Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (4): 621-625.doi: 10.3969/j.issn.2095-4344.2377
Previous Articles Next Articles
Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis
Ye Haimin1, Ding Linghua2, Kong Weihao2, Huang Zutai3, Xiong Long1
- 1Department of Orthopedics, Jiangxi Provincial People’s Hospital, Nanchang 330000, Jiangxi Province, China; 2People’s Hospital Affiliated to Nanchang University, Nanchang 330000, Jiangxi Province, China; 3Department of Orthopedics, Gannan Medical University, Ganzhou 341000, Jiangxi Province, China
-
Received:2020-03-07Revised:2020-03-12Accepted:2020-04-15Online:2021-02-08Published:2020-11-24 -
Contact:Xiong Long, Chief physician, Department of Orthopedics, Jiangxi Provincial People’s Hospital, Nanchang 330000, Jiangxi Province, China -
About author:Ye Haimin, Master candidate, Physician, Department of Orthopedics, Jiangxi Provincial People’s Hospital, Nanchang 330000, Jiangxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 81560355
CLC Number:
Cite this article
Ye Haimin, Ding Linghua, Kong Weihao, Huang Zutai, Xiong Long. Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 621-625.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
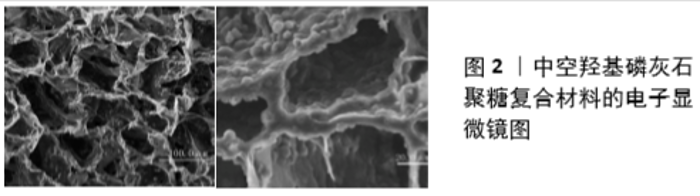
2.1 多级微管结构骨支架载体促进成骨 骨生长支架是骨组织工程构成的重要因素,它在调控种子细胞募集、血管再生及成骨微环境中发挥重要作用。作者在国家自然基金课题(81560355)资助下,将重组人骨形态发生蛋白2负载于中空羟基磷灰石微球中用于非负重区域兔桡骨骨缺损修复,发现其控释性、生物相容性、生物因子活性均良好,可激活多种信号通路参与成骨[7-9],并可保持生物活性因子活性,发挥良好成骨分化作用[8-11],但中空羟基磷灰石微球难于成形,作为骨生长支架载体有待优化。为此,在国家自然科学基金课题(81560377)资助下,成功制备了可注射中空羟基磷灰石微球/壳聚糖温敏水凝胶,该材料具有海绵状多孔结构(图2),它可以很好地同时负载趋化因子13、重组人骨形态发生蛋白2两种生物活性因子并显现出趋化因子13/重组人骨形态发生蛋白2两种生长因子释放时序、生物活性保持、募集效应、成骨分化诱导性能时空藕合[12],因此可用作生物因子的潜在载体;然而,中空羟基磷灰石微球/壳聚糖复合材料作为骨支架仍存在诸多缺点,如其孔隙与天然骨仍有较大差距,孔隙大小和孔隙互连仍不可控制等[8-9,13],这些性能将直接影响到内源性间充质干细胞的归巢、定植等。骨修复材料的孔隙不通或孔隙互连率低对早期血管长入有明显阻碍。这些缺点严重阻碍了该材料成骨能力的更大化发挥[13]。因此,如何构建理想支架促进骨缺损修复是亟待解决的问题,而模仿天然骨结构构建支架已成普遍认识。 鉴于目前3D打印技术空间分辨率有限且价格昂贵,打印后的支架需保持一定的生物相容性,完全模仿出与天然骨内在微细结构完全一样的骨生长支架目前还不能实现,选择某些特定关键结构在现有技术下进行模仿,是骨生长支架的研究焦点。支架内部微结构包括孔径、孔隙互连、孔隙率、孔隙形状、孔隙空间分布、微通道等会影响细胞的黏附、归巢、定植、增殖、分化、表型、基因表达等生物学行为[13-15]。瞄准天然骨上述相关的关键结构进行仿制是骨支架材料研发的一种方式。 人体组织中存在大量尺度各异的微纳米分级结构,如骨、肌腱等,这些结构在组织的生长和发育过程中发挥着非常重要的作用[16]。研究表明,微纳米分级结构材料可以有效调控细胞的黏附、迁移、增殖和分化等行为[17]。这是因为活体细胞对胞外基质的微观结构特性非常敏感,细胞会随着材料表面微米拓扑结构的变化发生结构重排,此时细胞在基质表面的黏附行为是通过许多受体分子相互聚集的方式实现[16,18];细胞在纳米拓扑结构表面的响应行为更为复杂,即细胞可能通过单个受体分子(而非相互聚集的受体分子)驱动的途径来控制细胞对纳米拓扑结构的行为响应,因此在组织工程支架表面仿生构建微纳米分级结构调控细胞行为和功能引起了极大的关注。WEINER等[19]研究认为骨是由羟基磷灰石/胶原仿生复合体纤维组成的具有高度复杂结构的复合材料,他们从7个等级的组织级别来描述骨组织的结构:第一级分别来自骨的独立晶体和部分未矿化的骨胶原纤维,第二级别为羟基磷灰石/胶原仿生复合体原纤维,第三级别为羟基磷灰石/胶原仿生复合体原纤维阵列,第四级别为羟基磷灰石/胶原仿生复合体原纤维阵列堆积类型,第五级别为人骨的单元,第六级别为松质骨与密质骨,最后是宏观尺度的第七级别:整块骨组织结构[20-22]。为模拟天然骨的内部结构,理想支架应具有互相连通的梯度孔隙结构,允许蛋白吸附、细胞扩散和营养物质、氧气、生长因子和废物等的有效运输,促进新生骨组织在支架内部的生长,直至支架完全降解并建立新骨以达到修复骨缺损的终极目标。 有研究表明,从微纳米到毫厘米梯度渐变、尺度各异的多级微管结构在骨组织再生中发挥了重要作用[17,23-24]。人们已经认识到内源性间充质干细胞的募集对修复过程非常有利,因为它们可以释放细胞因子和生长因子,促进细胞修复组织的功能,重塑受损的微环境[25-28]。已知干细胞归巢趋化因子如趋化因子12主要由炎症细胞分泌,以启动参与组织修复和重塑的细胞(如间充质干细胞)的迁移[29]。