Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (11): 1762-1768.doi: 10.3969/j.issn.2095-4344.2213
Previous Articles Next Articles
Acellular adipose tissue preparation: a soft-tissue filler allowing for allogeneic injection or in situ adipogenesis?
Nie Jiaying, Yi Yangyan, Zhu Yuanzheng
- Department of Plastic and Cosmetic Surgery, The Second Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China
-
Received:2019-05-11Revised:2019-05-23Accepted:2019-07-31Online:2020-04-18Published:2020-02-29 -
Contact:Yi Yangyan, Chief physician, Department of Plastic and Cosmetic Surgery, The Second Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China -
About author:Nie Jiaying, Master candidate, Department of Plastic and Cosmetic Surgery, The Second Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 81660326; the Natural Science Foundation of Jiangxi Province, No. 20171ACB20037
CLC Number:
Cite this article
Nie Jiaying, Yi Yangyan, Zhu Yuanzheng. Acellular adipose tissue preparation: a soft-tissue filler allowing for allogeneic injection or in situ adipogenesis? [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(11): 1762-1768.
share this article
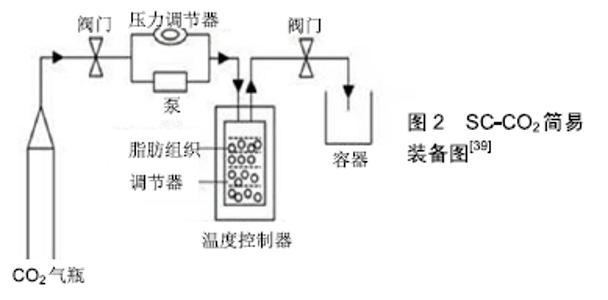
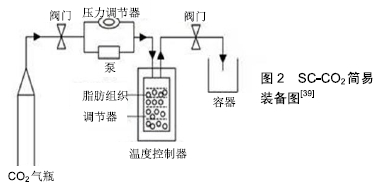
2.1 传统脱细胞脂肪组织的制作方案 一般来说,希望在不破坏细胞外基质的结构和功能成分的情况下使用尽可能温和的方法生成脱细胞支架。目前典型的脱细胞化方案包括物理方法(如冻融循环、均质化)、化学方法(如SDS、Triton x-100和EDTA)、生物方法(如胰蛋白酶、核酸酶)。然而,仅使用其中的任何一种都不能做到完全脱细胞化,而这些方法的组合已经被证明可以提高脱细胞效率和脂肪组织结构的完整性[30]。 FLYNN[35]是首次使用综合方案制作脱细胞脂肪组织的研究者。具体方案:①将脂肪组织与脱细胞液混合并在搅拌机中搅拌;②在低渗Tris溶液中进行3次冻融(-80-37 ℃)循环,每次循环历时1 h致使细胞膜破坏的同时促使细胞成分和脂质释放,促进后续过程的细胞成分和脂质的去除;③0.25%胰蛋白酶/0.1%EDTA溶液过夜孵化16 h致使裂解精氨酸和赖氨酸的碳侧肽键而除去细胞碎片[31];④使用99.9%的异丙醇极性萃取48 h,除去脂质成分;⑤继续使用0.25%胰蛋白酶/0.1%EDTA溶液孵化6 h使得细胞成分完全去除;⑥接下来使用核酸酶(DNase、RNase)和脂肪酶过夜孵化16 h裂解核酸序列和脂质以除去核酸和剩余的脂质;⑦99.9%异丙醇极性萃取8 h以做到完全去除脂质。该方案可使质量为25 g的大块脂肪组织完全脱细胞同时也保留了脱细胞脂肪组织中大量的Ⅳ型胶原(CIV)和层粘连蛋白。该方案损失的细胞外基质蛋白较少,最后获得的水合脱细胞脂肪组织体积与原脂肪组织块体积相近,质量占原脂肪组织块的30%-45%。 CHOI等[30]利用高速离心和SDS等化学试剂进行脱细胞化。SDS是一种阴离子型洗涤剂,对去除组织中的细胞成分非常有效,但SDS对脂肪组织细胞外基质的结构破坏和蛋白丢失影响较大,特别是层粘连蛋白和糖胺聚糖丢失十分严重。因此尽管该方案去除了细胞成分,但层粘连蛋白和糖胺聚糖含量却丢失严重。 YOUNG等[36]采用SDS和酶相结合的方案制作脱细胞脂肪组织。该方案成功的保留了Ⅳ型胶原蛋白和层粘连蛋白。但不足的是DNA去除不足,导致宿主小鼠对脱细胞脂肪组织发生了较严重的免疫排异反应。 BROWN等[37]用了非离子洗涤剂Triton X-100和异丙醇相结合的方案制备脱细胞脂肪组织。Triton X-100破坏脂质-脂质和脂质-蛋白质之间的相互作用,但对蛋白质-蛋白质的相互作用没有太大的影响,因此使得细胞外基质蛋白和结构得到了较好的保留[32]。最后再加入异丙醇去除脂质。该方案处理时间较短,脱细胞脂肪组织的天然胶原结构无较大破坏,同时可以检测到血管内皮生长因子F和糖胺聚糖, 但该方案完全去除了天然脂肪细胞外基质的关键成分-层粘连蛋白。 WANG等[38]方案是从他们先前肌筋膜脱细胞方法修改而来[33]。该方案与FLYNN等[35]的方案十分相似,如极性溶剂萃取。他们将Flynn中的胰蛋白酶改为使用Triton X-100溶液孵化。该方案保留了胶原蛋白和糖胺聚糖和血管内皮生长因子,同时在脱细胞化方面非常成功。