Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (29): 7592-7602.doi: 10.12307/2026.197
Previous Articles Next Articles
Lycium barbarum polysaccharide-mediated intestinal flora remodeling improves glycolipid abnormalities in type 2 diabetic rats
Wang Jingfeng, Feng Shuo, Cao Xuan, Li Xiaolin
- Harbin Sport University, Harbin 150008, Heilongjiang Province, China
-
Received:2025-06-30Revised:2025-09-29Online:2026-10-18Published:2026-03-04 -
Contact:Li Xiaolin, PhD, Professor, Doctoral supervisor, Harbin Sport University, Harbin 150008, Heilongjiang Province, China -
About author:Wang Jingfeng, PhD candidate, Harbin Sport University, Harbin 150008, Heilongjiang Province, China -
Supported by:Heilongjiang Provincial Natural Science Foundation, No. LH2024G003 (to LXL); Harbin Sport University Laboratory Platform Special Project, No. LAB2021-06 (to LXL)
CLC Number:
Cite this article
Wang Jingfeng, Feng Shuo, Cao Xuan, Li Xiaolin. Lycium barbarum polysaccharide-mediated intestinal flora remodeling improves glycolipid abnormalities in type 2 diabetic rats[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7592-7602.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

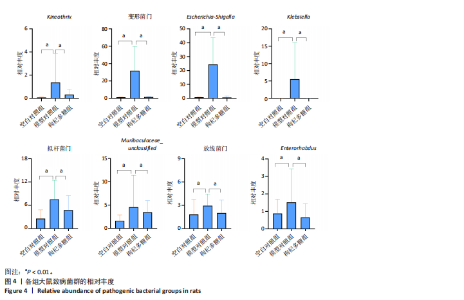
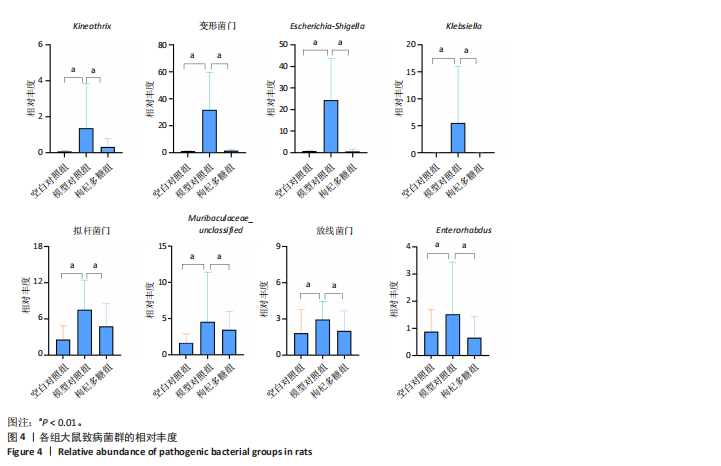
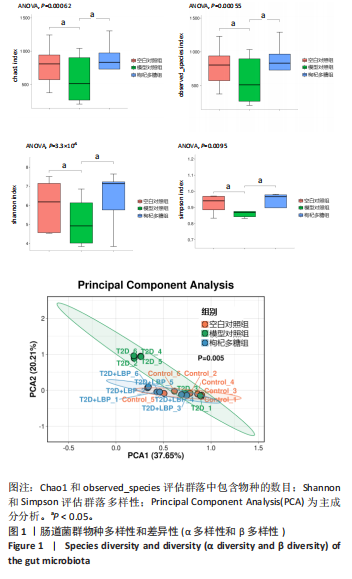
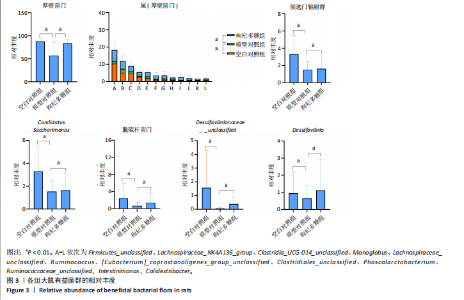
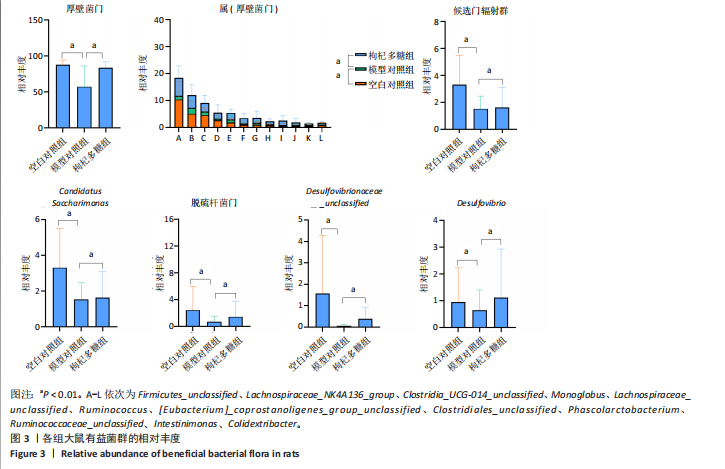
2.1 枸杞多糖对2型糖尿病大鼠肠道菌群多样性和丰度的影响 与空白对照组相比,模型对照组Chao1、Observed_species、Shannon显著降低(P < 0.01)。与模型对照组相比,枸杞多糖组Chao1、Observed_species、Shannon显著升高(P < 0.01)。根据主成分分析显示,空白对照组与模型对照组的菌群组成结构存在显著差异,模型对照组与枸杞多糖组的菌群组成结构存在显著差异,见图1。 门:与空白对照组相比,模型对照组厚壁菌门、候选门辐射群和脱硫杆菌门显著降低(P < 0.01),变形菌门、拟杆菌门和放线菌门显著升高(P < 0.01)。与模型对照组相比,枸杞多糖组厚壁菌门、候选门辐射群和脱硫杆菌门显著升高(P < 0.01),变形菌门、拟杆菌门和放线菌门显著降低(P < 0.01),见图2-4。 属:与空白对照组相比,模型对照组Firmicutes_unclassified、Lachnospiraceae_NK4A136_group、Clostridia_UCG-014_unclassified、Monoglobus、Lachnospiraceae_unclassified、Ruminococcus、Eubacterium]_coprostanoligenes_group_unclassified、Clostridiales_unclassified、Phascolarctobacterium、Ruminococcaceae_unclassified、Intestinimonas、Colidextribacter、Desulfovibrionaceae "
