Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (21): 5477-5484.doi: 10.12307/2026.726
Previous Articles Next Articles
Morphometric analysis of principal stress trabeculae in the proximal femur
Cao Meng, Li Lun, Tian Simiao, Cao Fang, Song Liqun, Zhao Dewei
- Department of Orthopedics, Zhongshan Hospital Affiliated to Dalian University, Dalian 116001, Liaoning Province, China
-
Accepted:2025-07-11Online:2026-07-28Published:2026-03-04 -
Contact:Zhao Dewei, MD, Chief physician, Doctoral supervisor, Department of Orthopedics, Zhongshan Hospital Affiliated to Dalian University, Dalian 116001, Liaoning Province, China -
About author:Cao Meng, Doctoral candidate, Attending physician, Department of Orthopedics, Zhongshan Hospital Affiliated to Dalian University, Dalian 116001, Liaoning Province, China
CLC Number:
Cite this article
Cao Meng, Li Lun, Tian Simiao, Cao Fang, Song Liqun, Zhao Dewei. Morphometric analysis of principal stress trabeculae in the proximal femur[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(21): 5477-5484.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
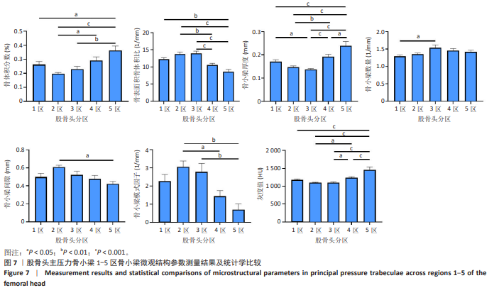
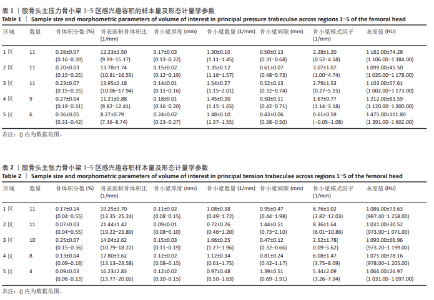
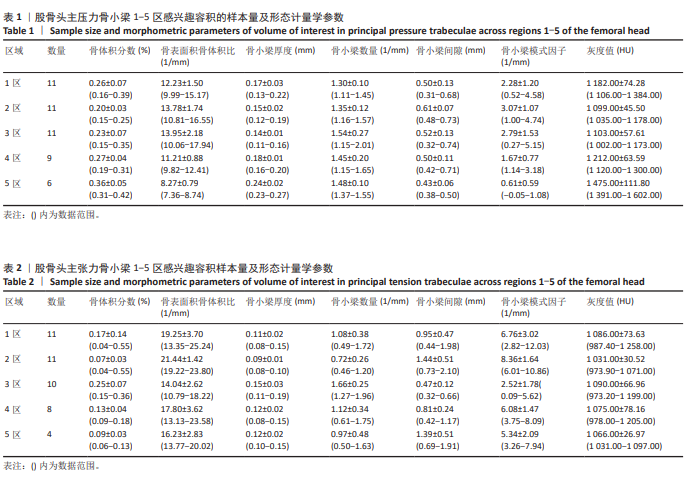
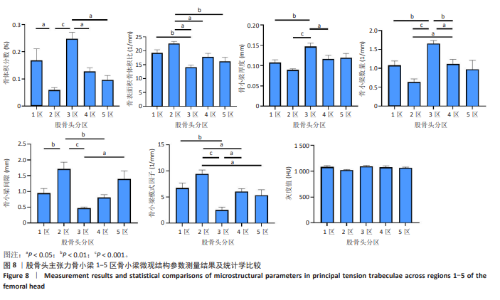
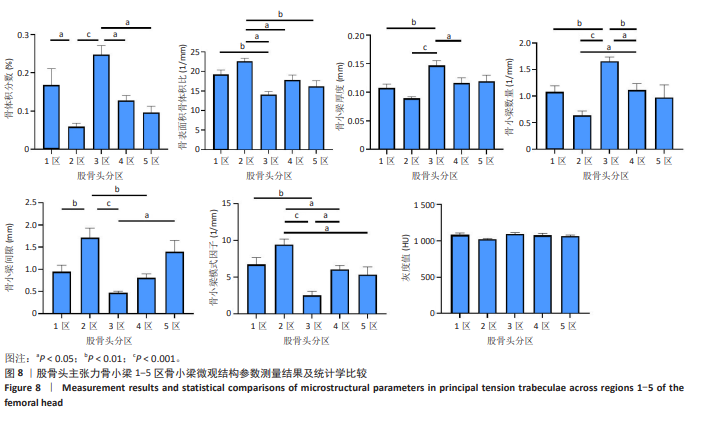
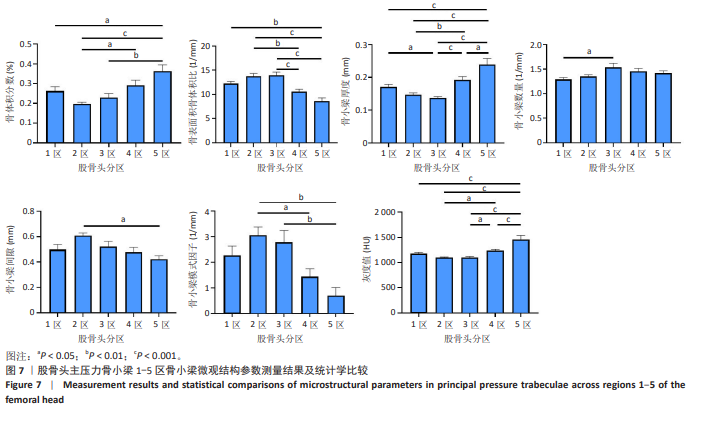
2.2 主压力骨小梁形态计量学统计学结果 如表1所示,在主压力骨小梁1-5区各感兴趣容积中,5区即主压力骨小梁的底部,具有最高的骨体积分数、骨小梁厚度、灰度值;4区次之;1区即主压力骨小梁的头部,具有第3高的骨体积分数、骨小梁厚度、灰度值;5区与1区在上述指标之间差异有显著性意义(P < 0.05,P < 0.001,P < 0.001),见图7;同时,5区具有最低的骨表面积骨体积比、骨小梁间隙、骨小梁模式因子。3区是主压力骨小梁的中间感兴趣容积,具有最大的骨表面积骨体积比、骨小梁数量以及最小的骨小梁厚度。由3区向压力骨小梁的底部和头部骨表面积骨体积比、骨小梁数量逐渐减小,骨小梁厚度逐渐增大。 2.3 主张力骨小梁形态计量学统计学结果 如表2所示,在主张力骨小梁1-5区中,3区即主压力、主张力骨小梁的交汇区,具有最高的骨体积分数、骨小梁厚度、骨小梁数量、灰度值,同时其具有最低的骨表面积骨体积比、骨小梁间隙、骨小梁模式因子。与之相反的是2区,具有最低的骨体积分数、骨小梁厚度、骨小梁数量、灰度值,但具有最高的骨表面积骨体积比、骨小梁间隙、骨小梁模式因子。除灰度值外,两区域之间的各项形态计量学参数相比差异均有显著性意义(P均< 0.001),见图8。"
| [1] HAMMER A. The paradox of Wolff’s theories. Ir J Med Sci. 2015;184(1): 13-22. [2] HAMMER A. Wolff: straight not curved. Ir J Med Sci. 2017;186(4): 939-946. [3] MANCUSO ME, WILZMAN AR, MURDOCK KE, et al. Effect of External Mechanical Stimuli on Human Bone: a narrative review. Prog Biomed Eng (Bristol). 2022;4(1):012006. [4] SAKASHITA M, YAMASAKI S, YAJI K, et al. Three-dimensional topology optimization model to simulate the external shapes of bone. PLoS Comput Biol. 2021;17(6):e1009043. [5] YANG P, LIN TY, XU JL, et al. Finite element modeling of proximal femur with quantifiable weight-bearing area in standing position. J Orthop Surg Res. 2020;15(1):384. [6] HAMMER A. The structure of the femoral neck: A physical dissection with emphasis on the internal trabecular