但不足的是,处理时间非常长,同时导致层粘连蛋白完全丢失。 SANO等[34]分别将纯物理方案(冻融)、洗涤剂和酶相结合的方案与Flynn方案进行对比,并对每种方案进行了相应的修改,以确定最佳方法。其中反复冻融的方案是最不成功的,即使将冻融次数增加至18次,细胞成分仍存在。只有延长Flynn方案处理时间才能实现完全脱细胞化,同时能检测到Ⅳ型胶原蛋白和层粘连蛋白的存在。最终获得的脱细胞脂肪组织体积与原脂肪块体积相近,质量占原脂肪块质量的30%-40%,这与Flynn所报道的相似。同时,SANO等发现即使延长Flynn方案的处理时间,也仅对质量小于0.8 g的脂肪组织才能实现脱细胞化。但他们认为该方案兼顾了脱细胞和保留细胞外基质蛋白,是经典的脱细胞脂肪组织制作方案。 2.2 改良脱细胞脂肪组织制作方案 从表2方案可以总结出,这些方案存在脱细胞时间过长和/或使用了Triton x-100和SDS等洗涤剂。对于洗涤剂,REN等[41]认为其对细胞外基质蛋白和结构损坏比较大。CEBOTARI等[42]也认为即使是低浓度的洗涤剂残留也存在细胞毒性,同时会抑制脱细胞脂肪组织功能。同时通过实验发现,即使是薄的组织,如阀瓣叶,也需要多次(超过6次)搅拌洗涤,才能完全去除洗涤剂[42]。因此残留化学试剂的清除也是一大难题。由于以上原因,WANG等[39]使用了超临界值二氧化碳设备(Supercritical carbon dioxide,SC-CO2)。该装备自20世纪70年代以来,一直用于提取海鱼中的鱼油和微藻中的脂质,且食品和药物管理局(the Food and Drug Administration,FDA)认为该装备是安全的。该装备大概示意图2[39]。大概步骤是将脂肪组织放入反应容器内,然后通过压力泵将CO2压力调节至18 000 kPa处理3 h。该装备可缩短脱细胞的时间,且除乙醇之外没有加入其他化学物质,因此,该方法可减少化学物质对细胞外基质蛋白的破坏以及制作过程中化学物质引入难以去除的问题[40]。同时 HE等[40]在FLYNN[35]的研究基础上使用了一种新的机械处理方案。该方案是将离心过的脂肪组织与等量的PBS混合后,用双接头接管连接两个内径为1.4 mm的10 mL注射器,将脂肪组织通过该装备以恒定速率10 mL/s转换1 min进行机械乳化。该方案除了降低了脱细胞过程脂质的去除时间,从而缩短了异丙醇的萃取的时间,减少了异丙醇对细胞外基质蛋白和结构的损坏。同时还得到了一种特殊的脂肪组织液体提取物,经过酶联免疫吸附试验证明该提取液中含有高浓度的血管生成因子。体内外实验表明将该提取液与脱细胞脂肪组织相结合后,其再细胞化能力比Flynn制作的脱细胞脂肪组织更高。 "

2.3 脂肪来源的干细胞辅助脱细胞脂肪组织移植 在FLYNN等[35]的研究中,与没有接种脂肪干细胞的脱细胞脂肪组织相比,接种脂肪干细胞的脱细胞脂肪组织过氧化物酶体增殖活化受体γ和CCAAT增强结合蛋白α表达增加。CERQUEIRA等[43]为脂肪干细胞通过旁分泌细胞因子调控宿主体内的内皮细胞,从而促进新生血管的形成。2013年POON等[44]在首次在小鼠脱细胞脂肪组织移植模型中播种了同种异体脂肪干细胞,通过8周的观察发现,单独移植脱细胞脂肪组织的组,只剩下原脱细胞脂肪组织体积的30%,而接种脂肪干细胞的脱细胞脂肪组织组,体积剩余原体积的48%,但遗憾的是没有与传统的脂肪移植进行对比。2014年,ADAM YOUNG团队[45]发现,脂肪细胞外基质水凝胶联合人脂肪干细胞在无胸腺小鼠模型中可刺激血管生成和脂肪生成。在2015年,HAN团队[46]在免疫能力强大的大鼠脱细胞脂肪组织移植模型中加入了异体脂肪干细胞,发现脱细胞脂肪组织在体内成脂和成血管能力加强的原因可能与异体脂肪干细胞介导了炎症反应,促进了成脂能力强的CD163+的M2型巨噬细胞生成,从而促进了脂肪和血管的生成。然而,由于脂肪干细胞可能存在致瘤的风险,导致该方案的临床应用存在巨大的安全问题[47]。 2.4 生长因子辅助脱细胞脂肪组织移植 CHOI等[48]、 TURNER等[49]研究表明脱细胞脂肪组织无论是体外或体内实验,其诱导成脂能力并不令人满意。2014年TING 等[50]认为导致这种情况的原因可能与血管生成不足有关,因此,其将碱性成纤维细胞生长因子、血管内皮生长因子、血小板源性生长因子BB“绑定”至脱细胞脂肪组织上,发现脂肪组织的体积有显著增加。但直接将生长因子“绑定”至脱细胞脂肪组织上可导致生长因子快速耗尽。而组织移植早期快速建立血供需要稳定而持久的生长因子刺激。LU等[51-52]为了弥补以上的不足,通过研究发现肝素可以增加碱性成纤维细胞生长因子的“绑定”数量,并控制碱性成纤维细胞生长因子的释放。结果表明,与肝素交联后的脱细胞脂肪组织再与碱性成纤维细胞生长因子“绑定”后,此种脱细胞脂肪组织的成脂能力和抗降解能力有明显上升。但ZENG等[53]、BERNARDES等[54]认为在多种肿瘤发生和发展过程中,生长因子(如碱性成纤维细胞生长因子)起着关键性的作用,如肺癌等,因此细胞因子的安全性仍受到不少学者的质疑。 2.5 添加脱细胞生物支架制备过程中损耗的细胞外基质蛋白 ZHOU等[55]通过对比髓核组织脱细胞前后细胞外基质蛋白的差异发现髓核脱细胞后糖胺聚糖与胶原蛋白的比值有所下降,其查阅文献发现主要是由于糖胺聚糖含量下降所引起。因此他们将糖胺聚糖蛋白加入至髓核脱细胞组织中,观察到脱细胞髓核支架(decellularized nucleus pulposus,dNP)的刚度与新鲜髓核组织的相似,且脂肪干细胞向髓核组织分化能力也有所增高。其可能机制可能是由于糖胺聚糖的生物学作用:①有很大的黏滞性,具有润滑和保护作用;②具有促进新生血管生成、组织再生的作用。但在这类方案在脱细胞脂肪组织移植中尚未见报道,若能对比脂肪组织脱细胞前后细胞外基质蛋白的差异,从而加强宿主细胞募集或调整脱细胞脂肪组织刚度的角度添加层粘连蛋白或糖胺聚糖等,可能对脱细胞脂肪组织移植后的血管及脂肪再生有一定的促进作用。 