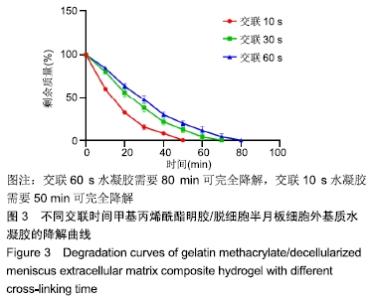
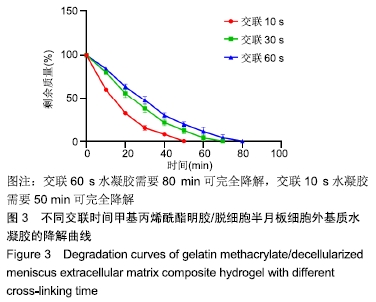
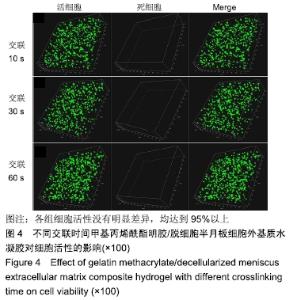
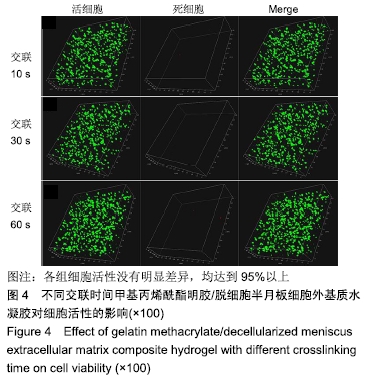
Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (16): 2493-2499.doi: 10.3969/j.issn.2095-4344.2286
Previous Articles Next Articles
Properties of gelatin methacrylate/decellularized meniscus extracellular matrix composite hydrogel with different crosslinking densities
Zhou Jian1, 2, Tian Zhuang1, 2, Tian Qinyu2, Luo Xujiang2, Peng Liqing2, Li Kun2, Sui Xiang2, Huang Jingxiang2, Liu Shuyun2, Guo Quanyi2, Yao Qi1
- 1Department of Joint Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing 100038, China; 2Institute of Orthopedics, Chinese PLA General Hospital, Beijing Key Lab of Regenerative Medicine in Orthopedics, Key Lab of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China
-
Received:2019-08-22Revised:2019-08-26Accepted:2019-12-26Online:2020-06-08Published:2020-03-24 -
Contact:Yao Qi, Chief physician, Professor, Department of Joint Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing 100038, China Guo Quanyi, Chief physician, Professor, Institute of Orthopedics, Chinese PLA General Hospital, Beijing Key Lab of Regenerative Medicine in Orthopedics, Key Lab of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China -
About author:Zhou Jian, Master candidate, Department of Joint Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing 100038, China; Institute of Orthopedics, Chinese PLA General Hospital, Beijing Key Lab of Regenerative Medicine in Orthopedics, Key Lab of Musculoskeletal Trauma & War Injuries, PLA, Beijing 100853, China -
Supported by:the National Key Research and Development Program of China, No. 2017YFC1103400; the Natural Science Foundation of Beijing, No. 7172203
CLC Number:
Cite this article
Zhou Jian, Tian Zhuang, Tian Qinyu, Luo Xujiang, Peng Liqing, Li Kun, Sui Xiang, Huang Jingxiang, Liu Shuyun, Guo Quanyi, Yao Qi. Properties of gelatin methacrylate/decellularized meniscus extracellular matrix composite hydrogel with different crosslinking densities[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2493-2499.
