Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (41): 6719-6724.doi: 10.3969/j.issn.2095-4344.2014.41.028
Proliferation, senescence and differentiation of mesenchymal stem cells: canonical and non-canonical regulations of Wnt signaling pathway
Shi Jian-ming1, 2, Wu Ya-hua3, Geng Shu-guo1, 2, Yin Ming1
- 1The Second Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China; 2Graduate School of Medicine, Nanchang University, Nanchang 330006, Jiangxi Province, China; 3Jiujiang University, Jiujiang 332000, Jiangxi Province, China
-
Revised:2014-08-29Online:2014-10-01Published:2014-10-01 -
Contact:Yin Ming, Professor, Doctoral supervisor, the Second Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China -
About author:Shi Jian-ming, Studying for master’s degree, the Second Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 81160226
CLC Number:
Cite this article
Shi Jian-ming, Wu Ya-hua, Geng Shu-guo, Yin Ming. Proliferation, senescence and differentiation of mesenchymal stem cells: canonical and non-canonical regulations of Wnt signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(41): 6719-6724.
share this article

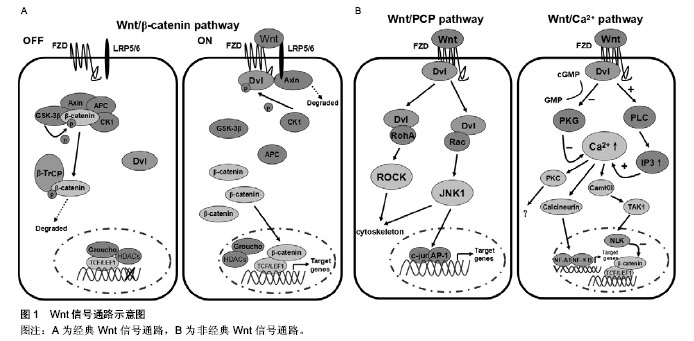
2.1 Wnt信号通路概述 Sharma(1973年)与Nusse(1982年)先后发现两种具有同源性的int21基因和wingless基因,两者被合并命名为Wnt基因族。随后又陆续在不同哺乳动物中发现了19种Wnt蛋白,Wnt蛋白通过分泌作用与位于细胞膜上的受体相结合,激活细胞内信号通路调节靶基因的表达,在胚胎的发育过程中对细胞的增殖、分化、死亡、迁移和极化均起到重要的作用。Wnt信号通路根据是否有β-连环蛋白(β-catenin)参与而分为经典Wnt信号通路(即Wnt/β-catenin信号通路)和非经典Wnt信号通路,后者又包括Wnt/PCP信号通路和Wnt/Ca2+信号通路。其中Wnt1、2、3、3a、8、8b等主要作用于经典Wnt信号通路,而Wnt4、5a、5b、6、7a、11等主要作用于非经典Wnt信号通路[4]。 2.1.1 经典Wnt信号通路 β-catenin是经典Wnt通路中的核心调控因子,其表达水平决定着通路的开放与否[5]。在未受到Wnt分子刺激的情况下,胞质中的β-catenin主要与结肠腺瘤息肉样蛋白(APC)及轴素多聚蛋白(Axin)结合,并被糖原合成酶激酶(GSK-3β)磷酸化,形成APC-Axin-GSK-3β-CK1-β-catenin复合体,随后被β-转导素重复蛋白(β-TrCP)介导的泛素/蛋白酶体途径降解,从而使胞浆内游离β-catenin处于相对较低水平[6]。