Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (23): 3756-3761.doi: 10.3969/j.issn.2095-4344.2014.23.025
Previous Articles Next Articles
Inflamed dental pulp stem cells: initial research and future development
Zhao Hua-xiang1, Zhao Shan-mei2, Xin Xin1, Zhang Bo1, Ma Ning-hu1, Li Mu-jia1, Zhang Meng-qi1, Li Ang3
- 1Xi’an Jiaotong University Hospital of Stomatology, Xi’an 710004, Shaanxi Province, China; 2Department of Periodontology, Xi’an Jiaotong University Hospital of Stomatology, Xi’an 710004, Shaanxi Province, China; 3the Central Laboratory of Xi’an Jiaotong University Hospital of Stomatology, Xi’an 710004, Shaanxi Province, China
-
Revised:2014-05-04Online:2014-06-04Published:2014-06-04 -
Contact:Li Ang, Professor, Chief investigator, Doctoral supervisor, the Central Laboratory of Xi’an Jiaotong University Hospital of Stomatology, Xi’an 710004, Shaanxi Province, China -
About author:Zhao Hua-xiang, Xi’an Jiaotong University Hospital of Stomatology, Xi’an 710004, Shaanxi Province, China -
Supported by:the Scientific Research Training and Practical Innovation Project for Postgraduate of Xi’an Jiaotong University, No.2013289
CLC Number:
Cite this article
Zhao Hua-xiang, Zhao Shan-mei, Xin Xin, Zhang Bo, Ma Ning-hu, Li Mu-jia, Zhang Meng-qi, Li Ang . Inflamed dental pulp stem cells: initial research and future development[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(23): 3756-3761.
share this article
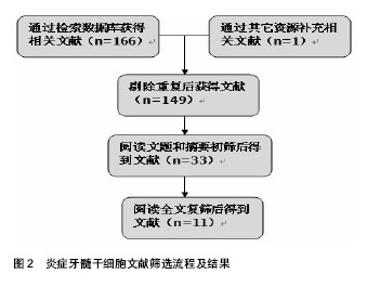
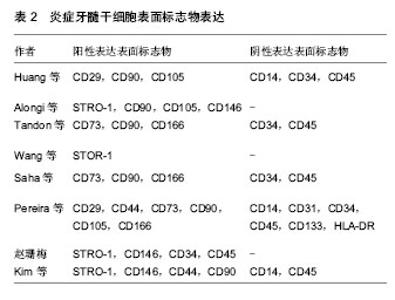
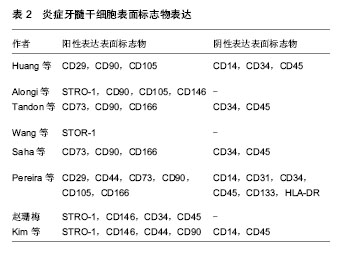
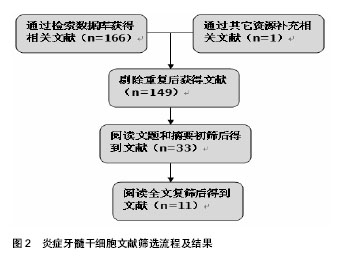
2.1 纳入研究的文献基本情况 初检得到文献167篇,其中英文文献131篇,中文文献36篇。经3位研究者(赵华翔、张博及辛欣)逐层筛选后,最终纳入11篇文献进行评价[10,14-23],文献筛选流程和结果见图2,纳入研究的文献见表1。 2.2 炎症牙髓干细胞的研究历史 2008年,Jiang等[24]报道了炎症牙髓组织比正常牙髓组织表达更多的SDF-1/CXCR4,而SDF-1/CXCR4作为一种趋化因子,在一些造血干/组细胞表面表达,并且促进骨髓基质干细胞的分化[25-26],这些提示了在炎症牙髓组织中也许存在着间充质干细胞。Huang等[14]于2009年从冠折露髓4 d的上颌恒切牙中分离并提取出了具有干细胞性质的细胞群落,并且首次应用拔髓提取而非传统的劈牙提取的方法成功分离出牙髓干细胞,这为今后从不可逆性牙髓炎的牙齿中提取牙髓奠定了技术基础。