Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (30): 4547-4553.doi: 10.3969/j.issn.2095-4344.2016.30.020
Previous Articles Next Articles
Hydroxyapatite/zirconia gradient composite scaffolds for bone tissue engineering: current uses and perspectives
Shao Rong-xue1, Huang Xiao-long1, Hu Hua-hui1, Quan Ren-fu2, Xie Shang-ju2, Li Xi3
- 1Zhejiang Chinese Medical University, Hangzhou 310053, Zhejiang Province, China; 2Department of Orthopedics, Xiaoshan Traditional Chinese Medical Hospital, Hangzhou 311201, Zhejiang Province, China; 3School of Materials Science and Engineering, Shanghai University, Shanghai 200072, China
-
Received:2016-05-05Online:2016-07-15Published:2016-07-15 -
Contact:Quan Ren-fu, Professor, Doctoral supervisor, Department of Orthopedics, Xiaoshan Traditional Chinese Medical Hospital, Hangzhou 311201, Zhejiang Province, China -
About author:Shao Rong-xue, Studying for doctorate, Zhejiang Chinese Medical University, Hangzhou 310053, Zhejiang Province, China -
Supported by:the Planned Project of Zhejiang Provincial Department of Science and Technology, No. 2012C33114
CLC Number:
Cite this article
Shao Rong-xue, Huang Xiao-long, Hu Hua-hui, Quan Ren-fu, Xie Shang-ju, Li Xi. Hydroxyapatite/zirconia gradient composite scaffolds for bone tissue engineering: current uses and perspectives[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(30): 4547-4553.
share this article
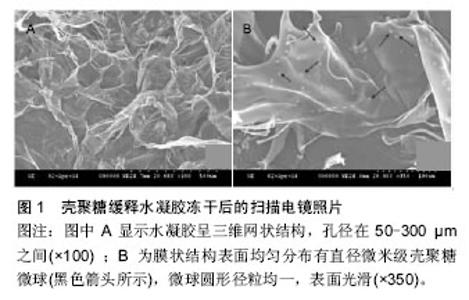
2.1 羟基磷灰石组织工程骨 目前临床常用治疗骨缺损的方法主要有自体骨移植、异体骨移植、异种骨移植及骨生物材料,但均存在诸多问题,自体取骨其本身对患者造成再次损伤,极大增加了患者的痛苦,且存在供区受损、功能障碍、甚至遗留慢性疼痛;异体骨和异种骨又存在来源有限且价格不菲、潜在传播疾病及免疫排斥反应等一系列问题;骨水泥又存在毒性反应等一系列问题,限制了其临床应用;而临床上目前应用的骨移植材料只能起到骨替代的作用,无法促使新生骨组织长入移植材料内部并融为一体,这也意味着材料在患者体内存在着耗损、老化的风险。大段骨移植后的骨折愈合是个复杂过程,随着愈合时间的延长增加了植入物致异物感染的概率,感染后则导致更大范围的假体周围骨溶解。那么,提高人工移植骨的成骨性能和骨融合率是提高骨移植手术成功的关键。 羟基磷灰石泡沫陶瓷具有良好的支架功能,可引导新生骨组织长入支架内部,达到自体骨组织与支架材料的严密整合,成功解决了当前密质材料的问题[6-7]。此外,羟基磷灰石[Ca10(PO4)6(OH)2]是一种生物活性材料,其结构与天然磷灰石矿物相近,具有与人体硬组织相似的结构和化学成分,是脊椎动物骨组织的主要无机成分,植入人体后,在体液作用下会被部分降解,释放出人体组织所必需的钙、磷元素并被人体组织吸收、利用,结合入新的组织,从而使羟基磷灰石植入物和人体骨组织获得良好的结合。羟基磷灰石能够提供更大的表面区域,以利于生物陶瓷与自体骨形成化合建[8]。 与医用生物陶瓷中的其他材料相比,羟基磷灰石与机体亲和性最为优良,具有良好的生物相容性、骨传导性及骨诱导性。Yoshikawa等[6]在植入泡沫陶瓷材料后6周观察可见骨组织长入多孔结构内,术后8周见多孔结构的内部布满新生骨组织。然而,材料孔径的大小直接影响新生骨细胞的黏附及骨组织、血管的长入,相互连接的孔为新生骨和血管提供一个引导和路径[9-11]。有学者指出,材料孔径大于 100 μm可为新生骨细胞及血管的生长提供一个支架,利于成骨细胞及其他骨细胞在其内部分化、爬行和扩增[12-14]。但材料的机械强度随着孔隙率的升高几乎呈直线下降[15]。利用泡沫浸渍技术制备的泡沫陶瓷平均抗压强度和弹性模量分别为(1.04±0.15) MPa和0.1 GPa,而松质骨的抗压强度和弹性模量分别为2-12 MPa 和0.3 GPa[16]。