Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (31): 4986-4993.doi: 10.3969/j.issn.2095-4344.2141
Previous Articles Next Articles
Pretreatment of unrelated umbilical cord blood transplantation without antithymocyte globulin for the treatment of acute myeloid leukemia and acute lymphoblastic leukemia: follow-up evaluation of 306 cases
Zhang Xuhan1, Wang Li2, Tang Baolin1, Wan Xiang1, Yao Wen1, Song Kaidi1, Sun Zimin1
- 1Department of Hematology, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China; 2Department of Rehabilitation Medicine, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230036, Anhui Province, China
-
Received:2020-02-29Revised:2020-03-06Accepted:2020-04-03Online:2020-11-08Published:2020-09-03 -
Contact:Sun Zimin, Chief physician, Professor, Department of Hematology, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China -
About author:Zhang Xuhan, Master, Attending physician, Department of Hematology, The First Affiliated Hospital of USCT, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei 230001, Anhui Province, China -
Supported by:the National Natural Science Foundation of Chin, No. 81470350
CLC Number:
Cite this article
Zhang Xuhan, Wang Li, Tang Baolin, Wan Xiang, Yao Wen, Song Kaidi, Sun Zimin. Pretreatment of unrelated umbilical cord blood transplantation without antithymocyte globulin for the treatment of acute myeloid leukemia and acute lymphoblastic leukemia: follow-up evaluation of 306 cases[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(31): 4986-4993.
share this article

Patient and transplant characteristics Patients and transplant characteristics are summarized in Table 1. Among 306 patients, 112 (36.6%) received transplantation for AML and 194 (63.1%) for ALL. Median age at UCBT was 13.87 years (range, 1-64 years) for patients with AML and 11.89 years (range, 1-50 years) for those with ALL. In the AML group, 65 patients (58%) were transplanted in CR1, 57 (18.7%) in CR2, and 25 (22.3%) in advanced disease status. For the ALL group, 103 patients (53%) were transplanted in CR1, 57 (29.4%) in CR2, and 24 (12.4%) in advanced disease status. Overall, 248 patients (81%) had high-risk disease at diagnosis. Among all patients, 31 (10.1%) received UCB that was 6/6 HLA matched, 152 (49.7%) with a 5/6 HLA match, and 123 (40.2%) with a 4/6 HLA match. A total 118 patients (38.5%) had matched donor ABO compatibility, 101 (33%) had major ABO mismatch, and 87 (28.4%) had minor ABO mismatch. There were 203 patients (66.3% of the entire population, of which 69 had AML and 134 had ALL) who received a conditioning regimen of BUCY2 in combination with fludarabine. Another 103 patients (33.3% of the total population, of which 43 had AML and 60 with ALL) received a conditioning regimen of total body irradiation cyclophosphamide plus cytarabine. For patients with AML, the median total nucleated cell dose and CD34+ cell dose was 4.25 × 107/kg (range: 1.71-17.27 × 107/kg) and 2.18 × 105/kg (range: 0.45-10.55 × 105/kg), respectively. For ALL patients, the median total nucleated cell dose and CD34+ cell dose was 4.22 × 107/kg (range: 1.69-13.5 × 107/kg) and 2.25 × 105/kg (range, 0.4-8.53 × 105/kg), respectively. "