当经典Wnt信号通路被激活时,Wnt分子与Frizzled受体(FZD) 及低密度脂蛋白受体相关蛋白5/6(LRP5/6)相结合,导致与FZD结合的胞内散乱蛋白(Dvl)被CK1过度磷酸化,并水解复合物分解限制组件Axin,从而抑制APC-Axin-GSK-3β-CK1对β-catenin的磷酸化,胞浆内未被磷酸化的游离β-catenin积聚后移入核内,并置换转录抑制剂Groucho/HDACs与转录因子T细胞因子(TCF)及淋巴样增强因子1(LEF1)结合,激活细胞周期蛋白D1(cyclinD1)、原癌基因(c-myc)、基质金属蛋白酶7(MMP-7)等下游靶基因的转录和翻译[7]。该信号通路的阻滞剂根据其作用方式不同可分为分泌性FZD相关蛋白类(sFRP)和Dickkopf蛋白类(Dkks),前者包括sFRP家族和Wnt抑制因子(WIF),可通过直接与FZD相关蛋白结合拮抗Wnt蛋白与FZD结合而发挥作用;后者包括Dkk-1、2、3、4,主要通过与LRP5/6结合从而抑制信号通路[8],见图1A。 2.1.2 非经典Wnt信号通路 非经典Wnt信号通路不仅不通过β-catenin发挥作用,甚至抑制β-catenin对通路的影响。Wnt/PCP信号通路和Wnt/Ca2+信号通路由非经典Wnt配子Wnt5a或Wnt11激活,信号转导体在FZD和Dvl[9]。激活的Wnt/PCP信号通路可通过Dvl活化Rho家族GTPases酶(RohA)和Rac蛋白,并进一步活化其下游的Rho激酶(ROCK)和JNK1激酶,与转录因子AP-1及c-jun的氨基末端区域结合后,引发下游靶基因的表达[10-11]。Wnt/Ca2+信号通路中内质网内的Ca2+释放入胞浆是关键环节,相关Wnt分子与FZD结合后活化胞浆中的Dvl,一方面可通过抑制阻止Ca2+释放的cGMP依赖性蛋白激酶(PKG)使内质网Ca2+释放增多,另一方面可通过激活磷脂酶C(PLC)和升高1,4,5-三磷酸肌醇(IP3)使胞浆中Ca2+增多,升高的Ca2+可激活Ca2+相关蛋白酶—蛋白激酶C(PKC)、钙调蛋白依赖性蛋白激酶Ⅱ(CamKⅡ)、钙调磷酸酶[12]。CamKⅡ进一步激活TGF-β激酶(TAK1)和NEMO激酶(NLK),后者可以磷酸化TCF从而拮抗经典Wnt信号通路的发生[13]。钙调磷酸酶还可进一步活化T细胞核因子(NF-AT)及核因子κB(NF-κB)等转录因子,最后诱导下游靶基因的表达[14],见图1B。 2.2 Wnt信号通路与间充质干细胞生物学特性的关系 2.2.1 Wnt信号通路与间充质干细胞增殖的关系 研究发现应用Wnt/β-catenin信号通路激活因子Wnt3a体外作用于大鼠间充质干细胞后能够显著促进其增殖[15]。Baek等[16]认为Wnt3a这一作用的产生可能与上调cyclinD1及c-myc的表达有关,cyclinD1和c-myc可驱动细胞周期进程,进而达到促进细胞增殖的效果。此外,Baksh等[17]发现伴随着Wnt/β-catenin信号通路相关受体LRP5表达量增加,间充质干细胞增殖加快。相反,Wnt5a可通过激活非经典Wnt信号途径,拮抗β-catenin/TCF信号,导致cyclinD1表达下降,最终抑制间充质干细胞增殖[18]。虽然Wnt3a在单独作用的情况下可以促进其增殖,但联合不同诱导剂后却起着促分化作用。还有学者认为在自分泌或旁分泌的方式下,Wnt/β-catenin信号通路激活能够抑制间充质干细胞增殖[19],这一观点得到了Gregory 等人的支持,Gregory 等[20]发现Dkk-1作为Wnt/β-catenin信号通路的阻滞剂是间充质干细胞重新进入细胞周期并进行增殖所必需的。De等[21]认为低水平激活的Wnt/β-catenin信号能够刺激间充质干细胞增殖,而高水平激活的Wnt/β-catenin信号反而抑制其增殖。