因此,多级微管结构支架的炎症反应明显移向解决期(即细胞极化向M2期),可能有利于其分泌趋化因子12,通过诱使间充质干细胞到达损伤部位加速组织愈合。体外实验证明,多级微管结构支架能更好地刺激骨髓间充质干细胞黏附和分化为成骨细胞系[23]。骨支架材料不同尺度的孔、管径结构有不同功能。小尺度微管(尺寸<10 μm)更容易被组织液浸渍,提供细胞更多的吸附位点及更大的表面积和更高的离子溶解度,支持甚至引导间充质干细胞成骨[30],为调控间充质干细胞成骨分化提供更多场所与更多的化学刺激物质;此外,表面积大可促进巨噬细胞黏附,加速细胞由表面向支架内部的迁移和增殖。中等尺度20-40 μm微管有利于原始巨噬细胞向M2型极化,并且能上调抗炎基因表达抑制宿主对移植物的免疫反应,这对于宿主细胞特别是间充质干细胞长入至关重要。大尺度微管(管径>100 μm)为细胞黏附、新血管形成和骨内生长提供了空间和通道[31],有利于细胞定植、着床、归巢,为形成细胞集落提供了场所。因此,无论大尺度还是小尺度微管对于骨支架都很重要[30],一方面,相互连通的大孔是连接支架表面和中心的通道,使组织和生物液可以进入整个支架有利于细胞的迁移、血管化和骨生长,例如:骨髓间充质干细胞会进入并定居在支架的大孔内[32-35]。另一方面,合适的微孔在新骨形成方面显示出特定的功能[36-37]。正如文献记载的那样,只有同时具有大孔和微孔的支架才能诱导骨形 成[38-40]。理想的骨组织工程支架应具有分级孔隙率,以提供不同的生物物理和生物化学效应。多级微管结构理论上可调控间充质干细胞的归巢,为间充质干细胞提供适宜的生长微环境,对于骨生长支架的结构优化起到重要作用,可为良好成骨效应带来重要保障。 2.2 多级微管结构骨支架载体成血管作用 具有良好成骨效应的骨再生材料为实现优异的骨修复疗效,必须有快速血管化功能支撑。血管新生是原有血管芽生新血管的过程,包括内皮细胞增生和迁移、蛋白溶解酶表达调控、细胞外基质破裂重建和内皮管腔形成,涉及一系列信号转导途径的激活。血管生成取决于内皮细胞周围血管生成促进因子和血管生成抑制因子的动态平衡,在血管生成促进因子的作用下产生对内皮细胞的刺激,随后蛋白水解酶对细胞外基质的降解,继而内皮细胞向细胞外基质的浸润、迁移、增殖,形成新的毛细血管,最后内皮外周细胞向局部的聚集使血管网得以稳定。根据血管再生病理生理机制的不同分为3种模式,即血管衍生、血管发生及动脉生成,它们在缺血后血运重建过程中均起着重要的作用[41-42]。血管衍生主要是指从原有的毛细后微血管网上芽生、套叠或被横跨的内皮细胞桥分隔(搭桥),分化出新生的、不具有完整中膜结构的毛细血管网过程,此过程是多环节作用的结果,包括某些特定的酶对原有血管基膜的降解、内皮细胞的增殖、迁移、有序排列及新生血管的重构和成熟。血管发生主要由特异的血管内皮祖细胞经过分裂、增殖,产生大量成熟的内皮细胞,再经过有序排列生成新的血管。血管内皮生长因子作为一种高度特异性的促血管内皮细胞生长因子,对血管生成的速度、血管的再内皮化有着重要作用。 多级微管结构可以促进血管再生,它可使黏附的巨噬细胞发生相应的形变,导致细胞骨架张力的增加,这种张力的变化会使得原始巨噬细胞向M2型转化极化,M2型巨噬细胞可上调血管内皮生长因子基因的表达,分泌血管内皮生长因子[43]。有一项里程碑式的研究证明了通道支架中的微孔大小对血管生成的影响[44-45]。血管生成在修复过程中对细胞生存和组织生长至关重要,它是由极化的巨噬细胞和招募的间充质干细胞分泌的生物活性分子刺激的严格协调生物反 应[46]。极化的巨噬细胞可分别激活间充质干细胞和血管内皮细胞的血管内皮生长因子信号通路,促进血管生成[43]。募集的间充质干细胞受多级微管结构的刺激可分泌血管生成因子[23],通过血小板衍生生长因子BB促进内皮祖细胞的迁移,并通过胰岛素样生长因子1和基质衍生因子1(SDF1/CXCL-12)促进血管形成[47-48]。不同的通道大小在体内诱导不同的血管化行为,大小为250 μm的通道可表达具有代表性的血管生成因子HIF1α、PLGF1和迁移因子CXCR4,有利于小血管的形成;而500 μm的通道可增强血管内皮生长因子的表达,有利于大血管的发育[49]。多级微管结构可以抑制免疫排斥反应,减少炎症因子生成,进而提供了促进血管生成的微环境。研究表明,募集定植的间充质干细胞能够与内皮细胞(或内皮祖细胞)相互作用促进血管形成,并且可以根据需要向血管内皮细胞分化[28,50]。多级微管内30-40 μm微通道最适合新生毛细血管长入[23],而对于更大的血管或骨骼内其他血液运输结构如滋养孔的构建,目前多级微管结构不能实现,因此在引入多级微管结构基础上继续创建更大尺度的空间管道网络结构,对实现骨修复材料的再血管化很重要[51]。对于更大尺度的空间管道网络结构特别是400 μm以上的多孔结构的构建,3D打印有其明显优势,采用3D打印技术构建多级微管3D打印多孔结构,实现骨组织再生过程中的血管生成理论上是可行的。多级微管结构不仅可实现间充质干细胞表面黏附,内部归巢、定植促进成骨,还可实现骨支架材料的血管化[23,47]。 目前多级微管结构可靠、稳定、重复性好的制备方法为WON等[23]所提出,该方法选用的材料成分为聚已内酯,聚已内酯因其良好的力学性能、生物相容性较好、熔点较低 (55 ℃)、可低温成型、可在体内缓慢降解、易于3D打印成孔隙互连的多孔结构等优点在医药领域广泛应用 [52],但聚已内酯存在亲水性差、降解过程中pH值变化过大,易影响生物因子的活性等缺点。大量研究结果表明,通过与无机纳米粒子复合,聚已内酯材料的力学性能、热稳定性、气体阻隔性和生物降解性等均可获得显著改善。相信随着对聚已内酯性能及应用的深入研究,聚已内酯将在医药领域得到更广泛的应用。"

| [1] LIU M, LV Y. Reconstructing Bone with Natural Bone Graft: A Review of In Vivo Studies in Bone Defect Animal Model.Nanomaterials (Basel). 