因此,单纯羟基磷灰石作为支架材料存在强度低、韧性差、脱粒等缺陷。将其制成多孔材料后,其抗弯强度与断裂韧性更进一步降低,其抗压强度和弹性模量比松质骨更差。故单一的羟基磷灰石材料无法满足人体承重骨代替的要求[17]。通过不同性能材料的加入,使二者相互补强,进而提高羟基磷灰石的力学强度、韧性等问题。羟基磷灰石作为修复材料的主体,与其他材料相复合形成的生物复合材料目前备受关注。 2.2 二氧化锆梯度复合羟基磷灰石组织工程骨 二氧化锆陶瓷材料具有较高的弯曲强度、断裂韧性和较低的弹性模量,且具有一定的生物相容性。现已有实验表明,在羟基磷灰石陶瓷材料中有选择地加入第二相颗粒,可以显著提高材料的断裂韧性。由于第二相颗粒如生物惰性材料二氧化锆具有良好的机械力学性能,基体上复合羟基磷灰石,增加了其生物相容性,用于承载种植体,将其与羟基磷灰石梯度复合制成材料后,当材料由于受到外加应力而产生裂纹时,第二相颗料能够有效吸收能量,防止微裂纹的进一步扩展,提高材料的机械力学性能,达到人体承重部位的使用要求。研究显示,羟基磷灰石/二氧化锆的抗弯曲强度、抗压强度约是单纯羟基磷灰石的10倍[18]。因此,用二氧化锆作为羟基磷灰石增强体可获得力学性能和生物相容性俱佳的生物复合材料。相反,材料的机械强度随着羟基磷灰石的含量及孔隙率的增加而逐渐降低[19]。正常成年人皮质骨的抗弯曲强度、抗压强度分别为50-150 MPa、100-230 MPa,抗骨折韧性、弹性模量分别为2-6 MPam1/2、7-30 GPa。羟基磷灰石/二氧化锆复合体相关参数优于皮质骨,可作为皮质骨的替代材料[20]。但是,若羟基磷灰石/二氧化锆复合体强度过大同时也带来应力遮蔽的问题[21],可以通过调节羟基磷灰石/二氧化锆复合泡沫陶瓷材料的成分比例及孔隙率来调节其强度,从而得到力学性能最贴近自然骨结构的高仿真人工骨支架材料,这也是羟基磷灰石/二氧化锆梯度复合泡沫陶瓷材料的最大优势所在。此外,羟基磷灰石/二氧化锆复合材料在体内、外实验中显示出完全的生物相容性,在骨组织、肌肉组织界面没有不良反应[22],细胞毒性试验和溶血试验的反应是微弱的、可接受的[23]。因此,二氧化锆梯度复合羟基磷灰石组织工程骨为骨移植提供良好的替代材料。 二氧化锆梯度复合羟基磷灰石组织工程骨的制备:采用聚氨酯作为泡沫模型材料,将其切割为长24 cm、半径9 cm半圆柱型大小,采用60 ℃、质量分数15%氢氧化钠溶液浸泡3.5 h,用清水反复揉搓3遍,然后晾干,再以表面活性剂继续对有机聚氨酯泡沫体材料进行表面处理,最后用5%聚乙烯醇浸泡24 h后干燥。采用箱式电阻炉对二氧化锆粉体进行预烧处理,缓慢升温至1 250 ℃后保温4 h,完成粉体预处理。浆料制备采用添加剂配比:聚乙烯醇、羧甲基纤维素的含量均为0.5%,加入10%硅溶胶,聚丙稀酸铵的含量为0.6%。首次挂浆加入二氧化锆的质量分数为60%。对原料进行完全混合研磨后,采用粉球质量比1∶2,转速300 r/min的高速球磨机研磨3 h以制取浆料,然后先将一定量去离子水和硅溶胶搅拌10 min,加入羧甲基纤维素和消泡剂辛醇和二氧化锆粉体,再搅拌3 h,等待挂浆。先将试样置于室温下18-24 h后自然干燥,而后放入烘箱中,在110 ℃条件下烘干12 h,使水分质量分数降至1%以下。然后进行第二、三次研磨、挂浆,2次加入二氧化锆粉料的质量分数分别为57%、54%,两次间隔需要经过相同工艺烘干。最后对上述挂浆后预制体试样采用了三段式烧结工艺进行烧结,制得长24 cm、半径 9 cm半圆柱型纯二氧化锆组织工程骨。 采取两步法浸涂法制备纯二氧化锆组织工程骨梯度羟基磷灰石涂层,第一次浸涂浆料配比为:二氧化锆粉体31.1%、羟基磷灰石粉体13.3%,磷酸乙酯1.4%,乙基纤维素0.2%,双蒸水54%。先将羟基磷灰石粉体加热至800 ℃后保温2 h,再将上述其余原料在50 ℃热水中混合,并搅拌5 h。把烧结出的纯二氧化锆组织工程骨浸入上述浆料,等完全浸透后取出、甩去多余浆料,并于100 ℃烘干5 h,再加热到900 ℃保温5 h,最后升温至1250 ℃保持1 h。第二次浸涂浆料配比为:二氧化锆粉体3.9%,羟基磷灰石粉体35.5%,磷酸乙酯1.4%,乙基纤维素0.2%,双蒸水59%,配置好浆料后重复上述步骤,最后制得二氧化锆梯度复合羟基磷灰石组织工程骨。 2.3 壳聚糖凝胶包裹骨形态发生蛋白2种植于羟基磷灰石/二氧化锆组织工程骨 骨形态发生蛋白是公认的骨诱导因子,成熟的成骨细胞中几乎所有的骨形态发生蛋白均能够刺激碱性磷酸酶活性,然而骨形态发生蛋白2是惟一能诱导多能造血干细胞和间充质干细胞中所有成骨细胞分化标志物的形态发生蛋白。目前在骨形态发生蛋白家族中已发现10多名成员,其中骨形态发生蛋白2能诱导多能造血干细胞和间充质干细胞中所有成骨细胞分化的形态发生蛋白。2002年重组人骨形态发生蛋白2被TFDA正式批准应用于腰椎前路融合中,标志着骨形态发生蛋白2在脊柱领域从基础实验转为临床应用的成功跨越,骨形态发生蛋白2有明显的成骨作用,可通过加快骨化过程来促进椎体内骨质生成。目前已经有多位学者认为骨形态发生蛋白将有希望挑战自体骨在脊柱融合中的金标准地位,使用骨形态发生蛋白方法可缩短手术时间和住院时间、减少出血,且无取骨部位疼痛。Lee等[24]植入种植重组人骨形态发生蛋白2的多孔陶瓷于SD大鼠的颅底缺损处,术后4周可见大量骨组织长入泡沫陶瓷孔隙中;而仅使用泡沫陶瓷材料组术后4周孔隙内仅有少量纤维组织,术后8周可见少量的成骨细胞长入。Burkus等[25]临床试验数据认为,骨形态发生蛋白在脊柱融合中比自体髂骨效果更好,骨形态发生蛋白2有明显的成骨作用,可通过加快骨化过程来促进椎体内骨质生成。