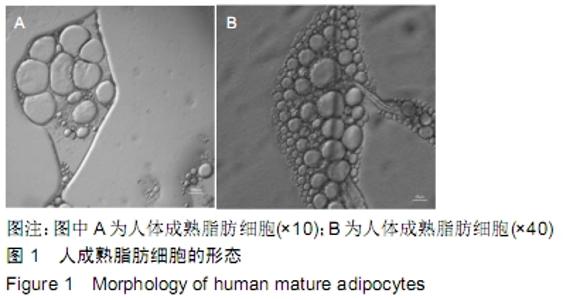
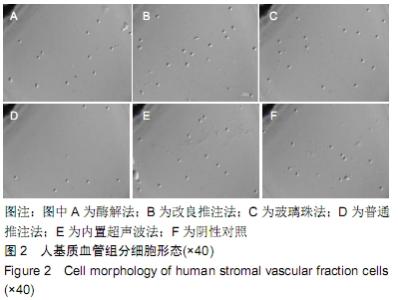
Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (13): 1976-1982.doi: 10.3969/j.issn.2095-4344.2045
Previous Articles Next Articles
Separation of adipose-derived stromal vascular fraction cells by a variety of physical methods: a comparative study
Lu Peiling1, Feng Chunchao1, Liang Miaomiao1, Wei Jiatian1, Huang Jing1, Cai Haiming1, Wu Guohui2, Zhang Linghua1, Nie Yunfei2
- 1Guangdong Provincial Key Laboratory of Protein Function and Regulation in Agricultural Organisms, College of Life Sciences, South China Agricultural University, Guangzhou 510642, Guangdong Province, China; 2Guangzhou Plastic Surgery Hospital of Chinese Family Physician, Guangzhou 510011, Guangdong Province, China
-
Received:2019-05-29Revised:2019-05-30Accepted:2019-07-15Online:2020-05-08Published:2020-03-07 -
Contact:Nie Yunfei, Master, Physician, Guangzhou Plastic Surgery Hospital of Chinese Family Physician, Guangzhou 510011, Guangdong Province, China Zhang Linghua, MD, Professor, Guangdong Provincial Key Laboratory of Protein Function and Regulation in Agricultural Organisms, College of Life Sciences, South China Agricultural University, Guangzhou 510642, Guangdong Province, China -
About author:Lu Peiling, Guangdong Provincial Key Laboratory of Protein Function and Regulation in Agricultural Organisms, College of Life Sciences, South China Agricultural University, Guangzhou 510642, Guangdong Province, China Feng Chunchao, Master candidate, Guangdong Provincial Key Laboratory of Protein Function and Regulation in Agricultural Organisms, College of Life Sciences, South China Agricultural University, Guangzhou 510642, Guangdong Province, China Lu Peiling and Feng Chunchao contributed equally this work. -
Supported by:the National Innovation and Entrepreneurship Training Program for College Students of China, No. 201810564021; the Natural Science Foundation of Guangdong Province, No. 2018A030313625
CLC Number:
Cite this article
Lu Peiling, Feng Chunchao, Liang Miaomiao, Wei Jiatian, Huang Jing, Cai Haiming, Wu Guohui, Zhang Linghua, Nie Yunfei. Separation of adipose-derived stromal vascular fraction cells by a variety of physical methods: a comparative study[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(13): 1976-1982.