2018;8(12).pii:E999.doi: 10.3390/nano8120999. [2] BALDWIN P, LI DJ, AUSTON DA, et al.Autograft, Allograft, and Bone Graft Substitutes: Clinical Evidence and Indications for Use in the Setting of Orthopaedic Trauma Surgery.J Orthop Trauma.2019;33(4):203-213. [3] GRIGORIAN SHAMAGIAN L, MADONNA R, TAYLOR D, et al. Perspectives on Directions and Priorities for Future Preclinical Studies in Regenerative Medicine.Circ Res.2019;124(6):938-951. [4] ORYAN A, ALIDADI S, MOSHIRI A, et al. Platelet-rich plasma for bone healing and regeneration. Expert Opin Biol Ther.2016;16(2):213-232. [5] WANG SJ, JIANG D, ZHANG ZZ, et al. Biomimetic Nanosilica-Collagen Scaffolds for In Situ Bone Regeneration: Toward a Cell-Free, One-Step Surgery.Adv Mater.2019;31(49):e1904341. [6] FILARDO G, ROFFI A, FEY T, et al. Vegetable hierarchical structures as template for bone regeneration: New bio-ceramization process for the development of a bone scaffold applied to an experimental sheep model.J Biomed Mater Res B Appl Biomater.2020;108(3):600-611. [7] LI JT, XIONG SL, DING LH, et al. The mechanism research of non-Smad dependent TAK1 signaling pathway in the treatment of bone defects by recombination BMP-2-loaded hollow hydroxyapatite microspheres/chitosan composite.J Mater Sci Mater Med.2019;30(12):130. [8] YAO AH, LI XD, XIONG L, et al. Hollow hydroxyapatite microspheres/chitosan composite as a sustained delivery vehicle for rhBMP-2 in the treatment of bone defects.J Mater Sci Mater Med.2015;26(1):5336. [9] XIONG L, ZENG JH, YAO AH, et al. BMP2-loaded hollow hydroxyapatite microspheres exhibit enhanced osteoinduction and osteogenicity in large bone defects.Int J Nanomedicine 2015;10:517-526. [10] XIAO W, BAL BS, RAHAMAN MN. Preparation of resorbable carbonate-substituted hollow hydroxyapatite microspheres and their evaluation in osseous defects in vivo.Mater Sci Eng C Mater Biol Appl.2016;60: 324-332. [11] TANAKA T, TAKAI Y, NAGASE A, et al. Protein adsorption characteristics of nanoparticle-assembled hollow microspheres of hydroxyapatite and their composites with PLLA microporous membranes.Heliyon. 2019;5(4):e01490. [12] ZENG JH, XIONG SL, DING LH, et al. Study of bone repair mediated by recombination BMP-2/ recombination CXC chemokine Ligand-13-loaded hollow hydroxyapatite microspheres/chitosan composite.Life Sci.2019;234:116743. [13] Zeng JH, Liu SW, Xiong L, et al. Scaffolds for the repair of bone defects in clinical studies: a systematic review.J Orthop Surg Res. 2018; 13(1):33. [14] LEE JH, KIM HW. Emerging properties of hydrogels in tissue engineering.J Tissue Eng. 2018;9:2041731418768285. [15] WANG K, WANG X, HAN C, et al. From Micro to Macro: The Hierarchical Design in a Micropatterned Scaffold for Cell Assembling and Transplantation. Adv Mater.2017;29(2).doi: 10.1002/adma.201604600. [16] ZHANG C, YUAN HH, LIU HH, et al. Well-aligned chitosan-based ultrafine fibers committed teno-lineage differentiation of human induced pluripotent stem cells for Achilles tendon regeneration. Biomaterials.2015;53:716-730. [17] HOLTHAUS MG, STOLLE J, TRECCANI L, et al. Orientation of human osteoblasts on hydroxyapatite-based microchannels.Acta Biomater. 