2.6 脱细胞脂肪组织水凝胶的制作方案 脱细胞脂肪组织支架在机体内的降解原因与基质金属蛋白酶或分解细胞外基质蛋白相关的细菌、酶渗入至脱细胞脂肪组织有关[51-52]。化学交联剂可通过增加脱细胞脂肪组织中的蛋白质含量而提高了脱细胞脂肪组织的抗降解能力[56]。WU等[56]通过在水凝胶中加入了1-乙基3-(3-二甲基胺丙基)盐酸二亚胺,体外实验表明,该水凝胶支持脂肪干细胞的生长和分化。大鼠模型表明,交联剂增加水凝胶对酶降解的抗性,且促进宿主细胞迁移、脂肪组织发育和血管形成。通过长期的动物实验发现,该水凝胶形成了高度血管化的脂肪组织。LI等[57]首先通过将脱细胞脂肪组织与硫醇-丙烯酸脂通过迈克尔加成反应聚合成水凝胶,然后再与聚乙二醇交联。结果表明该水凝胶提高了脂肪干细胞的增殖速度和向脂肪分化的能力。 丝素蛋白由于具有良好的生物相容性、在机体降解速度缓慢、炎症反应小和机械能力好等优点,在组织工程中得到了越来越多的应用。2007年,MAUNEY等[58]通过将丝素蛋白、胶原蛋白、聚乳酸3种支架做对比,检测三者的成脂和抗降解能力发现,3种支架在体外诱导成脂能力没有太多差距,但经过4周的体内实验发现,丝素蛋白支架仍然存在,而胶原蛋白和聚乳酸支架在植入后降解迅速。2009年,KANG等[59]也认为丝素支架由于降解速度缓慢,使得软组织持续生长成为可能。2017年,KAYABOLEN等[60]将脱细胞脂肪组织与丝素通过涡旋作用聚合成水凝胶提高脱细胞脂肪组织的刚度并增强其早期的血管再生能力,和远期成脂效率。然而,大多数交联剂存在成本高和有细胞毒性的缺点,因此受到大多数学者的质疑。 YOUNG等[36]、ZHAO等[61]在不添加交联剂的前提下,制作出脱细胞脂肪组织水凝胶。其水凝胶的制作大概分为两个部分:脂肪组织脱细胞化、凝胶化。①脂肪组织脱细胞化:YOUNG等使用的脱细胞方案是将洗涤剂(SDS、脱氧胆酸钠)和酶相结合;ZHAO等使用的脱细胞方案与BROWN等[37]相似;②凝胶化:使用的方法是将脱细胞脂肪组织冻干并碾磨成粉,再用胃蛋白酶和盐酸溶液溶解,最后用氢氧化钠中和。该凝胶在4 ℃时为流动的液体状,在37 ℃下60 min凝胶化。但POON等[44]认为在水凝胶的制备方案中加盐酸,可能会对细胞外基质蛋白造成破坏,因此他们制作了不需添加交联剂的水凝胶时改用较为温和的醋酸来参与蛋白的溶解与凝胶化。该水凝胶的制作大概分为3个部分:脱细胞化、蛋白萃取、凝胶化。主要思路为:①去除细胞成分降低免疫反应;②使用尿素缓冲液和胍-盐酸缓冲液将细胞外基质中的蛋白提取出来;③使用胃蛋白酶和醋酸将蛋白质溶解,加入NaOH将溶液调至中性并使其凝胶化。该水凝胶在4 ℃为液体状,在37 ℃温度下放置15 min左右即可成凝胶状。此方案避免了交联剂和酸性物质对细胞外基质蛋白的破坏,但此制作方案由于需要3次透析因而增加了成本,同时该方案制备也较繁琐。 "

| [1] HENKEL J, WOODRUFF MA, EPARI DR,et al. Bone Regeneration Based on Tissue Engineering Conceptions - A 21st Century Perspective. Bone Res.2013;1(3):216-248. [2] ATALA A, BAUER SB, SOKER S, et al. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet. 2006;367(9518): 1241-1246. [3] BIRCHALL MA, SEIFALIAN AM.Tissue engineering's green shoots of disruptive innovation. 2014;384(9940):288-290. [4] FULCO I, MIOT S, HAUG MD, et al. Engineered autologous cartilage tissue for nasal reconstruction after tumour resection: an observational first-in-human trial. Lancet. 2014;384(9940):337-346. [5] OLAUSSON M, PATIL PB, KUNA VK, et al. Transplantation of an allogeneic vein bioengineered with autologous stem cells: a proof-of-concept study. Lancet. 2012;380(9838):230-237. [6] RAYA-RIVERA AM, ESQUILIANO D, FIERRO-PASTRANA R, et al. Tissue-engineered autologous vaginal organs in patients: a pilot cohort study. Lancet. 2014;384(9940):329-336. [7] ELLIOTT RA JR, HOEHN JG. Use of commercial porcine skin for wound dressings. Plast Reconstr Surg. 1973;52(4):401-405. [8] BADYLAK SF, LANTZ GC, COFFEY A,et al. Small intestinal submucosa as a large diameter vascular graft in the dog. J Surg Res. 1989;47(1):74-80. [9] CHEN RN, HO HO, TSAI YT, et al. Process development of an acellular dermal matrix (ADM) for biomedical applications. Biomaterials. 