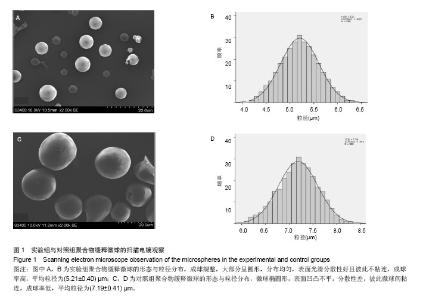
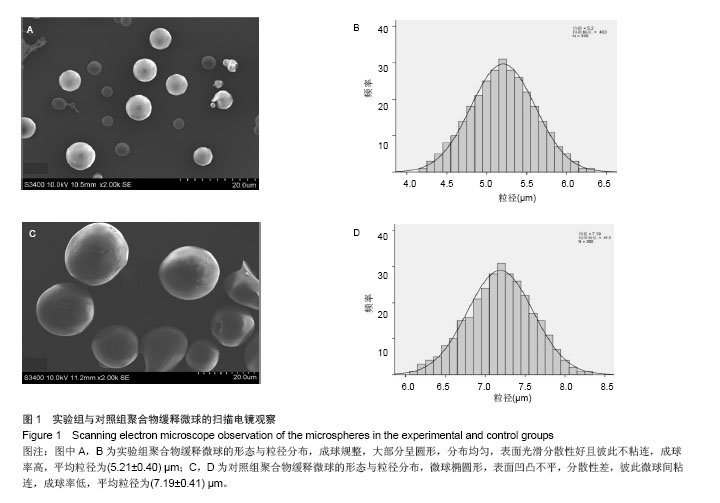
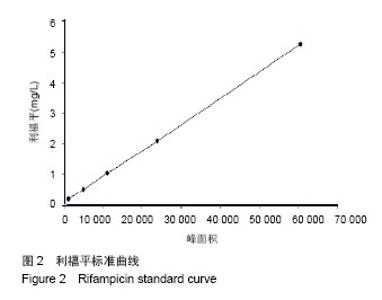
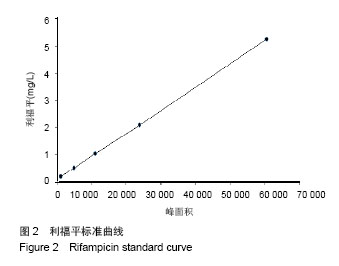
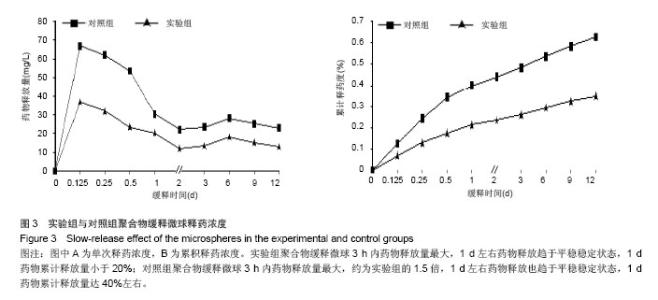
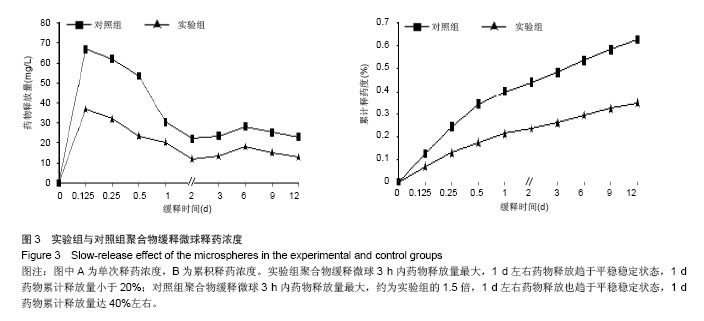
| [1]施建党,王自立.脊柱结核术后未愈及术后复发的原因探讨[J].中国矫形外科杂志,2005,13(15):1184-1186.
[2]Oga M,Arizono T,Takasita M,et al.Evaluation of the risk of instrumentation as a foreign body in spinal tuberculosis. Clinical and biologic study.Spine (Phila Pa 1976).1993; 18(13):1890-1894.
[3]Alcantar NA,Aydil ES,Israelachvili JN.Polyethylene glycol- coated biocompatible surfaces.J Biomed Mater Res.2000; 51(3):343-351.
[4]Wang Z,Ge Z,Jin W,et al.Treatment of spinal tuberculosis with ultrashort-course chemotherapy in conjunction with partial excision of pathologic vertebrae.Spine J.2007;7(6):671-681.
[5]戈朝晖,王自立,魏敏吉.脊柱结核病灶中抗痨药物浓度的测定[J].中华骨科杂志,2005,25(2):36-40.
[6]德向研,施建党,王自立,等.载三联抗结核药硫酸钙/氨基酸聚合物人工骨体内缓释实验研究[J].中国脊柱脊髓杂志,2013,23(6):成531-536.