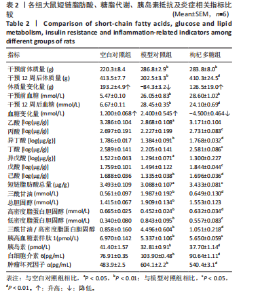
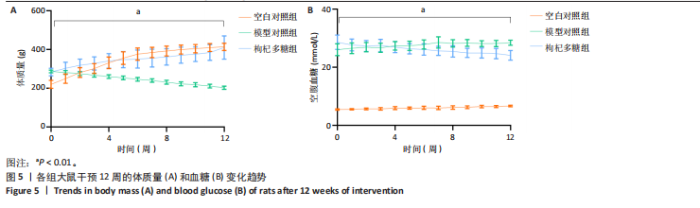
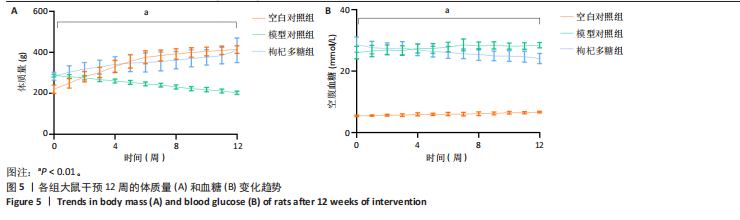
unclassified和Desulfovibrio显著降低(P < 0.01),Kineothrix、Escherichia-Shigella、Klebsiella、Muribaculaceae_unclassified和Enterorhabdus显著升高(P < 0.01)。与模型对照组相比,枸杞多糖组Firmicutes_unclassified、Lachnospiraceae_NK4A136_group、Clostridia_UCG-014_unclassified、Monoglobus、Lachnospiraceae_unclassified、Ruminococcus、Eubacterium]_coprostanoligenes_group_unclassified、Clostridiales_unclassified、Phascolarctobacterium、Ruminococcaceae_unclassified、Intestinimonas、Colidextribacter、Desulfovibrionaceae unclassified和Desulfovibrio显著升高(P < 0.01),Kineothrix、Escherichia-Shigella、Klebsiella、Muribaculaceae_unclassified和Enterorhabdus显著降低(P < 0.01),见图2-4。 2.2 枸杞多糖对2型糖尿病大鼠短链脂肪酸、糖脂代谢、胰岛素抵抗以及炎症的影响 与空白对照组相比,模型对照组乙酸、异丁酸、异戊酸、己酸以及短链脂肪酸总量均显著降低(P < 0.05或P < 0.01);与模型对照组相比,枸杞多糖组丙酸、异丁酸、丁酸、戊酸、己酸以及短链脂肪酸总量均显著升高(P < 0.05或P < 0.01),见表2。 造模后干预前,与空白对照组相比,模型对照组体质量显"
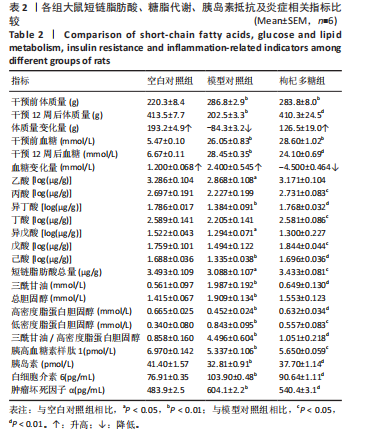

著升高;与模型对照组相比,枸杞多糖组体质量未见显著差异(P > 0.05);干预12周后,与空白对照组相比,模型对照组体质量显著降低,与模型对照组相比,枸杞多糖组体质量显著升高( P < 0.01),见表2;空白对照组与枸杞多糖组的体质量呈上升趋势,模型对照组体质量呈下降趋势(P < 0.01),见图5A,表2。造模后干预前,与空白对照组相比,模型对照组血糖显著升高;与模型对照组相比,枸杞多糖组血糖未见显著差异(P > 0.05);干预12周后,与空白对照组相比,模型对照组血糖显著升高;与模型对照组相比,枸杞多糖组血糖显著降低(P < 0.01),见表2;空白对照组与模型对照组的血糖呈上升趋势,枸杞多糖组的血糖呈下降趋势(P < 0.01),见图5B,表2。与空白对照组相比,模型对照组三酰甘油、总胆固醇、低密度脂蛋白胆固醇水平显著升高(P < 0.01),高密度脂蛋白胆固醇和胰高血糖素肽1水平显著降低(P < 0.01);与模型对照组相比,枸杞多糖组高密度脂蛋白胆固醇和胰高血糖素肽1水平显著升高(P < 0.05或P < 0.01),三酰甘油和低密度脂蛋白胆固醇水平显著降低(P < 0.05或P < 0.01),见表2。 与空白对照组相比,模型对照组三酰甘油/高密度脂蛋白胆固醇水平显著升高(P < 0.01),胰岛素水平显著降低(P < 0.01);与模型对照组相比,枸杞多糖组胰岛素水平显著升高(P < 0.01),三酰甘油/高密度脂蛋白胆固醇水平显著降低(P < 0.01),见表2。 与空白对照组相比,模型对照组白细胞介素6和肿瘤坏死因子α水平显著升高(P < 0.01);与模型对照组相比,枸杞多糖组白细胞介素6和肿瘤坏死因子α水平显著降低(P < 0.01),见表2。"

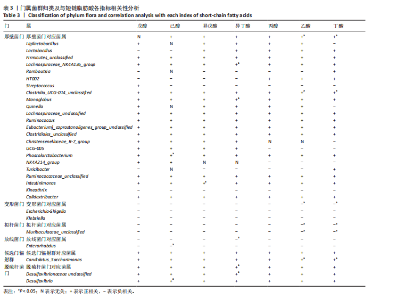
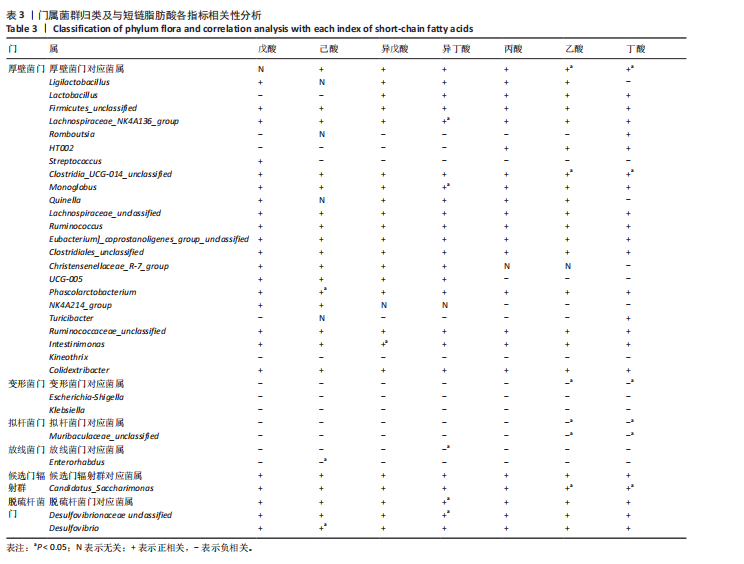
2.3 枸杞多糖对2型糖尿病大鼠肠道菌群与短链脂肪酸、糖脂代谢、胰岛素抵抗以及炎症指标相关性的影响 门:厚壁菌门与乙酸和丁酸呈显著正相关(P < 0.05);变形菌门与乙酸和丁酸呈显著负相关(P < 0.05);拟杆菌门与乙酸和丁酸呈显著负相关(P < 0.05);放线菌门与异丁酸呈显著负相关(P < 0.05);脱硫杆菌门与异丁酸呈显著负相关(P < 0.05),见图2D,表3。 属:Lachnospiraceae_NK4A136_group与异丁酸呈显著正相关(P < 0.05);Clostridia_UCG-014_unclassified与乙酸和丁酸呈显著正相关(P < 0.05);Monoglobus与异丁酸呈显著正相关(P < 0.05);Phascolarctobacterium与己酸呈显著正相关(P < 0.05); Intestinimonas与异戊酸呈显著正相关(P < 0.05);Muribaculaceae_unclassified与乙酸和丁酸呈显著正相关(P < 0.05);Enterorhabdus与己酸呈显著负相关(P < 0.05);Candidatus_Saccharimonas与乙酸和丁酸呈显著正相关(P < 0.05);Desulfovibrionaceae unclassified与异丁酸呈显著正相关(P < 0.05);Desulfovibrio与己酸呈显著正"

