Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (23): 3730-3736.doi: 10.3969/j.issn.2095-4344.2689
Previous Articles Next Articles
Effect of exercise-induced irisin on myocardial fibrosis
Yin Lian1, 2, Zhao Jin1, Lei Xuemei1, Li Miaomiao1, Wang Kun1, Zhang Tingran1, Luo Jiong1
- 1Research Centre for Exercise Detoxification, College of Physical Education, Southwest University, Chongqing 400715, China; 2Chongqing Medical and Health School, Chongqing 408000, China
-
Received:2019-09-30Revised:2019-10-09Accepted:2019-11-15Online:2020-08-18Published:2020-07-30 -
Contact:Luo Jiong, PhD, Professor, Research Centre for Exercise Detoxification, College of Physical Education, Southwest University, Chongqing 400715, China -
About author:Yin Lian, Master candidate, Research Centre for Exercise Detoxification, College of Physical Education, Southwest University, Chongqing 400715, China -
Supported by:the Scientific Research Project of Chongqing Administration of Sports, No. C201811
CLC Number:
Cite this article
Yin Lian, Zhao Jin, Lei Xuemei, Li Miaomiao, Wang Kun, Zhang Tingran, Luo Jiong. Effect of exercise-induced irisin on myocardial fibrosis[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(23): 3730-3736.
share this article
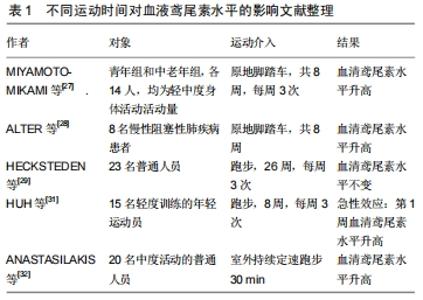
2.1 心肌纤维化概述 心肌纤维化是指心脏成纤维细胞的活化和过度增殖,导致胶原纤维过度积聚、胶原含量升高和胶原容积增高的病理性变化。随着心肌纤维化的发生,会出现心肌僵硬度增加、心肌顺应性降低、心肌的收缩和舒张功能发生障碍。此外,心肌纤维化会引起心肌组织的电传导异常,如传导延缓和传导阻滞等,增加心律失常发生甚至猝死的危险性[16]。心肌纤维化发生的细胞生物学机制是心肌组织中发生大量的成纤维细胞增殖,转化形成肌成纤维细胞造成胶原蛋白的合成能力增强,其发生机制有神经内分泌因素肾素-血管紧张素系统的调节,还有氧化应激和炎症反应的影响。其中氧化应激和炎症反应主要通过促进促纤维化细胞生长因子的表达和诱导成纤维化细胞转化为肌成纤维化细胞,进而导致心肌纤维化的发生。慢性发炎会降低心肌收缩能力、引起心肌纤维化,从而导致心肌重塑[10,17]。正常生理状况下,正常心脏组织不会持续出现高浓度肿瘤坏死因子α、白细胞介素1、白细胞介素6等炎性物质,而当心肌受到伤害或过度的机械应力时,这些炎性物质则会持续升高,其原因可能是肿瘤坏死因子α和白细胞介素6增加会直接引起心肌收缩力下降和引发心肌纤维化,其机制可能经由发炎物质活化转化生长因子β途径,进而活化纤维结合素(fibronectin),造成纤维化组织增加[18]。 氧化应激和炎症反应对心肌纤维化有促进作用,二者主要通过诱导纤维化关键因子转化生长因子β1的分泌促进心肌纤维化的发生。其主要途径为转化生长因子β1与细胞膜上的受体结合后,调节细胞内Smad分子的表达,从而主要通过3种途径介导心肌纤维化的发生:①转化生长因子β1可促进心肌成纤维细胞向肌成纤维细胞表型转化,从而促进细胞胶原蛋白的分泌能力;②通过促进结缔组织生长因子等细胞因子从而诱导心肌纤维细胞增殖进而使胶原蛋白的合成增加;③通过抑制基质金属蛋白酶活性,进而抑制胶原蛋白的降解[19]。目前心肌纤维化的主要治疗手段为药物治疗,然而选用药物治疗心肌纤维化会对机体产生一定的不良反应[20]。心肌纤维化可诱发或演变成多种心脏疾病,因此探寻一种无不良反应、对心肌纤维化的诱因均有良好效果的综合治疗手段显得尤为迫切。 2.2 运动诱发的鸢尾素在心肌纤维化中扮演的角色 2.2.1 运动产生鸢尾素 运动时骨骼肌是有分泌作用的器官之一,会释出具有不同作用的肌肉激素(myokine),能作用在肌肉本身或作用于其他器官,也可传递信息作为沟通桥梁,帮助调节代谢[21]。BOSTR?M等[12]研究发现了肌肉激素鸢尾素(irisin),并指出其能促发白色脂肪转化具有棕色脂肪的功能,增加产热,对人体健康有益。运动过程中会提高活性氧(ROS)的水平,活性氧可以激活p38丝裂原活化蛋白激酶(p38 MAPK),而p38丝裂原活化蛋白激酶又进一步调控过氧化物酶体增殖物激活受体γ辅助活化因子1α (peroxisome proliferator-activated receptorgamma coactivator-1 alpha,PGC-1α)的表现。PGC-1α上调穿膜蛋白FNDC5表达,而FNDC5于胞外被修剪释放的片段便是irisin[22]。