share this article

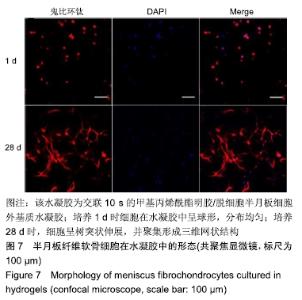
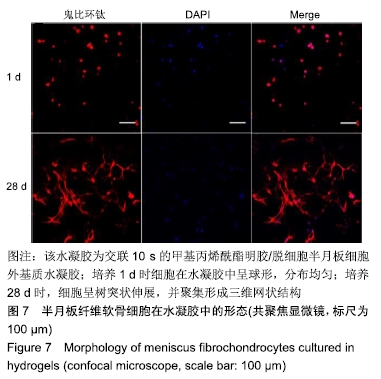
2.1 水凝胶的制备 dMECM浆液是由猪半月板通过湿法差速离心方法制备,见图1A,经课题组成员前期测试显示,dMECM保留了大部分的胶原和糖胺多糖,细胞成分绝大部分被去除[27-28]。半月板纤维软骨细胞取自1月龄新西兰白兔,通过重复多次Ⅱ型胶原酶消化的方法能够取得大量的原代细胞,细胞汇合度达到80%-90%进行传代,所有实验均取用P2-P5代细胞,该区间的细胞具有很好的形态(长梭形)和增殖能力,见图1B。GelMA并不能迅速且完全溶于PBS中,反复冻融及长时间置于60 ℃水浴锅溶解,有利于消除GelMA溶液中的泡沫及不溶成分。GelMA/dMECM预凝胶溶液具有一定的温敏性,其在37 ℃时呈液体状态,见图1C,在经过短暂的蓝光交联后能够形成稳定的胶体状态,见图1D。 "

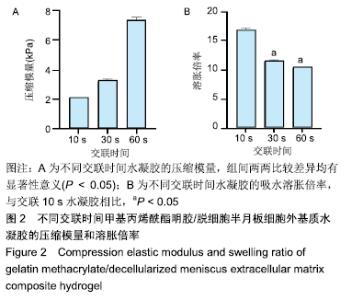
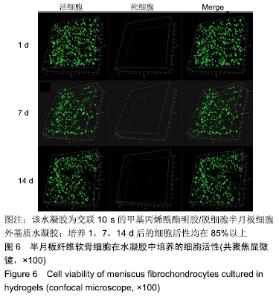
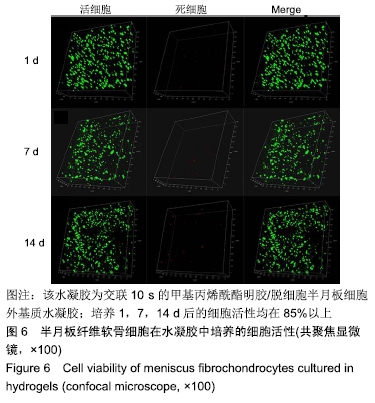
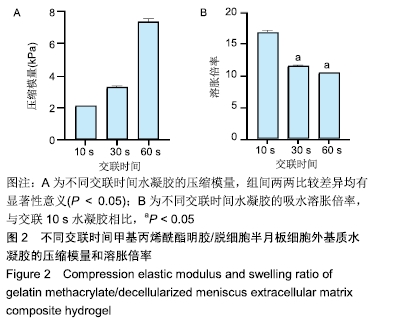
2.2 水凝胶的力学强度 由图2A可以看出3组之间力学性能差异明显,交联60 s水凝胶的压缩模量为(7.35±0.19) kPa,明显高于交联10,30 s的水凝胶(P < 0.05),并且交联30 s水凝胶的压缩模量高于交联10 s的水凝胶(P < 0.05)。证明随着交联时间的延长,水凝胶的压缩模量随之增高。 2.3 水凝胶的溶胀倍率 通过充分溶胀然后冻干方式对水凝胶的溶胀倍率进行测试,结果显示交联10 s水凝胶的溶胀倍率为16.81±0.31,明显高于交联30,60 s的水凝胶(P < 0.05),由此可见水凝胶的交联密度和溶胀性紧密相关;但是交联30,60 s水凝胶的溶胀倍率差异不大,说明30 s后的交联时间延长对溶胀的性能影响相对较小,见图2B。 "
| [1] FOX AJ, WANIVENHAUS F, BURGE AJ, et al.The human meniscus: a review of anatomy, function, injury, and advances in treatment.Clin Anat.2015;28(2):269-287. [2] WALKER PS, ARNO S, BELL C, et al. Function of the medial meniscus in force transmission and stability.J Biomech. 2015; 48(8):1383-1388. [3] CLARK CR, OGDEN JA. Development of the menisci of the human knee joint. Morphological changes and their potential role in childhood meniscal injury.J Bone Joint Surg Am. 1983; 65(4):538-547. [4] ARNOCZKY SP, WARREN RF. Microvasculature of the human meniscus.Am J Sports Med. 1982;10(2):90-95. [5] MAKRIS EA, HADIDI P, ATHANASIOU KA. The knee meniscus: Structure–function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials. 2011;32(30):7411-7431. [6] LEE KY, MOONEY DJ. Hydrogels for Tissue Engineering. 2001;101(7):1869-1879. [7] KHADEMHOSSEINI A, VACANTI JP, LANGER R. Progress in tissue engineering.Sci Am. 2009;300(5):64-71. [8] HUNT NC, GROVER LM. Cell encapsulation using biopolymer gels for regenerative medicine.Biotechnol Lett. 2010;32(6):733-742. [9] SELIKTAR D. Designing Cell-Compatible Hydrogels for Biomedical Applications.Science.2012;336(6085):1124-1128. [10] SPILLER KL, MAHER SA, LOWMAN AM. Hydrogels for the Repair of Articular Cartilage Defects. Tissue Eng Part B Rev. 2011;17(4):281-299. [11] DASH M, CHIELLINI E, CHIELLINI F, et al. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog Polym Sci.2011;36(8):981-1014. [12] MOXON SR, CORBETT NJ, FISHER K, et al. Blended alginate/collagen hydrogels promote neurogenesis and neuronal maturation. Mater Sci Eng C Mater Biol Appl. 2019;104:109904. [13] TAVAKOLI J, LAISAK E, GAO M, et al. AIEgen quantitatively monitoring the release of Ca2+ during swelling and degradation process in alginate hydrogels.Mater Sci Eng