Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (14): 2274-2279.doi: 10.3969/j.issn.2095-4344.3111
Previous Articles Next Articles
Mechanism of Runx2 gene in fracture healing
Wu Yukun1, Han Jie2, Wen Shuaibo1
- 1Ruikang Hospital Affiliated to Guangxi University of Chinese Medicine, Nanning 530011, Guangxi Zhuang Autonomous Region, China; 2Department of Joint and Sports Medicine, Ruikang Hospital Affiliated to Guangxi University of Chinese Medicine, Nanning 530011, Guangxi Zhuang Autonomous Region, China
-
Received:2020-05-06Revised:2020-05-12Accepted:2020-06-17Online:2021-05-18Published:2020-12-31 -
Contact:Han Jie, Associate chief physician, Master’s supervisor, Department of Joint and Sports Medicine, Ruikang Hospital Affiliated to Guangxi University of Chinese Medicine, Nanning 530011, Guangxi Zhuang Autonomous Region, China -
About author:Wu Yukun, Master candidate, Ruikang Hospital Affiliated to Guangxi University of Chinese Medicine, Nanning 530011, Guangxi Zhuang Autonomous Region, China -
Supported by:Guangxi Medical Hygiene Appropriate Technology Development, Promotion and Application Project, No. S2019070 (to HJ); “Qihuang Engineering” Project for High-level Talent Team Training Cultivation of Guangxi University of Chinese Medicine, No. 04B1804804 (to HJ); Youth Teacher Scientific Research Ability Improvement Project of Guangxi Universities, No. 2020KY07017 (to HJ)
CLC Number:
Cite this article
Wu Yukun, Han Jie, Wen Shuaibo. Mechanism of Runx2 gene in fracture healing[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(14): 2274-2279.
share this article

2.1 Runx2基因的结构及亚型 2.1.1 Runx2基因的结构 Runx2被称为核心结合因子al (core-bindingfactor al,Cbfa1)或多瘤病毒增强子结合蛋白(polyoma enhancer-binding protein2aA,Pebp2aA)。Runx蛋白是一类转录因子蛋白的统称,Runt域家族是由3个成员所组成的:Runx1(Cbfa2/Pebp2aB),Runx2(Cbfa1/Pebp2aA)以及Runx3(Cbfa3/Pebp2aC),这3个基因都含有一个runt结构域,即DNA结合结构域,与果蝇配对规则基因runt同源[3]。Runx蛋白在体外与转录辅激活因子核心结合因子b(Cbfb/多瘤增强子结合蛋白2b)形成异二聚体转录因子。Runx家族中3个基因所起的作用也不同:Runx1和Cbfb对于造血干细胞分化至关重要,Runx1和Cbfb基因的特殊易位是与急性髓系白血病相关;Runx2单侧缺失会导致颅骨锁骨发育不良;而Runx3在胃上皮细胞的生长调节和神经发生中起重要作用,被认为与胃癌有关[4-5](表1);此外,Runx1和Runx3是胸腺细胞发育所必需的[6]。 "

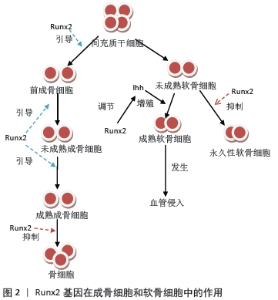
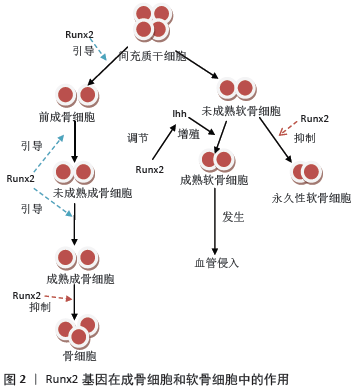