2004;25(13):2679-2686. [10] SCHMIDT CE, BAIER JM. Acellular vascular tissues: natural biomaterials for tissue repair and tissue engineering. Biomaterials. 2000;21(22):2215-2231. [11] UCHIMURA E, SAWA Y, TAKETANI S, et al. Novel method of preparing acellular cardiovascular grafts by decellularization with poly(ethylene glycol). J Biomed Mater Res A.2003;67:834-837. [12] DAHL SL, KOH J, PRABHAKAR V, et al.Decellularized native and engineered arterial scaffolds for transplantation. Cell Transplant.2003; 12:659-666. [13] CONKLIN BS, RICHTER ER, KREUTZIGER KL,et al.Development and evaluation of a novel decellularized vascular xenograft. Med Eng Phys 2002;24:173-183. [14] BORSCHEL GH, DENNIS RG, KUZON WM. Contractile skeletal muscle tissue-engineered on an acellular scaffold. Plast Reconstr Surg.2004;113:595-602. [15] LIN P, CHAN WC, BADYLAK SF, et al. Assessing porcine liverderived biomatrix for hepatic tissue engineering. Tissue Eng.2004;10:1046–53. [16] KROPP BP, EPPLEY BL, PREVEL CD,et al. Experimental assessment of small intestinal submucosa as a bladder wall substitute. Urology 1995;46:396-400. [17] BADER A, SCHILLING T, TEEBKEN OE, et al. Tissue engineering of heart valves-human endothelial cell seeding of detergent acellularized porcine valves. Eur J Cardiothorac Surg. 1998;14:279-284. [18] BOOTH C, KOROSSIS SA, WILCOX HE, et al. Tissue engineering of cardiac valve prostheses I:development and histological characterization of an acellular porcine scaffold.J Heart Valve Dis. 2002;11:457-462. [19] GRAUSS RW, HAZEKAMP MG, OPPENHUIZEN F, et al. Histological evaluation of decellularised porcine aortic valves: matrix changes due to different decellularisation methods. Eur J Cardiothorac Surg. 2005; 27:566-571. [20] KASIMIR MT, RIEDER E, SEEBACHER G, et al. Comparison of different decellularization procedures of porcine heart valves. Int J Artif Organs. 2003;26:421-427. [21] KOROSSIS SA, BOOTH C, WILCOX HE, et al. Tissue engineering of cardiac valve prostheses II:biomechanical characterization of decellularized porcine aortic heart valves.J Heart Valve Dis. 2002;11: 463-471. [22] RIEDER E, KASIMIR MT, SILBERHUMER G, et al. Decellularization protocols of porcine heart valves differ importantly in efficiency of cell removal and susceptibility of the matrix to recellularization with human vascular cells. J Thorac Cardiovasc Surg. 2004;127:399-405. [23] BOYCE M, RADTKE C, VOGT PM. The volumetric analysis of fat graft survival in breast reconstruction. Plast Reconstr Surg. 2013;132(5): 862e-863e. [24] CHACON AH. Fillers in dermatology: from past to present. Cutis. 