Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (21): 3387-3393.doi: 10.3969/j.issn.2095-4344.2606
Previous Articles Next Articles
Failure of articular cartilage repair
Zhang Yu, Yu Chengqiang, Wu Youcai, Ou Yufu, Wei Jianxun
- Department of Orthopedics, People’s Hospital of Guangxi Zhuang Autonomous Region, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Received:2019-09-28Revised:2019-10-08Accepted:2019-11-07Online:2020-07-28Published:2020-04-18 -
Contact:Wei Jianxun, Professor, Chief physician, Department of Orthopedics, People’s Hospital of Guangxi Zhuang Autonomous Region, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Zhang Yu, Master candidate, Department of Orthopedics, People’s Hospital of Guangxi Zhuang Autonomous Region, Nanning 530021, Guangxi Zhuang Autonomous Region, China
CLC Number:
Cite this article
Zhang Yu, Yu Chengqiang, Wu Youcai, Ou Yufu, Wei Jianxun. Failure of articular cartilage repair[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(21): 3387-3393.
share this article
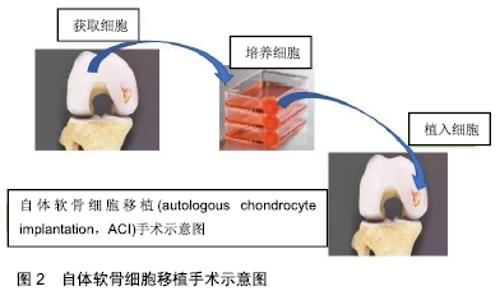
2.1 微骨折术(microfracture)和软骨镶嵌成形术(mosaicplasty) STEADMAN等[10]对26例年龄小于19岁(平均软骨缺损面积为1.77 cm2)且行微骨折术治疗的患者进行研究,并对26例患者中的22例进行了2年随访(未指定失败条件),至少在微骨折术后2年,有3例患者的Lysholm评分低于80分,3例患者均为女性,平均年龄18岁,其中2例接受髌骨微骨折术,1例接受滑车沟微骨折术,接受滑车沟微骨折术治疗的患者,在治疗1年后进行了一次修复,其他患者无需修复。SALZMANN等[11]对454例行微骨折术的患者(平均缺损面积2.97 cm2)进行了一项回顾性研究,在这项研究中,有与最初手术部位相关的疼痛,且接受了膝关节翻修手术的患者被定义为“失败”,并排除任何创伤史导致的最初微骨折术后膝关节再次手术的患者,关节软骨修复的失败率为27.1%(123例),首次微骨折术和再次手术之间的平均时间为1.6年,关节软骨修复失败的患者以前接受过的手术次数明显偏多。SOLHEIM等[12]前瞻性地收集了大约110例接受微骨折术治疗的患者(中位数缺损面积为 4 cm2),作者将软骨修复失败定义为通过新的手术来治疗软骨损伤,该研究人群中的有24例失败(21.82%)。 BENTLEY等[13]在一项随访10年以上的随机对照试验中,将100例有症状的关节软骨病变患者随机接受ACI或镶嵌成形术,比较两者的软骨修复失败率,其采用的软骨修复失败标准是:临床效果不佳,关节镜检查显示移植失败,或任何类型的软骨缺损都需要进行翻修手术。100例中42例患者(平均缺损面积为4 cm2)被纳入镶嵌成形术组,该组患者中软骨修复失败率较高(42例患者中23例修复失败,55%),显著高于ACI治疗组,差异有非常显著性意义(P < 0.001),其研究表明镶嵌成形术组在最初的2年中疗效相对满意,但在随后2年出现了较高的失败率,23例失败的患者中有9例接受ACI修复手术,3例接受基质诱导的自体软骨细胞植入术修复。ROBB等[14]对55例接受镶嵌成形术且平均随访5.9年的患者(平均缺损面积2.2 cm2)进行分析,软骨修复失败的标准为:移植的关节表面退化、任何随后的骨软骨再生或重建手术、任何形式的关节成形术,或牛津膝关节评分差,报告了6例失败的患者(10.91%),其中4例为翻修手术,2例为牛津膝关节评分差。 2.2 ACI ACI治疗示意图见图2。 "

MINAS等[15]分析了210例患者用骨膜覆盖的ACI治疗有症状的膝关节全厚度软骨缺损,并进行了至少10年的随访,他们将失败分为:经部分膝关节置换术或全膝关节置换翻修的移植物失败;修复性软骨移植失败;移植存活下来,但在同一膝关节的其他地方出现新的软骨缺损,需要额外的手术。结果显示手术失败率为25%(53例),术后19例患者行膝关节置换术,27例患者行软骨修复术,7例患者拒绝进一步治疗。对可能的失败危险因素的分析表明,髌骨、股骨移植比胫骨间室移植更早出现失败(10年为70例,10年为79%),但长期存活率没有显著差异(15年为70例,15年为73%);移植面积为15 cm2的患者存活率显著降低(P < 0.001);患者年龄小于30岁,15年存活率为84%,明显高于30-45岁和年龄超过45年的患者。