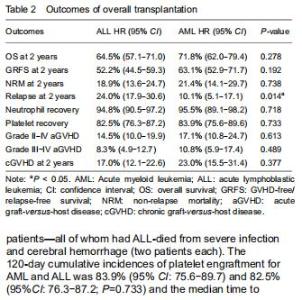
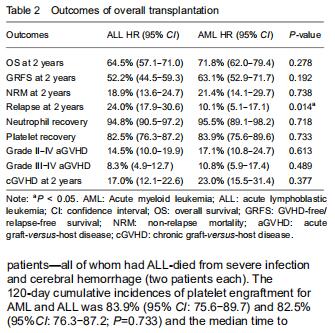
Outcomes of transplantation For the total population of patients with AML and ALL, the engraftment of neutrophil and platelet were similar. The 30-day cumulative incidences of neutrophil engraftment after UCBT in the AML and ALL groups were 95.5% (95% CI: 89.1-98.2) and 94.8% (95% CI: 90.5-97.2), respectively (P=0.718). The median time to neutrophil recovery was 17 (range, 12-30) days and 16 (range, 10-31) days, respectively (Table 2). Fourteen patients did not engraft after UCBT (AML: 5 and ALL: 9); four of those patients—all of whom had ALL-died from severe infection and cerebral hemorrhage (two patients each). The 120-day cumulative incidences of platelet engraftment for AML and ALL was 83.9% (95% CI: 75.6-89.7) and 82.5% (95%CI: 76.3-87.2; P=0.733) and the median time to platelet recovery was 36 (range, 17-121) and 36 (range, 11-196) days, respectively. "
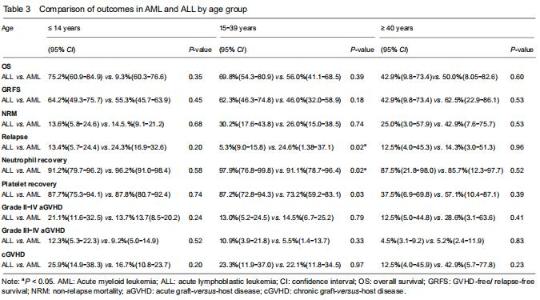
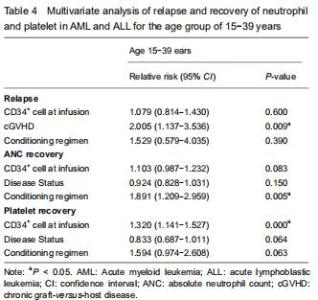
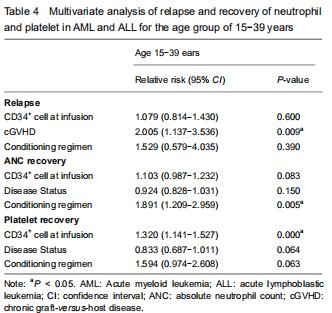
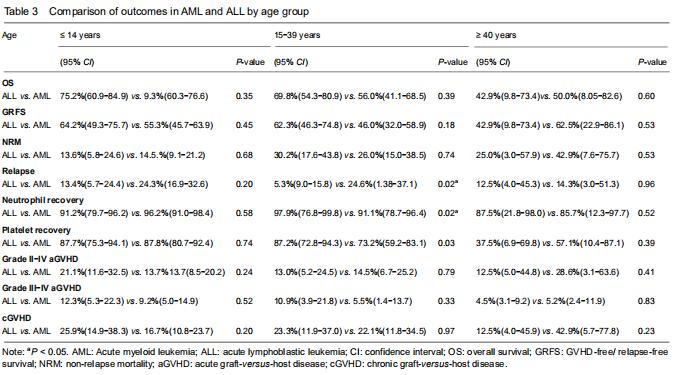
With respect to age groups (≤ 14 years, 15-39 years, ≥ 40 years) (Table 3), adolescents and young adults group had higher engraftment rates for AML than ALL. The 30-day cumulative incidence of neutrophil recovery was higher in patients with AML than in those with ALL (97.9%, 95% CI: 76.8-99.8, vs. 91.1%, 95% CI: 78.7-96.4, P=0.02). In multivariate analysis, CD34+ cell dose and conditioning regimen were independent factors influencing neutrophil recovery. The cumulative incidence of platelet recovery on day 120 was also higher in AML than in ALL (87.2%, 95% CI: 72.8-94.3, vs. 73.2%, 95% CI: 59.2-83.1; P=0.03). In multivariate analysis, CD34+ cell dose was the only independent factor influencing platelet recovery (Table 4). There was no difference between AML and ALL in the groups of children and old adults. "
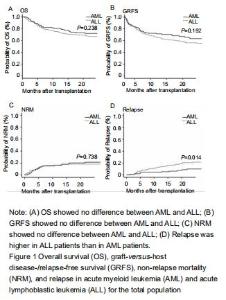
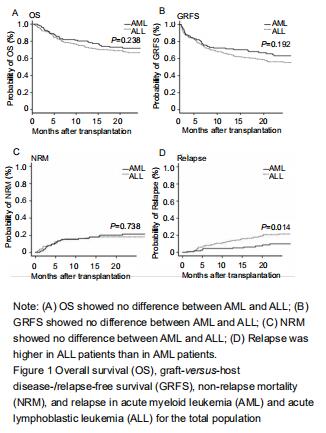
Incidences of acute and chronic GVHD For the total population of patients with AML and ALL, the cumulative incidences of acute GVHD (grade II-IV and grade III-IV) at 100 days were similar (grades II-IV: AML, 17.1% (95% CI: 10.8-24.7) and ALL, 14.5% (95% CI: 10.8-24.7), P=0.613; grades III-IV: AML, 11% (95% CI: 5.9-17.4) and children, 8% (95% CI: 4.9-12.7), P=0.489). The 2-years cumulative incidences of cGVHD in patients with AML and ALL were also similar (23%, (95% CI: 15.5-31.4) and 17%, (95% CI: 12.1-22.6, P=0.377). With grouping by age, cumulative incidences of aGVHD and cGVHD were still similar between AML and ALL in children, adolescents and young adults, and older adults. Relapse and non-relapse mortality, overall survival, and GRFS Fifty-four patients had relapse after UCBT (11 with AML and 43 with ALL). The cumulative incidence for relapse at 2 years in patients with ALL was higher than with AML (24%, 95% CI: 17.9-30.6, vs. 10.1% 95% CI: 5.1-17.1, P=0.01) (Figure 