2.1.2 Runx2基因的亚型 Runx2基因是骨骼发育所必需的转录因子,它对于成骨细胞分化以及骨形成是必不可少的,这已经在大量的体外实验中得到证实。Runx2基因大致分布220 kb,它包含8个术语的外显子,为1,2,3,4,5,6,6.1,7,内含子的大小是已知的,它有3种不同n端序列的亚型:分别是MRIPV为起始氨基酸序列的Ⅰ型,MASNS为起始氨基酸序列的Ⅱ型以及MLHSPH为起始氨基酸序列的Ⅲ型[7],但是MLHSPH的序列的亚型尚未得到相关实验的证明。Ⅰ型含有507个氨基酸,在间充质干细胞、骨祖细胞、软骨细胞和胸腺细胞中组成性表达;Ⅱ型包含521个氨基酸,表达仅限于骨细胞,由骨形态发生蛋白2在骨祖细胞中诱导,并在成骨细胞分化过程中增加[8]。 2.2 Runx2基因的调控和转录 Runx2是结合特定的DNA序列来调节许多基因的转录,从而控制成骨细胞从间充质干细胞向骨细胞的发育和成熟。Runx2的Ⅰ型和Ⅱ型都是以两个不同的启动子所驱动表达,为P1的远端启动子和P2的近端启动子,Ⅰ型开始转录在近端启动子P2,而Ⅱ型开始表达在远端启动子P1[9]。有实验表明Ⅰ型和Ⅱ型均在成骨和终末高营养软骨中表达,来源于P1的Ⅱ型还具有骨特异性。此外P2衍生的Ⅰ型也在早期肥大软骨细胞和前肥大软骨细胞中表达[10]。这两种异构体都是骨骼发育所必须的。P1衍生的Ⅱ型已被证明在分化成骨细胞和响应骨形态发生蛋白2中高度表达,在成骨分化过程中表达上调,在非骨组织中表达缺失[7];相比之下,来源于P2衍生的Ⅰ型常在成骨细胞、前成骨细胞甚至非成骨细胞间质细胞中表达,其Ⅰ型的表达在成骨过程中未发生改变,这两种异构体在几个靶基因的启动子上表现出相似但不完全相同的生物学活性[11]。 2.3 Runx2基因与骨代谢间的关系 骨折是指骨的力学完整性丧失, 同时也包括局部软组织与血管的损伤。骨组织创伤局部的修复是一个连续不断的愈合过程,一面清除破坏,一面再生修复,其本质就是骨折断端发生的骨重建塑型过程。由成骨细胞主导的骨形成与由破骨细胞主导、成骨细胞和破骨细胞共同调控的骨吸收,加上软骨细胞在骨重建塑性过程中起重要作用。Runx2作为骨特异性转录因子, 促进成骨细胞和软骨细胞转录和活化, 影响到成骨细胞介导的骨形成及破骨细胞介导的骨吸收,进而参与到骨折断端的重建塑型过程当中。 2.3.1 Runx2基因在成骨细胞中的作用 在Runx2基因缺陷的小鼠中缺乏成骨细胞标记基因的表达,小鼠体内完全缺失成骨细胞,这表明Runx2对于早期间充质干细胞向成骨细胞分化至关重要,是成骨细胞分化的重要转录因子[12]。为了检验Runx2在成骨细胞分化中的功能,最近有实验报道了在2.3-kb Col1a1启动子的控制下使用Cre转基因小鼠的Runx2条件敲除小鼠,该启动子将转基因表达导向已表达的成骨细胞,含有runt结构域一部分的外显子4的条件缺失不会导致显性表型[13];然而有条件的缺失外显子8的小鼠则会由于骨形成减少而导致骨质减少的发生,同时会产生截短的Runx2蛋白,后一组小鼠破骨细胞的生成也随之减少[14]。由于这些研究使用了相同的Cre转基因系,Cre转基因的表达水平似乎不是导致这些表型差异的原因。但是由于这些小鼠的遗传背景不同,这可能解释了所观察到的差异。还有实验通过使用2.3-kb Col1a1启动子在小鼠中过度表达Runx2,小鼠出现骨质疏松的情况,成骨细胞成熟以及成骨细胞向骨细胞的转化都受到严重抑制,从而引起骨形成减少而导致骨质的减少,同时还发现Runx2的表达最初在成骨前细胞中检测到,在胚胎和幼鼠中强烈表达,但在成年小鼠中逐渐降低,Runx2在未成熟成骨细胞中增加,然后在成骨细胞成熟过程中逐渐减少[15]。此外,在Runx2转基因小鼠中几乎没有骨细胞,这表明Runx2抑制成骨细胞向骨细胞的转化。因此,通过Runx2转基因小鼠的表现可以说明,维持Runx2的强烈表达在成骨细胞当中可抑制成骨细胞本身的成熟,并使成骨细胞处于未成熟阶段[16]。Runx2在成骨细胞分化的早期起到了促进作用,而随着成骨细胞分化成熟的过程中则逐渐起到抑制作用(图2)。 Runx2的功能在成骨细胞分化早期,体现在成骨细胞中所产生的主要骨基质蛋白基因(Col1a1、骨桥蛋白、骨涎蛋白和骨钙素),同时成骨细胞也决定了这些基因在Runx2中的DNA结合位点,Runx2激活这些骨基质蛋白基因在成骨细胞分化过程中的不同程度的表达 [12]。它还可以调节成骨细胞分化关键转录因子的Sp7蛋白(serine protease 7)以及骨基质基因Spp1、Ibsp、Bglap2的表达,间充质干细胞通过Runx2分化为不成熟的成骨细胞表达骨骼基质蛋白基因,在进入成骨细胞后根据细胞的成熟程度表达骨基质蛋白基因,未成熟的成骨细胞表达Spp1和Ibsp,而成熟的成骨细胞则表达Bglap2,最终成熟的成骨细胞被嵌入骨基质变成骨细胞[17]。其中Sp7对成骨细胞分化也是至关重要,因为Runx2在Sp7缺失的小鼠中正常表达,而Sp7在Runx2 缺失的小鼠中不表达,Sp7被认为是Runx2基因的下游[18]。有实验使用过度表达Runx2的小鼠肢芽细胞进行了微阵列分析,随后的微阵列分析数据也充分说明了Sp7在细胞中的表达能力显著提升,Sp7是成骨细胞生成必不可少的转录因子,其表达离不开Runx2的存在[19]。同时还有研究证明了miRNA外泌体也参与到Runx2对于成骨细胞的调控中,其中MicroRNA-137-3p可以直接作用于Runx2来防止激素诱导的股骨头坏死,促进成骨和血管生成[20]。还有学者发现泛素连接酶WWP2通过增强非蛋白水解、单泛素化的方法来催化Runx2从而增强它的转录激活活性,继而促进成骨分化[21]。 