由此可见,Wnt/β-catenin信号通路对间充质干细胞增殖起着双向调节作用,而具体的作用方向可能受培养环境及Wnt信号强度的影响。 2.2.2 Wnt信号通路与间充质干细胞衰老的关系 衰老是生物界的普遍现象,间充质干细胞与其他成体细胞一样也会发生衰老变化,衰老的间充质干细胞增殖和分化能力均会下降,严重制约了间充质干细胞的临床应用。研究发现,随着年龄增长,干细胞微环境变化引起的一系列信号通路改变是导致干细胞衰老的重要原因[22],其中Wnt信号通路被越来越多的研究证实参与了间充质干细胞衰老的调控。项晓霞等[23]分别用老年大鼠血清及年轻大鼠血清培养间充质干细胞,发现与年轻大鼠血清处理组比较,老年大鼠血清处理组的衰老细胞数量明显增多,β-catenin表达上调,并聚集于细胞核内,而在添加阻滞剂Dkk-1后衰老的细胞较未添加时减少,提示老年大鼠血清微环境可通过激活Wnt/β-catenin信号通路促进间充质干细胞的衰老。Zhang等[24]对不同年龄大鼠血清中Wnt3a的含量进行检测,结果显示老年大鼠血清中Wnt3a含量明显高于年轻大鼠血清,进一步证实了间充质干细胞衰老与Wnt/β-catenin信号通路的关系,他们还发现仅仅在高浓度Wnt3a作用下,高度激活的Wnt/β-catenin信号通路才可导致间充质干细胞发生衰老变化。另有研究发现,氯化锂作为一种GSK-3β抑制剂可以激活Wnt/β-catenin信号通路,在氯化锂的作用下,间充质干细胞β-半乳糖苷酶染色阳性细胞比例增加,细胞形态变大,成典型的细胞衰老表现[21]。c-myc作为Wnt/β-catenin信号通路的下游靶基因之一,以往认为被激活后将导致细胞癌变,近年来研究则发现,过度表达的c-myc也可引起细胞衰老[25],因此可以推测c-myc在Wnt/β-catenin信号通路引起的间充质干细胞衰老中发挥一定的作用。此外,刘荟敏等[26]利用3.5 Gy X射线照射建立的小鼠间充质干细胞衰老模型中发现非经典Wnt信号通路重要组分Wnt5a表达显著增高,提示非经典Wnt信号通路也可能参与了间充质干细胞衰老的过程。 2.2.3 Wnt信号通路与间充质干细胞分化的关系 Wnt信号通路在间充质干细胞定向分化中发挥着重要作用,在不同条件下Wnt信号通路的激活或抑制将决定间充质干细胞分化的方向。 向成骨细胞分化:目前对Wnt/β-catenin信号通路在间充质干细胞成骨分化过程中的作用报道不一。研究发现,在间充质干细胞成骨分化的过程中,Wnt信号通路相关组分sFRP2、sFRP3表达上调,而Wnt9a表达下调[27]。其中Wnt9a被认为与Wnt1具有类似的功能,能够激活Wnt/β-catenin信号通路,从而推断Wnt/β-catenin信号通路可能参与抑制间充质干细胞成骨分化。De等[21]、Cho等[28]的研究证实了上述观点,他们应用含Wnt3a配体的培养基培养间充质干细胞或向间充质干细胞转染外源性Wnt3a基因都呈抑制间充质干细胞成骨分化作用和下调成骨标志物的表达,如碱性磷酸酶、骨钙素、Ⅰ型胶原、矿化现象等,而Wnt/β-catenin信号通路阻滞剂sFRP3能够逆转其抑制成骨分化作用。但也有研究认为Wnt/β-catenin信号通路能够促进间充质干细胞向成骨细胞分化。Si等[29]发现大剂量的Wnt3a激活Wnt/ β-catenin信号通路后可以进一步激活CCN1/Cyr61基因,从而促进间充质干细胞成骨分化。Qiu等[19]还发现Wnt/β-catenin信号通路启动受体LRP5激活后可以上调Cbfa1/Runx2及碱性磷酸酶的表达,促进间充质干细胞成骨分化。有学者认为上述两种观点其实并不矛盾,Wnt/β-catenin信号通路对间充质干细胞成骨分化作用可能取决于细胞的分化阶段,当间充质干细胞已经启动向成骨细胞分化时,Wnt/β-catenin信号通路呈促进作用,但在分化的末期又会起着抑制成骨细胞分化成熟的作用[30]。非经典Wnt信号通路对间充质干细胞成骨分化作用研究相对较少,目前普遍认为非经典Wnt信号通路起着促间充质干细胞成骨分化作用[31-34]。 