share this article
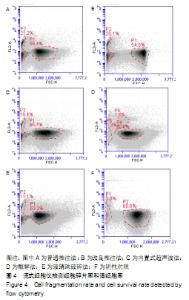
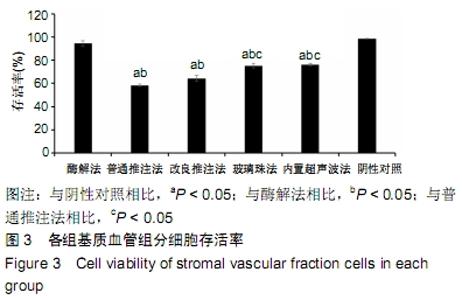
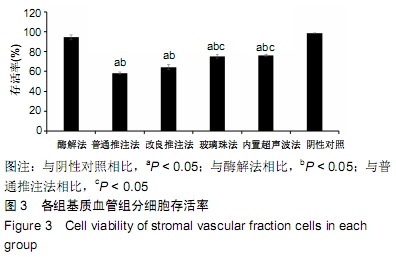
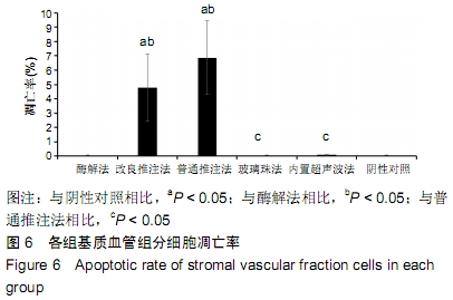
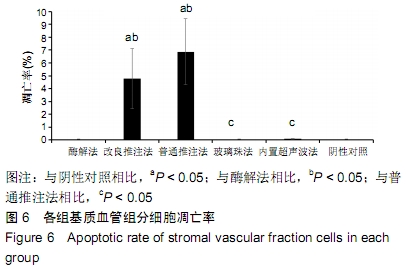
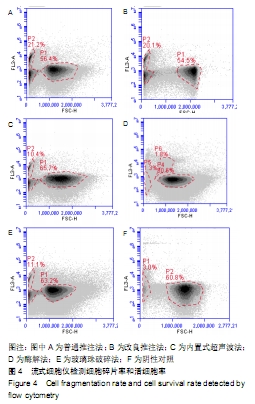
2.3 细胞增殖率 酶解法、改良推注法、普通推注法、玻璃珠法、内置超声波法、阴性对照组的细胞增殖率分别为0.036±0.005,0.021±0.005,0.024±0.004,0.035±0.003,0.032±0.004,0.032±0.003。酶解法因为无机械压力的原因增殖率最高,而玻璃珠法收集到的基质血管组分细胞比酶解法收集到的略低,普通推注法最低。普通推注法和改良推注法与阴性对照及酶解法相比差异有显著性意义(P < 0.05),玻璃珠法和内置超声波法与普通推注法相比差异也有显著性意义(P < 0.05)。 2.4 细胞活性 阴性对照由于没有经过任何处理,细胞量极低,故细胞活性也最低,为0.203±0.018。酶解法由于没有机械压力所以细胞活性最高,为0.959±0.016。玻璃珠法细胞活性为0.868±0.021,内置超声波法细胞活性为0.861±0.025,改良推注法细胞活性为0.651±0.011,普通推注法细胞活性为0.583±0.013,玻璃珠法及内置超声波法的细胞活性相差不大,与酶解法相比略低,但差异无显著性意义(P > 0.05),而改良推注法和普通推注法与酶解法、玻璃珠法、内置超声波法相比细胞活性更低,差异有显著性意义(P < 0.05),5种方法与阴性对照相比差异有非常显著性意义(P < 0.01)。 2.5 细胞碎片率及凋亡率 采用流式细胞仪进行检测,见图4,由结果可以看到酶解法、玻璃珠法、内置超声波法、改良推注法、普通推注法收集到的基质血管组分细胞中碎片比例分别为(2.35±1.48)%,(7.85±4.43)%,(10.63± 4.37)%,(24.49±4.65)%,(35.51±4.23)%,酶解法碎片比例略低于玻璃珠法,但差异无显著性意义(P > 0.05)。酶解法、玻璃珠法及内置超声波法细胞碎片比例要远远低于普通推注法和改良推注法,差异有显著性意义(P < 0.05),见图5。 "
| [1] CHUNG MT, ZIMMERMANN AS, PAIK KJ, et al. Isolation of human adipose-derived stromal cells using laser-assisted liposuction and their therapeutic potential in regenerative medicine. Stem Cells Transl Med. 2013;2(10):808-817. [2] ZUK PA.The adipose-derived stem cell: looking back and looking ahead. Mol Biol Cell. 2010;21(11):1783-1787. [3] ZUK PA, ZHU M, ASHJIAN P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12): 4279-4295. [4] 张仔昂,鲁峰.脂肪组织工程中生物支架材料的研究进展[J].中国美容整形外科志,2018,29(4):249-251,262. [5] PREMARATNE GU, MA LP, FUJITA M, et al. Stromal vascular fraction transplantation as an alternative therapy for ischemic heart failure: anti-inflammatory role. J Cardiothorac Surg. 2011; 6:43. [6] ATALAY S, CORUH A, DENIZ K. Stromal vascular fraction improves deep partial thickness burn wound healing. Burns. 2014;40(7): 1375-1383. [7] 侯崇超,周传德. SVF-gel联合自体颗粒脂肪在外生殖器整形中的临床应用[J].中国美容整形外科杂志, 2019;30(3):179-181,195. [8] 张杰,丁权威,王人彦.脂肪组织中的血管基质成分在骨科中的应用研究进展[J].现代实用医学,2017,29(8):1116-1117. [9] BATEMAN ME, STRONG AL, GIMBLE JM, et al. Concise Review: Using Fat to Fight Disease: A Systematic Review of Nonhomologous Adipose-Derived Stromal/Stem Cell Therapies. Stem Cells. 2018;36(9):1311-1328. [10] BAGLIONI S, FRANCALANCI M, SQUECCO R, et al. Characterization of human adult stem-cell populations isolated from visceral and subcutaneous adipose tissue. FASEB J. 2009; 23(10):3494-3505. [11] CHAU TA, MCCULLY ML, BRINTNELL W, et al. Toll-like receptor 2 ligands on the staphylococcal cell wall downregulate superantigen-induced T cell activation and prevent toxic shock syndrome. Nat Med. 2009;15(6):641-648. [12] ZUK PA, ZHU M, MIZUNO H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211-228. [13] BUNNELL BA, FLAAT M, GAGLIARDI C, et al. Adipose-derived stem cells: isolation, expansion and differentiation. Methods. 