GOMOLL等[16]研究表明,关于治疗伴有ACI的髌骨软骨病变,在110例患者(平均缺损面积为5.4 cm2)中,应用覆盖骨膜的ACI治疗的失败率为8.18%(9例),研究中设定的ACI软骨修复失败标准为:通过MRI和/或关节镜检查确认,并伴有疼痛,需要进行翻修手术。平均随访时间为7.5年,9例手术失败的患者中,4例接受了全膝关节置换术治疗,4例接受了部分膝关节置换术,1例拒绝进一步手术。FARR等[17]研究了38例(平均缺损面积为5.42 cm2)全层髌骨和/或滑车软骨损伤患者,其认定的ACI治疗失败的标准为:当患者在植入后进行手术,需要取出移植物,证实移植物部分或完全脱层,证实缺损填补丢失或侵犯软骨下骨质;平均随访3.1年,ACI治疗失败率为7.89%(3例),2例采用微骨折术治疗,另1例急性膝关节败血症取出了移植物。 2.3 骨软骨同种异体移植 LEVY等[18]评估了122例(平均移植物大小8.1 cm2)接受单独的股骨髁异体骨软骨移植的患者,移植物未经手术切除患者的中位随访时间是13.5年,大部分膝关节在异体骨软骨移植前接受过一次或多次手术,包括软骨修复手术。他们将异体骨软骨移植失败定义为移植物的修复或部分向全膝关节置换术的转化,根据这些失败标准,31个膝关节(24%)被归类为失败,平均失败时间为7.2年;15例失败患者接受了改良异体骨软骨移植,13例转为全膝关节置换,3例转为单髁膝关节置换;在logistic回归检验其他变量后,年龄和以往手术次数与异体骨软骨移植失败相关。 GRACITELLI等[19]报告了27例接受单独髌骨异体骨软骨移植的患者(28膝;平均移植面积10.1 cm2),当再次手术导致同种异体移植物移除时,记录为异体骨软骨移植失败。28个膝关节中有20个(71.4%)在最近的随访中进行了同种异体移植;平均随访时间为9.7年,28个膝盖中的8个(28.6%)被认为是异体骨软骨移植失败,平均时间为1.45年,其中4例转为全膝关节置换,2例转为髌股关节置换,1例转为异体骨软骨移植,1例转为髌骨切除。 SADR等[20]分析了异体骨软骨移植在治疗膝关节骨软骨炎中的应用,研究人群由135例患者组成(149个膝关节,平均移植物大小7.3 cm2),平均随访时间为6.3年,包括移植物移除在内的手术被认为是失败的,12个膝关节(8%)被归类为异体骨软骨移植失败,其中7个进行了异体骨软骨移植修复,3个进行了单髁膝关节置换,2个进行了全膝关节置换,平均失败时间为6.1年。 2.4 合成支架材料(Synthetic Scaffolds) DEGEN等[21]研究表明,61例年龄大于40岁且受股骨远端软骨病变影响的患者中,有9例患者软骨缺损位于股骨内侧或外侧髁,35例患者(平均缺损面积3.24 cm2)使用骨软骨支架塞治疗,4例(11.4%)平均3.15年后需要进行修复,其中2例患者接受了自体骨治疗,另外2例患者转为全膝关节置换。BERRUTO等[22]报告了MaioRegen在治疗大型膝关节骨软骨损伤中的应用,他们招募了49例患者参与研究(平均缺损面积为4.35 cm2),所有患者均前瞻性随访,至少随访2年,发现治疗失败5例,1例行单髁膝关节置换,1例行胫骨高位截骨术,1例行自体骨软骨移植,1例异体骨软骨移植伴股骨远端截骨。DHOLLANDER等[23]对20例软骨缺损患者进行研究(平均缺损大小0.83 cm2),发现其中1例患者的局灶性软骨缺损涉及股骨髁、髌骨或滑车,用骨软骨支架塞进行治疗,有15例患者完成了1年的随访,其中3例(20.0%)被认为是临床治疗失败,因此有指征进行翻修手术,随后的骨软骨缺损被髂嵴自体骨移植物填充。 2.5 软骨修复失败的治疗 HORTON等[24]报道了33个膝关节(平均移植面积9.5 cm2)在膝关节置换失败后进行翻修的膝关节行异体骨软骨移植的数据,所有患者至少2年前接受过手术,至少随访2年,术后平均随访10年,他们将所有经部分或全膝关节置换移植的患者归为翻修性同种异体移植失败。13例患者(39%)接受了与移植物相关的手术,被认为是失败的,其中1例转为单髁膝关节置换,12例转为全膝关节置换,平均失败时间为5.5年;在5年和10年时,翻修性移植物存活率分别为79%和61%;作者报告了在老年患者、骨关节炎患者和异体骨软骨移植时软骨疾病进展的患者中失败率增加,以及向部分或全膝关节置换转化的发生率增加,但是由于样本量太小,无法评估哪些变量导致同种异体移植的失败。 GRACITELLI等[25]分析比较了两组接受异体骨软骨移植的患者,一组为连续的46个膝关节系列,其主要的治疗为异体骨软骨移植(第1组),与另一组非连续的46个膝关节系列相匹配,后者在先前的软骨下骨髓刺激失败后接受了异体骨软骨移植(第2组);任何导致移植物移除的再次手术都被定义为软骨修复失败。在第1组46个膝关节中的11个(24%)进行了再次手术,在第2组46个膝关节中的20个(44%)进行了再次手术,两组再手术率有显著性差异(P < 0.04);第1组有5个膝关节(1%)出现异体骨软骨移植失败,2个膝关节进行了异体骨软骨移植修正,另外3个膝关节转为全膝关节置换;在第2组,7个膝关节(15%)被认为是失败的,其中3个进行了异体骨软骨移植修复,4个转为全膝关节置换,两组间的失败率无统计学意义(P < 0.53);10年随访发现同种异体移植的存活率相似 (87.4% vs. 86%);移植物保持原位的患者平均随访7.8年(第1组41膝)和11.3年(第2组39膝),两组患者的疼痛和功能从术前到最新随访均有改善(P < 0.001),两组患者在术前、最新随访时或从术前状态到随访的变化均无差异。此外,GRACITELLI等[26]分析了先前软骨修复失败后接受异体骨软骨移植治疗的163例患者(164膝,平均移植面积6.8 cm2),对患者至少随访2年,软骨下骨髓刺激是最常见的孤立性既往手术(88.4%),他们认为任何导致移植物移除的再手术都是失败的。