Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (19): 3071-3077.doi: 10.3969/j.issn.2095-4344.2068
Previous Articles Next Articles
Advances in stem cells research concerning tissue-engineered meniscus
Qing Wanyi, Wang Xingxing, Zhou Shengliang, Fu Weili
- West China Medical School/West China Hospital of Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2019-08-24Revised:2019-08-28Accepted:2019-10-09Online:2020-07-08Published:2020-04-09 -
Contact:Fu Weili, MD, Associate professor, West China Medical School/West China Hospital of Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Qing Wanyi, West China Medical School/West China Hospital of Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81972123 and 31741046; the Fundamental Research Funds for the Central Universities, No. 2015SCU04A40; the Innovative Spark Project of Sichuan University, No. 2018SCUH0034; 1·3·5 Project for Disciplines of Excellence of West China Hospital of Sichuan University, No. 2019HXFH039; Research Project for Sichuan Provincial Health and Family Planning Commission, No. 18ZD017
CLC Number:
Cite this article
Qing Wanyi, Wang Xingxing, Zhou Shengliang, Fu Weili. Advances in stem cells research concerning tissue-engineered meniscus[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3071-3077.
share this article
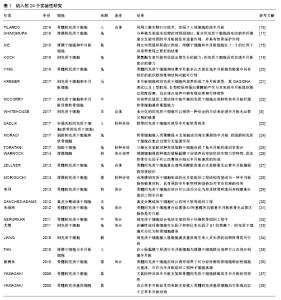
2.1 干细胞在组织工程半月板中的作用 在组织工程应用中,为了寻求最合适的种子细胞,学者们历年来研究过不同的细胞,例如干细胞、滑膜细胞、软骨细胞、人类半月板细胞等[16-38]。干细胞在其中是一个非常重要的角色,可以分泌组织工程必要的细胞因子、生长因子,且有很高的增殖能力和全能性,这些特性让干细胞更适合构建组织工程半月板[39]。一些实验有力地论证了干细胞能促进半月板修复[5,25]、诱导软骨再生[23]。研究人员还在前期进行了一些体外实验,证明了干细胞能在体外诱导分化为软骨细胞,成功构建组织工程半月板[29,31,33,36]。 干细胞能分泌促进内皮生长的递质,刺激祖细胞的自我增殖,抑制软骨细胞凋亡以及软骨退变,实现软骨再生和软骨保护[40]。此外,干细胞还可以通过分化和再生组织并产生细胞因子、生长因子,在修复半月板损伤方面发挥重要作用[41],见表1。 "


2.2 干细胞的分类 干细胞具有多向分化的特点,可分为胚胎干细胞和成体干细胞。成体干细胞例如骨髓间充质干细胞和脂肪来源干细胞是目前研究的热点[42]。干细胞来源广泛[5,19-20,22-23,27],骨髓、脂肪、胎盘、脐血、滑膜、外周血、肌腱、软骨等来源均可获得间充质干细胞[40]。骨髓间充质干细胞、脂肪来源干细胞、滑膜间充质干细胞、外周血源间充质干细胞等都已进行了临床、动物实验。间充质干细胞具有对多种间充质组织的多谱系可塑性、潜在的免疫调节和抗炎特性。其他来源的干细胞还包括滑膜干细胞[18]、真皮分离的成体干细胞[30]、脂肪来源的干细胞等[24-25]。另外,成人真皮分离的干细胞在软骨组织工程中应用良好,并且能够增殖分化成为其他间充质组织细胞[30]。 2.3 干细胞的来源 为了评估半月板维持软骨的稳定性,并确定半月板切除后骨关节炎发生情况,研究者制备了不同的半月板切除动物模型,包括兔[19-20,24-25,27]、猪[28]、大鼠[18,37-38]、羊[5]、牛[22,32]、马[21]、狗等[26,29],其中一些实验采用了灵长类动物猴及人体模型[23,34-35]。 JIANG等[34]实验应用间充质干细胞负载脱细胞真皮基质修复食蟹猴膝关节软骨缺损模型,其内侧半月板明显再生,20-24周软骨缺损明显改善,关节软骨退变、软骨下硬化减少,结果证明间充质干细胞负载脱细胞真皮基质治疗非人灵长类软骨再生的可行性。这些动物模型可以近似模拟发生在人类的创伤性骨关节炎的发病机制和病理方面,常常通过动物个体间比较或者动物自身双膝关节比较来进行实验。研究人员在做组织移植时通常选用同种异体移植或者自体移植,由于自体组织移植没有免疫排斥反应,成功率较高,比异体组织移植更广泛[43]。 马作为人类疾病的模型,步态被认为最像人类[44]。马站立的股胫关节角度大约是150°,而人类站立的股胫关节角度几乎是180°。膝关节过度伸展可能导致马内侧半月板上角损伤,类似于人类后内侧角过屈的情况。KREMER等[21]比较研究了三维共培养马间充质干细胞和半月板细胞分别种植在3个不同支架的情况,以期优化人类半月板组织工程的仿生学。结果显示半月板细胞和间充质干细胞共培养形成了半月板表型,其GAG/DNA表达以及Ⅰ型胶原、Ⅱ型胶原和蛋白聚糖的产生与本来的半月板组织相似程度很高,且在体外培养中拥有稳定性和可持续性。 