2008;45(2): 115-120. [14] MOUW JK, OU G, WEAVER VM. Extracellular matrix assembly: a multiscale deconstruction. Nat Rev Mol Cell Biol. 2014;15(12): 771-785. [15] OSINGA R, MENZI NR, TCHANG LA, et al. Effects of intersyringe processing on adipose tissue and its cellular components: implications in autologous fat grafting. Plast Reconstr Surg. 2015; 135(6):1618-1628. [16] YAO Y, DONG Z, LIAO Y, et al. Adipose Extracellular Matrix/Stromal Vascular Fraction Gel: A Novel Adipose Tissue-Derived Injectable for Stem Cell Therapy. Plast Reconstr Surg. 2017;139(4):867-879. [17] 陈信恺,任婧,袁艺,等.脂肪干细胞细胞在面部容量填充和年轻化中的应用[J].中国美容整形外科杂志,2018,29(9):542-544. [18] BOSCH J, HOUBEN AP, RADKE TF, et al. Distinct differentiation potential of "MSC" derived from cord blood and umbilical cord: are cord-derived cells true mesenchymal stromal cells. Stem Cells Dev. 2012;21(11):1977-1988. [19] MIDWOOD KS, WILLIAMS LV, Schwarzbauer JE. Tissue repair and the dynamics of the extracellular matrix. Int J Biochem Cell Biol. 2004;36(6):1031-1037. [20] SHAH FS, WU X, DIETRICH M, et al. A non-enzymatic method for isolating human adipose tissue-derived stromal stem cells. Cytotherapy. 2013;15(8):979-985. [21] TONNARD P, VERPAELE A, PEETERS G, et al. Nanofat grafting: basic research and clinical applications. Plast Reconstr Surg. 2013;132(4):1017-1026. [22] VAN DONGEN JA, STEVENS HP, HARMSEN MC, et al. Mechanical Micronization of Lipoaspirates: Squeeze and Emulsification Techniques. Plast Reconstr Surg. 2017;139(6): 1369e-1370e. [23] GARDIN C, BRESSAN E, FERRONI L, et al. In vitro concurrent endothelial and osteogenic commitment of adipose-derived stem cells and their genomical analyses through comparative genomic hybridization array: novel strategies to increase the successful engraftment of tissue-engineered bone grafts. Stem Cells Dev. 2012;21(5):767-777. [24] OSINGA R, MENZI NR, TCHANG LA, et al. Effects of intersyringe processing on adipose tissue and its cellular components: implications in autologous fat grafting. Plast Reconstr Surg. 2015; 135(6):1618-1628. [25] ZIMMERLIN L, DONNENBERG VS, PFEIFER ME, et al. Stromal vascular progenitors in adult human adipose tissue. Cytometry A. 2010;77(1):22-30. [26] 柳大烈,王晋煌.医疗美容中的吸脂术[J].中国医疗美容, 2012(3): 57-58. [27] PU LLQ. Fat Grafting for Facial Rejuvenation and Contouring: A Rationalized Approach. Ann Plast Surg. 2018;81(6S Suppl 1): S102-S108. [28] BANAS A. Purification of adipose tissue mesenchymal stem cells and differentiation toward hepatic-like cells. Methods Mol Biol. 2012;826: 61-72. [29] MOUW JK, OU G, WEAVER VM. Extracellular matrix assembly: a multiscale deconstruction. Nat Rev Mol Cell Biol. 2014;15(12): 771-785. [30] 冯春朝,卢沛伶,黄婧,等.改良推注方法制备脂肪移植SVF胶的实验研究[J].中国美容整形外科杂志,2019,30(6):338-341. [31] 魏嘉田,卢沛伶,黄婧,等.不同非接触式超声波处理获得脂肪源基质血管组分细胞方法的比较[J].中国组织工程研究,2019,23(33): 5347-5352. [32] YOSHIMURA K, SHIGEURA T, MATSUMOTO D, et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J Cell Physiol. 2006;208(1):64-76. [33] 宋正海.关于血细胞计数板的解读[J].生物学教学,2018,43(10): 68-69. [34] 袁清波.细胞分类计数方法的研究[D].成都:电子科技大学, 2017. [35] WATANABE M, UEHARA Y, YAMASHITA N, et al. Evaluation of a novel flow cytometry-based system for the detection of circulating tumor cells. Cancer Research. 