Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (29): 4731-4736.doi: 10.3969/j.issn.2095-4344.2790
Previous Articles Next Articles
Synovial macrophages in osteoarthritis: roles and features
Cao Jiangang, Chen Desheng
Tianjin Hospital, Tianjin 300221, China
-
Received:2020-02-10Revised:2020-02-15Accepted:2020-03-18Online:2020-10-18Published:2020-09-15 -
About author:Cao Jiangang, MD candidate, Associate chief physician, Tianjin Hospital, Tianjin 300221, China -
Supported by:the Funded Project of Wu Jieping
CLC Number:
Cite this article
Cao Jiangang, Chen Desheng. Synovial macrophages in osteoarthritis: roles and features[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4731-4736.
share this article
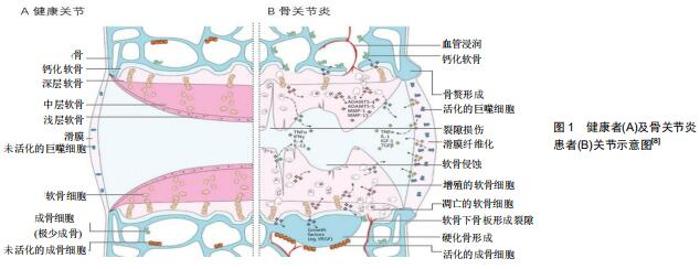
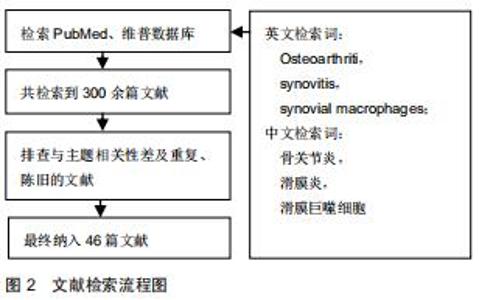
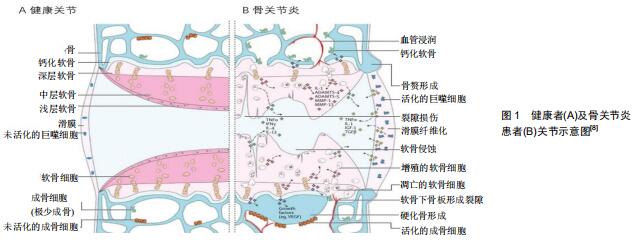
骨关节炎是最常见的关节病变,其可发生于几乎所有动关节,是一种累及关节软骨、软骨下骨、滑膜、韧带、关节囊及关节周围肌肉的复杂病症[1-3]。作为一种常见的致残性疾病,随着全球人口老龄化和肥胖率以及关节损伤个体数量的增加,在世界范围内估计,目前约有2.5亿人受骨关节炎影响,这不仅给患病个体带来巨大的生活障碍,给卫生保健系统带来新的挑战,还给社会带来了沉重的经济负担[4-5]。在老化、肥胖、性别、遗传、创伤等致病因素作用下,关节组织的损伤与修复之间的动态平衡被打破,致使软骨腐蚀缺损、骨赘形成、软骨下骨硬化、滑膜的肥厚增生,在临床上最初表现为关节的疼痛、肿胀、活动受限,最终导致整个关节的畸形和功能丧失,见图1[3,6-7]。最新的骨关节炎治疗理念强调以患者为中心,对其加强健康教育,使其在自我管理的基础上适当的锻炼和减肥,在这些温和治疗手段无效时再采取手术治疗[4]。预防、延缓进展和改善病情一直是各类骨关节炎研究的主要目的,然而至今尚无有效的治疗措施[8]。 "
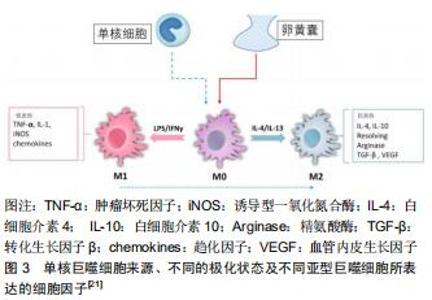
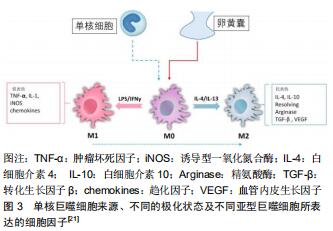
2.1.1 组织学研究 早在1991年ATHANASOU等[30]就对4例接受髋关节置换骨关节炎患者的关节滑膜组织抗原表位进行了分析,发现滑内膜细胞和内膜下细胞表达多种巨噬细胞抗体(CD11b,CD14,CD16,CD68)。这些抗原表位均是静息状态下组织巨噬细胞所表达的,不能反映滑膜中巨噬细胞的活化状态。此后,相继又有多个研究对骨关节炎患者滑膜巨噬细胞进行了分析,HAYWOOD等[32] (2003年)、BENITO等[33](2005年)、BONDESON等[34] (2006年)均发现与正常滑膜组织相比骨关节炎患者滑膜组织中巨噬细胞数量明显增多,类似的结果在小动物骨关节炎模型中亦得到证实,见图3[24,29,31]。上述研究均表明滑膜巨噬细胞在骨关节炎患者滑膜炎中发挥重要作用,但均未对不同极化状态的巨噬细胞进行标记分析。 "
|
[1] MARTEL-PELLETIER J, BARR AJ, CICUTTINI FM, et al. Osteoarthritis. Nat Rev Dis Primers.2016;2:16072.