2.3.2 Runx2在软骨细胞中的作用 小鼠的骨骼构造基本上由软骨组成,在缺失Runx2基因的小鼠中,在大部分骨骼中软骨细胞的成熟受到严重抑制,以至软骨细胞的增殖减少,小鼠的四肢长度都较短[22]。Col2a1在未成熟软骨细胞中表达,而Col10a1则在成熟软骨细胞中表达,同时在胫骨、腓骨、桡骨和尺骨中表达也受到限制,出现在终末分化的软骨细胞中表达的Spp1、Ibsp和基质金属蛋白酶13在整个骨骼中几乎缺失[17]。这些结果表明软骨细胞成熟需要Runx2基因从中进行调控。Ihh信号传导对软骨内骨骼成骨细胞的发育至关重要,在软骨发育过程中,Ihh主要由肥大前软骨细胞和早期肥大软骨细胞表达,Ihh信号传递给未成熟软骨细胞和上覆软骨膜细胞 [23]。在Runx2缺失的小鼠中,肥大前软骨细胞中表达的Ihh表达严重减少。在Ihh的启动子区域已经发现了几个Runx2结合基序,结果证实了Runx2是直接调控Ihh的表达[24-25]。因此,Runx2不仅通过Ihh调节软骨细胞成熟,还通过Ihh调节软骨细胞增殖。总而言之,Runx2调节Col10a1、Spp1、Ibsp和基质金属蛋白酶13在软骨细胞中的表达,它还能抑制软骨细胞获得永久性软骨细胞的表型,并且通过调节Ihh的表达来调节软骨细胞的增殖(图2)。 Runx2在非肥大软骨细胞中的表达是软骨细胞肥大过程的必要部分,软骨细胞肥大是软骨内骨化的必经步骤。在骨骼发育开始时,在骨骼中进行软骨内骨化的区域,未分化的间充质细胞发生了具有未来骨骼元素形状的凝聚现象,在这些间充质凝聚中的细胞分化为软骨细胞,表达特定的分子标记,而冷凝周围的未分化间充质细胞形成软骨膜,一旦这些软骨膜形成,其中心的软骨细胞进一步分化为肥大的软骨细胞[26]。软骨细胞肥大时,软骨膜细胞分化为成骨细胞,在软骨核周围形成骨领。然而在Runx2缺失的小鼠中成骨细胞分化被完全阻断,软骨膜中没有成骨细胞的存在,因此软骨膜中没有成骨细胞去影响软骨细胞的成熟[17]。这些结果表明Runx2还能通过调节软骨膜成骨细胞分化直接或间接地调节软骨细胞成熟。 Runx2是软骨细胞成熟过程中的一个重要调节因子,调节软骨细胞的增殖和凋亡[27]。ENOMOTO等[28]发现以外显子2为起始点的反义寡核苷酸在软骨细胞系ATDC5细胞中处理Runx2,可以抑制软骨细胞的成熟,但是在雏鸡的未成熟软骨细胞中反转录强制表达Runx2,诱导了软骨细胞的成熟。有实验在使用Col2a1启动子在软骨细胞中过度表达Runx2,证明其可以加速软骨细胞成熟和软骨内成骨[29]。软骨细胞的成熟有时候甚至发生在永久性软骨中,包括关节软骨、甲状软骨、环状软骨、气管软骨和椎间盘。相比之下,软骨细胞中dn-Runx2(显性负突变)的表达可减慢软骨细胞的成熟和软骨内成骨,Runx2可抑制软骨细胞获得永久软骨的特征[30]。由于Runx2和dn-Runx2仅在软骨细胞中表达,Runx2是直接调控软骨细胞的成熟。即使在软骨细胞成熟的Runx2缺失的小鼠限制性骨骼中,没有出现血管侵犯,这表明血管侵犯软骨也需要Runx2的参与[31]。 2.3.3 Runx2在破骨细胞中的作用 软骨内骨化过程中,软骨细胞成熟为肥大软骨细胞,终末肥大软骨细胞周围基质矿化,血管侵入钙化软骨以至软骨被骨替代,破骨细胞通过吸收钙化基质来加速这些过程导致骨髓形成。Runx2可以调节骨保护素和成骨细胞所表达的核因子κB受体激活因子配体 (receptor activator nuclear factor kappa-B ligand, Rankl) 来参与破骨细胞的分化过程,骨保护素和Rankl作为成骨细胞来源的旁分泌细胞因子,在体外的培养细胞中表达与Runx2密切相关,这些细胞因子对破骨细胞功能至关重要[32]。有研究证明17β-雌二醇在体内和体外可以减弱Runx2活性,消除了Runx2介导的Rankl的增加,强烈抑制Runx2所介导的成骨细胞驱动破骨细胞的生成。因此,由Runx2驱动的Rankl向骨髓前成骨细胞表面的转运的过程中可以与相邻的单核细胞上的受体Rank结合,促进破骨细胞的分化[33]。Runx2通过诱导Rankl和抑制骨保护素促进破骨细胞分化,还可以激活骨钙素、骨桥蛋白、骨涎蛋白和Ⅰ型胶原基因的转录与表达[34],但是Runx2与Rankl和骨保护素之间关系还需要进一步研究。还有研究发现溶酶体相关跨膜蛋白5(lysosomal-associated protein transmembrane5,LAPTM5)的表达是通过Runx2与其启动子区域的相互作用来调控的,并且LAPTM5参与了Rankl的转运。这些发现提示了成骨和破骨细胞形成之间可能存在耦合机制,其中Runx2可能通过调节溶酶体相关基因参与破骨细胞分化,从而调节RANKL的表达[35]。 2.4 Runx2基因与WNT信号通路 Wnt蛋白是一个由19个种分泌糖蛋白组成的家族,可与卷曲蛋白(Frizzled)家族的受体以及脂蛋白受体5/6(LRP5/6)相互作用,从而调节细胞多种多样的生物过程,如细胞的凋亡、增殖、分化等[36]。Wnt信号通路是由Wnt蛋白和膜蛋白受体结合的一组多下游通道的信号转导途径,分为经典、非经典两种途径,经典Wnt通路依赖β-catenin,而非经典通路则是β-catenin独立,Wnt信号通路是骨形成的关键调控因子,具有强大的诱导成骨功能[37-38]。