向脂肪细胞分化:一般情况下,间充质干细胞向成骨细胞及脂肪细胞分化之间保持着动态平衡,而Wnt信号通路对这种平衡的维持发挥着重要作用[35]。Platt等[36]发现激活的Wnt/β-catenin信号通路一方面可以通过促进成骨分化转录因子Runx2、Dlx5、Osx的表达而诱导间充质干细胞向成骨细胞分化,另一方面可以抑制成脂分化转录因子C/EBPα及PPAR-γ的表达而抑制间充质干细胞向脂肪细胞分化。此外,Fu等[37]发现Wnt/β-catenin信号通路下游靶基因cyclinD1及c-myc均具有抑制PPAR-γ表达作用。相反,通过Dkk-1或SFRP2来干扰Wnt/β-catenin信号通路可促进间充质干细胞分化成脂肪细胞[38]。Takada等[39]还发现激活非经典Wnt信号通路同样可以通过下调PPAR-γ表达而抑制间充质干细胞向脂肪细胞分化。 向神经细胞分化:间充质干细胞可以跨胚层向神经细胞分化,并历经神经干细胞、定向神经前体细胞、胶质前体细胞等阶段,近年来研究发现Wnt信号通路可能参与了这一过程。Wislet-Gendebien等[40]研究发现随着间充质干细胞向神经细胞分化,FZD1、FZD2、FZD5的表达量增加,而此3种蛋白均属于Wnt信号受体,提示Wnt信号通路可能参与了间充质干细胞向神经分化这一过程。阎文柱等[41]分别采用Wnt3a、Wht5a联合碱性成纤维细胞生长因子诱导间充质干细胞向神经细胞分化,前者能够增强碱性成纤维细胞生长因子诱导间充质干细胞成神经分化作用,而后者并无此作用,由此推断激活Wnt/β-catenin信号通路起着促间充质干细胞成神经分化作用,而非经典Wnt信号通路可能并不参与这一过程。这与何丁文等[42]的研究结果是一致的,他们在利用表皮生长因子联合碱性成纤维细胞生长因子诱导间充质干细胞向神经细胞分化的过程中发现β-catenin表达量上调,而后者是Wnt/β-catenin信号通路核心调控因子,其在细胞内的数量和状态对该途径有决定性影响。此外,Xu等[43]、林颢等[44]分别通过添加激活剂Wnt1或将携带目的基因Wnt3a的慢病毒转入骨髓间充质干细胞,建立能够持续激活Wnt/β-catenin信号通路的体外细胞模型,并成功诱导骨髓间充质干细胞向神经样细胞分化,从基因水平证实了Wnt/β-catenin信号通路参与促进骨髓间充质干细胞向神经细胞分化。"
| [1] Ding DC, Shyu WC, Lin SZ.Mesenchymal stem cells.Cell Transplant. 2011;20(1):5-14.
[2] Guo-ping W, Xiao-chuan H, Zhi-hui Y,et al.Influence on the osteogenic activity of the human bone marrow mesenchymal stem cells transfected by liposome-mediated recombinant plasmid pIRES-hBMP2-hVEGF165 in vitro.Ann Plast Surg. 2010;65(1):80-84.
[3] Lee PN, Pang K, Matus DQ,et al.A WNT of things to come: evolution of Wnt signaling and polarity in cnidarians.Semin Cell Dev Biol. 2006;17(2):157-167.
[4] Huelsken J, Behrens J.The Wnt signalling pathway.J Cell Sci. 2002;115(Pt 21):3977-3978.
[5] Yu Q, Liu L, Duan Y, et al.Wnt/β-catenin signaling regulates neuronal differentiation of mesenchymal stem cells.Biochem Biophys Res Commun. 2013;439(2):297-302.
