Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (28): 7267-7279.doi: 10.12307/2026.788
Previous Articles Next Articles
Molecular mechanisms of Toddalia asiatica against rheumatoid arthritis: bioinformatics and molecular dynamics simulation
Deng Qian1, Peng Zining1, Meng Fanyu1, Huang Yuanbo1, Liu Nian1, Yan Weitian2, Li Zhaofu1, Peng Jiangyun2
- 1The First Clinical Medical College of Yunnan University of Chinese Medicine, Kunming 650500, Yunnan Province, China; 2Yunnan Provincial Hospital of Traditional Chinese Medicine/The First Affiliated Hospital of Yunnan University of Chinese Medicine, Kunming 650021, Yunnan Province, China
-
Received:2025-09-20Revised:2025-11-15Online:2026-10-08Published:2026-02-09 -
Contact:Peng Jiangyun, MS, Chief physician, Doctoral supervisor, Yunnan Provincial Hospital of Traditional Chinese Medicine/The First Affiliated Hospital of Yunnan University of Chinese Medicine, Kunming 650021, Yunnan Province, China -
About author:Deng Qian, MD, The First Clinical Medical College of Yunnan University of Chinese Medicine, Kunming 650500, Yunnan Province, China Peng Zining, MD, The First Clinical Medical College of Yunnan University of Chinese Medicine, Kunming 650500, Yunnan Province, China Deng Qian and Peng Zining contributed equally to this paper. -
Supported by:National Natural Science Foundation of China, No. 81960863 (to PJY); Yunnan Provincial Clinical Medical Research Center for Traditional Chinese Medicine (Rheumatism), No. 202405AJ310004 (to PJY); Key Research and Development Program (Social Development Special Project) of Yunnan Provincial Department of Science and Technology, No. 202403AC100019 (to PJY); Special Project for Selection of High-level Scientific and Technological Talents and Innovation Teams in Yunnan Province, No. 202305AS350007 (to LZF)
CLC Number:
Cite this article
Deng Qian, Peng Zining, Meng Fanyu, Huang Yuanbo, Liu Nian, Yan Weitian, Li Zhaofu, Peng Jiangyun. Molecular mechanisms of Toddalia asiatica against rheumatoid arthritis: bioinformatics and molecular dynamics simulation[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7267-7279.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