已有研究表明,重组鸢尾素(R-鸢尾素)导致白色脂肪细胞的褐变,并经由细胞外信号相关激酶(ERK)和p38蛋白激酶降低肥胖小鼠的体质量[12]。除了骨骼肌外,AYDIN等[23]研究发现,心肌也表达高水平的FNDC5,运动后产生的鸢尾素多于骨骼肌。此外,人体研究发现鸢尾素与心脏健康紧密相关[24]。陈聪毅等[25]以动物模式仿真阻塞型睡眠呼吸中止症夜间的间歇性低氧现象,配合高脂饮食诱发肥胖建立肥胖诱发心肌纤维化模型,12周运动训练后,测得高脂饮食组心肌纤维化指标明显下降,其原因可能和运动诱发鸢尾素表达升高有关。 2.2.2 不同运动时间对血液鸢尾素水平的影响 BOSTR?M的研究中,同时进行了老鼠及人体的实验,实验结果均得到运动介入后血液中鸢尾素浓度提升;该研究结果发现,训练后血浆鸢尾素基础值显著增加,且血液中鸢尾素浓度的微幅提升,就能造成相对大量的解偶联蛋白1表达,进而增加能量消耗及改善葡萄糖耐受度。根据该研究结果,长期运动训练能提升鸢尾素浓度,并带来后续在肥胖、糖尿病等代谢疾病方面的益处。后面便出现较多关于运动介入对鸢尾素影响效益的研究[12,26],研究结果却不尽相同。 (1)长时间运动对血液鸢尾素的影响:较多研究结果显示长时间运动训练介入使血液鸢尾素水平提高。MIYAMOTO-MIKAMI等[27]研究轻中度活动量时发现,进行8周的原地脚踏车运动,年轻组血液鸢尾素水平无显著变化,而中老年组则显著上升。ALTER等[28]研究平均年龄较高的慢性阻塞性肺疾病患者,研究对象都为症状相对较严重的住院患者,介入期间分别为总住院期及2个月,试验分为3组,一组只进行物理治疗、二组除物理治疗外再加6 min的全身性震动运动、三组为30 min直立式原地踏车运动,8周后血液鸢尾素水平二组较一组高,三组血液鸢尾素基础值则显著上升。HECKSTEDEN等[29]则认为,样本存放天数可能会影响鸢尾素降解程度,并得出其变化之回归曲线,通过进行24周的阻力运动训练后鸢尾素基础值提升,但经过样本存放天数的调整修正后,显著差异便不复存在,这提示样本存放时间可能会影响长期运动介入的比较结果。上述长时间运动介入多关注健康层面,希望通过长期规律运动改善身体活动量,证实了运动对血液鸢尾素水平提升的结果。其他研究显示,进行24周的阻力运动训练后鸢尾素水平无明显提升,表明长时运动训练对血液鸢尾素水平无明显影响[30]。 (2)单次运动对血液鸢尾素的影响:相较于长期运动而言,较多试验证明单次运动对提升血液鸢尾素浓度有显著效果。HUH等[31]以15名年龄19-21岁的男性为研究对象,运动方式为2组2趟80 m冲刺,组间休息20 min,运动后30 min测量血清鸢尾素浓度上升。ANASTASILAKIS等[32]的研究对象为20名受试者,10男10女,年龄(20.0±0.1)岁,体质量指数为(23.4± 0.2) kg/m2,进行中度身体活动,运动方式为室外持续定速跑步30 min,试验后测得血清鸢尾素浓度升高且血清鸢尾素水平具有日夜节律。此外,在其他以跑步为干预方法的研究中,递增跑步强度至衰竭、原地脚踏车运动方式介入,HUH等以跑步、自由泳等运动方式介入,均在单次运动后测得血清鸢尾素浓度升高[33-34]。 部分试验结果显示单次运动对血液鸢尾素水平无显著影响。IJIRI等[35]对15名慢性阻塞性肺病患者进行 36 min原地脚踏车渐增强度运动,前30 min每6 s增加1 W,试验后测得血清鸢尾素无显着差异。HEW-BUTLER等[36]以跑步为运动介入方式,跑步机V4O2peak测验,每分钟增加0.8 km/h,试验设置跑步组和非跑步组,两组男女比例和年龄相近,运动后 15 min测量血液鸢尾素水平无显着差异。 TSUCHIYA等[37]进行总能量消耗相同的高、低强度跑步比较,运动后立即测得低强度组的鸢尾素浓度有显著变化,运动后3 h高、低强度组皆与基础值无显著差异,运动后6,19 h两组皆显著高于基础值,且高强度组又显著高于低强度组,结果说明单次运动对血液鸢尾素水平的影响可能和运动强度有关,相较于中低强度运动,高强度运动使鸢尾素浓度上升的幅度可能更大。 在研究运动时间对血液鸢尾素水平影响的文献中,选择的研究对象、运动方式、运动强度、运动频率不同(表1),这可能是导致得出的结果不相同的原因,也可能是因为试验对象的身体情况、年龄、健康状况影响了运动对血液鸢尾素的作用,但具体原因机制还尚未明确。 "
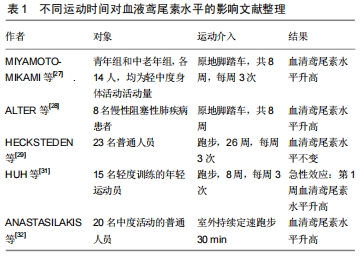
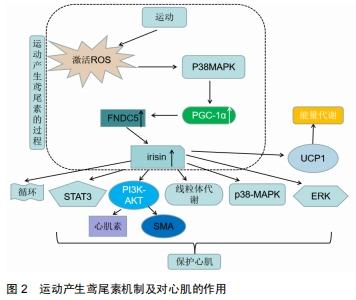
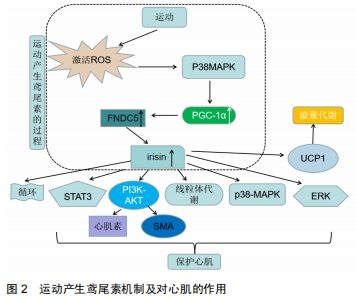
2.2.3 其他因素对血液鸢尾素水平的影响研究 有研究报告温度也可影响血液鸢尾素水平,LEE等[26]通过对10名健康成年人进行分级渐进式冷暴露后,发现冷暴露会增加循环鸢尾素的含量。其他研究发现禁食大鼠的FNDC5分泌降低,FNDC5裂解后即转化成鸢尾素,因此FNDC5分泌的降低会导致循环鸢尾素水平的下降[14]。因此除运动外,寒冷和饥饿可能会降低血液鸢尾素水平。 2.2.4 运动产生的鸢尾素对心肌的保护作用 图2显示:运动产生的鸢尾素可以对心肌产生保护效益。重组鸢尾素能激活PI3K-AKT信号通路,AKT是一种丝氨酸/苏氨酸蛋白激酶,可调节不同组织中的多种细胞功能,出生后在心脏发育过程中有至关重要的作用,该实验用H9C2细胞,H9C2细胞具有心脏组织的电生理学和生物化学特性[38],该细胞表面上存在鸢尾素特异性受体,通过该受体,鸢尾素可以激活心肌细胞中的ERK,p38-MAPK和STAT3信号通路,P38-MAPK和ERK涉及广泛的细胞过程,从细胞生长和增殖到凋亡[39]。已有研究表明,AKT通路可调节卵泡抑素在骨骼肌中的表达,心肌素释放和平滑肌肌动蛋白(SMA)的表达,心肌素(myocardin)是参与心血管组织特异表达的转录辅助因子,其活性的改变可影响人类主要的心血管疾病,这一过程和心肌纤维化的发生发展关系非常密切,而平滑肌肌动蛋白可启动心肌细胞分化和心脏生长[40-41]。LI 等[42-43]研究证明Ca2+信号可以激活心肌素的表达,而YOSHIDA等[44]研究表明心肌素是平滑肌的主要调节因子,介导出生后心脏生长和重塑,鸢尾素则可通过该两种途径调控心肌素的表达。 "