[6] Teo JL, Kahn M.The Wnt signaling pathway in cellular proliferation and differentiation: A tale of two coactivators.Adv Drug Deliv Rev. 2010;62(12):1149-1155.
[7] Clevers H, Nusse R.Wnt/β-catenin signaling and disease.Cell. 2012;149(6):1192-1205.
[8] Caraci F, Busceti C, Biagioni F,et al.The Wnt antagonist, Dickkopf-1, as a target for the treatment of neurodegenerative disorders.Neurochem Res. 2008;33(12):2401-2406.
[9] Komiya Y, Habas R.Wnt signal transduction pathways. Organogenesis. 2008;4(2):68-75.
[10] Nishita M, Itsukushima S, Nomachi A,et al.Ror2/Frizzled complex mediates Wnt5a-induced AP-1 activation by regulating Dishevelled polymerization.Mol Cell Biol. 2010; 30(14):3610-3619.
[11] Gebruers E, Cordero-Maldonado ML, Gray AI,et al.A phenotypic screen in zebrafish identifies a novel small-molecule inducer of ectopic tail formation suggestive of alterations in non-canonical Wnt/PCP signaling.PLoS One. 2013;8(12):e83293.
[12] De A.Wnt/Ca2+ signaling pathway: a brief overview.Acta Biochim Biophys Sin (Shanghai). 2011;43(10):745-756.
[13] Sassi N, Laadhar L, Allouche M,et al.WNT signaling and chondrocytes: from cell fate determination to osteoarthritis physiopathology.J Recept Signal Transduct Res. 2014;34(2): 73-80.
[14] Kim J, Kim DW, Chang W,et al.Wnt5a is secreted by follicular dendritic cells to protect germinal center B cells via Wnt/Ca2+/NFAT/NF-κB-B cell lymphoma 6 signaling.J Immunol. 2012;188(1):182-189.
[15] Olivares-Navarrete R, Hyzy SL, Park JH,et al.Mediation of osteogenic differentiation of human mesenchymal stem cells on titanium surfaces by a Wnt-integrin feedback loop. Biomaterials. 2011;32(27):6399-6411.
[16] Baek SH, Kioussi C, Briata P,et al.Regulated subset of G1 growth-control genes in response to derepression by the Wnt pathway.Proc Natl Acad Sci U S A. 2003;100(6): 3245-3250.
[17] Baksh D, Boland GM, Tuan RS. Cross-talk between Wnt signaling pathways in human mesenchymal stem cells leads to functional antagonism during osteogenic differentiation.J Cell Biochem. 2007;101(5):1109-1124.
[18] Baksh D, Tuan RS.Canonical and non-canonical Wnts differentially affect the development potential of primary isolate of human bone marrow mesenchymal stem cells.J Cell Physiol. 2007;212(3):817-826.
[19] Qiu W, Andersen TE, Bollerslev J,et al.Patients with high bone mass phenotype exhibit enhanced osteoblast differentiation and inhibition of adipogenesis of human mesenchymal stem cells.J Bone Miner Res. 2007;22(11):1720-1731.
[20] Gregory CA, Singh H, Perry AS,et al.The Wnt signaling inhibitor dickkopf-1 is required for reentry into the cell cycle of human adult stem cells from bone marrow.J Biol Chem. 2003; 278(30):28067-28078.
[21] De Boer J, Wang HJ, Van Blitterswijk C.Effects of Wnt signaling on proliferation and differentiation of human mesenchymal stem cells.Tissue Eng. 2004;10(3-4):393-401.
[22] Wallenfang MR.Aging within the Stem Cell niche.Dev Cell. 2007;13(5):603-604.
[23] 项晓霞,陈律,王骏浩,等. Wnt/β-catenin信号通路对间充质干细胞衰老的影响及其作用机制[J]. 浙江大学学报:医学版, 2012, 40(6): 630-640.
[24] Zhang DY, Wang HJ, Tan YZ. Wnt/β-catenin signaling induces the aging of mesenchymal stem cells through the DNA damage response and the p53/p21 pathway.PLoS One. 2011; 6(6):e21397.