Itoh等[26]采用羟基磷灰石/胶原蛋白混合材料制成13周内可被吸收的人工椎体,同时吸附人重组2型骨形态发生蛋白促进骨痂生长,能有效防止内植物塌陷。陈勇等[27]认为重组人骨形态发生蛋白2以磷酸钙骨水泥作为载体组成的复合骨水泥,不仅能够修复骨质疏松性椎体骨折,促进成骨活性及骨折愈合,还能够恢复骨折椎体的基本生物力学性能。因此,骨形态发生蛋白的运用将会在未来的骨科领域扮演一个非常重要的角色。但单独使用骨形态发生蛋白2将很快被吸收,因此需用成形材料作为其载体。而内源性骨形态发生蛋白在组织中的含量较微弱,仅在骨折早期发挥其促进骨生长的作用;外源性生长因子注入体内后,极易被体液稀释转运吸收,或被迅速降解导致局部因子浓度降低。此外,骨形态发生蛋白2的半衰期极短,注入体内呈爆发性释放[28],但骨折愈合是一个缓慢的过程,需要骨形态发生蛋白2缓慢而持久的释放,能在较长时间内保持有效的药物浓度,从而可获得良好的治疗效果。一些研究表明新骨形成依赖骨形态发生蛋白2的量[29],但大量注入骨形态发生蛋白2不仅会增加手术费用,而且会产生相应的不良反应[30-32]。因此,采用缓释技术构建有效的骨形态发生蛋白2缓释系统,延长骨形态发生蛋白2的释放和作用时间,是其充分发挥诱导成骨效应的关键。为了克服这一困难,一些学者设计出各种缓释系统来控制骨形态发生蛋白2的释放[33-38],其中以壳聚糖凝胶缓释系统应用最为广泛。 壳聚糖的化学名称为聚葡萄糖胺(1-4)-2-氨基- B-D葡萄糖,是几丁质经过脱乙酰作用得到的。由于它具有生物相容性、可降解性和骨传导性等优点,已被广泛应用到生物医学领域[33-36]。壳聚糖对药物的包封率和释放率受壳聚糖相对分子质量、脱乙酰度和浓度等的影响,研究表明,利用壳聚糖承载牛血清白蛋白,当壳聚糖相对分子质量从10 000提高到210 000,牛血清白蛋白的包封率提高约2倍,牛血清白蛋白的释放速率从73.9%降至17.6%;壳聚糖脱乙酰度从75.5%提高到92%,轻度增加牛血清白蛋白包封率和减慢牛血清白蛋白的释放速率[39]。壳聚糖包裹骨形态发生蛋白2可大幅度减缓骨形态发生蛋白2的释放、从而促进成骨。有学者应用壳聚糖干凝胶包裹骨形态发生蛋白2,使其持续释放达6周[40]。因此,壳聚糖凝胶缓释系统是骨形态发生蛋白2较好的包裹体系,可控制其缓慢、持续释放,从而促进骨缺损的修复(图1)。 "
| [1] 靳洪允.泡沫陶瓷材料的研究进展[J].现代技术陶瓷, 2005,3(4): 33-36. [2] Rhinelander FW,Rouweyha M,Milner JC.Microvascular and histogenic responses to implantation of a porous ceramic into bone.J Biomed Mater Res. 1971;5(1): 81-112. [3] Benum P,Lyng S,Alm T,et al. Porous ceramics as a bone substitute in the medial condyle of the tibia. An experimental study in sheep. Long-term observations. Acta Orthop Scand. 1977;48(2):150-154. [4] Niles JL,Coletti JM Jr,Wilson C.Biomechanical evaluation of bone-porous material interfaces.J Biomed Mater Res.1973; 7(2):231-251. [5] Furseth R,Pedersen KN.Ultrastructure of tissue in intrabony and subperiosteal porous ceramic implants.Int J Oral Surg. 1978;7(5):471-478. [6] Yoshikawa H,Myoui A.Bone tissue engineering with porous hydroxyapatite ceramics. J Artif Organs. 2005; 8:131-136. [7] Yoshikawa H,Tamai N,Murase T,et al.Interconnected porous hydroxyapatite ceramics for bone tissue engineering.J R Soc Interface.2009;6 Suppl 3:S341-348. [8] Tancred DC,McCormack BA,Carr AJ.A synthetic bone implant macroscopically identical to cancellous bone. Biomaterials. 1998;19:2303-2311. [9] Cavagna R,Daculsi G,Bouler JM.Macroporous calcium phosphate ceramic: a prospective study of 106 cases in lumbar spinal fusion. J Long Term Eff Med Implants. 