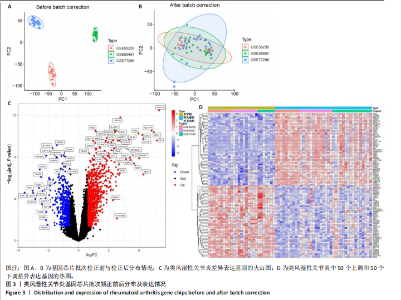
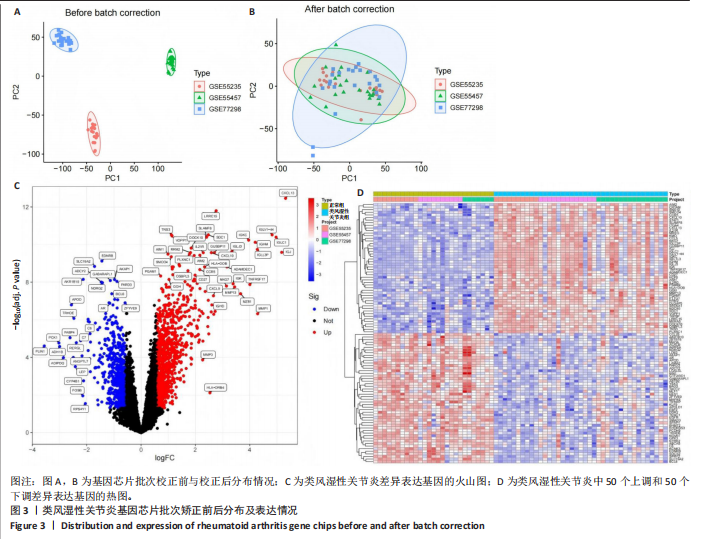
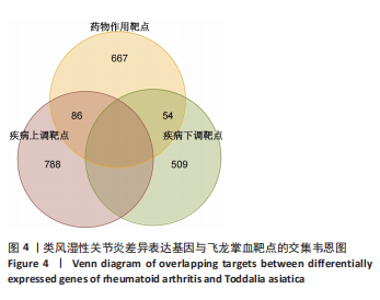
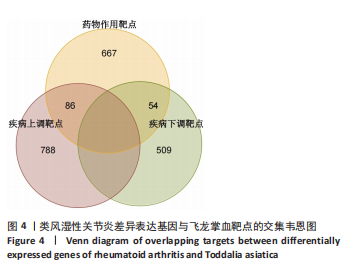
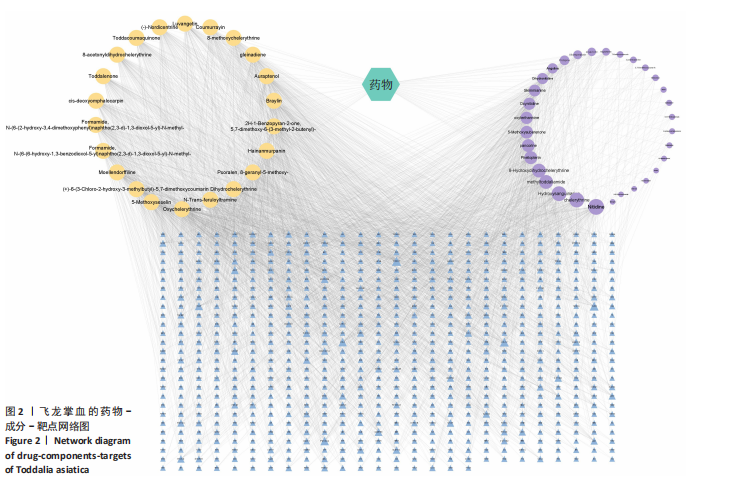
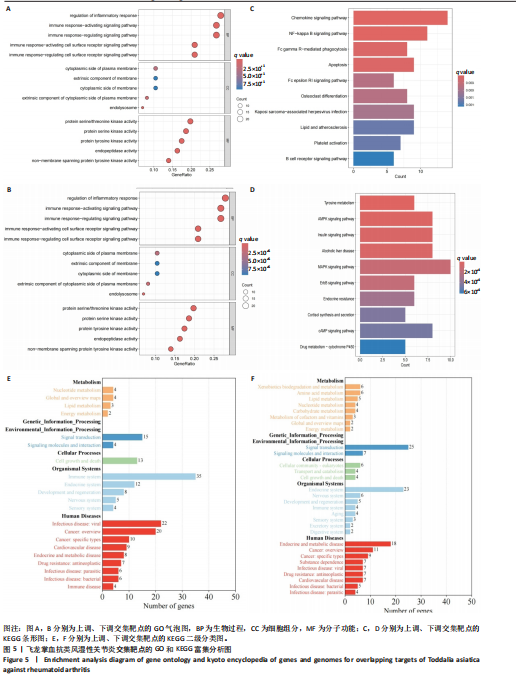
2.1 飞龙掌血的活性成分与靶点蛋白 通过文献回顾,鉴定出飞龙掌血的92种化学成分,其中87种符合筛选标准。排除无靶点的化合物后,最终预测飞龙掌血含54种有效活性成分,对应694个靶点蛋白。使用Cytoscape(v.3.8.0)可视化分析(图2),其中22种成分具有高度值,表明它们是飞龙掌血的主要活性成分。 2.2 类风湿性关节炎数据集的差异分析结果 将获取的类风湿性关节炎相关基因数据集中GSE77298、GSE55235和GSE55457作为训练集,GSE1919作为外部验证集,对训练集的基因数据进行批次校正处理(图3A,B)。根据筛选标准,共识别1 437个差异表达基因,包括563个下调基因和874个上调基因(图3C)。图3D展示了前50个最显著上调和下调的基因。 2.3 飞龙掌血作用于类风湿性关节炎的靶点 通过飞龙掌血活性成分靶点与类风湿性关节炎差异表达基因取交集,获得140个交集靶点,这些靶点在类风湿性关节炎发病过程中呈现显著差异表达,并与飞龙掌血活性成分存在潜在相互作用;进一步分析发现,其中包含86个靶点上调、54个靶点下调(图4)。 2.4 飞龙掌血抗类风湿性关节炎交集靶点的富集分析结果 对86个上调靶点的GO分析显示,它们参与976种生物学机制,主要通过860个生物过程,如炎症反应调控、免疫反应激活信号通路和免疫反应调节信号通路;定位于44个细胞组分,包括质膜胞质侧、膜外成分和膜胞质侧;涉及72个分子功能,如蛋白酪氨酸激酶活性、蛋白丝氨酸激酶活性和蛋白苏氨酸激酶活性(图5A)。对54个下调靶点的GO分析表明,它们涵盖539种生物学机制,主要通过471个生物过程,如激素代谢过程、伯醇代谢过程和视黄醇代谢过程;主要定位于10个细胞组分,如基底外侧质膜和RNA聚合酶Ⅱ转录调控复合物;涉及67个分子功能,包括激素结合、核受体活性和配体激活转录因子活性(图5B)。"
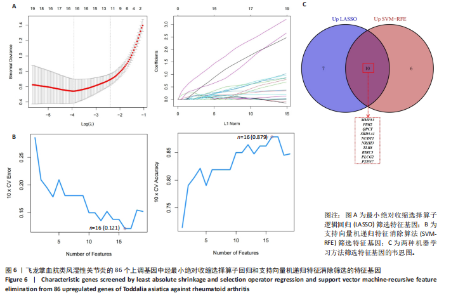
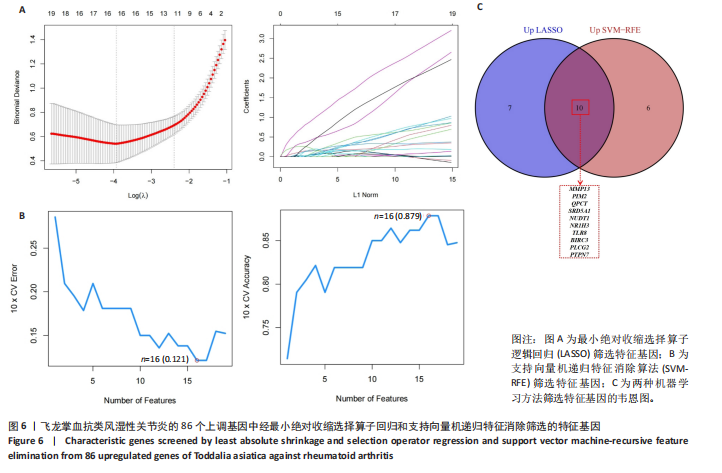
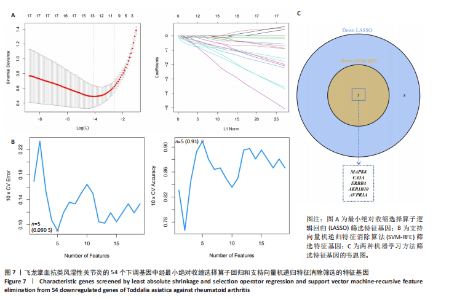
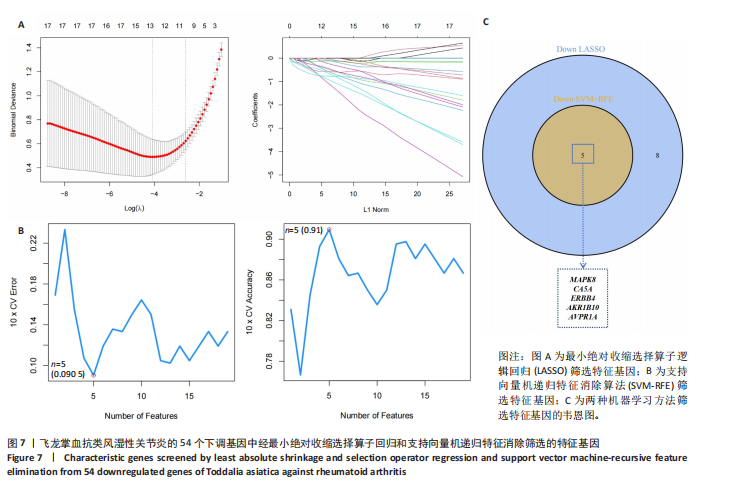
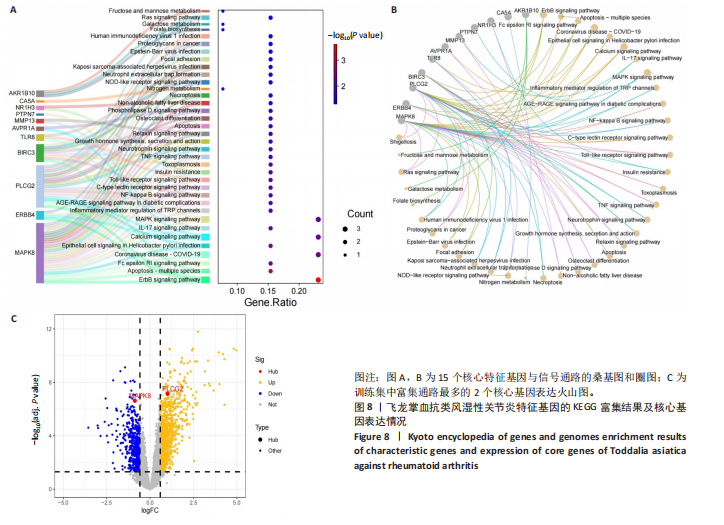
KEGG分析指出,86个上调靶点主要富集于53条信号通路,如趋化因子信号通路、核因子κB信号通路和细胞凋亡(图5C);54个下调靶点则富集于102条通路,包括酪氨酸代谢、AMP依赖的蛋白激酶信号通路和胰岛素信号通路(图5D)。基于KEGG通路二级分类图(图5E,F),推测飞龙掌血可能通过影响免疫、疾病、遗传和环境信息等多方面发挥抗类风湿性关节炎作用。 2.5 飞龙掌血抗类风湿性关节炎的特征基因 采用最小绝对收缩选择算子回归对飞龙掌血抗类风湿性关节炎相关的86个上调靶点和54个下调靶点深入分析,分别筛选出17个(图6A)和16个(图7A)误差最小的特征基因,其对模型贡献最大。同时运用支持向量机递归特征消除方法进一步验证筛选:对上调和下调靶点分别分析后,最终得到16个(图6B)和5个(图7B)误差最小的特征基因。 为更精准确定飞龙掌血抗类风湿性关节炎的特征基因,取最小绝对收缩选择算子回归和支持向量机递归特征消除2种方法的交集,最终筛选出10个上调特征基因(图6C):MMP13(基质金属蛋白酶13)、PIM2(Pim-2原癌基因)、QPCT(谷氨酰肽环转移酶)、SRD5A1(类固醇5α-还原酶1)、NUDT1(核苷二磷酸连接模块含有1)、NR1H3(核受体亚家族1H组成员3)、TLR8(Toll样受体8)、BIRC3(baculoviral IAP重复含有3)、PLCG2(磷脂酶Cγ2)和PTPN7(蛋白酪氨酸磷酸酶非受体型7);5个下调特征基因(图7C):MAPK8(丝裂原活化蛋白激酶8)、CA5A(碳酸酐酶5A)、ERBB4(erb-b2受体酪氨酸激酶4)、AKR1B10(醛糖还原酶相关蛋白1B10)和AVPR1A(精氨酸加压素受体1A)。 2.6 飞龙掌血抗类风湿性关节炎特征基因的KEGG富集结果 对飞龙掌血抗类风湿性关节炎的15个特征基因进行KEGG富集分析(图8A,B)显示,其主要通过ErbB信号通路、钙信号通路、"
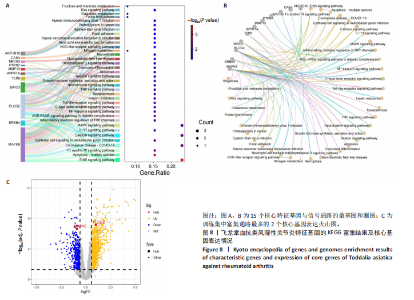
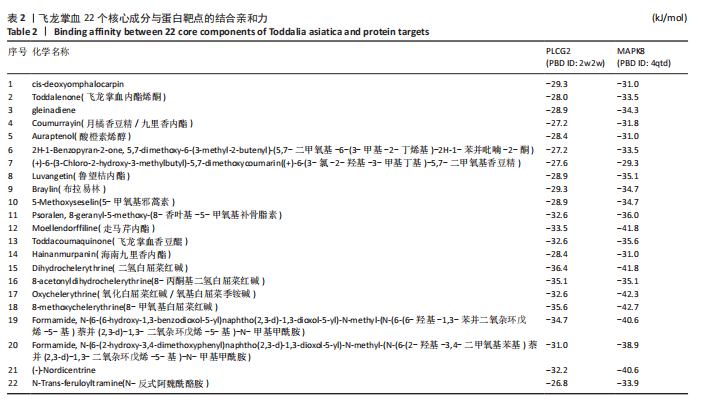
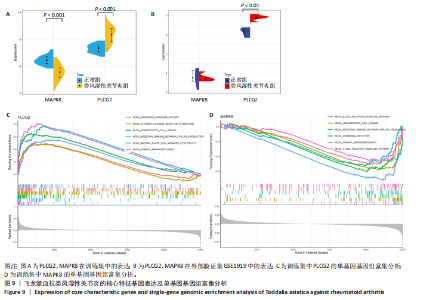
