Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (47): 6887-7005.doi: 10.3969/j.issn.2095-4344.2016.47.001
Construction and characteristics of poly(lactic-co-glycolic acid)/calcium silicate scaffolds with three-dimensional pores for bone tissue engineering
- 1School of Material Science and Engineering, South China University of Technology, Guangzhou 510640, Guangdong Province, China; 2National Engineering Research Center for Tissue Restoration and Reconstruction, Guangzhou 510006, Guangdong Province, China; 3Guangdong Provincial Key Laboratory of Biomedical Engineering, Guangzhou 510006, Guangdong Province, China
-
Received:2016-09-01Online:2016-11-18Published:2016-11-18 -
Contact:Wei Kun, Professor, School of Material Science and Engineering, South China University of Technology, Guangzhou 510640, Guangdong Province, China -
About author:Hu Lu, Master, School of Material Science and Engineering, South China University of Technology, Guangzhou 510640, Guangdong Province, China; National Engineering Research Center for Tissue Restoration and Reconstruction, Guangzhou 510006, Guangdong Province, China; Guangdong Provincial Key Laboratory of Biomedical Engineering, Guangzhou 510006, Guangdong Province, China -
Supported by:the Guangdong Provincial Science and Technology Program, No. 2015A020214005
CLC Number:
Cite this article
Hu Lu, Wei Kun, Zou Fen. Construction and characteristics of poly(lactic-co-glycolic acid)/calcium silicate scaffolds with three-dimensional pores for bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(47): 6887-7005.
share this article
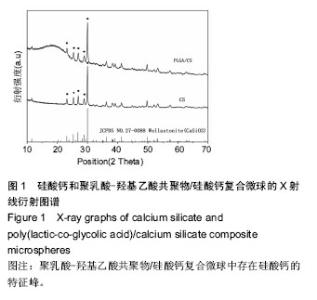
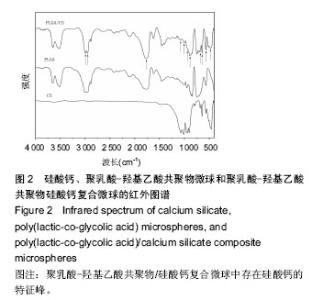
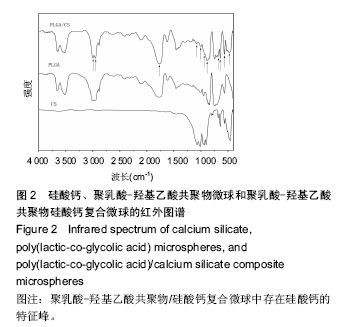
