Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (32): 5819-5826.doi: 10.3969/j.issn.2095-4344.2013.32.012
Previous Articles Next Articles
Low-frequency electromagnetic fields enhance the recovery of spinal cord injured rats undergoing bone mesenchymal stem cell transplantation
Feng Yu1, Bai Wen-fang2, Xu Wei-cheng2, Li Xin-ping2, Bai Li-ming2, Liang Ling3, Wang Xin3, Zhang Ming-sheng2
- 1Graduate School, Southern Medical University, Guangzhou 510515, Guangdong Province, China
2Department of Rehabilitation Medicine, Guangdong General Hospital, Guangdong Provincial Institute of Geriatric Medicine, Guangdong Academy of Medical Sciences, Guangzhou 510080, Guangdong Province, China
3the First Affiliated Hospital of Jinan University, Guangzhou 510630, Guangdong Province, China
-
Received:2013-05-07Revised:2013-05-13Online:2013-08-06Published:2013-08-06 -
Contact:Zhang Ming-sheng, M.D., Chief physician, Professor, Doctoral supervisor, Department of Rehabilitation Medicine, Guangdong General Hospital, Guangdong Provincial Institute of Geriatric Medicine, Guangdong Academy of Medical Sciences, Guangzhou 510080, Guangdong Province, China mszrch@163.com -
About author:Feng Yu★, Studying for master’s degree, Graduate School, Southern Medical University, Guangzhou 510515, Guangdong Province, China fengyu567810@126.com -
Supported by:the Natural Science Foundation of Guangdong Province, No. S2012010009308*
CLC Number:
Cite this article
Feng Yu, Bai Wen-fang, Xu Wei-cheng, Li Xin-ping, Bai Li-ming, Liang Ling, Wang Xin, Zhang Ming-sheng . Low-frequency electromagnetic fields enhance the recovery of spinal cord injured rats undergoing bone mesenchymal stem cell transplantation[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(32): 5819-5826.
share this article
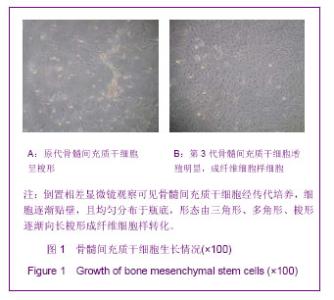
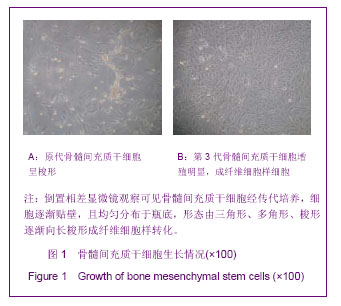
2.1 实验动物数量分析 在实验过程中,对照组、骨髓间充质干细胞组、电磁场组和电磁场+骨髓间充质干细胞组脊髓损伤大鼠在手术初期都不同程度的出现了神经损伤症状,手术当天BBB评分全部为0-2分,且有20只大鼠出现了血尿、乳糜尿。 在3周的观察期内,脊髓损伤对照组4只大鼠死亡,骨髓间充质干细胞组死亡7只,电磁场组死亡3只,电磁场+骨髓间充质干细胞组死亡6只。为保证实验例数,都已实时采用差额补充法补齐。 最终对照组、骨髓间充质干细胞组、电磁场组和电磁场+骨髓间充质干细胞组各16只大鼠进入结果分析。 2.2 骨髓间充质干细胞的形态 倒置相差显微镜下观察到骨髓间充质干细胞培养3 d时,细胞贴壁开始增殖,并向三角形、多角形、梭形等分化,见图1A。传代培养的第3代骨髓间充质干细胞均匀分布于瓶底,形态重新为长梭形成纤维细胞样,生长潜伏期较短,见图1B。"