Wnt3a和Wnt10a 可以通过Wnt信号通路激活Runx2转录因子表达,促进成骨细胞分化,从而促进骨形成[39]。有实验发现Runx2缺陷细胞中的典型Wnt信号传导受阻,Wnt10b表达程度减少80%,表明Runx2与Wnt10b的启动子结合,驱动Wnt10b的表达,通过增强β-catenin信号通路来抑制脂肪细胞生成,对成骨细胞的形成和维持至关重要[39]。WNT信号可能是胚胎中Runx2基因表达的重要激活因子,Wnt信号对Runx2的调控与骨形态发生蛋白信号转导途径一样重要,后者也诱导Runx2基因的表达。 2.5 Runx2基因与骨形态发生蛋白/转化生长因子β信号通路 转化生长因子β控制着大量的细胞功能,它们的活性对于调节众多的发育和稳态过程至关重要,骨形态发生蛋白是转化生长因子b超家族的成员,在骨形成和修复过程中作为细胞信号分子发挥着重要的调控作用[40-41]。骨形态发生蛋白有助于促进成骨分化,包括刺激骨结构蛋白如Ⅰ型胶原和骨钙素的表达,以及骨基质的矿化。Smad蛋白是骨形态发生蛋白和转化生长因子β信号的胞内效应蛋白,Smad1/5/8信号通路是一个成骨相关通路,骨形态发生蛋白通过Smad1/5/8信号通路的磷酸化作用与骨形态发生蛋白R2结合调节细胞的分化和生长等功能,Smad1/5/8蛋白转运到细胞核,与靶基因结合,调控骨髓间充质干细胞成骨分化[42]。在这方面,有一项研究首先证明Smad4在软骨细胞肥大过程中通过直接结合Runx2调节元件转录激活Runx2表达,提示Runx2在软骨生长和骨形成中的重要作用[43]。在表达Runx2的细胞中,Smad复合物被定向结合到含有核基质结合位点的Runx2,后者是活性转录位点,增强Runx2的转录表达[44]。在以往的研究中,Runx2被认为是转化生长因子β/骨形态发生蛋白信号转导的一个常见下游基因,尽管背后的机制还不清楚,但是Runx2与骨形态发生蛋白激活的smad合作,可以刺激骨髓间充质干细胞的成骨分化。 2.6 Runx2基因与甲状旁腺激素 甲状旁腺激素由甲状旁腺产生,在调节磷酸和钙代谢中起关键作用,Runx2的活性也受甲状旁腺激素和甲状旁腺相关肽(PTHrPs)的调节。在正常生理状态下,甲状旁腺激素是骨代谢的有效调节因子。治疗上甲状旁腺激素对骨形成的影响因其浓度和给药频率而大不相同,持续性暴露于甲状旁腺激素或高水平的甲状旁腺激素会降低骨密度,并抑制参与骨形成的Runx2基因的表达,相反间歇性或低水平的甲状旁腺激素增加骨量,可诱导骨合成[45]。甲状旁腺激素对Runx2表达的影响是剂量依赖性的,包括Runx2的上调和下调取决于甲状旁腺激素的浓度,所以甲状旁腺激素是骨代谢的双刃剑。有研究用1×10-8 mol/L 重组人甲状旁腺激素 (1-34) 诱导人根尖牙乳头干细胞,实验组中根尖牙乳头干细胞的Runx2表达明显增强,提示重组人甲状旁腺激素(1-34)可以刺激Runx2的表达,从而促进根尖牙乳头干细胞的成牙/成骨向分化[46]。采用逐层组装的方法在甲状旁腺激素组装在不同层(5,7和10)的钛表面上,培养出了骨髓间充质干细胞,证实甲状旁腺激素可以增强Runx2的表达,促进了成骨细胞的分化[47]。 2.7 Runx2基因与成纤维细胞生长因子 成纤维细胞生长因子蛋白是增强Runx2活性、促进体内骨形成的一个重要调节因子[48]。成骨细胞中成纤维细胞生长因子信号激活蛋白激酶c(PKC)、胞外信号调节激酶(ERK)和p38类MAP激酶,这些通路的激活使Runx2的羧基端磷酸化,增加了Runx2的DNA结合,增强了骨钙素启动子的表达[49-50]。PKC通路在很大程度上通过上调Runx2的表达来调节成纤维细胞生长因子2刺激的Runx2活性,同时它也在翻译后修饰Runx2蛋白,从而增强其转录活性,然而阻断PKC会抑制成纤维细胞生长因子2去诱导Runx2的表达,成纤维细胞生长因子2刺激的Runx2的DNA结合活性也被阻断,因此成纤维细胞生长因子/成纤维细胞生长因子受体信号可以增强Runx2的DNA结合和转录活性、刺激其表达,这一作用很大程度上受到PKC途径的调控[51]。有研究为了检测Runx2在成纤维细胞生长因子信号通路中的作用,在实验中发现在Runx2缺失小鼠中没有成骨祖细胞的增殖,成纤维细胞生长因子受体2和成纤维细胞生长因子受体3在成骨祖细胞增殖中的表达严重减少,说明Runx2对成骨祖细胞的增殖是必需的,Runx2通过调节其启动子诱导成纤维细胞生长因子受体2和成纤维细胞生长因子受体3的表达,促进成骨细胞祖细胞增殖[52]。 "
| [1] TZIOUPIS C, GIANNOUDIS PV.Prevalence of long-bone non-unions.Injury. 2007;38 Suppl 2:S3-9. [2] HAK DJ, FITZPATRICK D, BISHOP JA,et al. Delayed union and nonunions: epidemiology, clinical issues, and financial aspects.Injury. 