2010年,Alongi等[10]和Wang等[16]分别从临床诊断为不可逆性牙髓炎的恒牙牙髓中分离出干细胞;同年,Tandon等[15]从炎症乳牙牙髓中成功分离出了具有干细胞性质的细胞群落并进行了相关生物性能测试。其后,Saha等[17]详细描述了从炎症牙髓组织中提取分离干细胞的实验操作细节;Saha等[17]、Pereira等[19]及赵珊梅[21]分别进行了炎症牙髓干细胞与正常牙髓干细胞的生物学性能比较(包括自我扩增能力及多向分化能力)。Pereira等[18]、Wang等[22]和Kim等[23]在2012至2014年间探究了低强度激光(low-level laser therapy, LLLT)、矿物三氧化物(mineral trioxide aggregrate, MTA)及成纤维生长因子2(fibroblast growth factor-2, FGF-2)对于炎症牙髓干细胞的扩增和多向分化能力的影响;而Wang等[20]尝试从遭受缺血刺激后的牙髓干细胞中分离出侧群细胞(side population,SP)并进行生物学性能的分析。值得一提的是,在建立炎症牙髓干细胞动物模型方面,Wang等[22]于2013年利用人工暴露髓腔的方法成功建立了炎症牙髓干细胞的SD大鼠模型(Sprague–Dawley rats),并用于随后的实验研究。 2.3 炎症牙髓干细胞的来源与培养条件 迄今为止,研究者们已经成功从外伤露髓的恒切牙,临床诊断为不可逆性牙髓炎的非智齿恒磨牙、恒侧切牙、恒前磨牙以及人工制造的露髓动物模型中提取并分离得到了干细胞群落[10,14-22]。在不同的培养条件是否对炎症牙髓干细胞的培养存在影响这一问题上,学者们进行了诸多探究——Pereira等[18]发现低强度激光对炎症牙髓干细胞的扩增和分化能力并无明显影响。Wang等[22]发现低浓度(0.002-0.2 g/L)的矿物三氧化物对于炎症牙髓干细胞的扩增能力与对照组无差异,而高浓度的矿物三氧化物(>2 g/mL)则会减弱炎症牙髓干细胞的扩增能力。Kim等[23]证实经过成纤维生长因子2处理后的炎症牙髓干细胞在扩增能力上强于未经过成纤维生长因子2处理组;同时,他们发现在体外实验中,成纤维生长因子2抑制了细胞的成骨-成牙本质能力,却增强了细胞的成软骨能力,然而在成脂肪能力方面,各组实验数据并不统一;在体内组织工程植入物实验中,成纤维生长因子2使得炎症牙髓干细胞形成矿化组织的能力要远远强于未经过处理的对照组。 2.4 炎症牙髓干细胞的提取、初代培养及细胞扩增率 受临床操作、材料来源等条件的限制,不同于提取正常牙髓干细胞时所采用的拔牙后劈开提取完整牙髓的方法[2-3,27],大部分炎症牙髓干细胞的提取都需要采用口内直接拔髓的技术[10,14],这直接导致了原代培养材料来源容易受细菌污染。Alongi等[10]报道了经过抗生素处理的炎症牙髓干细胞的原代培养成功率(80%)显著高于未经过抗生素处理组的成功率(30%),作者前期的实验结果也证实了这一点[21]。因此,多种防止细菌污染的措施被应用到口内直接拔髓及原代培养操作过程中,包括患者在接受拔髓操作前使用洗必泰抗菌液漱口[15,17]、拔髓时使用橡皮障隔离[15,17,21]、多种联合抗生素培养等[16-17,21]。在细胞原代培养的成功率上,学者们报道差别较大,Huang等、Alongi等及Pereira 等[10,14,18]原代培养成功率超过80%,但Saha等[17]却认为从炎症牙髓中提取干细胞是一项技术敏感性工作[17]。原代培养单克隆形成被认为是成功分离干细胞的一项重要特征[10],在Wang等[16]和课题组前期的实验当 中[21],炎症牙髓细胞形成单克隆数/率显著小于正常牙髓组,而在Huang等[14]和Pereira等[19]的实验当中,炎症牙髓组织和正常牙髓组织之间并没有显著性差异。造成实验数据不统一的可能原因,一方面也许是炎症牙髓干细胞和正常牙髓干细胞之间可能存在的本身生物学性能的差异,另一方面牙髓干细胞供者与供牙的年龄、牙龄、机体状况等不同也是造成差异的潜在偏倚因素。在细胞扩增率检测实验当中,不同的检测方法被研究者们所采用,Alongi等[10]、Wang等[16]、Pereira等[18]及Kim等[23]发现炎症牙髓干细胞的扩增率低于正常牙髓组;而在另一项实验中,Pereira等[19]又得出了炎症牙髓组和正常牙髓组没有显著性差异的结论;相反的是,Huang等[14]发现从第3代到第7代培养当中,炎症牙髓干细胞的扩增率均要高于对应代数的正常牙髓干细胞组。 