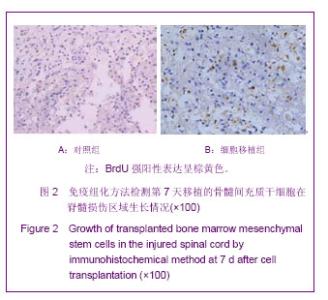
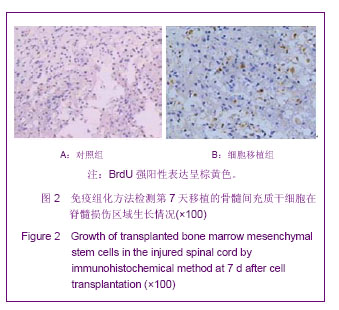
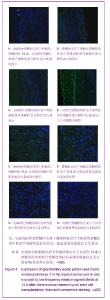
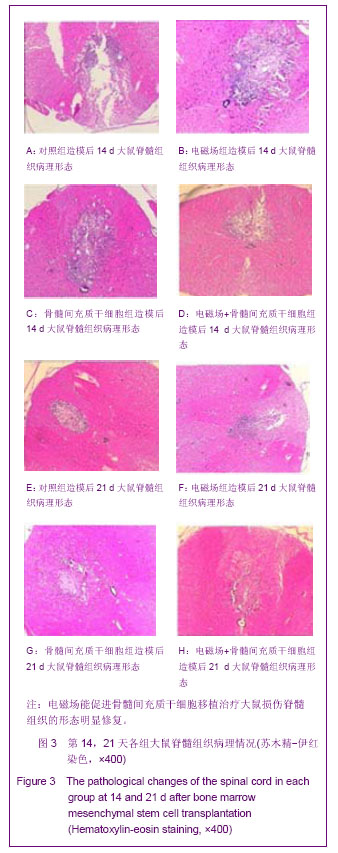
第7天,对照组和电磁场组脊髓组织结构破坏严重,损伤区有较多坏死液化吸收后形成的囊性空洞,神经细胞明显减少且变性、肿胀较重,周围有炎性细胞浸润,组织进一步肿胀,血流减少;骨髓间充质干细胞组和电磁场+骨髓间充质干细胞组损伤区囊性空洞面积较小、肿胀稍减轻,可见肥大的巨噬细胞和胶质细胞等,组织结构仍比较模糊。随着时间变化,如图3A-H所示,各组脊髓组织结构逐渐改善,第14天,除了对照组外,其他3组囊性空洞几乎闭合,被其它细胞组织所代替填充,骨髓间充质干细胞组组织水肿消失,仅有少量炎细胞浸润,电磁场+骨髓间充质干细胞组脊髓损伤结构已大部分修复,细胞排列有序,炎细胞基本消失。造模后第21天,除对照组外,其他各组损伤脊髓组织结构基本修复。 2.5 电磁场促进骨髓间充质干细胞移植治疗大鼠损伤脊髓中BrdU阳性细胞的生长及基质金属蛋白2的表达 免疫组化染色显示,对照组在脊髓损伤14 d和21 d脊髓损伤区见大量的胶质纤维酸性蛋白的表达;电磁场组和骨髓间充质干细胞组大鼠损伤脊髓中可见中等量胶质纤维酸性蛋白表达分布于损伤区周围;电磁场+骨髓间充质干细胞组大鼠损伤脊髓中胶质纤维酸性蛋白的表达最少,表明损伤局部胶质瘢痕组织最少,见图4A-D。 骨髓间充质干细胞组和电磁场+骨髓间充质干细胞组在脊髓损伤后7 d,大鼠损伤脊髓局部组织 基质金属蛋白2的表达量开始升高,14 d 最明显,见图4E-H。脊髓损伤后3-21 d,骨髓间充质干细胞组和电磁场+骨髓间充质干细胞组大鼠损伤脊髓组织中都可观察到BrdU阳性细胞,并与组织融合生长,电磁场+骨髓间充质干细胞组大鼠损伤脊髓中BrdU阳性细胞生长情况较好,对照组和电磁场组脊髓损伤部位未发现BrdU阳性细胞。"
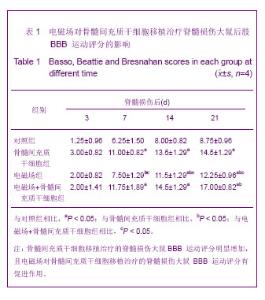
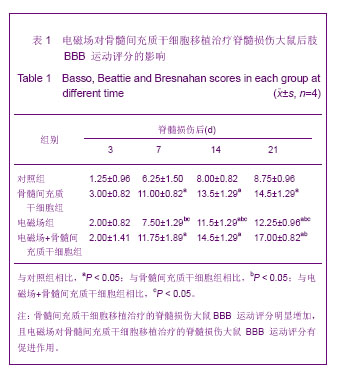
7 d后,各组BBB评分均有提高,电磁场+骨髓间充质干细胞组最高,骨髓间充质干细胞组次之,随之为电磁场组,对照组稍微改善,4组大鼠BBB评分差异有显著性意义(F=13.918,P=0.000),但对照组与电磁场组之间BBB评分差异无显著性意义(P=0.240),骨髓间充质干细胞组和电磁场+骨髓间充质干细胞组之间BBB评分差异无显著性意义(P=0.472);骨髓间充质干细胞移植后14 d,趋势同7 d,但除了骨髓间充质干细胞组和电磁场+骨髓间充质干细胞组之间BBB评分差异无显著性意义(P=0.258)外,其余各组之间差异都有显著性意义(P < 0.05);术后第21天,评分趋势同14 d,且效果更加显著,尤其是电磁场+骨髓间充质干细胞组,4组处理间BBB评分差异均有显著性意义(P < 0.01),见表1。"
| [1] 冯玉,张鸣生,李新平,等.骨髓间充质干细胞移植治疗脊髓损伤的实验研究现状[J].临床医学工程, 2011,18(10):1657-1659.[2] Vaquero J, Zurita M. Bone marrow stromal cells for spinal cord repair: a challenge for contemporary neurobiology. Histol Histopathol. 2009;24(1):107-116.[3] Furuya T, Hashimoto M, Koda M, et al. Treatment of rat spinal cord injury with a Rho-kinase inhibitor and bone marrow stromal cell transplantation. Brain Res. 2009;1295:192-202.[4] Ohta M, Suzuki Y, Noda T, et al. Bone marrow stromal cells infused into the cerebrospinal fluid promote functional recovery of the injured rat spinal cord with reduced cavity formation. Exp Neurol. 2004;187(2):266-278.[5] Bai WF, Zhang MS, Huang H, et al. Effects of 50 Hz electromagnetic fields on human epidermal stem cells cultured on collagen sponge scaffolds. Int J Radiat Biol. 2012; 88(7):523-530. [6] Wolf FI, Torsello A, Tedesco B, et al. 50-Hz extremely low frequency electromagnetic fields enhance cell proliferation and DNA damage: possible involvement of a redox mechanism. Biochim Biophys Acta. 2005;1743(1-2):120-129.