除此之外,鸢尾素对心脏的保护作用还可能和线粒体的功能有关,线粒体为各种心脏功能提供能量,心脏作为一种富含线粒体的组织,具有较高的O2消耗率和ATP产生/周转率,这是为维持其持续机械功,尤其是在运动后。XING等[45-46]实验证明鸢尾素增加了心肌细胞中的线粒体代谢。 2.2.5 运动产生鸢尾素对心肌纤维化机制的影响 (1)鸢尾素具有调节氧化应激和炎症反应的作用:研究发现,促炎细胞因子(如白细胞介素1和白细胞介素6)增加了黏附分子血管细胞黏附分子1(VCAM-1)和细胞间黏附分子1(ICAM-1)的表达,该实验证明鸢尾素在体内和体外均抑制单核细胞趋化蛋白1(MCP-1)、白细胞介素6、血管细胞黏附分子1和细胞间黏附分子1的表达,因此鸢尾素可通过抑制ROS/p38MAPK/NFKB信号通路的激活炎症基因的水平降低[47]。AKHMEDOV等[48]研究显示,过量产生的活性氧和活性氮淹没了抗氧化剂系统的能力,导致蛋白质、DNA和脂质的氧化损伤,而在心肌细胞中,内源性产生的活性氧的主要来源是线粒体。氧化受损的细胞分子会影响各种基本的心脏过程,包括能量代谢、兴奋收缩耦合、Ca2+稳态和心肌细胞死亡。以往研究研究表明,Nod样受体Pyin3(NLR family pyrin domain containing 3,NLRP3)炎症小体在介导氧化应激中起重要作用,氧葡萄糖剥夺激PC12神经细胞的氧化应激ROS-NLRP3炎症信号通路,而鸢尾素对氧葡萄糖剥夺诱导的氧化应激和炎症反应起逆转作用[49]。因此鸢尾素可通过抑制ROS/p38MAPK/NFKB信号通路、内源性活性氧和ROS-NLRP3炎症信号通路,从而影响心肌纤维化形成机制里的氧化应激反应和炎症反应过程。 (2)鸢尾素对线粒体稳态的调节:心脏为血液流动提供强大的动力,具有强大的能量需求,在病理情况下,心肌的收缩和代谢能力降低,并最终演变为慢性心力衰竭。在不同时期心脏的能量来源有所不同,生理情况下心脏主要能量来源于脂肪酸和葡萄糖,在正常心肌细胞中,线粒体游离脂肪酸氧化为心肌提供60%-70%的能量,糖酵解及糖的有氧氧化提供20%的能量,因此线粒体在心脏能量代谢中起到至关重要的作用[50]。生理条件下,为满足心脏的泵血功能线粒体需在活细胞中不断进行线粒体稳态重构,若线粒体稳态异常会导致线粒体生物合成异常、空间结构由融合趋向分裂和修复能力降低等,从而导致心肌能量代谢障碍和氧化应激。NAKAHIRA等[51]研究证实,线粒体稳态异常可导致心肌炎症反应,而研究发现鸢尾素诱导的心脏保护可能和线粒体功能改善有关。YE等[52]研究发现,鸢尾素对胰岛素抵抗的作用可能是通过p38-MAPK-PGC-1α通路的激活从而增强线粒体功能达到治疗作用。上述研究表明,鸢尾素通过抑制线粒体膜通透性转换孔的开放和线粒体肿胀保护线粒体的功能,从而保护心脏功能。 (3)鸢尾素对解偶联蛋白2(uncoupling protein 2,UCP2)的作用:解偶联蛋白2是一种线粒体内膜质子载体蛋白,广泛存在于心肌组织中,其基本生理功能是介导氧化磷酸化解偶联。解偶联蛋白2可使H+从线粒体的膜外直接进入其基质中,可提高呼吸链电子传递的速率,缩短活性中间体存在的时间,可减少电子通过活性中间体传递给O2的概率,从而导致线粒体活性氧的生成减少, 因此解偶联蛋白2在改善氧化应激损伤中发挥了重要作用[53]。心肌缺血预处理可提升解偶联蛋白2 mRNA的表达,使氧自由基损伤降低,从而使心肌线粒体对缺氧-复氧的耐受能力增强,进而减轻了心肌缺血再灌注损伤,这证明解偶联蛋白2是一种保护心肌缺血再灌注损伤的蛋白[54]。解偶联蛋白2具有氧化还原的生理调节功能,能有效减少氧自由基产生和免疫反应。研究发现,心衰组大鼠与正常对照组相比,其解偶联蛋白2的表达明显下降;然而,通过构建狗内毒素休克模型实验发现,与对照组相比,实验组心肌解偶联蛋白2 mRNA表达均明显增加,而且与ATP水平呈现负相关[55-56]。另外,GAO等[57]研究发现,在心衰终末期,解偶联蛋白2表达增高,加重心肌细胞的能量缺乏。综合以上研究结果,在心衰发生的不同时期解偶联蛋白2扮演的角色不同,心衰早期解偶联蛋白2的表达提高,此时对心肌起保护作用,而随着心衰的病程进展,解偶联蛋白2的表达不断增加,影响心肌能量代谢,扰乱心肌细胞兴奋-收缩耦联,此时对心肌起损伤作用。综上所述,鸢尾素可调控解偶联蛋白2的表达,应激条件下通过抑制或降低解偶联蛋白2的表达对心肌起保护作用[58]。 鸢尾素在运动改善心肌纤维化中有重要作用,其机制很有可能是通过抑制ROS/p38MAPK/NFKB信号通路、内源性活性氧和ROS-NLRP3炎症信号通路抑制氧化应激和炎症反应、抑制线粒体膜通透性转换孔的开放和线粒体肿胀及调控解偶联蛋白2的表达,抑制心肌纤维化的形成。 "

| [1]黄峻.中国心力衰竭流行病学特点和防治策略[J].中华心脏与心律电子杂志,2015,3(2):81-88. [2]NGUYEN TP, QU Z, WEISS JN. Cardiac fibrosis and arrhythmogenesis: the road to repair is paved with perils. J Mol Cell Cardiol. 2014;70:83-91. [3]GYöNGYöSI M, WINKLER J, RAMOS I, et al. Myocardial fibrosis: biomedical research from bench to bedside. Eur J Heart Fail. 2017; 19(2):177-191. [4]王友华,马美,田振军.心肌梗死后的心功能改善:有氧运动干预发挥效应新视角[J].中国运动医学杂志,2019,38(7):624-629. [5]贾丽晔,郭琪,王鹏程,等.