C Mater Biol Appl.2019;104:109951. [14] SEONG YJ, LIN G, KIM BJ, et al. Hyaluronic Acid-Based Hybrid Hydrogel Microspheres with Enhanced Structural Stability and High Injectability.ACS Omega.2019;4(9):13834-13844. [15] LI M, ZHANG X, JIA W, et al.Improving in vitro biocompatibility on biomimetic mineralized collagen bone materials modified with hyaluronic acid oligosaccharide.Mater Sci Eng C Mater Biol Appl.2019;104:110008.doi:10.1016/j.msec.2019.110008. [16] GARRETA E, ORIA R, TARANTINO C, et al. Tissue engineering by decellularization and 3D bioprinting.Mater Today. 2017;20(4):166-178. [17] KYLE S, JESSOP ZM, TARASSOLI SP, et al. Assessing printability of bioinks.In book: 3D Bioprinting for Reconstructive Surgery - Techniques and Applications, Edition: 1, Publisher: Woodhead Publishing, Editors: Daniel Thomas Zita Jessop Iain Whitaker,2017. [18] CHIMMENE D, LENNOX KK, KAUNAS RR, et al. Advanced Bioinks for 3D Printing: A Materials Science Perspective.Ann Biomed Eng.2016;44(6):2090-2102. [19] RUEDINGER F, LAVRENTIEVA A, BLUME C, et al. Hydrogels for 3D mammalian cell culture: a starting guide for laboratory practice.Appl Microbiol Biotechnol.2015;99(2):623-636. [20] VAN DEN BULCKE AI, BOGDANOV B, DE ROOZE N, et al. Structural and rheological properties of methacrylamide modified gelatin hydrogels.Biomacromolecules.2000;1(1): 31-38. [21] KLOTZ BJ, GAWLITTA D, ROSERBERG A, et al. Gelatin-Methacryloyl Hydrogels: Towards Biofabrication-Based Tissue Repair.Trends Biotechnol. 2016;34(5):394-407. [22] AUBIN H, NICHOL JW, HUTSON CB, et al. Directed 3D cell alignment and elongation in microengineered hydrogels. Biomaterials.2010;31(27):6941-6951. [23] WANG C, VARSHNEY RR, WANG DA. Therapeutic cell delivery and fate control in hydrogels and hydrogel hybrids.Adv Drug Deliv Rev.2010;62(7-8):699-710. [24] YUE K, TRUJILLO-DE SANTIAGO G, ALVAREZ MM, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels.Biomaterials. 2015;73: 254-271. [25] FAIRBANKS BD, SCHWARTZ MP, BOWMAN CN, et al. Photoinitiated polymerization of PEG-diacrylate with lithium phenyl-2,4,6-trimethylbenzoylphosphinate: polymerization rate and cytocompatibility. Biomaterials. 2009;30(35): 6702-6707. [26] WOLF MT, DALY KA, BRENNAN-PIERCE EP, et al. A hydrogel derived from decellularized dermal extracellular matrix. Biomaterials.2012;33(29):7028-7038. [27] 苑志国,卢世璧,郭全义,等.脱细胞半月板细胞外基质/脱钙骨基质双相半月板支架的制备及其生物相容性的研究[J].中国医药生物技术,2016,11(1):4-12. [28] 郭维民,卢世璧,郭全义,等.新型脱细胞半月板细胞外基质的制备及其生物相容性的研究[J].中国医药生物技术,2015,10(1):5-10. [29] GAO F, XU Z, LIANG Q, et al. Osteochondral Regeneration with 3D-Printed Biodegradable High-Strength Supramolecular Polymer Reinforced-Gelatin Hydrogel Scaffolds.Adv Sci (Weinh). 