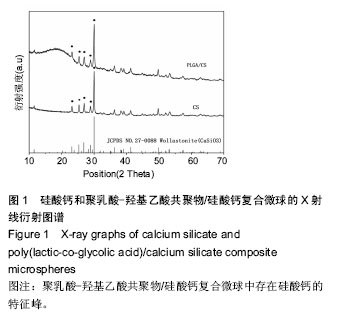
2.1 PLGA/硅酸钙复合微球的组成成分分析 图1中圆点标注的位置依次为(400)、(002)、(-202)、(202)、(320),对照X射线衍射标准JCPDS卡片数据可知,它们是硅酸钙的特征峰,在PLGA/硅酸钙谱图中,硅酸钙的特征峰均存在,说明微球中存在硅酸钙;由图2可以看出,在PLGA/硅酸钙复合微球的谱图中,1 762, 1 170 cm-1附近出现的2个峰分别为酯键和C-O键的特征吸收峰,而3 000,2 956 cm-1附近出现的2个较大的吸收峰,分别为-CH3和-CH2-的伸缩振动吸收,且PLGA的其他特征峰均存在,说明复合微球中含有PLGA;而在1 077,477 cm-1附近出现的2个吸收峰,分别为Si-O-Si键的伸缩振动吸收和弯曲振动吸收[24],1 006,945,891,682,643,567,477 cm-1附近出现的特征吸收峰与β-CaSiO3的谱图基本一致[25-26],说明复合微球中也存在硅酸钙。由于PLGA有机组分的存在,硅酸钙在复合微球中的特征峰没有单一组分的尖锐。"
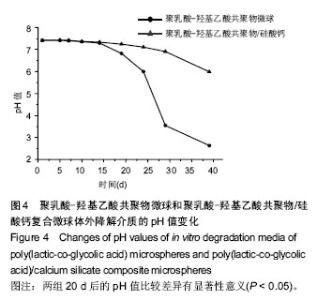
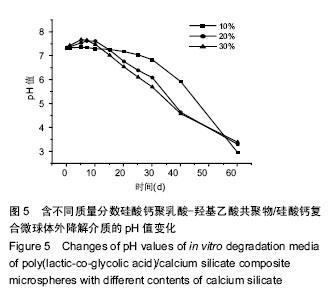
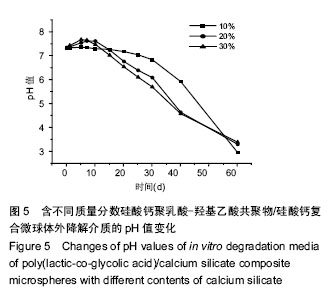
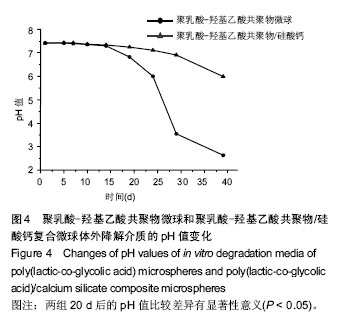
2.3 硅酸钙组分对PLGA降解pH值的影响 图4比较了PLGA微球和PLGA/硅酸钙复合微球的体外降解介质pH值变化情况,降解实验的前15 d,两组微球的pH值基本维持在7.4左右,且相差不大;15 d后,PLGA微球介质的pH值迅速下降,而PLGA/硅酸钙复合微球的下降较为缓慢;在第40天时,PLGA微球介质的pH值下降至2.6左右,而PLGA/硅酸钙复合微球的仍可维持在6左右,结果表明,硅酸钙组分的加入对PLGA的酸性降解产物有一定的中和作用。图5进一步比较了不同含量硅酸钙对PLGA微球体外降解介质pH值的影响,在降解的前15 d内,20%组和30%组的降解介质pH值都有一个明显的升高然后再降低的过程,而10%组基本维持在7.4左右;15 d后,3组pH值都开始缓慢降低,其中10%组下降最为缓慢;到降解的第60天,10%组的pH值降低至3左右,而20%和30%组的pH值比10%略高0.5。说明硅酸钙对PLGA微球体外降解介质pH值的中和作用主要发生在前15 d内。"

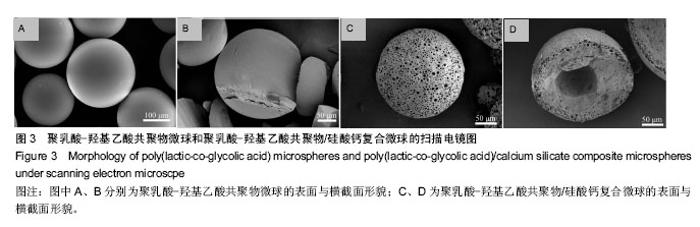
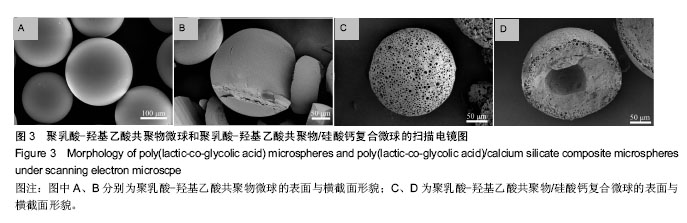
2.4 PLGA三维多孔支架和PLGA/硅酸钙复合多孔支架的结构特征 如图6所示,PLGA/硅酸钙支架的尺寸比PLGA支架略小(图6A);PLGA支架的纤维直径和孔径约为600 μm和800 μm(图6B),而PLGA/硅酸钙支架的纤维直径和支架孔径约为500 μm和750 μm(图6C),均小于PLGA支架,且两种支架的纤维直径均大于所用针头直径0.4 mm,说明在打印过程中出现了纤维挤出胀大现象,低温融合技术制备PLGA/硅酸钙复合多孔支架的过程中,支架尺寸又出现了一定程度的回缩;从支架的扫描电镜图片可以看出,PLGA支架保持良好的三维连通多孔结构(图6D),在PLGA/硅酸钙支架中,微球均匀地分散在支架的孔道之间,同时与支架保持较好的孔隙结构(图6E);在PLGA/硅酸钙支架200倍扫描电镜图中,微球已与支架融合,同时单一微球表面仍良好地保留着多孔形貌(图6F)。"

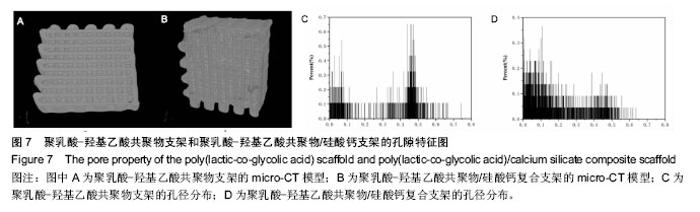
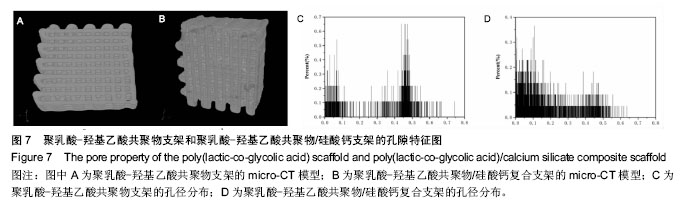