2.5 炎症牙髓干细胞的表面标志 国际细胞治疗学会指出间充质干细胞的一个重要特征就是阳性表达间充质表面标志物[28],阴性表达造血源性表面标志物。免疫组织化学、流式细胞术分析、免疫荧光等技术被应用于炎症牙髓干细胞的表面标志物的鉴定和与正常牙髓干细胞的比较,具体结果见表2[10,14-17,19,21-23]。在与正常牙髓来源的干细胞阳性表面标志物表达强度的比较上,Alongi等[10]观察到炎症牙髓干细胞比正常牙髓干细胞表达更强的STRO-1,CD90,CD105,CD146阳性表达,Kim等[23]观察到了正常牙髓干细胞组较炎症牙髓干细胞组表达更强的STRO-1,Perieira等[19]、赵珊梅[21]及Wang等[16]则报告了炎症牙髓干细胞和正常牙髓干细胞拥有相当的表面标志物阳性表达强度。 2.6 炎症牙髓干细胞的多向分化能力 纳入的文献都研究了炎症牙髓干细胞的多向分化能力[10,14-23],包括体外诱导后组织学观察、体外诱导后RT-PCR检测多向分化基因产物及体内组织工程植入物诱导实验。研究者们都认为炎症牙髓干细胞具有向成牙本质-成骨质分化[10,14-23]、成脂肪[10,14-15,17,19-21,23]、成软骨[14-15,17,19,21,23]、成神经[10,20]、成血管等方向发展的潜力[20]。Alongi 等[10]和课题组前期的研究[21]发现炎症牙髓干细胞的多向分化能力弱于正常牙髓干细胞,Tandon等[15]和Kim等[23]观察到炎症牙髓干细胞成牙本质-骨质、成软骨能力弱于正常牙髓组,然而在成脂肪能力上炎症牙髓组比正常牙髓组强或结果不统一。而Wang等[16]和Pereira等[19]则认为炎症牙髓干细胞和正常牙髓干细胞之间没有差异。在纳入的研究中,涉及成神经和成血管的诱导分化仅有Alongi等[10]和Wang等[20]进行过相关实验研究,考虑到牙髓组织中以血管和神经为主的解剖和组织结构[2,29],这也许是今后炎症牙髓干细胞多向分化诱导的研究方向之一。 2.7 炎症牙髓干细胞的免疫特征与亚群 干细胞是一类具有免疫调节功能的细胞[1,30-31],因其潜在的免疫抑制作用,干细胞在免疫治疗方面有着不可替代的重要地位[32],因而探究干细胞的免疫特征显得尤为重要。Dokic等[33]、Li等[34]和Vasandan等[35]探究了牙源性干细胞的免疫性质,然而,在本次纳入的对炎症牙髓干细胞的研究文献中,没有发现有任何学者对炎症牙髓干细胞的免疫性质进行研究[10,14-23]。干细胞又是一类具有多种亚群的祖细胞[36],因此分离和鉴别亚群是干细胞的研究方向之一,在这方面的研究工作中,Yang等[36]于2012年成功的从正常和炎症牙髓干细胞中分离出侧群细胞。 2.8 炎症牙髓干细胞的动物模型与临床应用前景 Wang 等[22]于2013年利用高速手机磨除SD大鼠的一侧切牙牙冠并在体外暴露72 h,成功建立炎症牙髓的啮齿类动物模型,同时在随后的实验中从这批动物模型中成功分离出了炎症牙髓干细胞并进行相关实验研究。动物模型成功建立使得炎症牙髓干细胞的获取不再拘泥于从患有不可逆性牙髓炎的临床患者口腔中提取牙髓,使得炎症牙髓干细胞的研究材料来源更加的广泛。同时,避免了因为供者与供牙的年龄、牙龄、机体状况等因素所造成的可能实验偏倚。然而不同于啮齿类动物牙齿不断生长的特性,人体的恒牙是终身不换的,因此二者牙髓干细胞之间的生物学特性是否相同值得商榷。人炎症牙髓干细胞与动物炎症牙髓干细胞之间的差异性即动物模型的转换性仍需要进一步的论证,更大型的哺乳类动物(如小型猪)的炎症牙髓干细胞模型的建立仍然是今后研究工作中需要进行的。 现有的研究已经证实了较高的增殖能力与多向分化能力使得牙髓干细胞可以在临床上应用于骨再生、神经再生、牙本质-牙髓再生等再生医学领域[5-8];作为具有免疫调节能力的干细胞使其在免疫系统等全身性疾病有着广泛应用[9]。虽然纳入的文献都没有涉及炎症牙髓干细胞在临床中应用的研究,但是作为牙髓干细胞的一种,它与正常牙髓干细胞一样有着良好的自我更新能力与多向分化潜能(异位成牙本质-成骨质、成脂肪、成软骨、成神经及成血管等),这些都是炎症牙髓干细胞能够于临床应用的基础。随着研究的进一步深入,炎症牙髓干细胞在临床中必将有着广阔而光明的应用前景。"
| [1] Uccelli A, Moretta L, Pistoia V.Mesenchymal stem cells in health and disease.Nat Rev Immunol. 2008;8(9):726-736.