68个膝关节(41.5%)在异体骨软骨移植后再次手术,31个膝关节(19.02%)被认为失败;164个膝关节中,18个失败的膝关节被转为膝关节置换,9个进行了异体骨软骨移植修正,2个转为单髁膝关节置换,1个转为髌股关节置换,1个转为关节融合术,平均失败时间为2.6年;异体骨软骨移植5年和10年存活率分别为87.8%和82%;与非失败患者相比,失败患者的体质量指数和既往手术次数都显著偏多。 JUNGMANN等[27]分析了413例接受ACI治疗的患者,其中88例再次干预,124例(30.02%)在膝关节同一位置至少接受过一次软骨修复治疗,主要是微骨折术或以前的ACI。两项logistic显示,对于既往行软骨下骨髓刺激患者,以移植部位的主动操作为特征的再干预需求风险增加。在NIETHAMMER等[28]研究评估的人群中,在143例接受第3代ACI治疗的软骨缺损患者中,有18例(19.9%)之前的软骨修复手术失败,经统计分析,在ACI之前进行过软骨修复的软骨缺损,经二次治疗后没有出现更多的并发症或更高的翻修率。VIJAYAN等[29]介绍了他们在ACI或基质诱导的自体软骨细胞移植失败后,对孤立软骨或骨软骨缺损的患者进行软骨修复手术的经验,他们研究的人群由22例患者(平均缺损面积4.46 cm2)组成,平均随访5.4年,主要手术为ACI 17例,基质诱导的自体软骨细胞移植5例,翻修手术包括13次基质诱导的自体软骨细胞移植,9次ACI,其定义的失败标准包括持续疼痛、目测类比评分(VAS)≥术前记录的疼痛及相关的“差”功能评分(modified Cincinnati knee score, Stanmore-Bentley score、VAS疼痛评分)、关节镜或放射治疗、移植失败的生物学确认。软骨修复后的3个临床结果评分均显示得到显著改善,1例患者在翻修手术3年后报告膝关节疼痛,关节镜下确认移植物分层,对患者进行第3次修复,基质诱导的自体软骨细胞移植程序治疗髌骨损伤,术后modified Cincinnati knee score“优秀”,VAS评分和Stanmore-Bentley score均为0,在最近随访6年中,记录了2例患者有膝关节疼痛及间歇性闭锁,关节镜检查证实其髌骨病变部位的移植物脱层,在翻修手术后的14个月和25个月,他们都接受了髌股关节置换术。在MINAS等[15]描述的上述人群中,210例(42.2%)经骨膜覆盖的ACI患者中,有89例曾接受过软骨下骨髓刺激治疗,ACI的生存显著降低,在ACI成功的患者中,先前接受骨髓刺激治疗的膝关节与ACI为主要软骨修复程序的膝关节的临床结果评分无差异。 "

| [1] SCHINHAN M, GRUBER M, DOROTKA R, et al. Matrix-associated autologous chondrocyte transplantation in a compartmentalized early stage of osteoarthritis. Osteoarthritis Cartilage. 2013;21(1):217-225. [2] MAKRIS EA, GOMOLL AH, MALIZOS KN, et al. Repair and tissue engineering techniques for articular cartilage.Nat Rev Rheumatol.2015;11:21-34. [3] ROSA D, DI DONATO SL, BALATO G, et al. How to manage a failed cartilage repair: a systematic literature review. Joints. 2017;5(2):93-106. [4] HUEY DJ, HU JC, ATHANASIOU KA. Unlike bone, cartilage regeneration remains elusive. Science. 2012;338(6109): 917-921. [5] LAMPLOT JD, SCHAFER KA, MATAVA MJ. Treatment of Failed Articular Cartilage Reconstructive Procedures of the Knee: A Systematic Review. Orthop J Sports Med.2018; 6(3):2325967118761871. [6] BROUWER RW, HUIZINGA MR, DUIVENVOORDEN T, et al. Osteotomy for treating knee osteoarthritis. Cochrane Database Syst Rev 2014;(12):CD004019. [7] GRACITELLI GC, MERIC G, BRIGGS DT, et al. Fresh osteochondral allografts in the knee: comparison of primary transplantation versus transplantation after failure of previous subchondral marrow stimulation. Am J Sports Med. 2015;43(4):885-891. [8] BIANT LC, MCNICHOLAS MJ, SPROWSON AP, et al.The surgical management of symptomatic articular cartilage defects of the knee: consensus statements from United Kingdom knee surgeons. Knee 2015;22(05):446-449. [9] DEVITT BM, BELL SW, WEBSTER KE, et al. Surgical treatments of cartilage