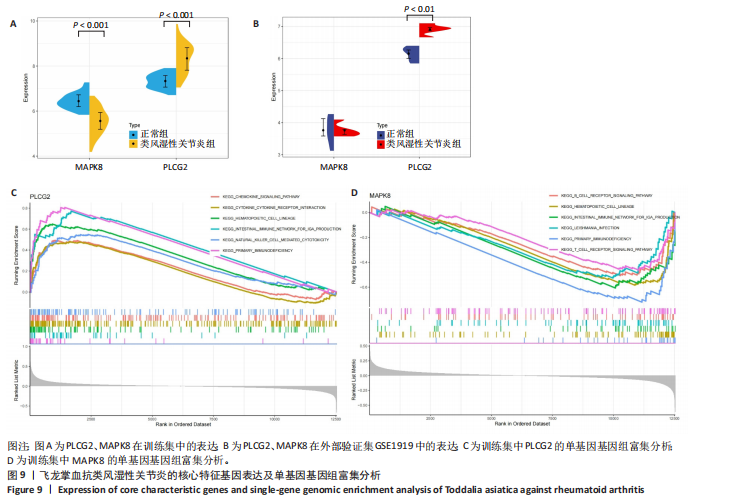
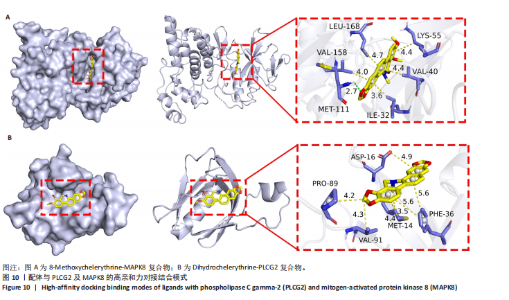
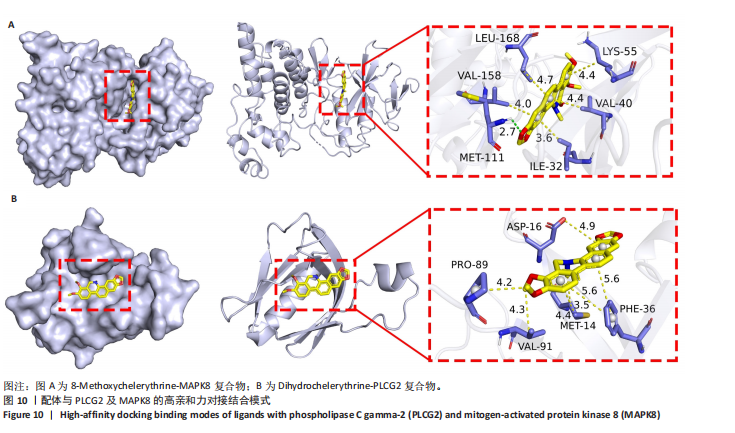
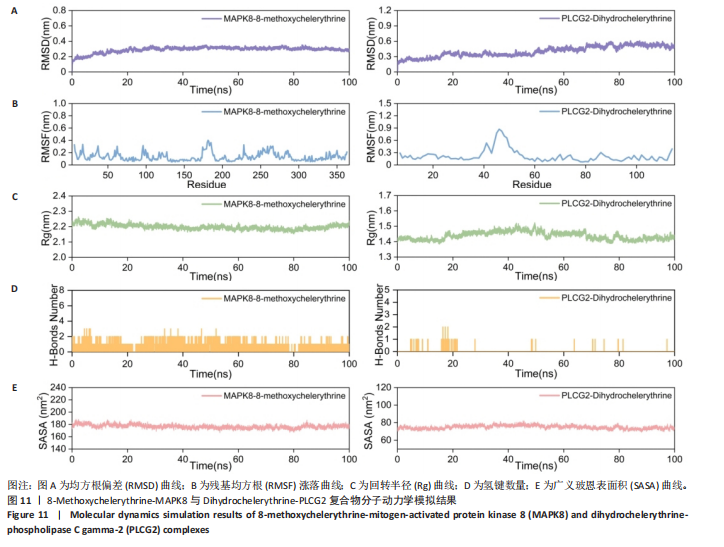
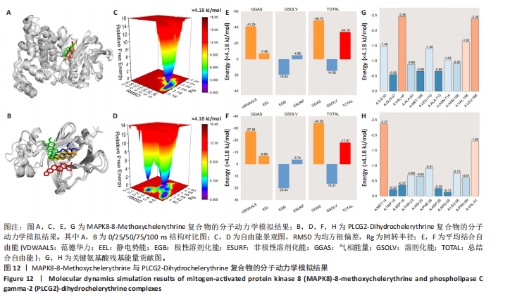
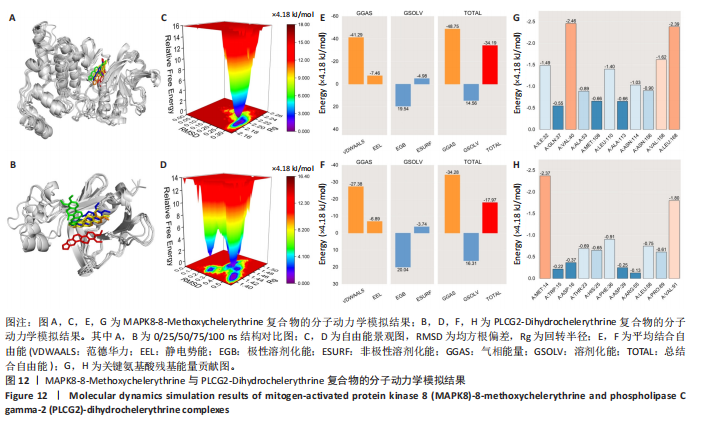
冠状病毒病-COVID-19及丝裂原活化蛋白激酶信号通路发挥作用。其中MAPK8和PLCG2两个基因在多个信号通路中富集,因此研究选择这2个基因进行深入分析。在疾病背景下,PLCG2被鉴定为上调基因,MAPK8为下调基因(图8C)。 2.7 飞龙掌血抗类风湿性关节炎核心特征基因表达及单基因基因组富集分析 在训练集中,类风湿性关节炎组MAPK8表达低于正常组(P < 0.001),PLCG2表达高于正常组(P < 0.001),见图9A。在外部验证集GSE1919中,类风湿性关节炎组PLCG2表达高于正常组(P < 0.01),并且表达趋势与训练集一致,两组间MAPK8无明显差异,见图9B。 通过单基因基因组富集分析进一步探索核心特征基因区分正常与疾病样本的潜在功能。PLCG2在类风湿性关节炎中表达升高,显著富集于免疫相关通路,包括细胞因子-细胞因子受体相互作用、趋化因子信号通路、造血细胞谱系、IgA生成肠道免疫网络、自然杀伤细胞介导的细胞毒性及原发性免疫缺陷(图9C)。 MAPK8与B细胞受体信号通路、造血细胞谱系、IgA生成肠道免疫网络、利什曼原虫感染、原发性免疫缺陷及T细胞受体信号通路相关(图9D)。表明PLCG2和MAPK8可能通过调控免疫炎症反应在类风湿性关节炎发病中起关键作用。 2.8 分子对接结果 飞龙掌血核心成分与类风湿性关节炎核心特征基因的分子对接能范围为-42.68 kJ/mol至-26.78 kJ/mol(表2)。配体与核心基因结合力强,主要依赖氢键、疏水作用及范德华力,表明天然条件下飞龙掌血核心成分可有效结合类风湿性关节炎核心特征基因PLCG2与MAPK8。图10详细展示药物成分与PLCG2/MAPK8的最佳受体结合模式,8-Methoxychelerythrine与MAPK8蛋白MET-111氨基酸残基形成1个氢键(绿色虚线),并与ILE-32、VAL-40等形成5个疏水作用(黄色虚线);Dihydrochelerythrine与PLCG2蛋白MET-14、ASP-16等形成7个疏水作用。 2.9 分子动力学模拟结果 为验证化合物与蛋白的结合亲和力及稳定性,对8-Methoxychelerythrine-MAPK8、Dihydrochelerythrine-PLCG2复合物进行100 ns 分子动力学模拟。从图11A可以看出,两复合物的均方根偏差曲线全程维持在1 nm内且无显著波动,表明结构高度稳定;图11B中,残基均方根涨落均≤1 nm,说明小分子对MAPK8/PLCG2氨基酸残基稳定性的影响极小;图11C中,回转半径曲线分别波动于2.2 nm和1.4 nm附近。MAPK8复合物轨迹平滑,提示结构紧密稳定;PLCG2复合物存在轻微波动,表明结合稳定性略弱;图11D中,8-Methoxychelerythrine-MAPK8间氢键数稳定在1–2个(波动平缓),凸显强氢键作用与高稳定性。Dihydrochelerythrine-PLCG2复合物几乎无氢键,但图10B的分子对接结果显示Dihydrochelerythrine通过与活性口袋内疏水残基形成范德华力。由于疏水作用的构象适应性较强,可减少因单点突变导致的结合口袋结构改变,并且分子对接结果提示复合物结合自由能绝对值较高,因此依旧维持良好稳定性。图11E中,MAPK8与PLCG2的广义玻恩表面积曲线分别稳定波动于180 nm2和70 nm2内,证实蛋白本征稳定性。 从图12A,B中可以看出,在0/25/50/75/100 ns五个时间点,8-Methoxychelerythrine和Dihydrochelerythrine始终结合于MAPK8、PLCG2的相同位点(小分子构象以红/绿/蓝/黄/橙色标示),进一步验证复合物的稳定性。根据图12C,MAPK8复合物的自由能景观形成单一主导能量簇,提示超强稳定性;PLCG2复合物存在多能量簇,但构象分析(图12D)证实小分子未解离,能量波动源于PLCG2右侧结构域的局部运动,不影响结合稳定性。根据图12E,F,MAPK8与PLCG2复合物的平均结合自由能分别为-143.05 kJ/mol和-75.19 kJ/mol,前者呈强结合亲和力,后者为中等亲和力。关键残基能量贡献分析显示(图12G,H),MAPK8的VAL-40(-10.29 kJ/mol)与LEU-168(-10.00 kJ/mol)、PLCG2的MET-14(-9.92 kJ/mol)与VAL-91(-7.53 kJ/mol)是主导结合的关键位点,这些残基与分子对接结果(图10)高度一致,证实模拟全程结合模式稳定。"
