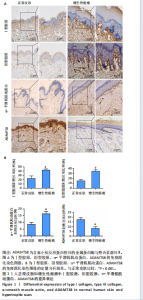
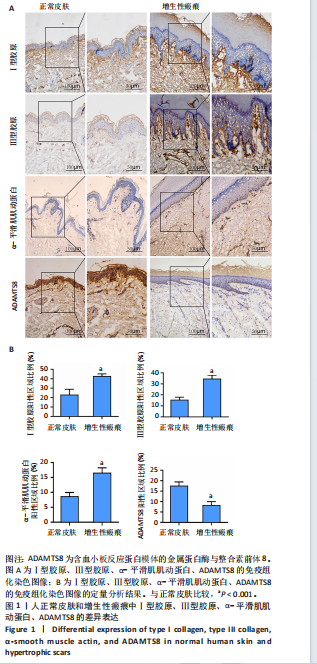
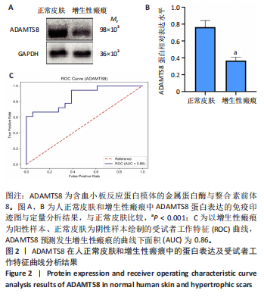
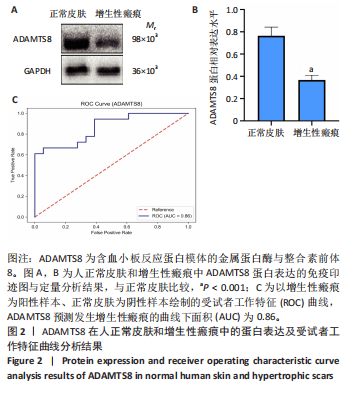
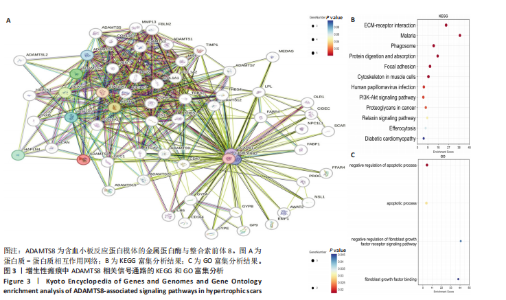
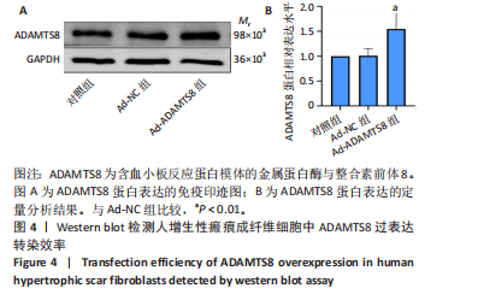
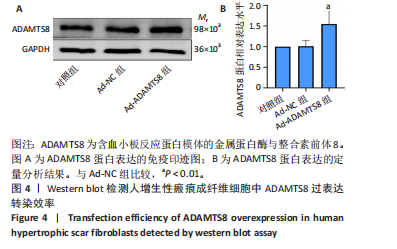
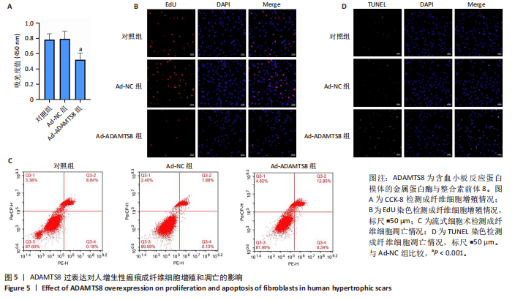
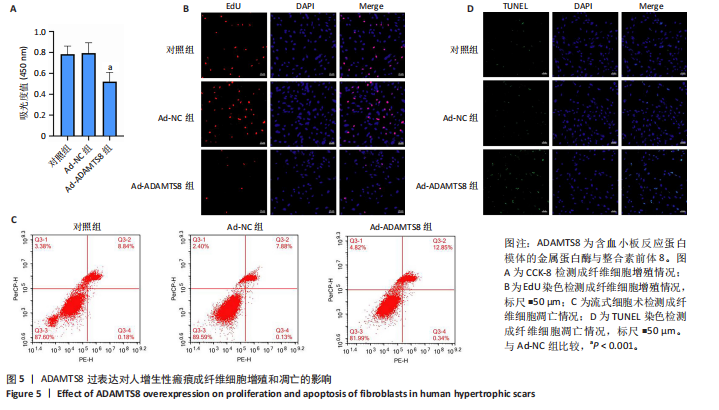
[1] 蔡景龙. 瘢痕防治的历史与现状[J].中华损伤与修复杂志(电子版) ,2017, 12(4):241-246.