system. Ann Anat. 2010; 192(3):168-177. [7] XU C, LI H, ZHANG C, et al. Quantitative Analysis of Primary Compressive Trabeculae Distribution in the Proximal Femur of the Elderly. Orthop Surg. 2024;16(8):2030-2039. [8] CHIBA K, BURGHARDT AJ, OSAKI M, et al. Heterogeneity of bone microstructure in the femoral head in patients with osteoporosis: an ex vivo HR-pQCT study. Bone. 2013;56(1):139-146. [9] JENKINS PJ, RAMAESH R, PANKAJ P, et al. A micro-architectural evaluation of osteoporotic human femoral heads to guide implant placement in proximal femoral fractures. Acta Orthop. 2013;84(5): 453-459. [10] METZNER F, NEUPETSCH C, FISCHER JP, et al. Influence of osteoporosis on the compressive properties of femoral cancellous bone and its dependence on various density parameters. Sci Rep. 2021;11(1): 13284. [11] WANG H, DING K, ZHANG Y, et al. A Controlled Variable Study of the Biomechanical Properties of the Proximal Femur before and after Cancellous Bone Removal. Orthop Surg. 2024;16(5):1215-1229. [12] WANG H, ZHANG Y, REN C, et al. Biomechanical properties and clinical significance of cancellous bone in proximal femur: A review. Injury. 2023:S0020-1383(23)00251-6. [13] FAZZALARI NL, FORWOOD MR, SMITH K, et al. Assessment of cancellous bone quality in severe osteoarthrosis: bone mineral density, mechanics, and microdamage. Bone. 1998;22(4):381-388. [14] FROST HM. Wolff’s Law and bone’s structural adaptations to mechanical usage: an overview for clinicians. Angle Orthod. 1994; 64(3):175-188. [15] SILVA RF, COELHO PG, GUSTAVO CV, et al. Functionally Graded Materials and Structures: Unified Approach by Optimal Design, Metal Additive Manufacturing, and Image-Based Characterization. Materials (Basel). 2024;17(18):4545. [16] RAABE D, SACHS C, ROMANO P. The crustacean exoskeleton as an example of a structurally and mechanically graded biological nanocomposite material. Acta Materialia. 2005;53(15):4281-4292. [17] BAR-ON B, BARTH FG, FRATZL P, et al. Multiscale structural gradients enhance the biomechanical functionality of the spider fang. Nat Commun. 2014;5:3894. [18] CAI J, WANG J, SUN C, et al. Biomaterials with stiffness gradient for interface tissue engineering. Biomed Mater. 2022;17(6). doi: 10.1088/1748-605X/ac8b4a. [19] GENIN GM, KENT A, BIRMAN V, et al. Functional grading of mineral and collagen in the attachment of tendon to bone. Biophys J. 2009;97(4): 976-985. [20] KWON H, BROWN WE, LEE CA, et al. Surgical and tissue engineering strategies for articular cartilage and meniscus repair. Nat Rev Rheumatol. 2019;15(9):550-570. [21] 祝晓忠, 梅炯, 倪明, 等. 股骨近端骨小梁结构的大体解剖和影像重建分析 [J]. 