[25] Grandori C, Wu KJ, Fernandez P,et al.Werner syndrome protein limits MYC-induced cellular senescence.Genes Dev. 2003;17(13):1569-1574.
[26] 刘荟敏. 3.5GyX射线对小鼠间充质干细胞的损伤及其机制的研究[D].长沙:华中科技大学, 2011.
[27] Ling L, Nurcombe V, Cool SM.Wnt signaling controls the fate of mesenchymal stem cells.Gene. 2009;433(1-2):1-7.
[28] Cho HH, Kim YJ, Kim SJ,et al.Endogenous Wnt signaling promotes proliferation and suppresses osteogenic differentiation in human adipose derived stromal cells.Tissue Eng. 2006;12(1):111-121.
[29] Si W, Kang Q, Luu HH,et al.CCN1/Cyr61 is regulated by the canonical Wnt signal and plays an important role in Wnt3A-induced osteoblast differentiation of mesenchymal stem cells.Mol Cell Biol. 2006;26(8):2955-2964.
[30] Eijken M, Meijer IM, Westbroek I,et al.Wnt signaling acts and is regulated in a human osteoblast differentiation dependent manner.J Cell Biochem. 2008;104(2):568-579.
[31] 焦雪峰,黄永灿,黄益洲,等.大鼠BMSCs自发钙化过程中成骨相关基因表达谱的分析[J].中国修复重建外科杂志, 2014,28(2): 133-141.
[32] Qiu W, Chen L, Kassem M. Activation of non-canonical Wnt/JNK pathway by Wnt3a is associated with differentiation fate determination of human bone marrow stromal (mesenchymal) stem cells.Biochem Biophys Res Commun. 2011;413(1):98-104.
[33] Sonomoto K, Yamaoka K, Oshita K,et al.Interleukin-1β induces differentiation of human mesenchymal stem cells into osteoblasts via the Wnt-5a/receptor tyrosine kinase-like orphan receptor 2 pathway.Arthritis Rheum. 2012;64(10): 3355-3363.
[34] Bolzoni M, Donofrio G, Storti P,et al.Myeloma cells inhibit non-canonical wnt co-receptor ror2 expression in human bone marrow osteoprogenitor cells: effect of wnt5a/ror2 pathway activation on the osteogenic differentiation impairment induced by myeloma cells.Leukemia. 2013;27(2): 451-463.
[35] Taipaleenmäki H, Abdallah BM, AlDahmash A,et al.Wnt signalling mediates the cross-talk between bone marrow derived pre-adipocytic and pre-osteoblastic cell populations. Exp Cell Res. 2011;317(6):745-756.
[36] Platt ID, El-Sohemy A.Regulation of osteoblast and adipocyte differentiation from human mesenchymal stem cells by conjugated linoleic acid.J Nutr Biochem. 2009;20(12): 956-964.
[37] Fu M, Rao M, Bouras T,et al.Cyclin D1 inhibits peroxisome proliferator-activated receptor gamma-mediated adipogenesis through histone deacetylase recruitment.J Biol Chem. 2005; 280(17):16934-16941.
[38] Christodoulides C, Lagathu C, Sethi JK,et al.Adipogenesis and WNT signalling.Trends Endocrinol Metab. 2009;20(1): 16-24.
[39] Takada I, Mihara M, Suzawa M,et al.A histone lysine methyltransferase activated by non-canonical Wnt signalling suppresses PPAR-gamma transactivation.Nat Cell Biol. 2007;9(11):1273-1285.
[40] Wislet-Gendebien S, Hans G, Leprince P,et al.Plasticity of cultured mesenchymal stem cells: switch from nestin-positive to excitable neuron-like phenotype.Stem Cells. 2005;23(3): 392-402.
[41] 阎文柱,秦书俭,刘学政,等.体外培养大鼠骨髓间充质干细胞向神经元样细胞分化: Wnt3a信号分子的诱导作用[J].中国组织工程研究,2010,14(14):2476-2480.