defects of the knee: Systematic review of randomised controlled trials. Knee.2017;24: 508-517. [10] STEADMAN JR, BRIGGS KK, MATHENY LM, et al. Outcomes following microfracture of full-thickness articular cartilage lesions of the knee in adolescent patients.J Knee Surg 2015;28(02):145-150. [11] SALZMANN GM, SAH B, SÜDKAMP NP, et al. Reoperative characteristics after microfracture of knee cartilage lesions in 454 patients. Knee Surg Sports Traumatol Arthrosc.2013; 21(2):365-371. [12] SOLHEIM E, ØYEN J, HEGNA J, et al. Microfracture treatment of single or multiple articular cartilage defects of the knee: a 5-year median follow-up of 110 patients.Knee Surg Sports Traumatol Arthrosc.2010;18(4):504-508. [13] BENTLEY G, BIANT LC, VIJAYAN S, et al., Carrington RW. Minimum ten-year results of a prospective randomised study of autologous chondrocyte implantation versus mosaicplasty for symptomatic articular cartilage lesions of the knee. J Bone Joint Surg Br.2012;94(4):504-509. [14] ROBB CA, EL SAYED C, MATHARU GS, et al. Survival of autologous osteochondral grafts in the knee and factors influencing outcome. Acta Orthop Belg. 2012;78(05): 643-651. [15] MINAS T, VON KEUDELL A, BRYANT T, et al.. The John Insall Award:a minimum 10-year outcome study of autologous chondrocyte implantation. Clin Orthop Relat Res.2014;472(01):41-51. [16] GOMOLL AH, GILLOGLY SD, COLE BJ, et al. Autologous chondrocyte implantation in the patella: a multicenter experience. Am J Sports Med.2014;42(5):1074-1081. [17] FARR J. Autologous chondrocyte implantation improves patellofemoral cartilage treatment outcomes. Clin Orthop Relat Res.2007;463:187-194. [18] LEVY YD, GÖRTZ S, PULIDO PA, et al. Do fresh osteochondral allografts successfully treat femoral condyle lesions? Clin Orthop Relat Res.2013;471(1):231-237. [19] GRACITELLI GC, MERIC G, PULIDO PA, et al, Bugbee WD. Fresh osteochondral allograft transplantation for isolated patellar cartilage injury. Am J Sports Med.2015;43(4): 879-884 [20] SADR KN, PULIDO PA, MCCAULEY JC, et al. Osteochondral allograft trans-plantation in patients with osteochondritis dissecans of the knee. Am J Sports Med.2016;44(11):2870-2875. [21] DEGEN RM, COLEMAN NW, CHANG B, et al, Williams RJ. Outcomes following structural grafting of distal femoral osteochondral injuries in patients aged 40 years and older. J Knee Surg. 2017;30(3):244-251 [22] BERRUTO M, DELCOGLIANO M, DE CARO F, et al. Treatment of large knee osteochondral lesions with a biomimetic scaffold: results of a multicenter study of 49 patients at 2-year follow-up. Am J Sports Med.2014;42(7): 1607-1617. [23] DHOLLANDER AA, LIEKENS K, ALMQVIST KF, et al. A pilot study of the use of an osteochondral scaffold plug for cartilage repair in the knee and how to deal with early clinical failures. Arthroscopy. 