[7] Yang Y, Li L, Wang YG, et al. Acute neuroprotective effects of extremely low-frequency electromagnetic fields after traumatic brain injury in rats. Neurosci Lett. 2012; 516(1):15-20. [8] Ito H, Bassett CA. Effect of weak, pulsing electromagnetic fields on neural regeneration in the rat. Clin Orthop Relat Res. 1983;(181):283-290.[9] Macias MY, Battocletti JH, Sutton CH, et al. Directed and enhanced neurite growth with pulsed magnetic field stimulation. Bioelectromagnetics. 2000;21(4):272-286.[10] Crowe MJ, Sun ZP, Battocletti JH, et al. Exposure to pulsed magnetic fields enhances motor recovery in cats after spinal cord injury. Spine (Phila Pa 1976). 2003;28(24):2660-2666.[11] 黄洁萍,翁金森,王锋,等.骨髓间充质干细胞移植对急性脊髓损伤大鼠神经功能恢复及Nogo-A表达的影响[J].中国康复医学杂志, 2009,24(7):582-587.[12] 邱家琦,张鸣生,李新平,等.低频电磁场对BDNF基因修饰的BMSCs增殖影响的实验研究[J].中国临床解剖学杂志, 2012, 30(1):74-78. [13] 王颖,冯世庆,赵鹏,等.大鼠脊髓损伤后不同时期移植人脐带间充质干细胞的修复效果观察[J].中国脊柱脊髓杂志, 2010, 20 (11): 918-925.[14] 于德水,吕刚,曹阳,等.BMSCs移植对大鼠脊髓损伤后VEGF基因和血管生成的影响[J].中国修复重建外科杂志,2011,25(7): 837-841.[15] Poirrier AL, Nyssen Y, Scholtes F, et al. Repetitive transcranial magnetic stimulation improves open field locomotor recovery after low but not high thoracic spinal cord compression-injury in adult rats. J Neurosci Res. 2004;75(2):253-261.[16] 王保金,刘长江.脊髓损伤模型的建立与应用[J].中国组织工程研究与临床康复, 2009,13(2):380-383.[17] Hagg T, Oudega M. Degenerative and spontaneous regenerative processes after spinal cord injury. J Neurotrauma. 2006;23(3-4):264-280.[18] Barnett SC, Riddell JS. Olfactory ensheathing cell transplantation as a strategy for spinal cord repair--what can it achieve? Nat Clin Pract Neurol. 2007;3(3):152-161.[19] Nandoe Tewarie RD, Hurtado A, et al. Bone marrow stromal cells for repair of the spinal cord: towards clinical application. Cell Transplant. 2006;15(7):563-577.[20] Alexanian AR, Fehlings MG, Zhang Z, et al. Transplanted neurally modified bone marrow-derived mesenchymal stem cells promote tissue protection and locomotor recovery in spinal cord injured rats. Neurorehabil Neural Repair. 