| [1] DI MATTEO A, BATHON JM, EMERY P. Rheumatoid Arthritis. Lancet. 2023; 402(10416):2019-2033. [2] SCUBLINSKY D, GONZALEZ CD. Quantifying Disease in Challenging Conditions: Incidence and Prevalence of Rheumatoid Arthritis. J Rheumatol. 2016;43(7):1263-1264. [3] RADU AF, BUNGAU SG. Management of Rheumatoid Arthritis: An Overview. Cells. 2021;10(11):2857. [4] BROWN P, PRATT AG, HYRICH KL. Therapeutic Advances in Rheumatoid Arthritis. BMJ. 2024;384:e070856. [5] KUMAGAI M, WATANABE A, YOSHIDA I, et al. Evaluation of Aculeatin and Toddaculin Isolated from Toddalia Asiatica as Anti-Inflammatory Agents in LPS-Stimulated RAW264 Macrophages. Biol Pharm Bull. 2018;41(1):132-137. [6] 秦黄冠,周刊,付宇蕾,等.飞龙掌血醇提物对类风湿性关节炎“肠-免疫-关节轴”中Th17/Treg平衡的影响[J].中国医院药学杂志,2023,43(10):1089-1094. [7] QIN H, FU Y, ZHOU K, et al. Toddalia Asiatica Extract Attenuates Adjuvant-Induced Arthritis by Modulating Colon Th17/Treg Balance and Colony Homeostasis. J Ethnopharmacol. 2023;313:116542. [8] ZHANG H, DAN W, HE Q, et al. Exploring the Biological Mechanism of Huang Yam in Treating Tumors and Preventing Antitumor Drug-Induced Cardiotoxicity Using Network Pharmacology and Molecular Docking Technology. Evid Based Complement Alternat Med. 2021;2021:9988650. [9] HE H, LI N, FAN Y, et al. Six New Coumarins from the Roots of Toddalia Asiatica and Their Anti-Inflammatory Activities. Chin J Nat Med. 2023;21(11):852-858. [10] 何文星,徐灿丽,罗淑娟,等.飞龙掌血化学成分及药理作用研究进展及其Q-Marker预测分析[J].中成药,2024,46(2):507-517. [11] ALBAAYIT SA, RASEDEE A, ABDULLAH N, et al. Methanolic Extract of Clausena Excavata Promotes Wound Healing via Antiinflammatory and Anti-Apoptotic Activities. Asian Pac J Trop bio. 2020;10:232. [12] NADAF S, DESAI R, MORE T, et al. Antiproliferative and Caspase-Mediated Apoptosis Inducing Effects of Murraya Koenigii Seeds against Cancer Cells. S Afr J Bot. 2020;132:328-337. [13] LIU YP, YAN G, XIE YT, et al. Bioactive Prenylated Coumarins as Potential Anti-Inflammatory and Anti-HIV Agents from Clausena Lenis. Bioorg Chem. 2020;97:103699. [14] LEE SW, LIM JM, MOHAN H, et al. Enhanced Bioactivity of Zanthoxylum Schinifolium Fermented Extract: Anti-Inflammatory Anti-Bacterial and Anti-Melanogenic Activity. J Biosci Bioeng. 