著正相关(P < 0.05);Kineothrix与低密度脂蛋白胆固醇呈显著正相关(P < 0.05);Colidextribacter与低密度脂蛋白胆固醇呈显著负相关(P < 0.05);Muribaculaceae_unclassified与三酰甘油呈显著正相关(P < 0.05),与胰高血糖素肽1呈显著负相关(P < 0.05);Candidatus_Saccharimonas与三酰甘油呈显著负相关(P < 0.05),与胰高血糖素肽1呈显著正相关(P < 0.05);Desulfovibrionaceae unclassified与高密度脂蛋白胆固醇呈显著正相关(P < 0.05),见图2C,表4。 门:放线菌门与三酰甘油/高密度脂蛋白胆固醇呈显著正相关(P < 0.05);脱硫杆菌门与三酰甘油/高密度脂蛋白胆固醇呈显著负相关(P < 0.05),见图2D,表4。 属:Lachnospiraceae_NK4A136_group与三酰甘油/高密度脂蛋白胆固醇呈显著负相关(P < 0.05);Monoglobus与三酰甘油/高密度脂蛋白胆固醇呈显著负相关(P < 0.05);Desulfovibrionaceae unclassified与三酰甘油/高密度脂蛋白胆固醇呈显著负相关(P < 0.05),见图2C,表4。 属:Firmicutes_unclassified与肿瘤坏死因子α呈显著负相关(P < 0.05);Monoglobus与白细胞介素6呈显著负相关(P < 0.05);Intestinimonas与白细胞介素6呈显著负相关(P < 0.05);Kineothrix与肿瘤坏死因子α呈显著正相关(P < 0.05);Colidextribacter 与肿瘤坏死因子α呈显著负相关(P < 0.05),见图2C,表4。"

| [1] YEHUALASHET AS, YIKNA BB. Microbial Ecosystem in Diabetes Mellitus: Consideration of the Gastrointestinal System. Diabetes Metab Syndr Obes. 2021;14:1841-1854. [2] ZHOU Z, SUN B, YU D, et al. Gut Microbiota: An Important Player in Type 2 Diabetes Mellitus. Front Cell Infect Microbiol. 2022;12:834485. [3] LI ZX, ZHUO JL, YANG N, et al. Effect of Lycium barbarum polysaccharide on osteoblast proliferation and differentiation in postmenopausal osteoporosis. Int J Biol Macromol. 2024;271(Pt 1):132415. [4] MA Q, ZHAI R, XIE X, et al. Hypoglycemic Effects of Lycium barbarum Polysaccharide in Type 2 Diabetes Mellitus Mice via Modulating Gut Microbiota. Front Nutr. 2022;9:916271. [5] DING H, YANG P, ZHANG XH, et al. Efficacy of Pretreatment with Lycium barbarum Polysaccharide in Various Doses in Influencing Splenic Immunity and Prognosis of Sepsis in Rats. Evid Based Complement Alternat Med. 2022;2022:9508603. [6] LIU RJ, HE YJ, LIU H, et al. Protective effect of Lycium barbarum polysaccharide on di-(2-ethylhexyl) phthalate-induced