2012;8(1):394-403. [18] RIVELINE D, ZAMIR E, BALABAN NQ, et al. Focal contacts as mechanosensors: externally applied local mechanical force induces growth of focal contacts by an mDia1-dependent and ROCK-independent mechanism.J Cell Biol.2001;153(6):1175-1186. [19] WEINER S, WAGNER D. The Material Bone: Structure-Mechanical Function Relations.Annu Rev Mater Res.2003;28:271-298. [20] LIU Y, LUO D, WANG T. Hierarchical Structures of Bone and Bioinspired Bone Tissue Engineering.Small.2016;12(34):4611-4632. [21] REZNIKOV N, SHAHAR R, WEINER S. Three-dimensional structure of human lamellar bone: The presence of two different materials and new insights into the hierarchical organization. Bone.2014;59:93-104. [22] RHO JY, KUHN L, ZIOUPOS P. Mechanical Properties and the Hierarchical Structure of Bone. Med Eng Phys.1998;20(2):92-102. [23] WON JE, LEE YS, PARK JH, et al. Hierarchical microchanneled scaffolds modulate multiple tissue-regenerative processes of immune-responses, angiogenesis, and stem cell homing.Biomaterials. 2020; 227:119548. [24] YANG H, ZHANG D, PENG WK, et al. Facile synthesis of copper doping hierarchical hollow porous hydroxyapatite beads by rapid gelling strategy.Mater Sci Eng C Mater Biol Appl. 2020;109:110531. [25] SU N, GAO PL, WANG K, et al. Fibrous scaffolds potentiate the paracrine function of mesenchymal stem cells: A new dimension in cell-material interaction.Biomaterials. 2017;141:74-85. [26] GNECCHI M, ZHANG ZP, NI AG, et al. Paracrine mechanisms in adult stem cell signaling and therapy.Circ Res.2008;103(11):1204-1219. [27] SINGER NG, CAPLAN AI. Mesenchymal Stem Cells: Mechanisms of Inflammation.Annu Rev Pathol.2011;6:457-478. [28] THEVENOT PT, NAIR AM, SHEN J, et al. The effect of incorporation of SDF-1alpha into PLGA scaffolds on stem cell recruitment and the inflammatory response.Biomaterials. 2010;31(14):3997-4008. [29] OGLE ME, SEGAR CE, SRIDHAR S, et al. Monocytes and macrophages in tissue repair: Implications for immunoregenerative biomaterial design.Exp Biol Med (Maywood). 2016;241(10):1084-1097. [30] PEI X, MA L, ZHANG B, et al. Creating hierarchical porosity hydroxyapatite scaffolds with osteoinduction by three-dimensional printing and microwave sintering. Biofabrication.2017;9(4):045008. [31] WANG XJ, LOU T, ZHAO WH, et al. The effect of fiber size and pore size on cell proliferation and infiltration in PLLA scaffolds on bone tissue engineering.J Biomater Appl.2016;30(10):1545-1551. [32] BILLIET T, GEVAERT E, DE SCHRYVER T, et al. The 3D printing of gelatin methacrylamide cell-laden tissue-engineered constructs with high cell viability.Biomaterials. 2014;35(1):49-62. [33] HONG Y, HUBER A, AKANARI K, et al. Mechanical properties and in vivo behavior of a biodegradable synthetic polymer microfiber-extracellular matrix hydrogel biohybrid scaffold.Biomaterials. 2011;32(13):3387-3394. [34] LIU X, WU S, YEUNG KWK, et al. Relationship between osseointegration and superelastic biomechanics in porous NiTi scaffolds. Biomaterials. 