2019;6(15):1900867. [30] LIU H, SHI X, WU D, et al. Injectable, Biodegradable, Thermosensitive Nanoparticles-Aggregated Hydrogel with Tumor-Specific Targeting, Penetration, and Release for Efficient Postsurgical Prevention of Tumor Recurrence.ACS Appl Mater Interfaces.2019;11(22):19700-19711. [31] WANG Y, MA M, WANG J, et al. Development of a photo-crosslinking, biodegradable GelMA/PEGDA hydrogel for guided bone regeneration materials.Materials. 2018;11(8): 1345. [32] KORNMULLER A, BROWUN CFC, YU C, et al. Fabrication of extracellular matrix-derived foams and microcarriers as tissue-specific cell culture and delivery platforms.J Vis Exp. 2017;(122).doi: 10.3791/55436. [33] YU C, KORNUMLLWE A, BROWN C, et al. Decellularized adipose tissue microcarriers as a dynamic culture platform for human adipose-derived stem/stromal cell expansion. Biomaterials. 2017;120:66-80. [34] YU C, BIANCO J, BROWN C, et al. Porous decellularized adipose tissue foams for soft tissue regeneration. Biomaterials. 2013;34(13):3290-3302. [35] PATI F, JANG J, HA DH, et al. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat Commun.2014;5:3935. [36] BELLIDO T. Osteocyte-driven bone remodeling.Calcif Tissue Int.2014;94(1):25-34. [37] BADYLAK SF, FREYTES DO, GILBERT TW. Reprint of: Extracellular matrix as a biological scaffold material: Structure and function.Acta Biomater.2015;23 Suppl:S17-26. [38] YU C, MA X, ZHU W, et al. Scanningless and continuous 3D bioprinting of human tissues with decellularized extracellular matrix.Biomaterials.2019;194:1-13.doi: 10.1016/j.biomaterials. 2018.12.009 [39] LIU Y, CHAN-PARK MB. A biomimetic hydrogel based on methacrylated dextran-graft-lysine and gelatin for 3D smooth muscle cell culture.Biomaterials.2010;31(6):1158-1170. [40] FAN C, WANG DA. Macroporous Hydrogel Scaffolds for Three-Dimensional Cell Culture and Tissue Engineering. Tissue Eng Part B Rev.2017;23(5):451-461. [41] MASON BN, CALIFANO JP, REINHART-KIN CA. Matrix Stiffness: A Regulator of Cellular Behavior and Tissue Formation//Engineering Biomaterials for Regenerative Medicine.Springer New York,2012:19-37. [42] STEIN S, HOSE S, WARNECKE D, et al.Meniscal Replacement With a Silk Fibroin Scaffold Reduces Contact Stresses in the Human Knee.J Orthop Resy.2019; 37(12): 2583-2592. [43] LEE M, WU BM, DUNN JC. Effect of scaffold architecture and pore size on smooth muscle cell growth.J Biomed Mater Res A.2008;87(4):1010-1016. [44] NAGANUMA T, TRAVERSA E. The effect of cerium valence states at cerium oxide nanoparticle surfaces on cell proliferation. Biomaterials.2014;35(15):4441-4453. |
| [1] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [2] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [3] | Liu Fang, Shan Zhengming, Tang Yulei, Wu Xiaomin, Tian Weiqun. Effects of hemostasis and promoting wound healing of ozone sustained-release hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3445-3449. |