[2] Gronthos S, Mankani M, Brahim J,et al.Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo.Proc Natl Acad Sci U S A. 2000;97(25):13625-13630.
[3] Huang GT, Sonoyama W, Chen J,et al.In vitro characterization of human dental pulp cells: various isolation methods and culturing environments.Cell Tissue Res. 2006;324(2):225-236.
[4] Yan X, Qin H, Qu C,et al.iPS cells reprogrammed from human mesenchymal-like stem/progenitor cells of dental tissue origin. Stem Cells Dev. 2010;19(4):469-480.
[5] d'Aquino R, De Rosa A, Lanza V,et al.Human mandible bone defect repair by the grafting of dental pulp stem/progenitor cells and collagen sponge biocomplexes.Eur Cell Mater. 2009; 18:75-83.
[6] Yamamoto A, Sakai K, Matsubara K,et al. Multifaceted neuro-regenerative activities of human dental pulp stem cells for functional recovery after spinal cord injury.Neurosci Res. 2014;78:16-20.
[7] Nakashima M, Iohara K, Sugiyama M.Human dental pulp stem cells with highly angiogenic and neurogenic potential for possible use in pulp regeneration.Cytokine Growth Factor Rev. 2009;20(5-6):435-440.
[8] Iohara K, Murakami M, Takeuchi N,et al.A novel combinatorial therapy with pulp stem cells and granulocyte colony-stimulating factor for total pulp regeneration.Stem Cells Transl Med. 2013;2(7):521-533.
[9] Leprince JG, Zeitlin BD, Tolar M,et al.Interactions between immune system and mesenchymal stem cells in dental pulp and periapical tissues.Int Endod J. 2012;45(8):689-701.
[10] Alongi DJ, Yamaza T, Song Y,et al.Stem/progenitor cells from inflamed human dental pulp retain tissue regeneration potential. Regen Med. 2010;5(4):617-631.
[11] Graziano A, d'Aquino R, Laino G,et al.Dental pulp stem cells: a promising tool for bone regeneration.Stem Cell Rev. 2008; 4(1):21-26.
[12] Telles PD, Machado MA, Sakai VT,et al.Pulp tissue from primary teeth: new source of stem cells.J Appl Oral Sci. 2011;19(3):189-194.