1999;9(4): 403-412. [10] Jones AC,Arns CH,Sheppard AP,et al.Assessment of bone ingrowth into porous biomaterials using MICRO-CT. Biomaterials.2007;28:2491-2504. [11] Karageorgiou V,Kaplan D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials.2005;26: 5474-5491. [12] Holmes RE,Wardrop RW,Wolford LM.Hydroxylapatite as a bone graft substitute in orthognathic surgery: histologic and histometric findings.J Oral Maxillofac Surg.1988;46:661-671. [13] Lecomte A,Gautier H,Bouler JM,et al.Biphasic calcium phosphate: a comparative study of interconnected porosity in two ceramics. J Biomed Mater Res B Appl Biomater. 2008; 84: 1-6. [14] Omae H,Mochizuki Y,Yokoya S,et al.Effects of interconnecting porous structure of hydroxyapatite ceramics on interface between grafted tendon and ceramics. J Biomed Mater Res A. 2006;79(2):329-337. [15] Suchanek W,Yoshimura M.Processing and properties of hydroxyapatite-based biomaterials for use as hard tissue replacement implants. J Mater Res. 1998;13: 94-117. [16] Liu B,Lin P,Shen Y,et al.Porous bioceramics reinforced by coating gelatin. J Mater Sci Mater Med. 2008;19: 1203-1207. [17] Linhart W,Briem D,Amling M,et al.Mechanical failure of porous hydroxyapatite ceramics 7.5 years after implantation in the proximal tibial.Der Unfallchirurg. 2004;107:154-157. [18] Silva VV,Domingues RZ.Hydroxyapatite-zirconia composites prepared by precipitation method.J Mater Sci Mater Med. 1997;8:907-910. [19] Curran DJ,Fleming TJ,Towler MR,et al.Mechanical properties of hydroxyapatite-zirconia compacts sintered by two different sintering methods. J Mater Sci Mater Med.2010;21: 1109-1120. [20] Matsuno T,Watanabe K,One K,et al.Microstructure and mechanical properties of sintered body of zirconia coated hydroxyapatite particles. Mater Sci Mater Med. 2000; 9: 573-576. [21] 宁聪琴,戴尅戎.硬组织替换用羟基磷灰石复合材料的研究进展[J].生物医学工程杂志, 2003,20(3):550-554. [22] Vaidhyanathan B,Kumar KA,Shetty JN,et al.Biological evaluation of a zirconia toughened apatitic composite implant. B Mater Sci.1997;20:135-140. [23] Quan R,Yang D,Wu X,et al.In vitro and in vivo biocompatibility of graded hydroxyapatite-zirconia composite bioceramic.J Mater Sci Mater Med. 2008; 19(1):183-187. [24] Lee JH,Ryu MY,Baek HR,et al. Effects of porous beta-tricalcium phosphate-based ceramics used as an E. coli-derived rhBMP-2 carrier for bone regeneration. J Mater Sci Mater Med.2013;24:2117-2127. [25] Burkus JK,Heim SE,Gornet MF,et al.The effectiveness of rhBMP-2 in replacing autograft: an integrated analys is of three human spines tudies.Orthopedics. 