| [4] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [5] | Li Xinping, Cui Qiuju, Zeng Shuguang, Ran Gaoying, Zhang Zhaoqiang, Liu Xianwen, Fang Wei, Xu Shuaimei. Effect of modification of β-tricalcium phosphate/chitosan hydrogel on growth and mineralization of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3493-3499. |
| [6] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| [7] | Gan Lili, Xiong Na, Liu Yanfei. Hydrogel as drug scaffold in skin wound repair: challenges of clinical application possibilities [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3578-3583. |
| [8] | Peng Chao, Liu Yunpeng, Hua Guojun, Yang Jiaji, Wang Xingliang, Wang Xiaolong. Imaging evaluation of the hip-knee-ankle angle and osteoarthritis progression before and after partial meniscectomy for degenerative medial meniscus posterior root tear [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3368-3373. |
| [9] | Chen Siyu, Li Yannan, Xie Liying, Liu Siqi, Fan Yurong, Fang Changxing, Zhang Xin, Quan Jiayu, Zuo Lin. Thermosensitive chitosan-collagen composite hydrogel loaded with basic fibroblast growth factor retards ventricular remodeling after myocardial infarction in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2472-2478. |
| [10] | Liu Feng, Zhang Yu, Wang Yanli, Luo Wei, Han Chaoshan, Li Yangxin. Application of temperature-sensitive chitosan hydrogel encapsulated exosomes in ischemic diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2479-2487. |
| [11] | Zhang Xin, Lu Ying, Yao Qingqiang, Zhu Yishen. Preparation and in vitro evaluation of self-assembling peptide hydrogel loaded with doxorubicin [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2488-2493. |
| [12] | Xu Changkui, Pu Xiaobing, Lu Yao, Chen Jiarong, Pan Lei. Safety and antibacterial properties of gentamicin-loaded silk fibroin in meniscus repair [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1545-1549. |
| [13] | Bai Xue, Wang Bin, He Sirong. Research focus and application advantages in encapsulating biomaterial for islet transplantation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1585-1591. |
| [14] | Li Jun, Yu Hao, Zhang Yong, Wei Yong, Xie Jia, Jing Juehua. Different types of hydrogel in bone defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1599-1603. |
| [15] | Jiang Zongrui, Zhang Zhiqi. Treatment of meniscus injury or degeneration: the effect and function of stem cells and artificial polymer scaffolds to form tissue engineering system [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(34): 5421-5427. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||