[2] BRANDT KD, RADIN EL, DIEPPE PA, et al. Yet more evidence that osteoarthritis is not a cartilage disease. Ann Rheum Dis. 2006;65:4.
[3] LOESER RF, GOLDRING SR, SCANZELLO CR, et al. Osteoarthritis: A disease of the joint as an organ. Arthritis Rheum. 2012;64(6):1697-1707.
[4] ROEMER FW, GUERMAZI A, FELSON DT, et al. Presence of MRI-detected joint effusion and synovitis increases the risk of cartilage loss in knees without osteoarthritis at 30-month follow-up: the MOST study.Ann Rheum Dis. 2011;70(10): 1804-1809.
[5] BORTOLUZZI A, FURINI F, SCIRE CA. Osteoarthritis and its management-Epidemiology, nutritional aspects and environmental factors. Autoimmun Rev.2018;17(11):1097-1104.
[6] FU K, ROBBINS SR, MCDOUGALL JJ.Osteoarthritis: the genesis of pain. Rheumatology(Oxford). Rheumatology (Oxford). 2018; 57(suppl_4):iv43-iv50.
[7] KRASNOKUTSKY S, ATTUR M, PALMER G, et al. Current concepts in the pathogenesis of osteoarthritis. Osteoarthritis Cartilage.2008;16 Suppl 3:S1-3.
[8] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182):1745-1759.
[9] FURMAN BD, KIMMERLING KA, ZURA RD, et al. Articular ankle fracture results in increased synovitis, synovial macrophage infiltration, and synovial fluid concentrations of inflammatory cytokines and chemokines. Arthritis Rheum. 2015;67(5): 1234-1239.
[10] AYRAL X, PICKERING EH, WOODWORTH TG, et al.Synovitis: a potential predictive factor of structural progression of medial tibiofemoral knee osteoarthritis-results of a 1 year longitudinal arthroscopic study in 422 patients. Osteoarthritis Cartilage. 2005;13(5):361-367.
[11] HENROTIN Y, LAMBERT C, RICHETTE P. Importance of synovitis in osteoarthritis: Evidence for the use of glycosaminoglycans against synovial inflammation. Semin Arthritis Rheum. 2014;43(5):579-587.
[12] LIU-BRYAN R. Synovium and the innate inflammatory network in osteoarthritis progression. Curr Rheumatol Rep. 2013;15(5):323.
[13] SCANZELLO CR, GOLDRING SR. The role of synovitis in osteoarthritis pathogenesis. Bone. 2012;51(2):249-257.
[14] BONDESON J, BLOM AB, WAINWRIGHT S, et al. The role of synovial macrophages and macrophage-produced mediators in driving inflammatory and destructive responses in osteoarthritis. Arthritis Rheum.2010;62(3):647-657.
[15] DE LANGE-BROKAAR BJE, IOAN-FACSINAY A, VAN OSCH GJVM, et al. Synovial inflammation, immune cells and their cytokines in osteoarthritis: a review. Osteoarthritis Cartilage. 2012;20(12):1484-1499.