中国修复重建外科杂志,2019,33(10):1254-1259. [22] DEY A, RAMONI M, YODO N. A Survey on Fused Filament Fabrication to Produce Functionally Gradient Materials. Materials (Basel). 2024; 17(15):3675. [23] JYOTI, GHOSH R. Printable functionally graded tibial implant for TAR: FE study comparing implant materials, FGM properties, and implant designs. Comput Biol Med. 2024;177:108645. [24] WANG Y, CHEN S, LIANG H, et al. A review of graded scaffolds made by additive manufacturing for tissue engineering: design, fabrication and properties. Biofabrication. 2025;17(2). doi: 10.1088/1758-5090/adba8e. [25] DING K, YANG W, ZHU J, et al. Titanium alloy cannulated screws and biodegradable magnesium alloy bionic cannulated screws for treatment of femoral neck fractures: a finite element analysis. J Orthop Surg Res. 2021;16(1):511. [26] LI M, ZHAO K, DING K, et al. Titanium Alloy Gamma Nail versus Biodegradable Magnesium Alloy Bionic Gamma Nail for Treating Intertrochanteric Fractures: A Finite Element Analysis. Orthop Surg. 2021;13(5):1513-1520. [27] WANG H, YANG W, DING K, et al. Biomechanical study on the stability and strain conduction of intertrochanteric fracture fixed with proximal femoral nail antirotation versus triangular supporting intramedullary nail. Int Orthop. 2022;46(2):341-350. [28] DING K, ZHU Y, LI Y, et al. Triangular support intramedullary nail: A new internal fixation innovation for treating intertrochanteric fracture and its finite element analysis. Injury. 2022;53(6):1796-1804. [29] DING K, ZHU Y, WANG H, et al. A comparative Study of Novel Extramedullary Fixation and Dynamic Hip Screw in the Fixation of Intertrochanteric Fracture: A Finite-Element Analysis. Front Surg. 2022;9:911141. [30] GUO L, ATAOLLAH NAGHAVI S, WANG Z, et al. On the design evolution of hip implants: A review. Mater Design. 2022;216:110552. [31] XIAO Z, WU L, WU W, et al. Multi-Scale Topology Optimization of Femoral Stem Structure Subject to Stress Shielding Reduce. Materials (Basel). 2023;16(8):3151. [32] LOHA T, BHATTACHARYA R, PAL B, et al. A novel design of hip-stem with reduced strain-shielding. Proc Inst Mech Eng H.2024;238(5):471-482. [33] LIU B, WANG H, ZHANG N, et al. Femoral Stems With Porous Lattice Structures: A Review. Front Bioeng Biotechnol. 2021;9:772539. [34] ZIAIE B, VELAY X, SALEEM W. Advanced porous hip implants: A comprehensive review. Heliyon. 