[13] Cook DJ, Mulrow CD, Haynes RB.Systematic reviews: synthesis of best evidence for clinical decisions.Ann Intern Med. 1997;126(5):376-380.
[14] Huang AH, Chen YK, Chan AW,et al. Isolation and characterization of human dental pulp stem/stromal cells from nonextracted crown-fractured teeth requiring root canal therapy. J Endod. 2009;35(5):673-681.
[15] Tandon S, Saha R, Rajendran R,et al.Dental pulp stem cells from primary and permanent teeth: quality analysis.J Clin Pediatr Dent. 2010;35(1):53-58.
[16] Wang Z, Pan J, Wright JT,et al.Putative stem cells in human dental pulp with irreversible pulpitis: an exploratory study.J Endod. 2010;36(5):820-825.
[17] Saha R, Tandon S, Rajendran R,et al.Dental pulp stem cells from primary teeth quality analysis: laboratory procedures.J Clin Pediatr Dent. 2011;36(2):167-173.
[18] Pereira LO, Longo JP, Azevedo RB.Laser irradiation did not increase the proliferation or the differentiation of stem cells from normal and inflamed dental pulp.Arch Oral Biol. 2012; 57(8):1079-1085.
[19] Pereira LO, Rubini MR, Silva JR,et al.Comparison of stem cell properties of cells isolated from normal and inflamed dental pulps.Int Endod J. 2012;45(12):1080-1090.
[20] Wang J, Wei X, Ling J,et al.Identification and characterization of side population cells from adult human dental pulp after ischemic culture.J Endod. 2012;38(11):1489-1497.
[21] 赵珊梅.人炎症及正常牙髓干细胞生物学性能的比较研究[D]. 西安:西安交通大学, 2012.
[22] Wang Y, Yan M, Fan Z,et al.Mineral trioxide aggregate enhances the odonto/osteogenic capacity of stem cells from inflammatory dental pulps via NF-κB pathway.Oral Dis. 2013. [Epub ahead of print]
[23] Kim J, Park JC, Kim SH,et al.Treatment of FGF-2 on stem cells from inflamed dental pulp tissue from human deciduous teeth.Oral Dis. 2014;20(2):191-204.
[24] Jiang HW, Ling JQ, Gong QM.The expression of stromal cell-derived factor 1 (SDF-1) in inflamed human dental pulp.J Endod. 2008;34(11):1351-1354.
[25] 杨志峰,杨清玲,陈昌杰.趋化因子SDF-1与受体CXCR4的研究进展[J].分子诊断与治疗杂志,2011, 3(1): 58-61.
[26] 冯柳,迟路湘. SDF-1促进BMSCs向血管内皮细胞分化[J]. 第三军医大学学报, 2013, 35(10): 951-955.
[27] Park SH, Hsiao GY, Huang GT.Role of substance P and calcitonin gene-related peptide in the regulation of interleukin-8 and monocyte chemotactic protein-1 expression in human dental pulp.Int Endod J. 2004;37(3):185-192.
[28] Dominici M, Le Blanc K, Mueller I,et al.Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement.Cytotherapy. 2006;8(4):315-317.
[29] 于世凤. 口腔组织病理学[M]. 6版.北京:人民卫生出版社, 2007: 64-69.
[30] Aggarwal S, Pittenger MF.Human mesenchymal stem cells modulate allogeneic immune cell responses.Blood. 2005; 105(4):1815-1822.
[31] Nauta AJ, Fibbe WE.Immunomodulatory properties of mesenchymal stromal cells.Blood. 2007;110(10):3499-3506.
[32] Parekkadan B, Milwid JM.Mesenchymal stem cells as therapeutics.Annu Rev Biomed Eng. 2010;12:87-117.
[33] Doki? J, Tomi? S, Cerovi? S,et al.Characterization and immunosuppressive properties of mesenchymal stem cells from periapical lesions.J Clin Periodontol. 2012;39(9): 807-816.
[34] Li Z, Jiang CM, An S,et al.Immunomodulatory properties of dental tissue-derived mesenchymal stem cells.Oral Dis. 2014;20(1):25-34.