[2] 武文博, 郭丽丽, 欧阳军. 病理性瘢痕的治疗研究进展及展望[J].中华灾害救援医学,2019,7(5):295-300.
[3] HUANG C, OGAWA R. Keloidal pathophysiology: Current notions. Scars Burn Heal. 2021;7:2059513120980320.
[4] JESCHKE MG, WOOD FM, MIDDELKOOP E, et al. Scars. Nat Rev Dis Primers. 2023;9(1):64.
[5] SLEMP AE, KIRSCHNER RE. Keloids and scars: a review of keloids and scars, their pathogenesis, risk factors, and management. Curr Opin Pediatr. 2006;18(4):396-402.
[6] RANDALL LJ, BAJAN S, TRAN TD, et al. Propolis compound inhibits profibrotic TGF-β1/SMAD signalling in human fibroblasts. Sci Rep. 2025;15(1):27260.
[7] FRECH FS, HERNANDEZ L, URBONAS R, et al. Hypertrophic Scars and Keloids: Advances in Treatment and Review of Established Therapies. Am J Clin Dermatol. 2023;24(2):225-245.
[8] LI Y, HAN Q, CHEN S, et al. Effect of intraarticular human umbilical cord mesenchymal stem cells transplantation on cartilage degradation and matrix metalloproteinases in OA rat model. BMC Musculoskelet Disord. 2025;26(1):538.
[9] YANG S, CHENG M, ZHANG S, et al. DNMT3A facilitates breast cancer progression via regulating ADAMTS8 mediated EGFR-MEK-ERK activation. PLoS One. 2025; 20(5):e0321889.
[10] GOTO S, GOTO S. Physical and Mechanobiological Basis of Biological Functions of Platelets. Thromb Haemost. 2025. doi: 10.1055/a-2676-4451.
[11] ROCKS N, PAULISSEN G, EL HOUR M, et al. Emerging roles of ADAM and ADAMTS metalloproteinases in cancer. Biochimie. 2008;90(2):369-379.
[12] WANG Z, LI W, CHEN S, et al. Role of ADAM and ADAMTS proteases in pathological tissue remodeling. Cell Death Discov. 2023;9(1):447.
[13] YAO Y, HU C, SONG Q, et al. ADAMTS16 activates latent TGF-β, accentuating fibrosis and dysfunction of the pressure-overloaded heart. Cardiovasc Res. 2020; 116(5):956-969.
[14] BADSHAH, II, BROWN S, WEIBEL L, et al. Differential expression of secreted factors SOSTDC1 and ADAMTS8 cause profibrotic changes in linear morphoea fibroblasts. Br J Dermatol. 2019;180(5):1135-1149.
[15] ZHA Y, LI Y, GE Z, et al. ADAMTS8 Promotes Cardiac Fibrosis Partly Through Activating EGFR Dependent Pathway. Front Cardiovasc Med. 2022;9:797137.
[16] 唐玉婷,贺茜,万瑀,等.紫草素调控MicroRNA-382-5p抑制人增生性瘢痕成纤维细胞纤维化[J]. 中国组织工程研究,2023,27(35):5642-5648.
[17] GAUGLITZ GG, KORTING HC, PAVICIC T, et al. Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Mol Med. 2011;17(1-2): 113-125.
[18] MONY MP, HARMON KA, HESS R, et al. An Updated Review of Hypertrophic Scarring. Cells. 2023;12(5):678.
[19] ARTZI O, KOREN A, NIV R, et al. A new approach in the treatment of pediatric hypertrophic burn scars: Tixel-associated topical triamcinolone acetonide and 5-fluorouracil delivery. J Cosmet Dermatol. 2020;19(1):131-134.
[20] KUBIAK R, LANGE B. Percutaneous collagen induction as an additive treatment for scar formation following thermal injuries: Preliminary experience in 47 children. Burns. 2017;43(5):1097-1102.
[21] HONDA A, KOIKE H, DOHI T, et al. CD206+ macrophages facilitate wound healing through interactions with Gpnmb(hi) fibroblasts. EMBO Rep. 2025;26(14):3679-3704.