[42] 何丁文,殷明,邬亚华,等. Wnt/β-catenin信号通路在大鼠BMSCs神经分化中的作用研究[J].中国生物工程杂志,2013, 33(3): 61-67.
[43] Xu Y, Gu Z, Shen B,et al. Roles of Wnt/β-catenin signaling in retinal neuron-like differentiation of bone marrow mesenchymal stem cells from nonobese diabetic mice.J Mol Neurosci. 2013;49(2):250-261.
[44] 林颢. Wnt信号通路对人骨髓间充质干细胞神经分化作用的研究[D].广州:广东医学院,2011. |
| [1] | Shang Xiao-pan, Li Tao, Lu Yu-tong, Deng Wei, Wang Wen-ju, Yang Yong-jin. Molecular mechanism of magnesium ions in bone metabolism: research and progress [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(48): 7280-7287. |
| [2] | Wang Jian-ji, Yang Long, Li Jing, Sun Qi, Zuo Wei-min, Ren Qi-feng, Sun Yu, Wu Zhan-yu, Zou Qiang, Ma Min-xian, Ye Chuan. Development and application of special-purpose grafter by femoral head decompression combined with bone marrow mesenchymal stem cells transplantation based on three-dimensional printing technology [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6636-6642. |
| [3] | Zhou Chang-yan, Zhou Qing-huan, Bian Jing, Chen Ke, Chen Wen. Bone marrow mesenchymal stem cells combined with calcium phosphate cement to repair articular cartilage defects in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1195-1199. |
| [4] | Xu Xiang, Yin He-ping. Platelet-rich plasma accelerates the proliferation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2144-2148. |
| [5] | Sha Wen-qiong, She Rui-lian, Wang Zi-neng, Ke Ru. Ultrastructure and phagocytotic function of human placental mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2230-2235. |
| [6] | Du Qing-hua, Cao Jun-kai, Dong Xi-xi, E Ling-ling, Wei Li-jun. Osteogenic differentiation of pluripotent stem cells induced by akermanite extracts [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2236-2242. |
| [7] | Wu Yan, Huang Lan . Bone morphogenetic protein 9-induced osteogenic differentiation of dental follicle cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2255-2260. |
| [8] | Rao Li-jia, Li Qi-meng, Li Jin-ling, Xu Qiong. Expression pattern of ten-eleven translocation family during differentiation of human dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2261-2266. |
| [9] | Gao Zhuo-yue, Liu Yong-qi, He Jian-xin, Wu Zhi-wei, Luo Ya-li, Su Yun, Zhang Li-ying, Zhang Qi, Wu You-ming, Zhou Ni-na. Regulatory effects of warming yang and invigorating qi treatment on the inflammatory balance and genetic stability of bone marrow mesenchymal stem cells under tumor microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2267-2272. |
| [10] | Yao Ming-zhi. Effects of Jiegu Qili Tablet on proliferation and mineralization of MC-3T3 cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(11): 1694-1698. |
| [11] | Han Xiang-zhen, He Hui-yu, Hu Yang, Ba Jiao-jiao, Wang Huan-huan, Mi Xue, Abulizi•Abudula. Recombinant lentiviral vector transfected sheep bone marrow mesenchymal stem cells and osteogenic gene expression changes [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 821-828. |
| [12] | Huang Jian-feng, Huang Ji-feng, Zhang Wei-cai. Bone marrow mesenchymal stem cells differentiate into neuron-like cells induced by combination of two cytokines [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 829-834. |
| [13] | Zou Bin, Zong Shao-hui, Zeng Gao-feng, Fang Ye, Gao Tai-hang. Effects of alpha-zearalanol on the osteogenic differentiation of mouse bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 835-840. |
| [14] | Yang Yi, Ding Wen-jing, Dong Wan-li. Autophagy-related gene Beclin-1 expression in neuron-like differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 841-846. |
| [15] | Su Xue-lian, Bao Guang-jie, Kang Hong, Liu Lin, Kong Nan-nan. Morphological changes of goat bone marrow mesenchymal stem cells differentiating into fibrochondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 860-865. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