2004; 27(7):723-728. [26] Itoh S,Kikuchi M,Koyama Y,et al.Development of an artificial vertebral body using a novel biomaterial, hydroxyapatite/ collagen composite.Biomaterials. 2002; 23: 3919-3926. [27] 陈勇,杜靖远,梁惠民,等.复合rhBMP-2的注射型磷酸钙人工骨在椎体成形术中的应用及生物力学评价[J].中华物理医学与康复杂志,2004,26(7):392-395. [28] Ruhe PQ,Boerman OC,Russel FG,et al.In vivo release of rhBMP-2 loaded porous calcium phosphate cement pretreated with albumin. J Mater Sci Mater Med. 2006; 17: 919-927. [29] Alam I,Asahina I,Ohmamiuda K, et al.Comparative study of biphasic calcium phosphate ceramics impregnated with rhBMP-2 as bone substitutes.J Mater Sci Mater Med.2001;54: 129-138. [30] Fu R,Selph S,McDonagh M,et al. Effectiveness and harms of recombinant human bone morphogenetic protein-2 in spine fusion: a systematic review and meta-analysis. Ann Intern Med. 2013;158(12):890-902. [31] Alt V,Chhabra A,Franke J,et al.An economic analysis of using rhBMP-2 for lumbar fusion in Germany, France and UK from a societal perspective.Eur Spine J 2009;18:800-806. [32] Benglis D,Wang MY,Levi AD.A comprehensive review of the safety profile of bone morphogenetic protein in spine surgery. Neurosurgery.2008;62:ONS423-31. [33] Abarrategi A,Garcia-Cantalejo J,Moreno-Vicente C,et al.Gene expression profile on chitosan/rhBMP-2 films: A novel osteoinductive coating for implantable materials. Acta Biomaterialia.2009;5:2633-2646. [34] Jun SH,Lee EJ,Yook SW,et al. A bioactive coating of a silica xerogel/chitosan hybrid on titanium by a room temperature sol-gel process. Acta Biomaterialia. 2010; 6:302-307. [35] Jun SH,Lee EJ,Kim HE,et al.Silica-chitosan hybrid coating on Ti for controlled release of growth factors.J Mater Sci Mater Med.2011;22:2757-27564. [36] Lee EJ,Shin DS,Kim HE,et al.Membrane of hybrid chitosan-silica xerogel for guided bone regeneration. Biomaterials.2009;30:743-750. [37] Chen L,Gu Y,Feng Y,et al. Bioactivity of porous biphasic calcium phosphate enhanced by recombinant human bone morphogenetic protein 2/silk fibroin microsphere.J Mater Sci Mater Med. 2014;25:1709-1719. [38] Hannink G,Geutjes PJ,Daamen WF,et al.Evaluation of collagen/heparin coated TCP/HA granules for long-term delivery of BMP-2.J Mater Sci Mater Med. 2013; 24 (2): 325-332. [39] Xu Y,Du Y.Effect of molecular structure of chitosan on protein delivery properties of chitosan nanoparticles.Int J Pharm. 