2012;72(8 Supplement):1735. [36] HORI G, SAREH A, ABBAS SL, et al. Influence of adipose derived mesenchymal stem cells on the effective inflammatory factors of diabetic wound healing in animal models. Journal of Mazandaran University of Medical Sciences. 2017;27(148):12-21. [37] BHAT IA, T B S, SOMAL A, et al. An allogenic therapeutic strategy for canine spinal cord injury using mesenchymal stem cells. J Cell Physiol. 2019;234(3):2705-2718. [38] EBISAWA K, KATO R, OKADA M, et al. Cell therapy for facial anti-aging. Med J Malaysia. 2008;63 Suppl A:41. [39] TABATABAEI QOMI R, SHEYKHHASAN M. Adipose-derived stromal cell in regenerative medicine: A review. World J Stem Cells. 2017;9(8): 107-117. [40] JANUS JR, JACKSON RS, LEES KA, et al. Human Adipose-Derived Mesenchymal Stem Cells for Osseous Rehabilitation of Induced Osteoradionecrosis: A Rodent Model. Otolaryngol Head Neck Surg. 2017;156(4):616-621. [41] HUANG SJ, FU RH, SHYU WC, et al. Adipose-derived stem cells: isolation, characterization, and differentiation potential. Cell Transplant. 2013;22(4):701-709. [42] LIN CS, XIN ZC, DENG CH, et al. Defining adipose tissue-derived stem cells in tissue and in culture. Histol Histopathol. 2010;25(6): 807-815. [43] MELIGA E, STREM BM, DUCKERS HJ, et al. Adipose-derived cells. Cell Transplant. 2007;16(9):963-970. [44] RAMAKRISHNAN VM, BOYD NL. The Adipose Stromal Vascular Fraction as a Complex Cellular Source for Tissue Engineering Applications. Tissue Eng Part B Rev. 2018;24(4):289-299. [45] BORA P, MAJUMDAR AS. Adipose tissue-derived stromal vascular fraction in regenerative medicine: a brief review on biology and translation. Stem Cell Res Ther. 2017;8(1):145. [46] MENZI N, OSINGA R, TODOROV A, et al. Wet milling of large quantities of human excision adipose tissue for the isolation of stromal vascular fraction cells. Cytotechnology. 2018;70(2): 807-817. [47] PRIGLINGER E, SCHUH CMAP, STEFFENHAGEN C, et al. Improvement of adipose tissue-derived cells by low-energy extracorporeal shock wave therapy. Cytotherapy. 2017;19(9): 1079-1095. [48] SCHENDEL SA. Autologous Adipose-Derived Tissue Matrix Part I: Biologic Characteristics. Aesthet Surg J. 2017;37(9):1062-1068. [49] SCHENDEL SA. Autologous Adipose-Derived Tissue Matrix Part II: Implantation Biology. Aesthet Surg J. 2017;37(9):1069-1074. |
| [1] | Liu Zhichao, Zhang Fan, Sun Qi, Kang Xiaole, Yuan Qiaomei, Liu Genzhe, Chen Jiang. Morphology and activity of human nucleus pulposus cells under different hydrostatic pressures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1172-1176. |
| [2] | Ye Dou, , Ma Xuexia , Guan Qian, , Luan Zuo , Yang Yinxiang , Wang Zhaoyan , Wang Qian , He Ying , Yao Ruiqin. Proportion and morphological characteristics of human oligodendrocyte precursor cells in different cell culture vessels [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(1): 44-49. |
| [3] | Qiao Xuesong, Chen Ni, Yang Fengying, Niu Yanmei. Exercises regulate bone marrow adipocytes derived from mesenchymal stem cells: roles and prospects [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(8): 1293-1298. |