2014;45 Suppl 2:S3-7. [3] WYSOKINSKI D, BLASIAK J, PAWLOWSKA E.Role of RUNX2 in Breast Carcinogenesis.Int J Mol Sci.2015;16: 20969-20993. [4] YOKOMIZO-NAKANO T,KUBOTA S,BAI J,et al.Overexpression of RUNX3 represses RUNX1 to drive transformation of myelodysplastic syndrome.Cancer Res.2020;80(12):2523-2536. [5] OTÁLORA-OTÁLORA BA, HENRÍQUEZ B, LÓPEZ-KLEINE L,et al.RUNX family: Oncogenes or tumor suppressors (Review). Oncol Rep. 2019;42: 3-19. [6] 黄耀星,余丹纯,孙小娟等.CD74与RUNX3在人胰腺癌细胞内存在相互作用[J].分子影像学杂志,2019,42(3):393-396. [7] Ito Y, Bae SC, Chuang LS. The RUNX family: developmental regulators in cancer. Nat Rev Cancer. 2015;15:81-95. [8] Bae SC,Lee YH.Phosphorylation, acetylation and ubiquitination: The molecular basis of RUNX regulation. Gene. 2006;366(1):58-66. [9] NIEKE S, YASMIN N, KAKUGAWA K, et al.Unique N-terminal sequences in two Runx1 isoforms are dispensable for Runx1 function. BMC Dev Biol. 2017;17(1):14. [10] OKURA H, SATO S, KISHIKAWA S,et al.Runx2-I isoform contributes to fetal bone formation even in the absence of specific N-terminal amino acids. PLoS ONE. 2014;9:e108294. [11] TAKAHASHI A, DE ANDRÉS MC, HASHIMOTO K, et al.DNA methylation of the RUNX2 P1 promoter mediates MMP13 transcription in chondrocytes. Sci Rep. 2017;7:7771. [12] KIM HJ, KIM WJ, RYOO HM. Post-Translational Regulations of Transcriptional Activity of RUNX2. Moleculer Cells. 2019;43(2):160-167. [13] TAKARADA T, HINOI E, NAKAZATO R, et al. An analysis of skeletal development in osteoblast-specific and chondrocyte-specific runt-related transcription factor-2 (Runx2) knockout mice. J Bone Miner Res. 2013;28:2064-2069. [14] ADHAMI MD, RASHID H, CHEN H, et al.Loss of Runx2 in committed osteoblasts impairs postnatal skeletogenesis. J Bone Miner Res. 2015;30:71-82. [15] KOMORI T. Animal models for osteoporosis. Eur J Pharmacol. 2015;759: 287-294. [16] MORIISHI T, FUKUYAMA R, ITO M, et al. Osteocyte network; a negative regulatory system for bone mass augmented by the induction of Rankl in osteoblasts and Sost in osteocytes at unloading. PLoS One. 2012;7(6):e40143. [17] KOMORI T. Roles of Runx2 in Skeletal Development. Adv Exp Med Biol. 2017;962:83-93. [18] KOMORI T. Regulation of Proliferation, Differentiation and Functions of Osteoblasts by Runx2. Int J Mol Sci. 2019;20(7):1694. [19] NISHIMURA R, WAKABAYASHI M, HATA K, et al. Osterix Regulates Calcification and Degradation of Chondrogenic