toxicity in rat liver. Environ Sci Pollut Res Int. 2021;28(18):23501-23509. [7] LIU Q, HAN Q, LU M, et al. Lycium barbarum polysaccharide attenuates cardiac hypertrophy, inhibits calpain-1 expression and inhibits NF-κB activation in streptozotocin-induced diabetic rats. Exp Ther Med. 2019; 18(1):509-516. [8] LAI W, WANG C, LAI R, et al. Lycium barbarum polysaccharide modulates gut microbiota to alleviate rheumatoid arthritis in a rat model. NPJ Sci Food. 2022;6(1):34. [9] HAN Q, WANG J, LI W, et al. Androgen-induced gut dysbiosis disrupts glucolipid metabolism and endocrinal functions in polycystic ovary syndrome. Microbiome. 2021;9(1):101. [10] MAYNERIS-PERXACHS J, CARDELLINI M, HOYLES L, et al. Iron status influences non-alcoholic fatty liver disease in obesity through the gut microbiome. Microbiome. 2021;9(1):104. [11] LIU L, ZHANG J, CHENG Y, et al. Gut microbiota: A new target for T2DM prevention and treatment. Front Endocrinol (Lausanne). 2022;13:958218. [12] 韩云琰,江华,刘献萍,等.基于肠道菌群和代谢组学研究牡蛎多糖改善小鼠2型糖尿病的作用机制[J].食品工业科技,2026,47(3):420-431. [13] FRANK J, GUPTA A, OSADCHIY V, et al. Brain-Gut-Microbiome Interactions and Intermittent Fasting in Obesity. Nutrients. 2021;13(2):584. [14] ZHENG Y, LEY SH, HU FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018; 14(2):88-98. [15] YI C, HUANG S, ZHANG W, et al. Synergistic interactions between gut microbiota and short chain fatty acids: Pioneering therapeutic frontiers in chronic disease management. Microb Pathog. 2025;199:107231. [16] PALMAS V, PISANU S, MADAU V, et al. Gut microbiota markers associated with obesity and overweight in Italian adults. Sci Rep. 2021;11(1):5532. [17] LI SX, GUO Y. Gut microbiome: New perspectives for type 2 diabetes prevention and treatment. World J Clin Cases. 2023;11(31):7508-7520. [18] MOKKALA K, HOUTTU N, CANSEV T, et al. Interactions of dietary fat with the gut microbiota: Evaluation of mechanisms and metabolic consequences. Clin Nutr. 2020;39(4):994-1018. [19] ARAGÓN-VELA J, SOLIS-URRA P, RUIZ-OJEDA FJ, et al. Impact of Exercise on Gut Microbiota in Obesity. Nutrients. 