运动疗法对心血管疾病患者的影响和作用机理研究进展[J].中国康复理论与实践,2016,22(9):1041-1044. [6]KATSURA H, KANEMARU A, YAMADA K, et al. Long-term effectiveness of an inpatient pulmonary rehabilitation program for elderly COPD patients: Comparison between young-elderly and old-elderly groups. Respirology. 2004;9(2):230-236. [7]OLDRIDGE NB, GUYATT GH, FISCHER ME, et al. Cardiac rehabilitation after myocardial infarction. Combined experience of randomized clinical trials.JAMA. 1988;260(7):945-950. [8]YANCY CW, JESSUP M, BOZKURT B, et al. 2013 ACCF/AHA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines.Circulation. 2013; 128(16):1810-1852. [9]DERUMEAUX G, ICHINOSE F, RAHER MJ, et al. Myocardial alterations in senescent mice and effect of exercise training: a strain rate imaging study.Circ Cardiovasc Imaging. 2008;1(3):227-234. [10]LI Y, CAI X, GUAN Y, et al. Adiponectin Upregulates MiR-133a in Cardiac Hypertrophy through AMPK Activation and Reduced ERK1/2 Phosphorylation.PLoS One. 2016;11(2):e0148482. [11]王世强,李丹,刘奥峰.运动改善疾病所致心肌纤维化研究进展[J].中国运动医学杂志,2019,38(3):220-227. [12]BOSTRöM P, WU J, JEDRYCHOWSKI M P, et al. A PGC1-alpha- dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481(7382):463-468. [13]WRANN CD, WHITE JP, SALOGIANNNIS J, et al. Exercise Induces Hippocampal BDNF through a PGC-1 alpha/FNDC5 Pathway. Cell Metab. 2013;18(5):649-659. [14]ROCA-RIVADA A, CASTELAO C, SENIN L L, et al. FNDC5/irisin is not only a myokine but also an adipokine.PLoS One. 2013;8(4):e60563. [15]MATSUO Y, GLEITSMANN K, MANGER N, et al. Fibronectin type III domain containing 5 expression in skeletal muscle in chronic heart failure-relevance of inflammatory cytokines. J Cachexia Sarcopenia Muscle. 2015;6(1):62-72. [16]NGUYEN TP, QU Z, WEISS JN. Cardiac fibrosis and arrhythmogenesis: The road to repair is paved with perils. J Mol Cell Cardiol. 2014;70:83-91 [17]YUE Y, MENG K, PU Y, et al. Transforming growth factor beta (TGF-beta) mediates cardiac fibrosis and induces diabetic cardiomyopathy. Diabetes Res Clin Pract. 2017;133:124-130. [18]RAO Z, WANG S, BUNNER WP, et al. Exercise induced Right Ventricular Fibrosis is Associated with Myocardial Damage and Inflammation. Korean Circ J. 2018;48(11):1014-1024. [19]KUUSISTO J, KARJA V, SIPOLA P, et al. Low-grade inflammation and the phenotypic expression of myocardial fibrosis in hypertrophic cardiomyopathy. Heart. 2012l;98(13):1007-1013. [20]CALLIS TE, PANDYA K, SEOK HY, et al. MicroRNA-208a is a regulator of cardiac hypertrophy and conduction in mice. J Clin Invest. 