人和动物有共性也有差别,差别不仅体现在生理病理,更重要的是心理上的差异。有创伤的人类通常会停止使用受影响的肢体,而处于相同情况的动物通常不会。因此,在动物模型中,疾病进展通常要快得多[41]。人体模型对于临床转化的意义更大,在绝大多数情况下,不能未经人体试验直接将动物实验的结果延伸到人体上。与小型动物模型相比,大型动物模型和人体模型更接近人体状态,因此需多进行大动物模型以填补人体试验和动物实验之间的证据,以推进临床试验及应用。 2.4 细胞共培养 通常来说,选择机体原有的半月板细胞作为种子细胞是一种相容性佳的方式,然而为组织工程获得足够数量半月板细胞很困难。半月板细胞与间充质干细胞共培养可减少半月板细胞的需求量,且能促进间充质干细胞向纤维软骨表型分化,减少过度生长,有利于纤维基质的形成。 研究者尝试共同培养几种细胞作为组织工程的种子细胞[18,21-22,24,35],将干细胞与半月板细胞等混合培养然后移植入动物模型。XIE等[18]证明在1∶3的比例下,滑膜干细胞/半月板细胞共培养体系比两者单独培养或者其他比例有更好的效果。但并不是所有的联合培养都呈现更合适的结果,MORADI等[24]的结论为单独关节软骨细胞种植在壳聚糖组4支架可以成功再生撕裂伤半月板,而脂肪来源间充质干细胞在愈合过程中无明显用处。TAN等[35]的实验证明了半月板细胞与滑膜干细胞在小肠黏膜下层联合培养对于半月板组织工程和再生的优势;与滑膜干细胞共培养产生了细胞存活率更高并能分化为软骨表型的基于半月板细胞的组织结构,该结构表现出了较高的糖胺聚糖、Ⅱ型胶原和Sox9表达,而相对较低的Ⅰ型胶原表达。MCCORRY等[22]证实共培养可作为组织工程中平衡间充质干细胞合成特性和半月板纤维软骨细胞基质重塑能力的一种方法。 共培养的结果不一,可能与实验设计有关,也与不同干细胞来源和动物模型相关,具体情况如何待进一步研究。 2.5 组织工程支架 间充质干细胞可以修复全层软骨缺损,但不能产生足够的生物力学再生组织[34],因此研究者常常配合不同组织工程支架移植于动物模型体内,如聚氨酯支架[19]、丝素半月板多孔支架[20]、聚乙烯醇/壳聚糖支架[24]、透明质酸胶原蛋白为基础的支架等[27],这些支架与种子细胞复合促进了半月板再生。 支架分为天然支架和人工支架两大类。天然支架主要来源于组织衍生基质的天然成分,如胶原蛋白、软骨膜组织和透明质酸。合成支架材料由聚氨酯、聚己内酯、聚乳酸、聚乙醇酸、聚乳酸-聚乳酸等合成高分子材料组成。多孔支架适用于组织工程,孔隙大小及其相互连通性、三维表面环境等有利于促进细胞间的接触[45]。半月板组织可由合成的高分子支架材料或上述所有材料的混合/复合材料替代[46-48]。然而合成材料制成的支架在高排斥、不可预测的降解或肿胀方面存在局限性。合成材料具有易获得性、易加工性、可定制支架性能和结构等特点,在未来的组织工程中会具有更广泛的用途[45]。 目前可供临床使用的支架有胶原蛋白支架、聚氨酯支架、聚氨酯/聚已酸内酯支架、蚕丝支架、聚已酸内酯支架、聚乙烯醇支架、异体脱细胞支架,其中胶原蛋白支架是唯一被FDA批准的无细胞支架[49]。 2.6 细胞因子和外泌体 支架材料[47]、细胞因子等都是半月板组织工程中的重要因素。生长因子是组织/细胞培养中的信号蛋白分子。生长因子与细胞中各自的受体结合,促进细胞分化和增殖、细胞迁移和基质合成[50-51]。不同生长因子治疗半月板均有促进细胞增殖、迁移和排列,增加胶原蛋白和蛋白聚糖的产量,促进无血管区撕裂的愈合以及血管形成的作用。转化生长因子刺激合成特定蛋白聚糖、细胞外基质和Ⅱ型胶原蛋白。碱性成纤维细胞生长因子可促进半月板细胞的增殖和细胞外基质的生成[51]。 外泌体已成为细胞间通讯的关键介质[52]。外泌体被定义为细胞分泌的一种纳米级囊泡结构,含有核酸和蛋白质等多种具有生物活性的成分,可通过多种方式发挥调节作用,在多种类型的细胞中均发现外泌体的存 在[53]。不同细胞分泌的外泌体具有不同的组分,不同生理条件下外泌体的成分亦不相同,因而在机体的生命活动中分别发挥不同的作用,这些差别可以用来分辨其来源和特性[52]。与基于干细胞的应用相比,在再生医学中使用无细胞疗法,如间充质干细胞来源外泌体具有明显的优势:①外泌体的应用解决了与活细胞和增殖细胞移植相关的几个安全问题,包括免疫相容性、致瘤性、栓子形成和感染的传播;②可以在不使用可能有毒的冷冻保存剂的情况下长期储存,而不会降低产品的效力[54-56];③使用间充质干细胞来源分泌体,由于避免了侵入性细胞收集程序[57],因此更经济、更实用。 "

| [1] KAZEMI M, LI LP, SAVARD P, et al. Creep behavior of the intact and meniscectomy knee joints. J Mech Behav Biomed Mater. 2011;4(7):1351-1358. [2] NAIMARK MB, KEGEL G, O'DONNELL T, et al. Knee Function Assessment in Patients With Meniscus Injury: A Preliminary Study of Reproducibility, Response to Treatment, and Correlation With Patient-Reported Questionnaire Outcomes. Orthop J Sports Med. 2014;2(9):2325967114550987. [3] SHIMOMURA K, ROTHRAUFF BB, TUAN RS. Region-Specific Effect of the Decellularized Meniscus Extracellular Matrix on Mesenchymal Stem Cell-Based Meniscus Tissue Engineering. Am J Sports Med. 2017;45(3):604-611. [4] CUCCHIARINI M, MCNULTY AL, MAUCK RL, et al. Advances in combining gene therapy with cell and tissue engineering-based approaches to enhance healing of the meniscus. Osteoarthritis Cartilage. 