2021;13(11):3999. [20] PERRY RJ, PENG L, BARRY NA, et al. Acetate mediates a microbiome-brain-β-cell axis to promote metabolic syndrome. Nature. 2016;534(7606):213-217. [21] HE J, ZHANG P, SHEN L, et al. Short-Chain Fatty Acids and Their Association with Signalling Pathways in Inflammation, Glucose and Lipid Metabolism. Int J Mol Sci. 2020;21(17):6356. [22] MA Q, LI Y, LI P, et al. Research progress in the relationship between type 2 diabetes mellitus and intestinal flora. Biomed Pharmacother. 2019;117: 109138. [23] 杨启航,蒲锐,陈子扬,等.肠道菌群代谢物在肥胖调控中的作用与机制[J].中国组织工程研究,2024,28(2):308-314. [24] SAWICKI CM, LIVINGSTON KA, OBIN M, et al. Dietary Fiber and the Human Gut Microbiota: Application of Evidence Mapping Methodology. Nutrients. 2017;9(2):125. [25] LA FATA G, WEBER P, MOHAJERI MH. Probiotics and the Gut Immune System: Indirect Regulation. Probiotics Antimicrob Proteins. 2018; 10(1):11-21. [26] BÄUMLER AJ, SPERANDIO V. Interactions between the microbiota and pathogenic bacteria in the gut. Nature. 2016;535(7610):85-93. [27] PERDIJK O, AZZONI R, MARSLAND BJ. The microbiome: an integral player in immune homeostasis and inflammation in the respiratory tract. Physiol Rev. 2024;104(2):835-879. [28] RATAJCZAK W, RYŁ A, MIZERSKI A, et al. Immunomodulatory potential of gut microbiome-derived short-chain fatty acids (SCFAs). Acta Biochim Pol. 2019;66(1):1-12. [29] 唐甜,索化夷,李键,等.膳食营养与宿主遗传通过肠道菌群调节能量代谢研究进展[J].食品与机械,2019,35(3):225-230. [30] SOWMIYA T, SILAMBANAN S. Association of Gut Microbiota and Diabetes Mellitus. Curr Diabetes Rev. 2023;19(7):e211122211066. [31] GUO Q, LI Y, DAI X, et al. Polysaccharides: The Potential Prebiotics for Metabolic Associated Fatty Liver Disease (MAFLD). Nutrients. 2023; 15(17):3722. [32] PI Y, FANG M, LI Y, et al. Interactions between Gut Microbiota and Natural Bioactive Polysaccharides in Metabolic Diseases: Review. Nutrients. 2024; 16(17):2838. [33] REN F, MENG C, CHEN W, et al. Ganoderma amboinense polysaccharide prevents obesity by regulating gut microbiota in high-fat-diet mice. Food Bioscience. 