2020;129(5):638-645. [15] 郝小燕,彭琳,叶兰,等.飞龙掌血生物总碱抗炎镇痛作用的研究[J].中西医结合学报,2004,2(6):450-452. [16] TAKOMTHONG P, WAIWUT P, YENJAI C, et al. Structure-Activity Analysis and Molecular Docking Studies of Coumarins from Toddalia Asiatica as Multifunctional Agents for Alzheimer’s Disease. Biomedicines. 2020;8(5):107. [17] CUCA-SUAREZ LE, DELLA-MONACHE F, COY-BARRERA E. Cytotoxic Constituents from Bark and Leaves of Amyris Pinnata Kunth. Rec Nat Prod. 2015;9:441-445. [18] LIN TT, HUANG YY, TANG GH, et al. Prenylated Coumarins: Natural Phosphodiesterase-4 Inhibitors from Toddalia Asiatica. J Nat Prod. 2014;77(4): 955-962. [19] RAFAEL S, COSTA, OTÁVIO P, et al. In Vitro Antileishmanial and Antitrypanosomal Activity of Compounds Isolated from the Roots of Zanthoxylum Tingoassuiba. Rev Bras Farmacogn. 2018;28(5):551-558. [20] LIN Y, LIU S, SUN Y, et al. CCR5 and Inflammatory Storm. Ageing Res Rev. 2024; 96:102286. [21] POON IKH, RAVICHANDRAN KS. Targeting Efferocytosis in Inflammaging. Annu Rev Pharmacol Toxicol. 2024;64:339-357. [22] HARGREAVES CE, ROSE-ZERILLI MJJ, MACHADO LR, et al. Fcγ Receptors: Genetic Variation Function and Disease. Immunol Rev. 2015;268(1):6-24. [23] GRILLET B, PEREIRA RVS, VAN DAMME J, et al. Matrix Metalloproteinases in Arthritis: Towards Precision Medicine. Nat Rev Rheumatol. 2023;19(6):363-377. [24] GAUDENZI C, SCHIOPPA T, PASSARI M, et al. Extracellular microRNAs Induce Dendritic Cell-Dependent Joint Inflammation and Potentiate Osteoclast Differentiation via TLR7/8 Engagement. J Autoimmun. 2024;145:103189. [25] KEHLEN A, HAEGELE M, BÖHME L, et al. N-Terminal Pyroglutamate Formation in CX3CL1 Is Essential for Its Full Biologic Activity. Biosci Rep. 2017;37(4): BSR20170712. [26] BENDER P, EGGER A, WESTERMANN M, et al. Expression of Human and Porphyromonas Gingivalis Glutaminyl Cyclases in Periodontitis and Rheumatoid Arthritis-A Pilot Study. Arch Oral Biol. 2019;97:223-230. [27] MARTIN CS, CRASTIN A, SAGMEISTER MS, et al. Inflammation Dynamically Regulates Steroid Hormone Metabolism and Action within Macrophages in Rheumatoid Arthritis. J Autoimmun. 2024;147:103263. [28] CHOJNACKA K, MRUK DD. The Src Non-Receptor Tyrosine Kinase Paradigm: New Insights into Mammalian Sertoli Cell Biology. Mol Cell Endocrinol. 