[35] Vasandan AB, Shankar SR, Prasad P,et al.Functional differences in mesenchymal stromal cells from human dental pulp and periodontal ligament.J Cell Mol Med. 2014;18(2): 344-354.
[36] Yang R, Liu Y, Kelk P,et al.A subset of IL-17(+) mesenchymal stem cells possesses anti-Candida albicans effect.Cell Res. 2013;23(1):107-121.
[37] 霍永标,凌均棨.乳牙牙髓干细胞库的构建及其研究进展[J].国际口腔医学杂志, 2011, 38(2):188-191.
[38] Nakashima M, Akamine A.The application of tissue engineering to regeneration of pulp and dentin in endodontics. J Endod. 2005;31(10):711-718.
[39] Kinaia BM, Chogle SM, Kinaia AM,et al.Regenerative therapy: a periodontal-endodontic perspective.Dent Clin North Am. 2012; 56(3):537-547. |
| [1] | Wang Jian-ji, Yang Long, Li Jing, Sun Qi, Zuo Wei-min, Ren Qi-feng, Sun Yu, Wu Zhan-yu, Zou Qiang, Ma Min-xian, Ye Chuan. Development and application of special-purpose grafter by femoral head decompression combined with bone marrow mesenchymal stem cells transplantation based on three-dimensional printing technology [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6636-6642. |
| [2] | Zhou Chang-yan, Zhou Qing-huan, Bian Jing, Chen Ke, Chen Wen. Bone marrow mesenchymal stem cells combined with calcium phosphate cement to repair articular cartilage defects in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1195-1199. |
| [3] | He Yan-ping, Ma De-chun, Li Lei, Zhang Li, Zheng Shuang, Dong Ke-xin. Hemocompatibility and surface modification of artificial blood vessel materials [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1272-1276. |
| [4] | Wu Qiong, Hu Wei-ping. Clinical common problems after restoration with telescopic crown-retained removable partial dentures [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1301-1305. |
| [5] | Xu Xiang, Yin He-ping. Platelet-rich plasma accelerates the proliferation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2144-2148. |
| [6] | Ma Fa-ku, Wang Huan, Liu Bin, Yang Yan-li, Su Qin-jun, Qian Zhen, Dong Liang. Expression of CD44+/C-myc+ cancer stem cells and its relationship with the prognosis of patients in colorectal tumors [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2161-2166. |
| [7] |
Chen Ping, Wang Jian.
Enrichment of lung cancer stem cells and expression of related markers
[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2167-2171.
|
| [8] | Fan Zhi-gang, Fan Hong-liang. Oxidative stress response in diabetic nephropathy rats following injection of embryonic stem cells via the tail vein [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2199-2204. |
| [9] | Wang Yong, Zhao Wei, Feng Jian-zhou, Chen Xiao-chun. Nerve growth factor-modified adipose derived stem cells for repair of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2224-2229. |
| [10] | Sha Wen-qiong, She Rui-lian, Wang Zi-neng, Ke Ru. Ultrastructure and phagocytotic function of human placental mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2230-2235. |
| [11] | Du Qing-hua, Cao Jun-kai, Dong Xi-xi, E Ling-ling, Wei Li-jun. Osteogenic differentiation of pluripotent stem cells induced by akermanite extracts [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2236-2242. |
| [12] | Wu Yan, Huang Lan . Bone morphogenetic protein 9-induced osteogenic differentiation of dental follicle cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2255-2260. |
| [13] | Rao Li-jia, Li Qi-meng, Li Jin-ling, Xu Qiong. Expression pattern of ten-eleven translocation family during differentiation of human dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2261-2266. |
| [14] | Gao Zhuo-yue, Liu Yong-qi, He Jian-xin, Wu Zhi-wei, Luo Ya-li, Su Yun, Zhang Li-ying, Zhang Qi, Wu You-ming, Zhou Ni-na. Regulatory effects of warming yang and invigorating qi treatment on the inflammatory balance and genetic stability of bone marrow mesenchymal stem cells under tumor microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2267-2272. |
| [15] | Wang Fang, Chen Shao-wei. In vitro culture of embryos and establishment of embryonic stem cell lines [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2273-2277. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||