2024;10(18):e37818. [35] SOLIMAN MM, ISLAM MT, CHOWDHURY MEH, et al. Advancement in total hip implant: a comprehensive review of mechanics and performance parameters across diverse novelties. J Mater Chem B. 2023;11(44):31. [36] SOLA A, BELLUCCI D, CANNILLO V. Functionally graded materials for orthopedic applications - an update on design and manufacturing. Biotechnol Adv. 2016;34(5):504-531. [37] SAFAVI S, YU Y, ROBINSON DL, et al. Additively manufactured controlled porous orthopedic joint replacement designs to reduce bone stress shielding: a systematic review. J Orthop Surg Res. 2023;18(1):42. [38] NAGHAVI SA, TAMADDON M, GARCIA-SOUTO P, et al. A novel hybrid design and modelling of a customised graded Ti-6Al-4V porous hip implant to reduce stress-shielding: An experimental and numerical analysis. Front Bioeng Biotechnol. 2023;11:1092361. [39] MüLLER P, SYNEK A, STAUß T, et al. Development of a density-based topology optimization of homogenized lattice structures for individualized hip endoprostheses and validation using micro-FE. Sci Rep. 2024;14(1):5719. [40] BANDYOPADHYAY A, MITRA I, AVILA JD, et al. Porous metal implants: processing, properties, and challenges. Int J Extrem Manuf. 2023;5(3): 032014. [41] ZHAI Y, ZHANG H, WANG J, et al. Research progress of metal-based additive manufacturing in medical implants. Rev Adv Mater Sci. 2023; 62:20230148. |
| [1] | Jiang Xinghai, Song Yulin, Li Dejin, Shao Jianmin, Xu Junzhi, Liu Huakai, Wu Yingguo, Shen Yuehui, Feng Sicheng. Vascular endothelial growth factor 165 genes transfected into bone marrow mesenchymal stem cells to construct a vascularized amphiphilic peptide gel module [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1903-1911. |
| [2] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Advance in the mechanisms underlying miRNAs in steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1207-1214. |
| [3] | Liao Long, Zhao Zepeng, Li Zongyuan, Yu Qinglong, Zhang Tao, Tang Jinyuan, Ye Nan, Xu Han, Shi Bo. Establishment and validation of a model for femoral head necrosis after internal fixation of femoral neck fracture using logistic regression and SHAP analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 626-633. |
| [4] | Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanism by which vascular endothelial growth factor A targets regulation of angiogenesis in the treatment of steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 671-679. |
| [5] | Zhou Zixiang, Zhao Baoxiang. Research progress in the relationship between nontraumatic necrosis of the femoral head and lipid metabolism and its treatment [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(3): 680-690. |
| [6] | Cheng Yan, Yang Yidan, Li Zhipeng, Liu Youwen, Guo Jiayi, Yue Chen. Mechanism of Gugutou Huaisiyu Capsule in alleviating peripheral pain sensitization in rats with steroid-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7512-7519. |