2015;415:133-142. [29] MCDONELL LM, KERNOHAN KD, BOYCOTT KM, et al. Receptor Tyrosine Kinase Mutations in Developmental Syndromes and Cancer: Two Sides of the Same Coin. Hum Mol Genet. 2015;24(R1):R60-66. [30] TRISTANO AG. Tyrosine Kinases as Targets in Rheumatoid Arthritis. Int Immunopharmacol. 2009;9(1):1-9. [31] TRIPOLINO C, CIAFFI J, PUCINO V, et al. Insulin Signaling in Arthritis. Front Immunol. 2021;12:672519. [32] URSINI F, RUSSO E, RUSCITTI P, et al. The Effect of Non-TNF-Targeted Biologics and Small Molecules on Insulin Resistance in Inflammatory Arthritis. Autoimmun Rev. 2018;17(4):399-404. [33] URSINI F, D’ANGELO S, RUSSO E, et al. Serum Complement C3 Strongly Correlates with Whole-Body Insulin Sensitivity in Rheumatoid Arthritis. Clin Exp Rheumatol. 2017;35(1):18-23. [34] CHEN M, ZHU J, LUO H, et al. The Journey towards Physiology and Pathology: Tracing the Path of Neuregulin 4. Genes Dis. 2024;11(2):687-700. [35] GUO M, WANG T, GE W, et al. Role of AKR1B10 in Inflammatory Diseases. Scand J Immunol. 2024;100(2):e13390. [36] SANGHAVI N, INGRASSIA JP, KOREM S, et al. Cardiovascular Manifestations in Rheumatoid Arthritis. Cardiol Rev. 2024;32(2):146-152. [37] CHO MH, CHO JH, EUN Y, et al. Rheumatoid Arthritis and Risk of Lung Cancer: A Nationwide Cohort Study. J Thorac Oncol. 2024;19(2):216-226. [38] PARK SK, RYOO JH, KIM MH, et al. Association Between Eight Autoimmune Diseases and Thyroid Cancer: A Nationwide Cohort Study. Thyroid. 2024; 34(2):206-214. [39] DIEPERINK SS, NØRGAARD M, MEHNERT F, et al. Rheumatoid Arthritis and Risk of Osteoarticular Infection and Death Following Staphylococcus Aureus Bacteraemia: A Nationwide Cohort Study. Rheumatology (Oxford). 2024;63(11):2989-2996. [40] JEON KH, HAN K, JUNG J, et al. Rheumatoid Arthritis and Risk of Depression in South Korea. JAMA Netw Open. 2024;7(3):e241139. [41] MCARTHUR GA. Combination Therapies to Inhibit the RAF/MEK/ERK Pathway in Melanoma: We Are Not Done Yet. Front Oncol. 2015;5:161. [42] JOHNSON GL, LAPADAT R. Mitogen-Activated Protein Kinase Pathways Mediated by ERK JNK and P38 Protein Kinases. Science. 