| [4] | Han Yanfu, Tao Ran, Sun Tianjun. Autophagy has an early protective effect against fibroblast injury induced by advanced glycation end products [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(35): 5619-5624. |
| [5] | Yin Lingni, Chen Dexuan. The role of PI3K/Akt pathway in hypoxia-induced cell proliferation and endothelial cell differentiation of adipose-derived stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(19): 3004-3009. |
| [6] | Nie Jiaying, Yi Yangyan, Zhu Yuanzheng. Acellular adipose tissue preparation: a soft-tissue filler allowing for allogeneic injection or in situ adipogenesis? [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(11): 1762-1768. |
| [7] | Li Zifei, Wang Qian, Luan Jie, Mu Dali, Liu Chunjun, Xin Minqiang, Fu Su, Xu Boyang, Liu Wenyue, Chen Lin, Cheng Hao, Wang Chenglong, Kang Deni, Li Shangshan, Qi Jun. Ammonium chloride lysis method versus non-lysis method for isolation of human adipose-derived stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(1): 45-50. |
| [8] | Sun Heyuan, Yan Weiqing. Adipose-derived stem cell transplantation for acute kidney injury caused by crush injury [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1364-1369. |
| [9] | Wang Disheng, Kong Liusha, Wang Jia, Li Xia. Erythropoietin preconditioned adipose-derived mesenchymal stem cell transplantation for treating diabetic nephropathy in rats [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1370-1376. |
| [10] | Zhang Deguo, Tan Xueying, Tang Nan, Huang Fei, Shi Guangjun. Human adipose-derived mesenchymal stem cell transplantation via the tail vein for acute liver failure in rats [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1416-1421. |
| [11] | Yin Yating, Zhang Aijun, Wang Hao, Wang Pingping, Li Jianhua, Wen Minmin, Jin Peisheng. Clinical application of autogenous adipose-derived stromal vascular fraction-assisted fat transplantation in the improvement of facial skin quality [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1428-1433. |
| [12] | Huang Kun, Liu Chengli, Han Gonghai, Liu Rui, Chen Weiwei, Ding Xiao, Tang He, Peng Jiang. Repair of liver injury in rats by adipose-derived mesenchymal stem cells combined with porous chitosan microspheres [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(5): 716-722. |
| [13] | Tian Meng, Han Hongling. Utilization of adiponectin-modified adipose stem cell transplantation in the rat with nephrotic syndrome [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(5): 723-728. |
| [14] | Feng Fang, Dong Chenming, Chen Yu, Qi Yan. Adipose tissue-derived mesenchymal stem cell transplantation for treating septic rats with acute respiratory distress syndrome and the underlying mechanism [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(5): 741-748. |
| [15] | Jia Daping, Ma Li, Wu Liangliang, Zhao Yu. Variables affecting the retained volume of adipose tissue after transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(35): 5652-5657. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||