2016;24(8):1330-1339. [5] WHITEHOUSE MR, HOWELLS NR, PARRY MC, et al. Repair of Torn Avascular Meniscal Cartilage Using Undifferentiated Autologous Mesenchymal Stem Cells: From In Vitro Optimization to a First-in-Human Study. Stem Cells Transl Med. 2017;6(4): 1237-1248. [6] ROMANAZZO S, VEDICHERLA S, MORAN C, et al. Meniscus ECM-functionalised hydrogels containing infrapatellar fat pad-derived stem cells for bioprinting of regionally defined meniscal tissue. J Tissue Eng Regen Med. 2018;12(3): e1826-e1835. [7] QI Y, CHEN G, FENG G. Osteoarthritis prevention and meniscus regeneration induced by transplantation of mesenchymal stem cell sheet in a rat meniscal defect model. Exp Ther Med. 2016; 12(1):95-100. [8] PARK A, BARRERA-RAMIREZ J, RANASINGHE I, et al. Use of Statins to Augment Progenitor Cell Function in Preclinical and Clinical Studies of Regenerative Therapy: a Systematic Review. Stem Cell Rev Rep. 2016;12(3):327-339. [9] WU Y, ZHOU J, BI L, et al. Effects of bone marrow mesenchymal stem cells on the cardiac function and immune system of mice with endotoxemia. Mol Med Rep. 2016;13(6):5317-5325. [10] TARNG YW, HUANG BF, SU FC. A novel recirculating flow-perfusion bioreactor for periosteal chondrogenesis. Int Orthop. 2012;36(4): 863-868. [11] WANG N, GRAD S, STODDART MJ, et al. Particulate cartilage under bioreactor-induced compression and shear. Int Orthop. 2014;38(5): 1105-1111. [12] HUANG H, WANG S, GUI J, et al. A study to identify and characterize the stem/progenitor cell in rabbit meniscus. Cytotechnology. 2016;68(5):2083-2103. [13] VIZOSO FJ, EIRO N, CID S, et al. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int J Mol Sci. 2017;18(9): E1852. [14] QI Y, YANG Z, DING Q, et al. Targeted transplantation of iron oxide-labeled, adipose-derived mesenchymal stem cells in promoting meniscus regeneration following a rabbit massive meniscal defect. Exp Ther Med. 2016;11(2):458-466. [15] BAEK J, SOVANI S, CHOI W, et al. Meniscal Tissue Engineering Using Aligned Collagen Fibrous Scaffolds: Comparison of Different Human Cell Sources. Tissue Eng Part A. 2018;24(1-2): 81-93. [16] FILARDO G, PETRETTA M, CAVALLO C, et al. Patient-specific meniscus prototype based on 3D bioprinting of human cell-laden scaffold. Bone Joint Res. 2019;8(2):101-106. [17] SHIMOMURA K, ROTHRAUFF BB, HART DA, et al. Enhanced repair of meniscal hoop structure injuries using an aligned electrospun nanofibrous scaffold combined with a mesenchymal stem cell-derived tissue engineered construct. Biomaterials. 