2.5 Micro-CT构建三维多孔支架模型和孔隙特征 利用Micro-CT对支架扫描重建,得到支架的结构模型和孔径分布图(图7),PLGA支架的平均孔隙率、孔隙连通率及平均孔径分别为31.35%、100%、0.32 mm,PLGA/CS支架的平均孔隙率、孔隙连通率及平均孔径分别为24.09%、99.88%、0.17 mm。由图7可知,两支架的孔隙均完全连通,孔隙在支架中分布较均匀;PLGA支架的孔径主要分布在0.4-0.5 mm,同时还出现较多的小于0.1 mm的孔洞(图7C),这部分小孔可能是由于边缘效应造成的;而在PLGA/CS支架中,由于多孔微球的加入,除了0.4-0.5 mm孔洞外,更多的出现了小于0.2 mm的孔洞(图7D);对两支架的孔隙率、孔隙连通率和平均孔径加以比较发现,PLGA/硅酸钙支架的孔隙率和平均孔径均比PLGA支架小,而两支架的孔隙连通率几乎都为100%。以上结果说明,利用低温融合技术将PLGA/硅酸钙复合多孔微球与PLGA三维多孔支架成功组装成具有完全连通的梯度多级孔结构的复合支架。"
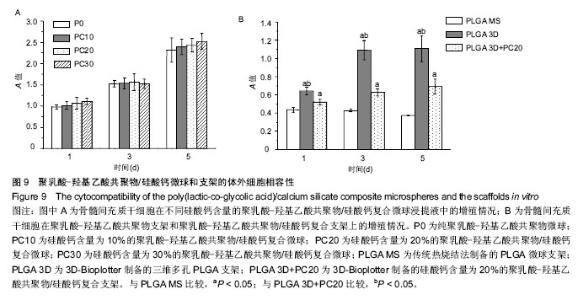
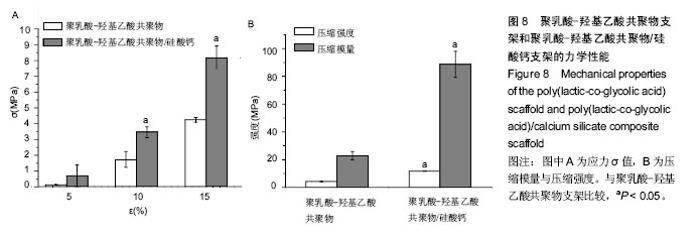
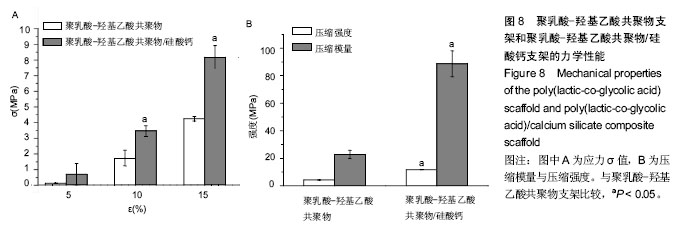
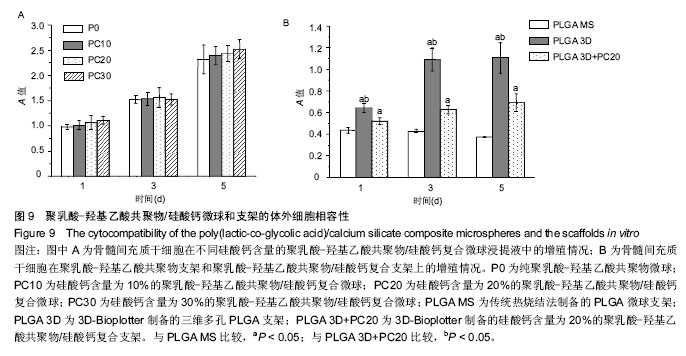
2.7 材料的体外细胞相容性 图9A比较了骨髓间充质干细胞在不同硅酸钙含量PLGA/硅酸钙微球浸提液中的增殖情况,第1-5天,所有组别吸光度一直升高,随着微球中硅酸钙含量的增加,浸提液对细胞增殖率的作用总体呈略微增加趋势,但与不含硅酸钙的PLGA微球比较差异均无显著性意义,说明细胞可以在不同硅酸钙含量的PLGA微球浸提液中正常生长,硅酸钙组分的加入对骨髓间充质干细胞的增殖有一定的促进作用,但效果不显著;图9B为骨髓间充质干细胞在PLGA支架和PLGA/硅酸钙支架上的增殖情况,以传统热烧结法制备的PLGA微球支架作为对照组,由图可知,细胞在3组支架上均可稳定生长,说明3组支架均具有良好的生物相容性;从单个时间点来看,3D-Bioplotter制备PLGA组的细胞数明显高于其他两组,PLGA/硅酸钙组的细胞数也显著高于传统热烧结法制备的PLGA微球支架,说明PLGA三维多孔支架和PLGA/硅酸钙复合多孔支架比传统热烧结法制备的PLGA微球支架更有利于骨髓间充质干细胞的增殖,其原因可能是高的孔隙率、孔隙连通率和较大的孔径更有利于细胞的长入。 "
| [1]Hutmacher DW.Scaffols in tissue engineering bone and cartilage.Biomaterials.2000; 21(24):2529-2543. [2]Gentile P,Chiono V,Carmagnola I,et al.An overview of poly (lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering.Int J Mol Sci.2014;15(3): 3640-3659. [3]Danhier F,Ansorena E,Silva JM,et al.PLGA-based nanoparticles: an overview of biomedical applications.J Control Release.2012;161(2):505-522. [4]Zolnik BS,Burgess DJ.Effect of acidic pH on PLGA microsphere degradation and release. J Control Release. 