[16] 戴冽,许赤多,汤美安,等.佐剂性关节炎大鼠滑膜巨噬细胞在关节破坏中的作用[J].中国病理生理杂志,2003,19(7):950-953
[17] GINHOUX F, JUNG S. Monocytes and macrophages: developmental pathways and tissue homeostasis. Nat Rev Immunol. 2014;14(6):392-404.
[18] YONA S, KIM KW, WOLF Y, et al.Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity. 2013;38(1):79-91.
[19] HASHIMOTO D, CHOW A, NOIZAT C,et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity. 2013; 38(4):792-804.
[20] LAPENNA A, DE PALMA M, LEWIS CE. Perivascular macrophages in health and disease. Nat Rev Immunol.2018; 18(11):689-702.
[21] XIE J, HUANG Z, YU X,et al. Clinical implications of macrophage dysfunction in the development of osteoarthritis of the knee. Cytokine Growth Factor Rev. 2019;46:36-44.
[22] GORDON S, PLUDDEMANN A. Tissue macrophages: heterogeneity and functions. BMC Biol. 2017;15(1):53.
[23] MOSSER DM, EDWARDS JP.Exploring the full spectrum of macrophage activation. Nat Rev Immunol.2008;8(12):958-969.
[24] KRAUS VB, MCDANIEL G, HUEBNER JL, et al. Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthritis Cartilage. 2016;24(9):1613-1621.
[25] PISCAER TM, MULLER C, MINDT TL, et al. Imaging of activated macrophages in experimental osteoarthritis using folate-targeted animal single-photon-emission computed tomography/computed tomography. Arthritis Rheum.2011; 63(7):1898-1907.
[26] DE VISSER HM, KORTHAGEN NM, MULLER C,et al. Imaging of Folate Receptor Expressing Macrophages in the Rat Groove Model of Osteoarthritis: Using a New DOTA-Folate Conjugate. Cartilage. 2018;9(2):183-191.
[27] DAGHESTANI HN, PIEPER CF, KRAUS VB. Soluble macrophage biomarkers indicate inflammatory phenotypes in patients with knee osteoarthritis. Arthritis Rheum.2015;67(4): 956-965.
[28] WU CL, MCNEILL J, GOON K, et al. Conditional Macrophage Depletion Increases Inflammation and Does Not Inhibit the Development of Osteoarthritis in Obese Macrophage Fas-Induced Apoptosis-Transgenic Mice. Arthritis Rheum. 2017;69(9):1772-1783.
[29] ZHANG H, LIN C, ZENG C,et al. Synovial macrophage M1 polarisation exacerbates experimental osteoarthritis partially through R-spondin-2. Ann Rheum Dis.2018;77(10):1524-1534.
[30] ATHANASOU NA, QUINN J.Immunocytochemical analysis of human synovial lining cells: phenotypic relation to other marrow derived cells.Ann Rheum Dis.1991;50(5):311-315.
[31] BLOOM O, HERMAN PE, SPUNGEN AM. Systemic inflammation in traumatic spinal cord injury. Exp Neurol. 2020;325:113143.
[32] HAYWOOD L, MCWILLIAMS DF, PEARSON CI, et al. Inflammation and angiogenesis in osteoarthritis. Arthritis Rheum. 2003;48(8):2173-2177.
[33] BENITO MJ, VEALE DJ, FITZGERALD O, et al. Synovial tissue inflammation in early and late osteoarthritis. Ann Rheum Dis. 2005;64(9):1263-1267.
[34] BONDESON J, WAINWRIGHT SD, LAUDER S, et al. The role of synovial macrophages and macrophage-produced cytokines in driving aggrecanases, matrix metalloproteinases, and other destructive and inflammatory responses in osteoarthritis. Arthritis Res Ther. 2006;8(6):R187.
[35] MANFERDINI C, PAOLELLA F, GABUSI E, et al. From osteoarthritic synovium to synovial-derived cells characterization: synovial macrophages are key effector cells. Arthritis Res Ther. 2016;18:83.
[36] VAN DEN BOSCH MH, BLOM AB, SCHELBERGEN RF, et al. Alarmin S100A9 Induces Proinflammatory and Catabolic Effects Predominantly in the M1 Macrophages of Human Osteoarthritic Synovium. J Rheumatol. 2016;43(10):1874-1884.
[37] SHEN J, HILGENBRINK AR, XIA W, et al. Folate receptor-beta constitutes a marker for human proinflammatory monocytes. J Leukoc Biol.2014;96(4):563-870.