2015; 96(5):E17-19. [25] CHIANG YZ, PIERONE G, AL-NIAIMI F. Dermal fillers: pathophysiology, prevention and treatment of complications. J Eur Acad Dermatol Venereol.2017;31(3):405-413. [26] VAN NIEUWENHOVE I, TYTGAT L, RYX M, et al. Soft tissue fillers for adipose tissue regeneration: From hydrogel development toward clinical applications.Acta Biomater.2017;63:37-49. [27] DZIKI JL, WANG DS, PINEDA C, et al. Solubilized extracellular matrix bioscaffolds derived from diverse source tissues differentially influence macrophage phenotype.J Biomed Mater Res A. 2017;105(1):138-147. [28] YU Y, ALKHAWAJI A, DING Y, et al. Decellularized scaffolds in regenerative medicine. Oncotarget.2016;7(36):58671-58683. [29] LIN CY, LIU TY, CHEN MH, et al. An injectable extracellular matrix for the reconstruction of epidural fat and the prevention of epidural fibrosis. Biomed Mater.2016;11(3):035010. [30] CHOI JS, KIM BS, KIM JY, et al. Decellularized extracellular matrix derived from human adipose tissue as a potential scaffold for allograft tissue engineering. J Biomed Mater Res A.2011;97(3):292-299. [31] GILBERT TW, SELLARO TL, BADYLAK SF.Decellularization of tissues and organs. Biomaterials.2006;27(19):3675-3683. [32] SEDDON AM, CURNOW P, BOOTH PJ. Membrane proteins, lipids and detergents: not just a soap opera. Biochim Biophys Acta. 2004; 1666(1-2):105-117. [33] WANG L, JOHNSON JA, CHANG DW, et al. Decellularized musculofascial extracellular matrix for tissue engineering. Biomaterials. 2013;34(11):2641-2654. [34] SANO H, ORBAY H, TERASHI H, et al. Acellular adipose matrix as a natural scaffold for tissue engineering. J Plast Reconstr Aesthet Surg. 2014;67(1):99-106. [35] FLYNN LE. The use of decellularized adipose tissue to provide an inductive microenvironment for the adipogenic differentiation of human adipose-derived stem cells. Biomaterials.2010;31(17):4715-4724. [36] YOUNG DA, IBRAHIM DO, HU D, et al.Injectable hydrogel scaffold from decellularized human lipoaspirate. Acta Biomater. 2011;7(3): 1040-1049. [37] BROWN BN, FREUND JM, HAN L, et al.Comparison of three methods for the derivation of a biologic scaffold composed of adipose tissue extracellular matrix. Tissue Eng Part C Methods.2011;17(4):411-421. [38] WANG L, JOHNSON JA, ZHANG Q, et al. Combining decellularized human adipose tissue extracellular matrix and adipose-derived stem cells for adipose tissue engineering.Acta Biomater. 