Matrices through Matrix Metalloproteinase 13 (MMP13) Expression in Association with Transcription Factor Runx2 during Endochondral Ossification. J Biol Chem. 2012;287(40):33179-33190. [20] KONG L, ZUO R, WANG M, et al.Silencing MicroRNA-137-3p, which Targets RUNX2 and CXCL12 Prevents Steroid-induced Osteonecrosis of the Femoral Head by Facilitating Osteogenesis and Angiogenesis. Int J Biol Sci. 2020;16(4):655-670. [21] ZHU W, HE X, HUA Y, et al. The E3 ubiquitin ligase WWP2 facilitates RUNX2 protein transactivation in a mono-ubiquitination manner during osteogenic differentiation. J Biol Chem. 2017;292:11178-11188. [22] Komori T. Runx2, an inducer of osteoblast and chondrocyte differentiation. Histochem Cell Biol. 2018;149:313-323. [23] Kurio N, Saunders C, Bechtold TE, et al. Roles of Ihh signaling in chondroprogenitor function in postnatal condylar cartilage. Matrix Biol. 2018;67:15-31. [24] Liao L, Zhang S, Zhou GQ, et al. Deletion of Runx2 in condylar chondrocytes disrupts TMJ tissue homeostasis. J Cell Physiol. 2019;234: 3436-3444. [25] Liao L, Jiang H, Fan Y, et al. Runx2 is required for postnatal intervertebral disc tissue growth and development. J Cell Physiol. 2019; 234(5):6679-6687. [26] Provot S, Schipani E. Molecular mechanisms of endochondral bone development. Biochem Biophys Res Commun. 2005;328(3):658-665. [27] Jiang Q, Qin X, Yoshida CA, et al.Antxr1, Which is a Target of Runx2, Regulates Chondrocyte Proliferation and Apoptosis. Int J Mol Sci. 2020;21(7):2425. [28] ENOMOTO H, ENOMOTO-IWAMOTO M, IWAMOTO M, et al. Cbfa1 Is a Positive Regulatory Factor in Chondrocyte Maturation. J Biol Chem. 2000;275(12):8695-8702. [29] CATHELINE SE, HOAK D, CHANG M, et al.Chondrocyte-Specific RUNX2 Overexpression Accelerates Post-traumatic Osteoarthritis Progression in Adult Mice. J Bone Miner Res. 2019;34(9):1676-1689. [30] UETA C, IWAMOTO M, KANATANI N, et al. Skeletal malformations caused by overexpression of Cbfa1 or its dominant negative form in chondrocytes. J Cell Biol. 2001;153:87-100. [31] HIMENO M, ENOMOTO H, LIU W, et al. Impaired vascular invasion of Cbfa1-deficient cartilage engrafted in the spleen.J Bone Miner Res. 2002;17:1297-305. [32] HOFBAUER LC, KÜHNE CA, VIERECK V. The OPG/RANKL/RANK system in metabolic bone diseases. J Musculoskelet Neuronal Interact. 