2019;192:346-354. [18] XIE X, ZHU J, HU X, et al. A co-culture system of rat synovial stem cells and meniscus cells promotes cell proliferation and differentiation as compared to mono-culture. Sci Rep. 2018;8(1): 7693. [19] KOCH M, ACHATZ FP, LANG S, et al. Tissue Engineering of Large Full-Size Meniscus Defects by a Polyurethane Scaffold: Accelerated Regeneration by Mesenchymal Stromal Cells. Stem Cells Int. 2018;2018:8207071. [20] YING XZ, QIAN JJ, PENG L, et al. Model research on repairing meniscus injury in rabbits using bone marrow mesenchymal stem cells and silk fibroin meniscus porous scaffold. Eur Rev Med Pharmacol Sci. 2018;22(12):3689-3693. [21] KREMER A, RIBITSCH I, REBOREDO J, et al. Three-Dimensional Coculture of Meniscal Cells and Mesenchymal Stem Cells in Collagen Type I Hydrogel on a Small Intestinal Matrix-A Pilot Study Toward Equine Meniscus Tissue Engineering. Tissue Eng Part A. 2017;23(9-10):390-402. [22] MCCORRY MC, BONASSAR LJ. Fiber development and matrix production in tissue-engineered menisci using bovine mesenchymal stem cells and fibrochondrocytes. Connect Tissue Res. 2017;58(3-4):329-341. [23] SADLIK B, JAROSLAWSKI G, GLADYSZ D, et al. Knee Cartilage Regeneration with Umbilical Cord Mesenchymal Stem Cells Embedded in Collagen Scaffold Using Dry Arthroscopy Technique. Adv Exp Med Biol. 2017;1020:113-122. [24] MORADI L, VASEI M, DEHGHAN MM, et al. Regeneration of meniscus tissue using adipose mesenchymal stem cells-chondrocytes co-culture on a hybrid scaffold: In vivo study. Biomaterials. 2017;126:18-30. [25] TORATANI T, NAKASE J, NUMATA H, et al. Scaffold-Free Tissue-Engineered Allogenic Adipose-Derived Stem Cells Promote Meniscus Healing. Arthroscopy. 2017;33(2):346-354. [26] WARNOCK JJ, SPINA J, BOBE G, et al. Culture of canine synoviocytes on porcine intestinal submucosa scaffolds as a strategy for meniscal tissue engineering for treatment of meniscal injury in dogs. Vet J. 2014;199(1):49-56. [27] ZELLNER J, HIERL K, MUELLER M, et al. Stem cell-based tissue-engineering for treatment of meniscal tears in the avascular zone. J Biomed Mater Res B Appl Biomater. 2013;101(7): 1133-1142. [28] MORIGUCHI Y, TATEISHI K, ANDO W, et al. Repair of meniscal lesions using a scaffold-free tissue-engineered construct derived from allogenic synovial MSCs in a miniature swine model. Biomaterials. 2013;34(9):2185-2193. [29] 李丹,周广东,刘浥,等.骨髓间充质干细胞体外构建组织工程化半月板[J]. 组织工程与重建外科杂志,2013,9(5):251-255,278. [30] SANCHEZ-ADAMS J, ATHANASIOU KA. Dermis isolated adult stem cells for cartilage tissue engineering. Biomaterials. 2012; 33(1):109-119. [31] 朱现玮,徐卫袁,张兴祥,等.多孔型丝素蛋白/羟基磷灰石复合骨髓间充质干细胞修复兔半月板无血运区软骨损伤[J].中国组织工程研究, 2012,16(29): 5375-5378. [32] NERURKAR NL, HAN W, MAUCK RL, et al. Homologous structure-function relationships between native fibrocartilage and tissue engineered from MSC-seeded nanofibrous scaffolds. Biomaterials. 2011;32(2):461-468. [33] 尤微,熊建义,曹清丽,等.骨髓间充质干细胞构建组织工程半月板软骨种子细胞[J].