2002; 298(5600):1911-1912. [43] DING C. Drug Evaluation: VX-702 a MAP Kinase Inhibitor for Rheumatoid Arthritis and Acute Coronary Syndrome. Curr Opin Investig Drugs. 2006;7(11):1020-1025. [44] CAO W, LI Y, ZENG Z, et al. Terpinen-4-Ol Induces Ferroptosis of Glioma Cells via Downregulating JUN Proto-Oncogene. Molecules. 2023;28(12):4643. [45] LUO C, NIE C, ZENG Y, et al. LINC01564 Promotes the TMZ Resistance of Glioma Cells by Upregulating NFE2L2 Expression to Inhibit Ferroptosis. Mol Neurobiol. 2022;59(6):3829-3844. [46] CHAE JJ, PARK YH, PARK C, et al. Connecting Two Pathways through Ca2+ Signaling: NLRP3 Inflammasome Activation Induced by a Hypermorphic PLCG2 Mutation. Arthritis Rheumatol. 2015;67(2):563-567. [47] EEKTIMMERMAN F, SWEN JJ, DEN BROEDER AA, et al. Genome-Wide Association Study of Methotrexate-Induced Liver Injury in Rheumatoid Arthritis Patients. Clin Pharmacol Ther. 2023;113(4):916-923. [48] 陈龙,陈小燕,张淼,等.飞龙掌血茎和根不同炮制品对小鼠的急性毒性试验[J].湖北农业科学,2024,63(1):90-95. |
| [1] | Chen Yulin, He Yingying, Hu Kai, Chen Zhifan, Nie Sha Meng Yanhui, Li Runzhen, Zhang Xiaoduo , Li Yuxi, Tang Yaoping. Effect and mechanism of exosome-like vesicles derived from Trichosanthes kirilowii Maxim. in preventing and treating atherosclerosis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1768-1781. |
| [2] | Zhou Sirui, Xu Yukun, Zhao Kewei. Ideas and methods of anti-melanogenesis of Angelica dahurica extracellular vesicles [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(7): 1747-1754. |
| [3] | Peng Zhiwei, Chen Lei, Tong Lei. Luteolin promotes wound healing in diabetic mice: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1398-1406. |
| [4] | Guo Ying, Tian Feng, Wang Chunfang. Potential drug targets for the treatment of rheumatoid arthritis: large sample analysis from European databases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1549-1557. |
| [5] | Wu Zhilin, , He Qin, Wang Pingxi, Shi Xian, Yuan Song, Zhang Jun, Wang Hao . DYRK2: a novel therapeutic target for rheumatoid arthritis combined with osteoporosis based on East Asian and European populations [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(6): 1569-1579. |