2003;250(1):215-226. [40] Jun SH,Lee EJ,Jang TS,et al.Bone morphogenic protein-2 (BMP-2) loaded hybrid coating on porous hydroxyapatite scaffolds for bone tissue engineering.J Mater Sci Mater Med. 2013;24(3):773-782. [41] Park J,Ries J,Gelse K,et al.Bone regeneration in critical size defects by cell-mediated BMP-2 gene transfer: a comparison of adenoviral vectors and liposomes. Gene Ther. 2003; 10 (13):1089-1098. [42] Tsuda H,Wada T,Yamashita T,et al.Enhanced osteoinduction by mesenchymal stem cells transfected with a fiber-mutant adenoviral BMP2 gene.J Gene Med. 2005;7(10):1322-1334. [43] Ohgushi H,Goldberg VM,Caplan AI.Heterotopic osteogenesis in porous ceramics induced by marrow cells.J Orthop Res. 1989;7:568-578. [44] Ohgushi H,Dohi Y,Tamai S,et al.Osteogenic differentiation of marrow stromal stem cells in porous hydroxyapatite ceramics. J Biomed Mater Res. 1993; 27:1401-1407. [45] Strong M,Farrugia A,Rebulla P.Stem cell and cellular therapy developments. Biologicalsn. 2009;37(2):103-107. [46] Takahashi K,Tanabe K,Ohnuki M,et al.Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell.2007;131:861-872. [47] Liu H, Ye Z, Kim Y,et al.Generation of endoderm- derived human induced pluripotent stem cells from primary hepatocytes. Hepatology (Baltimore, Md).2010; 51: 1810 -1819. [48] Kim JB,Greber B,Arauzo-Bravo MJ,et al.Direct reprogramming of human neural stem cells by OCT4. Nature. 2009;461:649-643. [49] Yu J,Vodyanik MA,Smuga-Otto K,et al.Induced pluripotent stem cell lines derived from human somatic cells.Science (New York, NY).2007;318:1917-1920. [50] Park IH,Zhao R,West JA,et al.Reprogramming of human somatic cells to pluripotency with defined factors. Nature. 2008;451:141-146. [51] Yan X,Qin H,Qu C,et al.iPS cells reprogrammed from human mesenchymal-like stem/progenitor cells of dental tissue origin.Stem Cells Dev. 2010;19(4):469-480. [52] Liu Y,Goldberg AJ,Dennis JE,et al.One-step derivation of mesenchymal stem cell (MSC)-like cells from human pluripotent stem cells on a fibrillar collagen coating.PloS One.2012;7:e33225. [53] Villa-Diaz LG,Brown SE,Liu Y,et al.Derivation of mesenchymal stem cells from human induced pluripotent stem cells cultured on synthetic substrates. Stem cells (Dayton, Ohio).2012;30: 1174-1181. [54] Yang H,Aprecio RM,Zhou X,et al.Therapeutic effect of TSG-6 engineered iPSC-derived MSCs on experimental periodontitis in rats: a pilot study. PloS One.2014;9(6):e100285 |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||