2011;32(2):330-338. [35] XU C, SU P, CHEN X, et al. Biocompatibility and osteogenesis of biomimetic Bioglass-Collagen-Phosphatidylserine composite scaffolds for bone tissue engineering. Biomaterials.2011;32(4):1051-1058. [36] BARBIERI D, YUAN H, LUO X, et al. Influence of polymer molecular weight in osteoinductive composites for bone tissue regeneration.Acta Biomater.2013;9(12):9401-9413. [37] GROEN N, YUAN H, HEBELS DGAJ, et al. Linking the Transcriptional Landscape of Bone Induction to Biomaterial Design Parameters.Adv Mater.2017;29(10).doi:10.1002/adma.201603259 [38] LEE VK, DAI GH. Printing of Three-Dimensional Tissue Analogs for Regenerative Medicine. Ann Biomed Eng.2017;45(1):115-131. [39] ZHOU C, YE X, FAN Y, et al. Biomimetic fabrication of a three-level hierarchical calcium phosphate/collagen/hydroxyapatite scaffold for bone tissue engineering. Biofabrication.2014;6(3):035013. [40] Li X, Ding J, Wang J, et al.Biomimetic biphasic scaffolds for osteochondral defect repair. Regen Biomater.2015;2(3):221-228. [41] BUSCHMANN I, SCHAPER W. Arteriogenesis Versus Angiogenesis: Two Mechanisms of Vessel Growth.News Physiol Sci.1999;14:121-125. [42] BUSCHMANN I, SCHAPER W. The pathophysiology of the collateral circulation (arteriogenesis).J Pathol.2000;190(3):338-342. [43] PAN HH, XIE YT, ZHANG ZQ, et al. Immunomodulation effect of a hierarchical macropore/nanosurface on osteogenesis and angiogenesis.Biomed Mater.2017;12(4):045006. [44] MADDEN LR, MORTISEN DJ, SUSSMAN EM, et al. Proangiogenic scaffolds as functional templates for cardiac tissue engineering.Proc Natl Acad Sci U S A.2010;107(34):15211-15216. [45] YU T, LIU Q, JIANG T, et al. Channeled β-TCP Scaffolds Promoted Vascularization and Bone Augmentation in Mandible of Beagle Dogs.Adv Funct Mater.2016;26(37):6719-6727. [46] RATNER BD. Reducing capsular thickness and enhancing angiogenesis around implant drug release systems.J Control Release. 2002;78 (1-3): 211-218. [47] TIAN T, ZHANG T, LIN Y, et al. Vascularization in Craniofacial Bone Tissue Engineering.J Dent Res.2018;97(9):969-976. [48] DARNELL M, YOUNG S, GU L, et al. Substrate Stress-Relaxation Regulates Scaffold Remodeling and Bone Formation In Vivo.Adv Healthc Mater. 2017;6(1):10.1002/adhm.201601185. [49] YU T, DONG C, SHEN ZH, et al. Vascularization of plastic calcium phosphate cement in vivo induced by in-situ-generated hollow channels. Mater Sci Eng C Mater Biol Appl. 2016;68:153-162. [50] ARINZEH TL, PETER SJ, ARCHAMBAULT MP, et al. Allogeneic mesenchymal stem cells regenerate bone in a critical-sized canine segmental defect.J Bone Joint Surg Am. 2003;85(10):1927-1935. [51] XING HL, WANG X, XIAO G, et al. Hierarchical assembly of nanostructured coating for siRNA-based dual therapy of bone regeneration and revascularization.Biomaterials. 2020;235:119784. [52] JANG CH, LEE J, KIM G. Synergistic effect of alginate/BMP-2/Umbilical cord serum-coated on 3D-printed PCL biocomposite for mastoid obliteration model.J Ind Eng Chem.2019;72:432-441. |