2009;119(9):2772-2786. [21]PEDERSEN BK, FEBBRAIO MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8(8): 457-465. [22]SANCHIS-GOMAR F, PEREZ-QUILIS C. The p38-PGC-1alpha- irisin-betatrophin axis: Exploring new pathways in insulin resistance. Adipocyte. 2014;3(1):67-68. [23]AYDIN S, KULOGLU T, AYDIN S, et al.Cardiac, skeletal muscle and serum irisin responses to with or without water exercise in young and old male rats: cardiac muscle produces more irisin than skeletal muscle. Peptides. 2014;52:68-73. [24]ARONIS KN, MORENO M, POLYZOS SA, et al. Circulating irisin levels and coronary heart disease: association with future acute coronary syndrome and major adverse cardiovascular events. Int J Obes (Lond). 2015;39(1):156-161. [25]陈聪毅,王冠捷.运动诱发鸢尾素对肥胖合并间歇性低氧引起心肌纤维化的影响[J].大专体育学刊,2017,19(3)DOI:10.5297/ser.1903.006 [26]LEE P, LINDERMAN JD, SMITH S, et al.Irisin and FGF21 are cold-induced endocrine activators of brown fat function in humans.Cell Metab. 2014;19(2):302-309. [27]MIYAMOTO-MIKAMI E, SATO K, KURIHARA T, et al. Endurance training-induced increase in circulating irisin levels is associated with reduction of abdominal visceral fat in middle-aged and older adults. PLoS One. 2015;10(3):e0120354. [28]ALTER P, VAN DE SAND K, NELL C, et al. Airflow limitation in COPD is associated with increased left ventricular wall stress in coincident heart failure. Respir Med. 2015;109(9):1131-1137. [29]HECKSTEDEN A, WEGMANN M, STEFFEN A, et al. Irisin and exercise training in humans - Results from a randomized controlled training trial. BMC Med. 2013;11:235. [30]SCHARHAG-ROSENBERGER F, MEYER T, WEGMANN M, et al. Irisin does not mediate resistance training-induced alterations in resting metabolic rate. Med Sci Sports Exerc. 2014;46(9):1736-1743. [31]HUH JY, MOUGIOS V, SKRAPARLIS A, et al. Irisin in response to exercise in humans with and without metabolic syndrome.J Clin Endocrinol Metab. 2015;100(3):E453-457. [32]ANASTASILAKIS AD, POLYZOS SA, SARIDAKIS ZG, et al. Circulating irisin in healthy, young individuals: day-night rhythm, effects of food intake and exercise, and associations with gender, physical activity, diet, and body composition.J Clin Endocrinol Metab. 2014;99(9): 3247-3255. [33]DASKALOPOULOU SS, COOKE AB, GOMEZ YH, et al.Plasma irisin levels progressively increase in response to increasing exercise workloads in young,healthy, active subjects. Eur J Endocrinol. 2014; 171(3):343-352. [34]NORHEIM F, LANGLEITE TM, HJORTH M, et al. The effects of acute and chronic exercise on PGC-1α, irisin and browning of subcutaneous adipose tissue in human. FEBS J. 2014;281(3):739-749. [35]IJIRI N, KANAZAWA H, ASAI K, et al. Irisin, a newly discovered myokine, is a novel biomarker associated with physical activity in patients with chronic obstructive pulmonary disease. Respirology. 