2007;122(3):338-344. [5]Liu H,Slamovich EB,Webster TJ.Less harmful acidic degradation of poly(lactic-co-glycolic acid) bone tissue engineering scaffolds through titania nanoparticle addition.Int J Nanomedicine.2006;1(4):541-545. [6]Cheng D,Cao X,Gao H,et al.Superficially porous poly (lactic-co-glycolic acid)/calcium carbonate microsphere developed by spontaneous pore-forming method for bone repair.RSC Adv.2013;3(19):6871-6878. [7]Ege D,Cameron R,Best S.The degradation behavior of nanoscale HA/PLGA and alpha-TCP/PLGA composites. Bioinspir Biomim Nan.2014;3(2):85-93. [8]Cao J,Shi S,Geng H,et al.Neutralizing ability of bioactive glass (BG) on the PLGA microsphere degradation.Lat Am J Pharm.2015;34(3):468-473. [9]Lynn A,Nakamura T,Patel N,et al. Composition- controlled nanocomposites of apatite and collagen incorporating silicon as an osseopromotive agent.J Biomed Mater Res A. 2005;74(3):447-453. [10]Liu X,Ding C,Chu PK.Mechanism of apatite formation on wollastonite coatings in simulated body fluids. Biomaterials.2004;25(10):1755-1761. [11]Lin K,Zhai W,Ni S,et al.Study of the mechanical property and in vitro biocompatibility of CaSiO3 ceramics.Ceram Int.2005;31(2):323-326. [12]Xue W,Liu X,Zheng X,et al.In vivo evaluation of plasma-sprayed wollastonite coating. Biomaterials. 2005;26(17):3455-3460. [13]Wu C,Chang J.A review of bioactive silicate ceramics. Biomed Mater.2013, 8(3):032001. [14]Shirazi FS,Mehrali M,Oshkour AA,et al.Mechanical and physical properties of calcium silicate/alumina composite for biomedical engineering applications.J Mech Behav Biomed Mater.2014;30:168-175. [15]Nam YS,Park TG.Porous biodegradable polymeric scaffolds prepared by thermally induced phase separation.J Biomed Mater Res.1999;47(1): 8-17. [16]Cai Q,Yang J,Bei J,et al. A novel porous cells scaffold made of polylactide-dextran blend by combining phase-separation and particle-leaching techniques. Biomaterials. 