2021;42:101107. [34] CAO C, WANG Z, GONG G, et al. Effects of Lycium barbarum Polysaccharides on Immunity and Metabolic Syndrome Associated with the Modulation of Gut Microbiota: A Review. Foods. 2022;11(20):3177. [35] CAO C, WANG L, AI C, et al. Impact of Lycium barbarum arabinogalactan on the fecal metabolome in a DSS-induced chronic colitis mouse model. Food Funct. 2022;13(16):8703-8716. [36] WANG X, WANG X, JIANG H, et al. Marine polysaccharides attenuate metabolic syndrome by fermentation products and altering gut microbiota: An overview. Carbohydr Polym. 2018;195:601-612. [37] ZHANG X, ZHANG N, KAN J, et al. Anti-inflammatory activity of alkali-soluble polysaccharides from Arctium lappa L. and its effect on gut microbiota of mice with inflammation. Int J Biol Macromol. 2020;154:773-787. [38] TANCA A, ABBONDIO M, PALOMBA A, et al. Potential and active functions in the gut microbiota of a healthy human cohort. Microbiome. 2017;5(1):79. [39] 郑志,姜林娟,朱绍辉,等.益生菌-海带复合发酵液对肥胖大鼠肠道菌群的影响及抗肥胖效果[J].郑州大学学报(医学版),2022,57(3):308-314. [40] 杨雨,肖定福,邢月腾,等.外源性营养物质与肠道微生物群-肠-脑轴互作途径及机制研究进展[J].动物营养学报,2023,35(9):5578-5588. [41] WILLIAMS BA, ZHANG D, LISLE AT, et al. Soluble arabinoxylan enhances large intestinal microbial health biomarkers in pigs fed a red meat-containing diet. Nutrition. 2016;32(4):491-497. |
| [1] | Wen Xiaolong, Weng Xiquan, Feng Yao, Cao Wenyan, Liu Yuqian, Wang Haitao. Effects of inflammation on serum hepcidin and iron metabolism related parameters in patients with type 2 diabetes mellitus: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1294-1301. |
| [2] | Yan Chengbo, Luo Qiuchi, Fan Jiabing, Gu Yeting, Deng Qian, Zhang Junmei. Effect of type 2 diabetes mellitus on orthodontic tooth movement and bone microstructure parameters on the tension side in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 824-831. |
| [3] | Gu Fucheng, Yang Meixin, Wu Weixin, Cai Weijun, Qin Yangyi, Sun Mingyi, Sun Jian, Geng Qiudong, Li Nan. Effects of Guilu Erxian Glue on gut microbiota in rats with knee osteoarthritis: machine learning and 16S rDNA analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 1058-1072. |
| [4] |
Zhao Yanan, Cao Liquan, Tan Sijie.