1). The median time to relapse was 12.7 (range, 1.1-59.6) months among AML and 10 (range, 1.5-34.8) months among ALL. In the analysis of age-group, both cumulative incidence of relapse and median time to relapse were significantly different between AML and ALL in adolescents and young adults group (relapse: 25%, 95% CI: 1.4-37.1, vs. 9%, 95% CI: 5.3-15.8, P=0.02); median time: 20.4 (range, 18.6-59.6) months vs. 8.1 (range, 2.8-17.7) months). There were no differences between AML and ALL in both children group and old adult group. In multivariate analysis, cGVHD was an independent factor influencing relapse. Sixty patients died without relapse, 23 with AML and 37 with ALL. The median time to non-relapse mortality in AML and ALL was 301 (range: 45-1 044) days and 121 (range: 3-1 392) days, respectively. The cumulative incidence of non-relapse mortality at 2 years was 21.4%(95% CI: 14.1-29.7) for AML and 18.1%(95% CI: 13.0-23.9) for ALL (P=0.738). Causes of death included pulmonary infection (n=30), hemorrhage (n=4), multiorgan failure (n=22), and other causes (n=4). There was no significant difference in non-relapse mortality between AML and ALL by age groups. The median follow-up duration was 21.6 (range, 3-80.7) months. Overall survival at 2 years in AML was similar to that in ALL (72%, 95% CI: 62-79.4, vs. 66%, 95% CI: 59.5-70.3, P=0.263). There was also no difference in overall survival between AML and ALL by age groups. The GRFS at 2 years in AML and ALL was 63.1% (95% CI: 52.9-71.7) and 56.2% (95% CI: 48.7-63.0), respectively (P=0.192). GRFS also showed no difference between AML and ALL by age groups. "
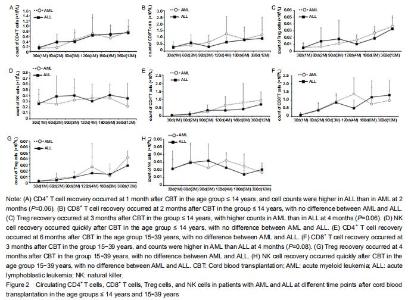
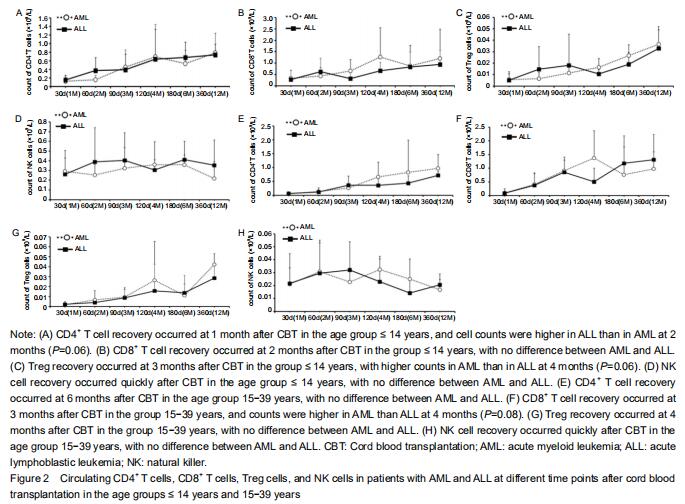
Immune reconstitution Considering the effect of age on the development of the immune system, the immune reconstitution was compared among age groups. In the comparison of outcomes between AML and ALL, we analyzed age groups ≤ 14 years and 15-39 years with respect to CD4+ T cells, CD8+ T cells, natural killer cells, and Treg cells at 1, 2, 3, 4,6, 12, and 18 months after CBT (Figure 2). The results showed that CD4+, CD8+, and Treg cells recovered with time after CBT whereas natural killer cells recovered more quickly (within 1 month) in patients in both age groups. In the group ≤ 14 years, CD4+ T cell counts in AML were lower than in ALL at 2 months after CBT whereas Treg cell counts in AML were higher than in ALL at 4 months post CBT. There was no difference seen in CD8+ and natural killer cells between AML and ALL. The proportion of CD4/CD8 T cells was lowest at 2 months and recovered at 4 months after CBT. In the age group 15-39 years, CD8+ T cell counts in AML at 4 months post CBT were higher than in ALL whereas CD4+, Treg, and natural killer cells showed no difference between AML and ALL. The proportion of CD4+/CD8+ T cells was lowest at 3 months and recovered at 12 months post CBT. "