2004; 4(3):268-275. [33] MARTIN A, XIONG J, KOROMILA T, et al. Estrogens antagonize RUNX2-mediated osteoblast-driven osteoclastogenesis through regulating RANKL membrane association. Bone. 2015;75: 96-104. [34] ENOMOTO H, SHIOJIRI S, HOSHI K, et al. Induction of osteoclast differentiation by Runx2 through receptor activator of nuclear factor-kappa B ligand (RANKL) and osteoprotegerin regulation and partial rescue of osteoclastogenesis in Runx2-/- mice by RANKL transgene. J Biol Chem. 2003;278:23971-23977. [35] GENG YM, LIU CX, LU WY, et al. LAPTM5 is transactivated by RUNX2 and involved in RANKL trafficking in osteoblastic cells. Mol Med Rep. 2019;20(5):4193-4201. [36] MOHAMMED MK, SHAO C, WANG J, et al. Wnt/β-catenin signaling plays an ever-expanding role in stem cell self-renewal, tumorigenesis and cancer chemoresistance. Genes Dis. 2016;3:11-40. [37] HUYBRECHTS Y, MORTIER G, BOUDIN E, et al.WNT Signaling and Bone: Lessons From Skeletal Dysplasias and Disorders. Front Endocrinol (Lausanne). 2020;11:165. [38] CHEN Y, HU Y, YANG L, et al. Runx2 alleviates high glucose-suppressed osteogenic differentiation via PI3K/AKT/GSK3β/β-catenin pathway. Cell Biol Int. 2017;41:822-832. [39] WU M, WANG Y, SHAO JZ, et al. Cbfβ governs osteoblast-adipocyte lineage commitment through enhancing β-catenin signaling and suppressing adipogenesis gene expression. Proc Natl Acad Sci U.S.A. 2017;114:10119-10124. [40] YU X, SHEN G, REN H, et al. TGFβ-induced factor homeobox 2 blocks osteoblastic differentiation through targeting pSmad3/HDAC4/H4ac/Runx2 axis. J Cell Physiol. 2019;234:21284-21293. [41] YANG B, LIN X, TAN J, et al. Root bark of Sambucus Williamsii Hance promotes rat femoral fracture healing by the BMP-2/Runx2 signaling pathway. J Ethnopharmacol. 2016;191:107-114. [42] LIU DD, ZHANG JC, ZHANG Q, et al. TGF‐β/BMP signaling pathway is involved in cerium‐promoted osteogenic differentiation of mesenchymal stem cells. J Cell Biochem. 2013;114(5):1105-1114. [43] YAN J, LI J, HU J, et al. Smad4 deficiency impairs chondrocyte hypertrophy via the Runx2 transcription factor in mouse skeletal development. J Biol Chem. 2018;293:9162-9175. [44] KOPF J, PAARMANN P, HIEPEN C, et al. BMP growth factor signaling in a biomechanical context. Biofactors. 