2015;20(4):612-617. [36]HEW-BUTLER T, LANDIS-PIWOWAR K, BYRD G, et al.Plasma irisin in runners and nonrunners: no favorable metabolic associations in humans. Physiol Rep. 2015;3(1). pii: e12262. [37]TSUCHIYA Y, ANDO D, GOYoto K, et al. High-intensity exercise causes greater irisin response compared with low-intensity exercise under similar energy consumption. Tohoku J Exp Med. 2014;233(2): 135-140. [38]XIE C, ZHANG Y, TRAN TD, et al. Irisin Controls Growth, Intracellular Ca2+ Signals, and Mitochondrial Thermogenesis in Cardiomyoblasts. PLoS One. 2015;10(8):e0136816. [39]WANG YB. Mitogen-activated protein kinases in heart development and diseases. Circulation. 2007;116(12):1413-1423. [40]WINBANKS CE, WEEKS KL, THOMSON RE, et al. Follistatin-mediated skeletal muscle hypertrophy is regulated by Smad3 and mTOR independently of myostatin. J Cell Biol. 2012; 197(7):997-1008. [41]谢平,李汇华.心肌素与心血管疾病的研究进展[J].基础医学与临床,2008, 28(10):1103-1106. [42]CLEMENT S, STOUFFS M, BETTOL E, et al.Expression and function of alpha-smooth muscle actin during embryonic-stem-cell-derived cardiomyocyte differentiation. J Cell Sci. 2007;120(Pt 2):229-238. [43]LI M, WANG N, GONG HQ, et al. Ca²⁺ signal-induced cardiomyocyte hypertrophy through activation of myocardin.Gene. 2015;557(1):43-51. [44]YOSHIDA T, SINHA S, DANDRE F, et al. Myocardin is a key regulator of CArG-dependent transcription of multiple smooth muscle marker genes. Circ Res. 2003;92(8):856-864. [45]XING W, ZHANG TC, CAO D, et al.Myocardin induces cardiomyocyte hypertrophy. Circ Res. 2006;98(8):1089-1097. [46]WANG H, ZHAO YT, ZHANG S, et al. Irisin plays a pivotal role to protect the heart against ischemia and reperfusion injury. J Cell Physiol. 2017;232(12):3775-3785. [47]ZHANG YZ, MU Q, ZHOU Z, et al. Protective Effect of Irisin on Atherosclerosis via Suppressing Oxidized Low Density Lipoprotein Induced Vascular Inflammation and Endothelial Dysfunction. PLoS One. 2016;11(6):e0158038. [48]AKHMEDOV AT, RYBIN V, MARIN-GARCIA J. Mitochondrial oxidative metabolism and uncoupling proteins in the failing heart. Heart Fail Rev. 2015;20(2):227-249. [49]PENG J, DENG X, HUANG W, et al. Irisin protects against neuronal injury induced by oxygen-glucose deprivation in part depends on the inhibition of ROS-NLRP3 inflammatory signaling pathway. Mol Immunol. 