2012;28(02):225-233. [24] HORTON MT, PULIDO PA, MCCAULEY JC, et al. Revision osteochondral allograft transplantations: do they work? Am J SportsMed.2013;41(11):2507-2511 [25] GRACITELLI GC, MERIC G, BRIGGS DT, et al. Fresh osteochondral allografts in the knee: comparison of primary transplantation versus transplantation after failure of previous subchondral marrow stimulation. Am J Sports Med.2015;43(4):885-891 [26] GRACITELLI GC, MERIC G, PULIDO PA, et al. Osteochondral allograft transplantation for knee lesions after failure of cartilage repair surgery.Cartilage.2015; 6(2): 98-105 [27] JUNGMANN PM, SALZMANN GM, SCHMAL H, et al. Autologous chondrocyte implantation for treatment of cartilage defects of the knee: what predicts the need for reintervention? Am J Sports Med. 2012;40(1):58-67. [28] NIETHAMMER TR, VALENTIN S, FICKLSCHERER A, et al. Revision surgery after third generation autologous chondrocyte implantation in the knee. Int Orthop.2015; 39(8):1615-1622. [29] VIJAYAN S, BENTLEY G, RAHMAN J, et al. Revision cartilage cell transplantation for failed autologous chondrocyte transplantation in chronic osteochondral defects of the knee. Bone Joint J. 2014;96-B(1):54-58. [30] BERIS AE, LYKISSAS MG, KOSTAS-AGNANTIS I, et al. Treatment of full-thickness chondral defects of the knee with autologous chondrocyte implantation: a functional evaluation with long-term follow-up. Am J Sports Med. 2012;40(3):562-567. [31] ROSA D, BALATO G, CIARAMELLA G, et al.Long-term clinical results and MRI changes after autologous chondrocyte implantation in the knee of young and active middle aged patients. J Orthop Traumatol.2016;17(1): 55-62. [32] MURPHY RT, PENNOCK AT, BUGBEE WD. Osteochondral allograft transplantation of the knee in the pediatric and adolescent population. Am J Sports Med.2014;42(3): 635-640. [33] FRANK RM, LEE S, LEVY D, et al. Osteochondral allograft transplantation of the knee: analysis of failures at 5 years. Am J Sports Med.2017;45(4):864-874. [34] JOSHI N, REVERTE-VINAIXA M, DÍAZ-FERREIRO EW, et al. Synthetic resorbable scaffolds for the treatment of isolated patellofemoral cartilage defects in young patients: magnetic resonance imaging and clinical evaluation. Am J Sports Med.2012;40(6):1289-1295. [35] BRIGGS DT, SADR KN, PULIDO PA, et al.. The use of osteochondral allograft transplantation for primary treatment of cartilage lesions in the knee. Cartilage.2015; 6(4):203-207. [36] BERT JM. Abandoning microfracture of the knee: has the time come? Arthroscopy. 