[38] VAN LENT PL, BLOM AB, VAN DER KRAAN P, et al. Crucial role of synovial lining macrophages in the promotion of transforming growth factor beta-mediated osteophyte formation. Arthritis Rheum. 2004;50(1):103-111.
[39] BLOM AB, VAN LENT PL, HOLTHUYSEN AE,et al. Synovial lining macrophages mediate osteophyte formation during experimental osteoarthritis. Osteoarthritis Cartilage. 2004; 12(8):627-635.
[40] LI J, HSU HC, YANG P, et al. Treatment of arthritis by macrophage depletion and immunomodulation: testing an apoptosis-mediated therapy in a humanized death receptor mouse model. Arthritis Rheum. 2012;64(4):1098-1109.
[41] BLOM AB, VAN LENT PL, LIBREGTS S, et al. Crucial role of macrophages in matrix metalloproteinase-mediated cartilage destruction during experimental osteoarthritis: involvement of matrix metalloproteinase 3.Arthritis Rheum.2007;56(1):147-157.
[42] TOPOLUK N, STECKBECK K, SIATKOWSKI S, et al. Amniotic mesenchymal stem cells mitigate osteoarthritis progression in a synovial macrophage-mediated in vitro explant coculture model. J Tissue Eng Regen Med. 2018; 12(4):1097-1110.
[43] TAKANO S, UCHIDA K, INOUE G, et al. Nerve growth factor regulation and production by macrophages in osteoarthritic synovium. Clin Exp Immunol. 2017;190(2):235-243.
[44] SAMAVEDI S, DIAZ-RODRIGUEZ P, ERNDT-MARINO JD, et al. A Three-Dimensional Chondrocyte-Macrophage Coculture System to Probe Inflammation in Experimental Osteoarthritis. Tissue Eng Part A. 2017;23(3-4):101-114.
[45] UTOMO L, BASTIAANSEN-JENNISKENS YM, VERHAAR JA, et al. Cartilage inflammation and degeneration is enhanced by pro-inflammatory (M1) macrophages in vitro, but not inhibited directly by anti-inflammatory (M2) macrophages. Osteoarthritis Cartilage. 2016;24(12):2162-2170.
[46] DAI M, SUI B, XUE Y, et al.Cartilage repair in degenerative osteoarthritis mediated by squid type II collagen via immunomodulating activation of M2 macrophages, inhibiting apoptosis and hypertrophy of chondrocytes. Biomaterials. 2018;180:91-103. |
| [1] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [2] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [3] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [4] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [5] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [6] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [7] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [8] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [9] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [10] | Zheng Li, Li Dadi, Hu Weifan, Tang Jinlong, Zhao Fengchao. Risk assessment of contralateral knee arthroplasty after unilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 374-379. |
| [11] | Lü Jiaxing, Bai Leipeng, Yang Zhaoxin, Miao Yuesong, Jin Yu, Li Zhehong, Sun Guangpu, Xu Ying, Zhang Qingzhu. Evaluation of internal fixation with proximal femoral nail antirotation in elderly knee osteoarthritis patients with femoral intertrochanteric fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 391-396. |
| [12] | Luo Anyu, Liu Hanlin, Xie Xiaofei, Huang Chen. Effect of antioxidant mixture on structural degeneration of an osteoarthritis rat model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3625-3629. |
| [13] | Gao Kun, Chen Dayu, Zhang Yong, Liu Weidong, Sun Shufen, Lai Wenqiang, Ma Dujun, Wu Yihong, Lin Zhanpeng, Jiang Yinglu, Yu Weiji. Achyranthes bidentata alcohol extract inhibits extracellular matrix degradation of the cartilage by regulating synovial fibroblast exosomes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3636-3640. |
| [14] | Liu Jinfu, Zeng Ping, Nong Jiao, Fan Siqi, Feng Chengqin, Huang Jiaxing. Integrative analysis of biomarkers and therapeutic targets in synovium of patients with osteoarthritis by multiple microarrays [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3690-3696. |
| [15] | Chen Feng, Zhang Xiaoyun, Chen Yueping, Liao Jianzhao, Li Jiajun, Song Shilei, Lai Yu. Molecular mechanism of anhydroicaritin in the treatment of osteoarthritis: an analysis based on network pharmacology and bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3704-3710. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||