海南医学,2011,22(14): 4-7. [34] JIANG L, MA A, SONG L, et al. The combination of autologous mesenchymal stem cells, acellular dermal matrix and growth factors for cartilage regeneration in experimental cartilage defect model in nonhuman primates. Transplantation. 2010;90(2s): 466. [35] TAN Y, ZHANG Y, PEI M. Meniscus reconstruction through coculturing meniscus cells with synovium-derived stem cells on small intestine submucosa--a pilot study to engineer meniscus tissue constructs. Tissue Eng Part A. 2010;16(1):67-79. [36] 蔡贵泉,崔一民,陈晓东.骨髓间充质干细胞向纤维软骨细胞表型的诱导分化[J].中国组织工程研究与临床康复,2010,14(2):218-222. [37] YAMASAKI T, DEIE M, SHINOMIYA R, et al. Transplantation of meniscus regenerated by tissue engineering with a scaffold derived from a rat meniscus and mesenchymal stromal cells derived from rat bone marrow. Artif Organs. 2008;32(7):519-524. [38] YAMASAKI T, DEIE M, SHINOMIYA R, et al. Meniscal regeneration using tissue engineering with a scaffold derived from a rat meniscus and mesenchymal stromal cells derived from rat bone marrow. J Biomed Mater Res A. 2005;75(1):23-30. [39] PILLAI MM, GOPINATHAN J, SELVAKUMAR R, et al. Human Knee Meniscus Regeneration Strategies: a Review on Recent Advances. Curr Osteoporos Rep. 2018;16(3):224-235. [40] WANG M, YUAN Z, MA N, et al. Advances and Prospects in Stem Cells for Cartilage Regeneration. Stem Cells Int. 2017;2017: 4130607. [41] DEPONTI D, DI GIANCAMILLO A, SCOTTI C, et al. Animal models for meniscus repair and regeneration. J Tissue Eng Regen Med. 2015;9(5):512-527. [42] NIU W, GUO W, HAN S, et al. Cell-Based Strategies for Meniscus Tissue Engineering. Stem Cells Int. 2016;2016:4717184. [43] ZHANG Z, GUO W, GAO S, et al. Native tissue-based strategies for meniscus repair and regeneration. Cell Tissue Res. 2018;373(2): 337-350. [44] MADRY H, OCHI M, CUCCHIARINI M, et al. Large animal models in experimental knee sports surgery: focus on clinical translation. J Exp Orthop. 2015;2(1):9. [45] VADODARIA K, KULKARNI A, SANTHINI E, et al. Materials and structures used in meniscus repair and regeneration: a review. Biomedicine (Taipei). 2019;9(1):2. [46] BULGHERONI P, MURENA L, RATTI C, et al. Follow-up of collagen meniscus implant patients: clinical, radiological, and magnetic resonance imaging results at 5 years. Knee. 2010; 17(3):224-229. [47] MANDAL BB, PARK SH, GIL ES, et al. Multilayered silk scaffolds for meniscus tissue engineering. Biomaterials. 2011;32(2): 639-651. [48] SCOTTI C, HIRSCHMANN MT, ANTINOLFI P, et al. Meniscus repair and regeneration: review on current methods and research potential. Eur Cell Mater. 2013;26:150-170. [49] BILGEN B, JAYASURIYA CT, OWENS BD. Current Concepts in Meniscus Tissue Engineering and Repair. Adv Healthc Mater. 2018;7(11):e1701407. [50] ELI N, ORAGUI E, KHAN W. Advances in meniscal tissue engineering. Ortop Traumatol Rehabil. 