2013;9(11):8921-8931. [39] WANG JK, LUO B, GUNETA V, et al. Supercritical carbon dioxide extracted extracellular matrix material from adipose tissue. Mater Sci Eng C Mater Biol Appl.2017;75:349-358. [40] HE Y, LIN M, WANG X, et al.Optimized adipose tissue engineering strategy based on a neo-mechanical processing method. Wound Repair Regen.2018;26(2):163-171. [41] REN H, SHI X, TAO L, et al. Evaluation of two decellularization methods in the development of a whole-organ decellularized rat liver scaffold. Liver Int.2013;33(3):448-458. [42] CEBOTARI S, TUDORACHE I, JAEKEL T, et al.Detergent decellularization of heart valves for tissue engineering: toxicological effects of residual detergents on human endothelial cells. Artif Organs. 2010;34(3):206-210. [43] CERQUEIRA MT, MARQUES AP, REIS RL. Using stem cells in skin regeneration: possibilities and reality. Stem Cells Dev. 2012;21(8): 1201-1214. [44] POON CJ, PEREIRA E COTTA MV, SINHA S, et al. Preparation of an adipogenic hydrogel from subcutaneous adipose tissue.Acta Biomater. 2013;9(3):5609-5620. [45] ADAM YOUNG D, BAJAJ V, CHRISTMAN KL. Award winner for outstanding research in the PhD category, 2014 Society for Biomaterials annual meeting and exposition, Denver, Colorado, April 16-19, 2014: Decellularized adipose matrix hydrogels stimulate in vivo neovascularization and adipose formation. J Biomed Mater Res A. 2014;102(6):1641-1651. [46] HAN TT, TOUTOUNJI S, AMSDEN BG, et al. Adipose-derived stromal cells mediate in vivo adipogenesis, angiogenesis and inflammation in decellularized adipose tissue bioscaffolds.Biomaterials.2015; pii: S0142-9612(15)00735-8. [47] BAJEK A, GURTOWSKA N, OLKOWSKA J, et al. Adipose-Derived Stem Cells as a Tool in Cell-Based Therapies, Arch Immunol Ther Exp (Warsz).2016;64(6):443-454. [48] CHOI JS, YANG HJ, KIM BS, et al. Human extracellular matrix (ECM) powders for injectable cell delivery and adipose tissue engineering.J Control Release.2009;139(1):2-7. [49] TURNER AE, YU C, BIANCO J, et al. The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells. Biomaterials. 2012;33(18):4490-4499. [50] TING AC, CRAFT RO, PALMER JA, et al.The adipogenic potential of various extracellular matrices under the influence of an angiogenic growth factor combination in a mouse tissue engineering chamber. Acta Biomater.2014;10(5):1907-1918. [51] LU Q, LI M, ZOU Y, et al. Delivery of basic fibroblast growth factors from heparinized decellularized adipose tissue stimulates potent de novo adipogenesis.J Control Release.2014;174:43-50. [52] ZHANG