| [1] TANAKA M, MIYAMURA K, TERAKURA S, et al. Comparison of cord blood transplantation with unrelated bone marrow transplantation in patients older than fifty years. Biol Blood Marrow Transplant. 2015;21(3):517-525. [2] RUGGERI A, LABOPIN M, SANZ G, et al. Comparison of outcomes after unrelated cord blood and unmanipulated haploidentical stem cell transplantation in adults with acute leukemia. Leukemia. 2015;29(9): 1891-900. [3] PAGE KM, LABOPIN M, RUGGERI A, et al. Factors associated with long-term risk of relapse after unrelated cord blood transplantation in children with acute lymphoblastic leukemia in remission. Biol Blood Marrow Transplant. 2017;23(8):1350-1358. [4] DALLE JH, BALDUZZI A, BADER P, et al. Allogeneic stem cell transplantation from HLA-mismatched donors for pediatric patients with acute lymphoblastic leukemia treated according to the 2003 BFM and 2007 International-BFM studies: impact of disease risk on outcomes. Biol Blood Marrow Transplant. 2018;24(9):1848-1855. [5] TANG X, CHEN J, FANG J, et al. Similar outcomes of allogeneic hematopoietic cell transplantation from unrelated donor and umbilical cord blood vs. sibling donor for pediatric acute myeloid leukemia: multicenter experience in China. Pediatr Transplant. 2015;19(4):413-421. [6] RUGGERI A,VOLT F, LOCATELLI F, et al. Unrelated cord blood transplantation for acute leukemia diagnosed in the first year of life:outcomes and risk factor analysis. Biol Blood Marrow Transplant. 2017;23(1):96-102. [7] RAIOLA AM, DOMINIETTO A, DI GRAZIA C, et al. Unmanipulated haploidentical transplants compared with other alternative donors and matched sibling grafts. Biol Blood Marrow Transplant. 2014;20(10): 1573-1579. [8] RUGGERI A, CICERI F, GLUCKMAN E, et al. Alternative donors hematopoietic stem cells transplantation for adults with acute myeloid leukemia:Umbilical cord blood or haploidentical donors? Best Pract Res Clin Haematol. 2010;23(2):207-216. [9] TONG J, XUAN L, SUN Y, et al. Umbilical cord blood transplantation without Antithymocyte Globulin results in similar survival but better quality of life compared with unrelated peripheral blood stem cell transplantation for the treatment of acute leukemia-a retrospective study in China. Biol Blood Marrow Transplant. 2017;23(9):1541-1548. [10] TRAMA A, BOTTA L, FOSCHI R, et al. Survival of European adolescents and young adults diagnosed with cancer in 2000-07:population-based data form EUROCARE-5. Lancet Oncol. 2016;17(7):896-906. [11] ZHENG C, TANG B, ZHU X, et al. Pre-engraftment bloodstream infections in acute leukemia patients undergoing unrelated cord blood transplantation following intensified myeloablative conditioning without ATG. Ann Hematol. 2017;96(1):115-124. [12] SUN Z, LIU H, LUO C, et al. Better outcomes of modified myeloablative conditioning without antithymocyte globulin versus myeloablative conditioning in cord blood transplantation for hematological maliganacies: a retrospective (development) and a prospective (validation) study. Int J Cancer. 2018;143(3):699-708. [13] PRZEPIORKA D, CHAN KW, CHAMPLIN RE, et al. Prevention of graft-versus-host disease with anti-CD5 ricin A chain immunotoxin after CD3-depleted HLA-nonidentical marrow transplantation in pediatric leukemia patients. Bone Marrow Transplant. 