| [7] | He Long, Gao Shuang, Chen Chao, Qin Guozhong, Ran Qingsen, Wang Zhengchun, Yang Yafeng, Ren Hang, Qiu Yunkai, Yang Yang, Li Wei. A new strategy for preventing and treating orthopedic diseases by regulating ferroptosis through signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7619-7631. |
| [8] | Wang Weiwei, Ding Qiang, Rong Xiangbin, Guo Liang, Zhao Canbin, Tao Hongcheng, Niu Chicheng, Liu Jinfu, Zeng Ping. Bushen Tongshi Pills improves osteogenic disorders in alcoholic femoral head necrosis rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6165-6173. |
| [9] | Wan Ziyi, Jiang Mengyu, Zhou Yuehui, Xue Yuxuan, Wei Yangwenxiang, Zhou Chi. Articular cartilage lesions at different stages of steroid-induced osteonecrosis of the femoral head: characteristics and mechanisms of crescent sign formation [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(22): 5659-5670. |
| [10] | Liang Yingjie, Yuan Lingli, Geng Chunhui, Zhang Zhongchuan, Zheng Wenming, Hu Tengfei, Tang Haoxu, Zhang Kunkun. Finite element analysis of core decompression with ceramic rod implantation in osteonecrosis of the femoral head during the peri-collapse stage [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(21): 5429-5436. |
| [11] | Huang Zhijian, Zhang Cheng, He Haijun, Dong Yawei, Sun Yifei, Gao Rui, Du Pengcheng. Correlation analysis between acetabular coverage and the onset of idiopathic osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(21): 5485-5493. |
| [12] | Chai Jinlian, Liang Xuezhen, Sun Tiefeng, Li Shudong, Li Wei, Li Guangzheng, Yu Huayun, Wang Ping. Mechanistic insights into how Cervi Cornus Colla regulates the intestinal flora-bile acid metabolic pathway to alleviate steroid-induced osteonecrosis of the femoral head in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(18): 4568-4581. |
| [13] | Lin Yong, Yang Xiaoqiang, Lin Kun, Yang Fan, He Mincong, Wei Qiushi. Immune microenvironment and inflammatory repair of cystic degeneration in steroid-induced osteonecrosis of the femoral head: a single-cell sequencing analysis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(17): 4309-4317. |
| [14] | Yao Kexin, Yang Yidan, Li Yapeng, Zhu Xuanye, Wang Qiuyuan, Guo Jiayi, Liu Youwen, Yue Chen. A controlled analysis of phenomenon of acupoint sensitization in osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(15): 3913-3919. |
| [15] | Tang Yuntao, Wu Zhonghan, Wang Jingkun, Li Tao, Lu Jingtao, Mao Shitan, Tang Jian, Xu Xinzhong. Relationship of femoral artery, vein and nerve projections with femoral head positions in patients with femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(15): 3953-3959. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||