2015;31(3):501-505. [37] ZEDDE P, CUDONI S, GIACHETTI G, et al. Subchondral bone remodeling: comparing nanofracture with microfracture. an ovine in vivo study. Joints.2016;4(2): 87-93. [38] BENTLEY G, BHAMRA JS, GIKAS PD, et al. Repair of osteochondral defects in joints--how to achieve success. Injury. 2013;44 Suppl 1:S3-10. |
| [1] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [2] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [3] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [4] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Liu, Zhou Qingzhu, Gong Zhuo, Liu Boyan, Yang Bin, Zhao Xian. Characteristics and manufacturing techniques of collagen/inorganic materials for constructing tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 607-613. |
| [7] | Ye Haimin, Ding Linghua, Kong Weihao, Huang Zutai, Xiong Long. Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 621-625. |
| [8] | Song Kaikai, Zhang Kai, Jia Long. Microenvironment and repair methods of peripheral nervous system injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 651-656. |
| [9] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [10] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [11] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [12] | Gan Lili, Xiong Na, Liu Yanfei. Hydrogel as drug scaffold in skin wound repair: challenges of clinical application possibilities [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3578-3583. |
| [13] | Ren Wenbo, Liao Yuanpeng. Visualization analysis of traumatic osteoarthritis research hotspots and content based on CiteSpace [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3374-3381. |
| [14] | Jiang Lingkai, Wei Lei, Dong Zhengquan, Gu Xiaodong, Li Pengcui. Comparison of the effects of arthroscopic double-row suture fixation and suture bridge fixation for rotator cuff injury: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3431-3437. |
| [15] | Lu Mingfeng, Zhao Lilian, Xing Jisi, He Lilei, Xu Ting, Wang Changbing. Posttraumatic progression of cartilage degeneration following anterior cruciate ligament reconstruction: a second-look arthroscopic analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(2): 222-227. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||