1995;16:737-741. [14] FLOWERS ME, KANSU E, SULLIVAN KM. Pathophysiology and treatment of graft-versus-host disease. Hematol Oncol Clin North Am. 1999;13: 1091-1112. [15] HUGUET F, LEGUAY T, RAFFOUX E, et al. Pediatric-inspired therapy in adults with Philadelphia chromosome-negative actute lymphoblastic leukemia: the GRAALLL-2003 study. J Clin Oncol. 2009;27:911-918. [16] DOHNER H, ESTEY E, GRIMWADE D, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129(4):424-447. [17] GRAY RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Stat. 1988,16:1141-1154. [18] KANDA Y. Investigation of the freely available easy-to-use software ‘ESR’ for medical statistics. Bone Marrow Transplant. 2013;48:452-458. [19] BOROWITZ MJ, WOOD BL, DEVIDAS M, et al. Prognostic significance of minimal residual disease in high risk B-ALL: a report from Children’s Oncology Group study AALL0232. Blood. 2015;126:964-971. [20] ESPARZA SD, SAKAMOTO KM. Topics in pediatric leukemia-acute lymphoblastic leukemia. MedGenMed. 2005;7:23. [21] BAKHTIAR S, SALZMANN-MANRIQUE E, Hutter M, et al. AlloHSCT in pediatric ALL and AML in complete remission: improvement over time impacted by accrediatation? Bone Marrow Transplant. 2019;54(5):737-745. [22] ARAKAWA Y, KATO M, KOH K, et al. Unrelated cord blood and bone marrow transplantation in pediatric leukemia. Pediatric Int. 2014;56(4): 647-650. [23] SIEGEL RL, MILLER KD, JEMAL A. Cancer Statistics, 2017. CA Cancer J Clin. 2017;67:7-30. [24] GIANNOTTI F, LABOPIN M, SHOUVAL R, et al. Haploidentical transplantation is associated with better overall survival when compared to single cord blood transplantation: an EBMT-Eurocord study of acute leukemia patients conditioned with thiotepa, busulfan, and fludarabine. J Hematol Oncol. 2018;11(1):110. [25] KONUMA T, TSUKADA N, KANDA J, et al. Comparison of transplant outcomes from matched sibling bone marrow or peripheral blood stem cell and unrelated cord blood in patients 50 years or older. Am J Hematol. 2016, 91(5):E284-E292. [26] LOU X, ZHAO C, CHEN H. Unrelated donor umbilical cord blood transplant versus unrelated hematopoietic stem cell transplant in patients with acute leukemia: a meta-analysis and systematic review. Blood Rev. 2018;32(3): 192-202. [27] LAWITSCHKA A, PETERS C. Long-term effects of myeloablative allogeneic hematopoietic stem cell transplantation in pediatric patients with acute lymphoblastic leukemia. Curr Oncol Rep. 2018;20(9):74. [28] NISHIMOTO T, SATOH T, TAKEUCHI T, et al. Critical role of CD4(+)CD25(+) regulatory T cells in preventing murine autoantibody-mediated thrombocytopenia. Exp Hematol. 2012;40:279-289. [29] AKAHOSHI Y, KANDA J, GOMYO A, et al. Risk factors and impact of secondary failure of platelet recovery after allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2016;22(9):1678-1683. [30] WARLICK ED, PEFFAULT DE LATOUR R, SHANLEY R, et al. Allogeneic hematopoietic cell transplantation outcomes in acute myeloid leukemia: similar outcomes regardless of donor type. Bio Blood Marrow Transplant. 