2017;91:185-194. [50]DOENST T, NGUYEN TD, ABEL ED, et al. Cardiac metabolism in heart failure-implications beyond ATP production. Circ Res. 2013; 113(6):709-724. [51]NAKAHIRA K, HASPEL JA, RATHINAM VA, et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol. 2011r;12(3):222-230. [52]YE X, SHEN Y, NI C, et al. Irisin reverses insulin resistance in C2C12 cells via the p38-MAPK-PGC-1 alpha pathway. Peptides. 2019;119: 170120. [53]MA ST, ZHANG Y, WANG Q, et al. Ablation of uncoupling protein 2 exacerbates salt-induced cardiovascular and renal remodeling associated with enhanced oxidative stress. Int J Cardiol. 2014;175(1): 206-210. [54]MCLEOD CJ, AZIZ A, HOYT RF JR, et al. Uncoupling proteins 2 and 3 function in concert to augment tolerance to cardiac ischemia.J Biol Chem. 2005;280(39):33470-33476. [55]HASLIP M, DOSTANIC I, HUANG Y, et al. Endothelial uncoupling protein 2 regulates mitophagy and pulmonary hypertension during intermittent hypoxia.Arterioscler Thromb Vasc Biol. 2015;35(5):1166-1178. [56]MA S, WANG Q, ZHANG Y, et al. Transgenic overexpression of uncoupling protein 2 attenuates salt-induced vascular dysfunction by inhibition of oxidative stress. Am J Hypertens. 2014;27(3):345-354. [57]GAO YH, GAO HB, DI NN, et al. Effects of beta3-adrenergic receptor antagonist on myocardial UCP2 expression and energy metabolism in chronic heart failure rats. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2013l;29(4):376-379, 384. [58]CHEN K, XU Z, LIU Y, et al.Irisin protects mitochondria function during pulmonary ischemia/reperfusion injury. Sci Transl Med. 2017;9(418). pii: eaao6298. |
| [1] | Cui Wei, Cui Di, Ouyang Ting, Li Xiang, Wei Huiting, Xue Weiyue, Zhou Gang, Qiu Ye. Inhibiting NOX alleviates alcoholic liver damage and lipid metabolism disorder [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-8. |
| [2] | Chen Xianghe, Liu Bo, Yang Kang, Lu Pengcheng, Yu Huilin. Treadmill exercise improves the myocardial fibrosis of spontaneous type 2 diabetic mice: an exploration on the functional pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1210-1215. |
| [3] | Tang Wenjing, Wu Siyuan, Yang Chen, Tao Xi. Inflammatory responses in post-stroke depression [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1278-1285. |
| [4] | Yin Tingting, Du Dayong, Jiang Zhixin, Liu Yang, Liu Qilin, Li Yuntian. Granulocyte colony-stimulating factors improve myocardial fibrosis in rats with myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 730-735. |
| [5] | Zhao Yuwei, Gao Yuting, Li Zhen, Hao Huiqin . Mechanism of Ermiao San in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 742-748. |