S, LU Q, CAO T, et al. Adipose Tissue and Extracellular Matrix Development by Injectable Decellularized Adipose Matrix Loaded with Basic Fibroblast Growth Factor. Plast Reconstr Surg.2016; 137(4): 1171-1180. [53] ZENG F, HARRIS RC.Epidermal growth factor,from gene organization to bedside.Semin Cell Dev Biol.2017;28:2-11. [54] BERNARDES VF, GLEBER-NETTO FO, SOUSA SF, et al, EGF insaliva and tumor samples of oral squamous cell carcinoma.Appl Immunohistochem Mol Morphol.2017;19(6):528-533. [55] ZHOU X, WANG J, HUANG X, et al. Injectable decellularized nucleus pulposus-based cell delivery system for differentiation of adipose-derived stem cells and nucleus pulposus regeneration.Acta Biomater. 2018;81: 115-128. [56] WU I, NAHAS Z, KIMMERLING KA, et al. An injectable adipose matrix for soft-tissue reconstruction. Plast Reconstr Surg. 2012;129(6):1247-1257. [57] LI S, POCHE JN, LIU Y, et al. Hybrid Synthetic-Biological Hydrogel System for Adipose Tissue Regeneration. Macromol Biosci. 2018; 18(11):e1800122. [58] MAUNEY JR, NGUYEN T, GILLEN K, et al.Engineering adipose-like tissue in vitro and in vivo utilizing human bone marrow and adipose-derived mesenchymal stem cells with silk fibroin 3D scaffolds. Biomaterials.2007;28(35):5280-5290. [59] KANG JH, GIMBLE JM, KAPLAN DL. In vitro 3D model for human vascularized adipose tissue. Tissue Eng Part A.2009;15(8):2227-2236. [60] KAYABOLEN A, KESKIN D, AYKAN A, et al. Native extracellular matrix/fibroin hydrogels for adipose tissue engineering with enhanced vascularization. Biomed Mater.2017;12(3):035007. [61] ZHAO Y, FAN J, BAI S. Biocompatibility of injectable hydrogel from decellularized human adipose tissue in vitro and in vivo.J Biomed Mater Res B Appl Biomater. 2019;107(5):1684-1694. [62] MORI S, KIUCHI S, OUCHI A, et al. Characteristic expression of extracellular matrix in subcutaneous adipose tissue development and adipogenesis; comparison with visceral adipose tissue." Int J Biol Sci. 2014;10(8):825-833. |
| [1] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [2] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [3] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [4] | Dai Min, Wang Shuai, Zhang Nini, Huang Guilin, Yu Limei, Hu Xiaohua, Yi Jie, Yao Li, Zhang Ligang. Biological characteristics of hypoxic preconditioned human amniotic mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3004-3008. |