2015;21(2):357-363. [31] YANADA M, KUROSAWA S, YAMAGUCHI T, et al. Effect of related donor availability on outcome of AML in the context of related and unrelated hematopoietic cell transplantation. Bone Marrow Transplant. 2013;48: 390-395. [32] JINDRA P, MUZIK J, INDRAK K, et al. The outcome of allogeneic HSCT in older AML patients is determined by disease biology and not by the donor type: an analysis of 96 allografted AML patients ≥50 years from the Czech acute leukaemia clinical register (alert). Neoplasma. 2013;60: 576-583. [33] MO XD, TANG BL, ZHANG XH, et al. Comparison of outcomes after umbilical cord blood and unmanipulated haploidentical hematopoietic stem cell transplantation in children with high-risk acute lymphoblastic leukemia. Int J Cancer. 2016;139(9):2106-2115. [34] BENADIBA J, ANSARI M, KRAJINOVIC M, et al. Pharmacokinetics-adapted Busulfan-based myeloablative conditioning before unrelated umbilical cord blood transplantation for myeloid malignancies in children. PLoS One. 2018;13(4):e0193862. [35] RANTI J, KURKI S, SALMENNIEMI U, et al. Early CD8+-recovery independently predicts low probability of disease relapse but also associates with severe GVHD after allogeneic HSCT. PLoS One. 2018; 13(9):e0204136. [36] HUTTUNEN P, TASKINEN M, SIITONEN S, et al. Saarinen-Pihkala UM. Impact of very early CD4(+)/CD8(+) T cell counts on the occurrence of acute graft-versus-host disease and NK cell counts on outcome after pediatric allogeneic hematopoietic stem cell transplantation. Pediatr Blood Cancer. 2015;62(3):522-528. [37] HILLHOUSE EE, THIANT S, MOUTUOU MM, et al. Double negative T cells levels correlate chronic graft-versus-host-disease severity. Biol Blood Marrow Transplant. 2019;25(1):19-25. [38] CUPIT MC, DUNCAN C, SAVANI BN, et al. Childhood to adult transition and long-term follow-up after blood and marrow transplantation. Bone Marrow Transplant. 2016;51(2):176-181. [39] WOOD WA, LEE SJ. Malignant hematologic diseases in adolescents and young adults. Blood. 2011;117:5803-5815. [40] NSIR SS, GIRI S, NUNNERY S, et al. Outcomes of adolescents and young adults compared with pediatric patients with acute myeloid and promyelocytic leukemia. Clin Lymphoma Myeloma Leuk. 2017;17:126-132. [41] PULEWKA K, WOLFF D, HERZBERG PY, et al. Physical and psychosocial aspects of adolescent and young adults after allogeneic hematopoietic stem cell transplantation: results form a prospective multicenter trial. J Cancer Res Clin Oncol. 2017;143(8):1613-1619. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [5] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [6] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [7] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [8] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [9] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [10] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [11] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [12] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [13] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [14] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [15] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||