[22] FLECKNER M, DÖHMEN NK, SALZ K, et al. Exposure of Primary Human Skin Fibroblasts to Carbon Dioxide-Containing Solution Significantly Reduces TGF-β-Induced Myofibroblast Differentiation In Vitro. Int J Mol Sci. 2024;25(23):13013.
[23] KIM WS, PARK BS, SUNG JH, et al. Wound healing effect of adipose-derived stem cells: a critical role of secretory factors on human dermal fibroblasts. J Dermatol Sci. 2007;48(1):15-24.
[24] KOHLHAUSER M, MAYRHOFER M, KAMOLZ LP, et al. An Update on Molecular Mechanisms of Scarring-A Narrative Review. Int J Mol Sci. 2024;25(21):11579.
[25] KO DS, BAEK SE, HA M, et al. Osteopontin is a key regulator of vascular smooth muscle cell proliferation in the outflow vein of arteriovenous fistulas. Microvasc Res. 2023;149:104570.
[26] SANTAMARIA S, MARTIN DR, DONG X, et al. Post-translational regulation and proteolytic activity of the metalloproteinase ADAMTS8. J Biol Chem. 2021; 297(5):101323.
[27] KEMBERI M, SALMASI Y, SANTAMARIA S. The Role of ADAMTS Proteoglycanases in Thoracic Aortic Disease. Int J Mol Sci. 2023;24(15):12135.
[28] WANG W, ZHANG H, HOU C, et al. Internal modulation of proteolysis in vascular extracellular matrix remodeling: role of ADAM metallopeptidase with thrombospondin type 1 motif 5 in the development of intracranial aneurysm rupture. Aging (Albany N Y). 2021;13(9):12800-12816.
[29] TANG BL. ADAMTS: a novel family of extracellular matrix proteases. Int J Biochem Cell Biol. 2001;33(1):33-44.
[30] BURKHARD T, MINNS AF, SANTAMARIA S. Expression and Purification of Recombinant ADAMTS8. Methods Mol Biol. 2024;2747:55-66.
[31] DRILON A, SUGITA H, SIMA CS, et al. A prospective study of tumor suppressor gene methylation as a prognostic biomarker in surgically resected stage I to IIIA non-small-cell lung cancers. J Thorac Oncol. 2014;9(9):1272-1277.
[32] LEE HC, CHANG CY, WU KL, et al. The Therapeutic Potential of ADAMTS8 in Lung Adenocarcinoma without Targetable Therapy. J Pers Med. 2022;12(6);902.
[33] DUNN JR, REED JE, DU PLESSIS DG, et al. Expression of ADAMTS-8, a secreted protease with antiangiogenic properties, is downregulated in brain tumours. Br J Cancer. 2006;94(8):1186-1193.
[34] WU Z, SHI Y, REN S, et al. ADAMTS8 Inhibits Progression of Esophageal Squamous Cell Carcinoma. DNA Cell Biol. 2020. doi: 10.1089/dna.2020.6053.
[35] ZHANG K, TIAN R, WANG G, et al. ADAMTS8 Inhibits Cell Proliferation and Invasion, and Induces Apoptosis in Breast Cancer. Onco Targets Ther. 2020;13: 8373-8382.
[36] ZHAO X, YANG C, WU J, et al. ADAMTS8 targets ERK to suppress cell proliferation, invasion, and metastasis of hepatocellular carcinoma. Onco Targets Ther. 2018; 11:7569-7578.
[37] KOŽIK B, ČORBO T, POJSKIĆ N, et al. Identifying Molecular Modulators of the Vascular Invasion in Rectal Carcinoma: Role of ADAMTS8 and Its Co-Dependent Genes. Int J Mol Sci. 2025;26(13):6261.
[38] YEO HJ, HA M, SHIN DH, et al. Development of a Novel Biomarker for the Progression of Idiopathic Pulmonary Fibrosis. Int J Mol Sci. 2024;25(1):599.
[39] ZHANG Y, HU K, QU Z, et al. ADAMTS8 inhibited lung cancer progression through suppressing VEGFA. Biochem Biophys Res Commun. 2022;598:1-8.
[40] OMURA J, SATOH K, KIKUCHI N, et al. ADAMTS8 Promotes the Development of Pulmonary Arterial Hypertension and Right Ventricular Failure: A Possible Novel Therapeutic Target. Circ Res. 2019;125(10):884-906.
|