378-7387. |
| [9] | Su Jiemao, Qi Yansong, Kong Keyu, Zhai Zanjing, Xu Yongsheng. Role of chondrocyte ferroptosis in the pathogenesis of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6282-6288. |
| [10] | Li Zijing, Chen Xuwu, Ouyang Xinye, Wang Maoyuan. Mitophagy impairment mediated muscular atrophy: insights from the Drosophila model [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 5897-5905. |
| [11] | Zhou Menghan, Liu Shuning, Jiang Tao, Sun Zhuangzhuang, Cao Lingling, Su Xin, Yu Cheng, Guo Junpeng. Systematic druggable genome-wide Mendelian randomization identifies therapeutic targets for major depressive disorder [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5886-5896. |
| [12] | Xu Peng, Jiang Wei, Yu You, Lei Zhengliang, Tian Yang, Zhang Jie, Liu Luchang. Cobalt chloride-induced hypoxic environment accelerates knee cartilage degeneration in New Zealand rabbits [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5650-5658. |
| [13] | Han Jie, Hu Tianfa, Wu Yachao, Nong Bin, Yu Kailong. Forkhead box transcription factor O3 affects bone metabolism and participates in the pathological processes of various bone-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5770-5781. |
| [14] | Liao Xingzhuan, Li Guangdi, Wu Yabin, Liu Xingyu, Wan Jiajia. Molecular mechanisms underlying non-coding RNA regulation of ferroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4713-4725. |
| [15] | Chen Xinlong, Meng Tao, Wang Yaomin, Zhang Kefan, Li Jian, Shi Hui, Zhang Chenchen. Ferroptosis inhibitors in the treatment of osteoarthritis: diversity and multitarget characteristics [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4166-4179. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||