| [1] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [2] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [3] | Zou Gang, Xu Zhi, Liu Ziming, Li Yuwan, Yang Jibin, Jin Ying, Zhang Jun, Ge Zhen, Liu Yi. Human acellular amniotic membrane scaffold promotes ligament differentiation of human amniotic mesenchymal stem cells modified by Scleraxis in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1037-1044. |
| [4] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Intravenous, topical tranexamic acid alone or their combination in total knee arthroplasty: a meta-analysis of randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 948-956. |
| [5] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [6] | Zhan Fangbiao, Cheng Jun, Zou Xinsen, Long Jie, Xie Lizhong, Deng Qianrong. Intraoperative intravenous application of tranexamic acid reduces perioperative bleeding in multilevel posterior spinal surgery: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 977-984. |
| [7] | Zhang Bin, Sun Lihua, Zhang Junhua, Liu Yusan, Cui Caiyun. A modified flap immediate implant is beneficial to soft tissue reconstruction in maxillary aesthetic area [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 707-712. |
| [8] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [9] | Fu Shuanhu, Qin Kai, Lu Dahan, Qin Haibiao, Gu Jin, Chen Yongxi, Qin Haoran, Wei Jiading, Wu Liang, Song Quansheng. Lumbar spinal tuberculosis implanted with artificial bone with streptomycin sulfate and percutaneous pedicle screw under transforaminal endoscopy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 493-498. |
| [10] | Li Wenjing, Li Haobo, Liu Congna, Cheng Dongmei, Chen Huizhen, Zhang Zhiyong. Comparison of different bioactive scaffolds in the treatment of regenerative pulp of young permanent teeth [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 499-503. |
| [11] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [12] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [13] | Li Chenjie, Lü Linwei, Song Yang, Liu Jingna, Zhang Chunqiu. Measurement and statistical analysis of trabecular morphological parameters of titanium alloy peri-prosthesis under preload [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 516-520. |
| [14] | Sun Qi, Zhou Yanan, Dong Xin, Li Ning, Yan Jiazhen, Shi Haojiang, Xu Sheng, Zhang Biao. Metal-ceramic interface characteristics of Co-Cr alloy fabricated by selective laser melting [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 521-525. |
| [15] | Liu Yang, Gong Yi, Fan Wei. Anti-hepatoma activity of targeted Pluronic F127/formononetin nanocomposite system in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 526-531. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||