[7]Hirota K,Hasegawa T,Nakajima T,et al.Delivery of rifampicin-PLGA microspheres into alveolar macrophages is promising for treatment of tuberculosis.J Control Release. 2010;142(3):339-346.
[8]劳丽春.药物缓释载体材料类型及其临床应用[J].中国组织工程研究与临床康复,2010,14(47):8865-8868.
[9]金丽霞.药物缓释载体材料在医药领域中的研究及应用[J].中国组织工程研究与临床康复,2011,15(25):4699-4702.
[10]刘艳,张志鹏,索绪斌,等.缓控释制剂体外释放度的研究进展[J].时珍国医国药,2011,22(3):701-703.
[11]Fournier E,Passirani C,Montero-Menei CN,et al. Biocompatibility of implantable synthetic polymeric drug carriers: focus on brain biocompatibility. Biomaterials. 2003; 24(19):3311-3331.
[12]丁小力,王自立,戈朝晖,等.载异烟肼利福平聚乳酸纳米粒的制备及体外释药[J].中国组织工程研究,2012,16(16):2909-2912.
[13]叶美娣,郑彩虹.乳酸-羟基乙酸共聚物(PLGA)微球控释系统的突释[J].中国现代应用药学,2005,22(5):20-23.
[14]Veronese FM,Pasut G.PEGylation: Posttranslational bioengineering of protein biotherapeutics.Drug Discov Today Technol.2008;5(2-3):e57-e64.
[15]Hong SJ,Yu HS,Kim HW.Tissue engineering polymeric microcarriers with macroporous morphology and bone-bioactive surface.Macromol Biosci.2009;9(7):639-645.
[16]黄玲玲,姜志华.PLA的合成及改性研究进展[J].合成树脂及塑料, 2010,27(6):65-69.
[17]张洁琼,曹健,李艳芳,等.聚乳酸的合成•生物降解及应用研究现状[J].安徽农业科学,2010,50(31):17384-17386.
[18]曲文强,夏文龙,孙斌,等.生物降解材料聚羟基乙酸及其共聚物研究进展[J].山东化工,2014,43(3):34-36.
[19]Hirota K,Kawamoto T,Nakajima T,et al.Distribution and deposition of respirable PLGA microspheres in lung alveoli. Colloids Surf B Biointerfaces.2013;105:92-97.
[20]赵先英,张涛,刘毅敏,等.乳酸-羟基乙酸共聚物多肽蛋白类药物微球控释系统的突释与控制[J].西南国防医药,2011,21(8): 920-922.
[21]Hao J,Hu ZM,ZhengQX,et al. Biomineralization of the Surface of PLGA-(ASP-PEG) Modified with the K_(16) and RGD-containing Peptide.Journal of Wuhan University of Technology(Mater Sci Ed).2009;24(5):768-771.
[22]汪求精,孙新林,祝爱萍,等. PLGA/PEG缓释微球的制备与载药性能的研究[J].中华神经医学杂志,2009,8(7):674-677.
[23]王希,张文强,邱蔚芬,等.乳酸-羟基乙酸共聚物缓释微球的制备、性能及应用[J].中国组织工程研究与临床康复,2009,13(12): 2353-2356.
[24]Ito F,Fujimori H,Honnami H,et al.Effect of polyethylene glycol on preparation of rifampicin-loaded PLGA microspheres with membrane emulsification technique. Colloids Surf B Biointerfaces. 2008;66(1):65-70.
[25]Wu G,Chen L,Li H,et al.Comparing microspheres with different internal phase of polyelectrolyte as local drug delivery system for bone tuberculosis therapy. Biomed Res Int.2014;2014:297808.
[26]黄术.RFP-PLGA骨缓释复合体的初步实验研究[D].中南大学, 2013.
[27]谢鑫鑫,王俊腾,秦利芳,等.聚乙二醇化聚乳酸羟基乙酸纳米粒的制备及体外抗黏性能评价[J].中国医院药学杂志,2012,32(21): 1727-1731.
[28]Jeffery H,Davis SS,O'Hagan DT.The preparation and characterization of poly(lactide-co-glycolide) microparticles. II. The entrapment of a model protein using a (water-in-oil)-in- water emulsion solvent evaporation technique.Pharm Res. 1993; 10(3):362-368.
[29]张海龙,高玲美,邵洪伟.聚乳酸载药微球的制备及应用研究进展[J].西北药学杂志,2010,25(2):158-160.
[30]林丽丽.聚乳酸载药系统的研究进展[J].广东化工,2012,39(6): 90-91.
[31]Hasan AS,Socha M,Lamprecht A,et al.Effect of the microencapsulation of nanoparticles on the reduction of burst release.Int J Pharm.2007;344(1-2):53-61.
[32]Liu P,Zhu Q,Jiang J.Distribution of three antituberculous drugs and their metabolites in different parts of pathological vertebrae with spinal tuberculosis. Spine (Phila Pa 1976). 2011;36(20):E1290-E1295. |