2002;23(23):4483-4492. [17]Bose S,Vahabzadeh S,Bandyopadhyay A.Bone tissue engineering using 3D printing. Mater Today. 2013; 16(12):496-504. [18]Cox SC,Thornby JA,Gibbons GJ,et al.3D printing of porous hydroxyapatite scafolds intended for use in bone tissue engineering applications.Mater Sci Eng C Mater Biol Appl.2015;47:237-247. [19]Castilho M,Rodrigues J,Pires I,et al.Fabrication of individual alginate-TCP scaffolds for bone tissue engineering by means of powder printing. Biofabrication. 2015;7(1):015004. [20]Tarafder S,Balla VK,Davies NM,et al. Microwave-sintered 3D printed tricalcium phosphate scaffolds for bone tissue engineerig.J Tissue Eng Regen Med.2013;7(8):631-641. [21]Inzana JA,Olvera D,Fuller SM,et al.3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration.Biomaterials. 2014;35(13):4026-4034. [22]Wu C,Fan W,Zhou Y,et al.3D-printing of highly uniform CaSiO3 ceramic scaffolds: preparation, characterization and in vivo osteogenesis.J Mater Chem.2012;22(24):12288-12295. [23]Xu WK,Wang LY,Ling Y,et al.Enhancement of compressive strength and cytocompatibility using apatite coated hexagonal mesoporous silica/poly(lactic acid-glycolic acid) microsphere scaffolds for bone tissue engineering.Rsc Adv.2014; 4(26):13495-13501. [24]法默.矿物的红外光谱[M]. 应育浦,汪寿松,李春庚,等译.北京:科学出版社,1982. [25]方敏.纳米CaSiO3 发光材料的性能及特殊形貌研究[D].上海师范大学,2007. [26]袁曦明,石岚,何应律,等.人工合成硅灰石的发光性质与发光机理的初步研究[J].地球科学: 中国地质大学学报, 1994,19(2):175-180. [27]Quelch KJ,Melick R,Bingham PJ,et al.Chemical composition of human bone.Arch Oral Biol. 1983; 28(8):665-674. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||