2011;13(4):319-326. [51] IONESCU LC, LEE GC, HUANG KL, et al. Growth factor supplementation improves native and engineered meniscus repair in vitro. Acta Biomater. 2012;8(10):3687-3694. [52] CHEW E, PRAKASH R, KHAN W. Mesenchymal stem cells in human meniscal regeneration: A systematic review. Ann Med Surg (Lond). 2017;24:3-7. [53] BEER L, MILDNER M, ANKERSMIT HJ. Cell secretome based drug substances in regenerative medicine: when regulatory affairs meet basic science. Ann Transl Med. 2017;5(7):170. [54] EIRÓ N, SENDON-LAGO J, SEOANE S, et al. Potential therapeutic effect of the secretome from human uterine cervical stem cells against both cancer and stromal cells compared with adipose tissue stem cells. Oncotarget. 2014;5(21):10692-10708. [55] BERMUDEZ MA, SENDON-LAGO J, EIRO N, et al. Corneal epithelial wound healing and bactericidal effect of conditioned medium from human uterine cervical stem cells. Invest Ophthalmol Vis Sci. 2015;56(2):983-992. [56] BERMUDEZ MA, SENDON-LAGO J, SEOANE S, et al. Anti-inflammatory effect of conditioned medium from human uterine cervical stem cells in uveitis. Exp Eye Res. 2016;149: 84-92. [57] OSUGI M, KATAGIRI W, YOSHIMI R, et al. Conditioned media from mesenchymal stem cells enhanced bone regeneration in rat calvarial bone defects. Tissue Eng Part A. 2012;18(13-14): 1479-1489. [58] KWON H, BROWN WE, LEE CA, et al. Surgical and tissue engineering strategies for articular cartilage and meniscus repair. Nat Rev Rheumatol. 2019;15(9):550-570. [59] GUO W, XU W, WANG Z, et al. Cell-Free Strategies for Repair and Regeneration of Meniscus Injuries through the Recruitment of Endogenous Stem/Progenitor Cells. Stem Cells Int. 2018;2018: 5310471. [60] DERKUS B, EMREGUL KC, EMREGUL E. A new approach in stem cell research-Exosomes: Their mechanism of action via cellular pathways. Cell Biol Int. 2017;41(5):466-475. [61] ZHANG B, WU X, ZHANG X, et al. Human umbilical cord mesenchymal stem cell exosomes enhance angiogenesis through the Wnt4/β-catenin pathway. Stem Cells Transl Med. 2015;4(5): 513-522. [62] BAGLIO SR, ROOIJERS K, KOPPERS-LALIC D, et al. Human bone marrow- and adipose-mesenchymal stem cells secrete exosomes enriched in distinctive miRNA and tRNA species. Stem Cell Res Ther. 2015;6:127. [63] HOFER HR, TUAN RS. Secreted trophic factors of mesenchymal stem cells support neurovascular and musculoskeletal therapies. Stem Cell Res Ther. 2016;7(1):131. [64] WANG Y, YU D, LIU Z, et al. Exosomes from embryonic mesenchymal stem cells alleviate osteoarthritis through balancing synthesis and degradation of cartilage extracellular matrix. Stem Cell Res Ther. 2017;8(1):189. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||