2014;40:171-187. [45] QIN L, RAGGATT LJ, PARTRIDGE NC. Parathyroid hormone: a double-edged sword for bone metabolism. Trends Endocrinol Metab. 2004;15: 60-65. [46] 庄颖,庞希瑶,蔡琴,等.rhPTH(1-34)间断刺激对人根尖牙乳头干细胞增殖及成牙成骨分化的影响[J].口腔生物医学,2018,9(3): 125-128. [47] LAI K, XI Y, MIAO X, et al. PTH coatings on titanium surfaces improved osteogenic integration by increasing expression levels of BMP-2/Runx2/Osterix. Rsc Advances. 2017;7(89):56256-56265. [48] QIN X, JIANG Q, MIYAZAKI T, et al. Runx2 regulates cranial suture closure by inducing hedgehog, Fgf, Wnt and Pthlh signaling pathway gene expressions in suture mesenchymal cells.Hum Mol Genet. 2019;28:896-911. [49] Kim JM, Yang YS, Park KH, et al. The ERK MAPK Pathway Is Essential for Skeletal Development and Homeostasis.Int J Mol Sci. 2019;20(8): 1803. [50] Li Y, Ge C, Franceschi RT. MAP Kinase-Dependent RUNX2 Phosphorylation Is Necessary for Epigenetic Modification of Chromatin During Osteoblast Differentiation. J Cell Physiol. 2017;232(9): 2427-2435. [51] KIM HJ, KIM JH, BAE SC, et al. The protein kinase C pathway plays a central role in the fibroblast growth factor-stimulated expression and transactivation activity of Runx2. J Biol Chem. 2003;278(1):319-326. [52] KAWANE T, QIN X, JIANG Q, et al. Runx2 is required for the proliferation of osteoblast progenitors and induces proliferation by regulating Fgfr2 and Fgfr3. Sci Rep. 2018;8(1):13551. |
| [1] | Min Youjiang, Yao Haihua, Sun Jie, Zhou Xuan, Yu Hang, Sun Qianpu, Hong Ensi. Effect of “three-tong acupuncture” on brain function of patients with spinal cord injury based on magnetic resonance technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [8] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [9] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [10] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [11] | Tan Jingyu, Liu Haiwen. Genome-wide identification, classification and phylogenetic analysis of Fasciclin gene family for osteoblast specific factor 2 [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1243-1248. |
| [12] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [13] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [14] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [15] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||