| [5] | Fan Feiyan, Zhang Yunke. Effect and mechanism of traditional Chinese medicine for replenishing qi and promoting blood combined with bone marrow mesenchymal stem cells in promoting angiogenesis in ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2060-2069. |
| [6] | Tao Guilu, Chu Tongbin, Zhang Lei. Proliferation and migration of endothelial progenitor cells promoted by bone marrow mesenchymal stem cells conditioned medium with rosiglitazone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 56-60. |
| [7] | Yuan Yiming, Wang Yan, Chen Chengcheng, Zhao Mingyue, Pei Fei. Efficacy of exosomes in peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 5079-5084. |
| [8] |
Geng Kang, Ding Xiaobin, Tian Xinli, Wang Xue, Yang Yuting, Yan Hong.
Electrical stimulation promotes wound healing and angiogenesis in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4152-4156. |
| [9] |
Xiang Haidong, Cheng Dongmei, Guo Han, Gao Qi .
Changes in the proliferation and angiogenesis of human dental pulp stem cells after treated with prostaglandin E1 combined with basic fibroblast growth factor [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(25): 4006-4011. |
| [10] | Guo Wei, Lu Shan, Fan Hong, Li Jun. Alternative treatment for liver failure repair: current status and development countermeasures [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(20): 3248-3255. |
| [11] | Cao Baichuan, Zeng Gaofeng, Gao Yunbing, Deng Guiying, Cen Zhongxi, Zhang Chuanyang, Guo Yande, Zong Shaohui. Heat-shock endothelial cells induce differentiation of bone marrow mesenchymal stem cells into vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3023-3028. |
| [12] | Li Junyan, Fang Hang, Feng Xiaofeng, Cai Daozhang. Expression and role of retinoblastoma RB1-inducible coiled-coil 1 in the subchondral bone of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(17): 2642-2647. |
| [13] | Cao Yang, Hu Ping, Tian Min, Wei Fang, Gu Qing, Lü Hongbin. Effects of 6-phosphofructokinase-2/fructose-2,6-bisphosphatase 3 on tubule formation of human umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(14): 2217-2222. |
| [14] | Chen Ting, Li Xinzhu, Xu Wenan. Role of angiogenesis in dental pulp regeneration: exosomes and angiogenic factors#br# [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(14): 2263-2270. |
| [15] | Sun An, Bi Xiangyu, Han Xiangzhen, Zhou Qiqi, He Huiyu. Effects of vascular endothelial growth factor combined with platelet-derived growth factor-BB on the angiogenic and proliferative abilities of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(1): 1-6. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||