A new perspective on exercise for the prevention and treatment of type 2 diabetes mellitus: pyroptosis#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7673-7679.
|
| [5] | Fan Zhiliang, Chai Yihui, Chen Guanglei, Li Qian, Gu Chunsong, Chen Yunzhi, Li Wen, Wu Damei, Pu Xiang. Pathogenesis and potential therapeutic targets of idiopathic pulmonary fibrosis: analysis of data from a large-scale genome-wide association study [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7465-7474. |
| [6] | Wang Wei, Chen Jun, Jia Shaohui, Xue Xinxuan, Dong Kunwei. Mechanisms by which exercise regulates gut microbiota in the prevention and treatment of non-alcoholic fatty liver disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6328-6336. |
| [7] | Zhang Zheng, Zhang Yibo, Xu Bin, Yan Shichao, Guo Hui. Sarcopenia and non-alcoholic fatty liver disease: analysis of the gut microbiota [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6000-6009. |
| [8] | Li Yiguang, Guo Haonan, Ding Xiaotao, Yuan Mengyao, Jiang Lijin, Fan Xinfeng, Feng Yan. Visual analysis of research hotspots in the field of gut microbiota in the elderly at home and abroad [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6071-6080. |
| [9] | Chai Jinlian, Liang Xuezhen, Sun Tiefeng, Li Shudong, Li Wei, Li Guangzheng, Yu Huayun, Wang Ping. Mechanistic insights into how Cervi Cornus Colla regulates the intestinal flora-bile acid metabolic pathway to alleviate steroid-induced osteonecrosis of the femoral head in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4568-4581. |
| [10] | Wu Yilin, Tian Hongying, Sun Jiale, Jiao Jiajia, Zhao Zihan, Shao Jinhuan, Zhao Kaiyue, Zhou Min, Li Qian, Li Zexin, Yue Changwu. Intervention effect and mechanism of Compound Herba Gueldenstaedtiae in a mouse model of breast hyperplasia [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4377-4389. |
| [11] | Zou Yuxiong, Liu Xiaomeng, Liu Ying, Zhu Yue, Li Shuming, Guo Fangyang, Yu Xinyu, Nie Heyun, Liu Qian, Ao Meiying. Cerebral palsy decoction improves cerebral palsy in male and female young rats: mechanisms based on the “gut-brain-muscle” axis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4054-4066. |
| [12] | Niu Qi, Chen Junji, Tu Haining, Mo Weibin, Zhong Yujin, Li Mingliang. Effect of swimming exercise combined with probiotic intervention on anti-inflammatory and apoptotic gene expression in renal tissue of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(16): 4105-4114. |
| [13] | Yao Lijuan, Wang Yinfeng, Ma Zhennan, Chen Leqin. Exercise-induced extracellular vesicles: action and mechanisms in occurrence and development of insulin resistance [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3412-3423. |
| [14] | Wu Fangjia, Lei Senlin, Li Xianhui, Yang Yang. Aerobic and resistance exercise interventions in a mouse model of nonalcoholic fatty liver disease: correlation between gut microbiota and irisin [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(12): 3029-3043. |
| [15] | Sun Long, Wu Haiyang, Tong Linjian, Liu Rui, Yang Weiguang, Xiao Jian, Liu Lice, Sun Zhiming. Regulatory mechanism of leptin in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(12): 3100-3108. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||