| [6] | Chen Na, Wang Xiaohan, Zhang Yunke. Effect and mechanism of mesenchymal stem cells on aging-related ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3914-3920. |
| [7] | Yang Demeng, Wang Changgeng, Hu Xinyuan, Ao Fei. Protective effect of eucommia alcohol extract on articular cartilage of osteoarthritis model rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3756-3761. |
| [8] | Deng Bowen, Li Xiaoye, Jiang Shengyuan, Liu Gan, Zhao Yi, Zhang Houjun, He Feng, Mu Xiaohong. Mechanisms of Huangqi Guizhi Wuwu Decoction in the treatment of cervical spondylosis and anxiety disorder based on the principle of “treating different diseases with the same method”: a network pharmacology analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3650-3656. |
| [9] | Wang Sihan, Chen Junji, Mo Weibin. Effects of Dendrobium officinale flavonoid on oxidative stress and autophagy in the liver of an exhaustive exercise rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3212-3219. |
| [10] | Li Anan, Jiang Tao, Zhan Min, Cai Yuning, Song Min, Li Congcong, Lin Wenzheng, Zhang Jiayuan, Liu Wengang. Pharmacological mechanism of Shenling Baizhu San in the treatment of knee osteoarthritis based on network pharmacology and molecular docking [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 197-204. |
| [11] | Li Wanhai, Dong Yuhang, Shi Chao, Jiang Yiyao, Liu Ge, Diao Wenjie, Wu Zhen. Ligustrazine furoxan complex protects against renal ischemia-reperfusion injury in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 205-210. |
| [12] | Gu Qingfang, Guo Minfang, Liu Xiaoqin, Mu Bingtao, Li Weimei, Song Lijuan, Chai Zhi, Ma Cungen, Yu Jiezhong. Mechanism by which bone marrow mesenchymal stem cells improve cognitive function in APP/PS1 double transgenic mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 2964-2969. |
| [13] | Li Chuanhong, Yu Xing, Yang Yongdong, Zhao He. Microglia in spinal cord injury: M1/M2 phenotypic polarization and neurotoxic/neuroprotective effects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(14): 2265-2272. |
| [14] | Zhu Chunhui, Zhang Yi, Song Huanghe, Liang Wenwei. Protective effect of astaxanthin on tert-butyl hydrogen peroxide-induced chondrocyte damage [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(11): 1648-1655. |
| [15] | Xu Lin, Qi Hongshun, Ge Rucun, Li Peipei, Zhen Lixiao, Feng Xiaoya. Effects of minimally invasive surgery combined with metformin on inflammatory responses in rabbits with intracerebral hemorrhage [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(11): 1741-1746. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||