| [6] | Zheng Yin, Wu Zhenhua, Zhang Cheng, Ruan Kexin, Gang Xiaolin, Ji Hong. Safety and efficacy of immunoadsorption therapy for rheumatoid arthritis: a network meta-analysis and systematic review [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1260-1268. |
| [7] | Liu Fengzhi, Dong Yuna, Tian Wenyi, Wang Chunlei, Liang Xiaodong, Bao Lin. Gene-predicted associations between 731 immune cell phenotypes and rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1311-1319. |
| [8] | Chen Yixian, Chen Chen, Lu Liheng, Tang Jinpeng, Yu Xiaowei. Triptolide in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 805-815. |
| [9] | Bao Zhuoma, Hou Ziming, Jiang Lu, Li Weiyi, Zhang Zongxing, Liu Daozhong, Yuan Lin. Effect and mechanism by which Pterocarya hupehensis skan total flavonoids regulates the proliferation, migration and apoptosis of fibroblast-like synoviocytes [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(4): 816-823. |
| [10] | Qi Yuxin, Dang Yifan, Dai Liming, Zhang Xiaoling. Function and molecular mechanism of physcion in regulating bone homeostasis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7237-7244. |
| [11] | Zhang Hongrui, Wu Ruiqi, Wang Wenchi, Peng Qinglin, Cui Wei. Molecular mechanisms of active compounds from Tripterygium wilfordii in prevention and treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7332-7339. |
| [12] | Zan Junhao, Hu Shujuan, Yuan Xinguo, Pu Rui. Exercise regulation of pyroptosis for the prevention and treatment of bone metabolic disorders [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7347-7354. |
| [13] | Li Huaying, Li Hao, Peng Wuxun, Dong Wentao. Mechanism of cuproptosis in the diagnosis and treatment of orthopedic-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6275-6281. |
| [14] | Zhang Xiaomin, Du Pengyang, Zhang Xiuping, Xue Guofang. Potential targets of glucagon-like peptide 1 receptor agonist ticagrelor in the treatment of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6122-6133. |
| [15] | Zhou Wen, Yang Hongwei. Molecular mechanism and natural drug screening for ferroptosis-targeted therapy in rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(23): 6051-6061. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||