2011; 25(9):873-880. [21] Wei X, Wen Y, Zhang T, et al. Effects of bone marrow mesenchymal stem cells with acellular muscle bioscaffolds on repair of acute hemi-transection spinal cord injury in rats. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2012;26(11): 1362-1368.[22] Ryu HH, Kang BJ, Park SS, et al. Comparison of mesenchymal stem cells derived from fat, bone marrow, Wharton's jelly, and umbilical cord blood for treating spinal cord injuries in dogs. J Vet Med Sci. 2012;74(12):1617-1630.[23] Yazdani SO, Pedram M, Hafizi M, et al. A comparison between neurally induced bone marrow derived mesenchymal stem cells and olfactory ensheathing glial cells to repair spinal cord injuries in rat. Tissue Cell. 2012;44(4):205-213.[24] Karamouzian S, Nematollahi-Mahani SN, Nakhaee N, et al. Clinical safety and primary efficacy of bone marrow mesenchymal cell transplantation in subacute spinal cord injured patients. Clin Neurol Neurosurg. 2012;114(7):935-939. [25] Karaoz E, Kabatas S, Duruksu G, et al. Reduction of lesion in injured rat spinal cord and partial functional recovery of motility after bone marrow derived mesenchymal stem cell transplantation. Turk Neurosurg. 2012;22(2):207-217. [26] Xu C. Transplantation of neural stem cells and bone marrow mesenchymal stem cells in treatment of spinal cord injury. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2012;26(2): 186-189.[27] Mahmood A, Lu D, Chopp M. Intravenous administration of marrow stromal cells (MSCs) increases the expression of growth factors in rat brain after traumatic brain injury. J Neurotrauma. 2004;21(1):33-39.[28] Wright KT, El Masri W, Osman A, et al. Concise review: Bone marrow for the treatment of spinal cord injury: mechanisms and clinical applications. Stem Cells. 2011;29(2):169-178.[29] Novikova LN, Brohlin M, Kingham PJ, et al. Neuroprotective and growth-promoting effects of bone marrow stromal cells after cervical spinal cord injury in adult rats. Cytotherapy. 2011;13(7):873-887. [30] Greenebaum B, Sutton CH, Vadula MS, et al. Effects of pulsed magnetic fields on neurite outgrowth from chick embryo dorsal root ganglia. Bioelectromagnetics. 1996;17(4):293-302.[31] Kang ES, Ha KY, Kim YH. Fate of transplanted bone marrow derived mesenchymal stem cells following spinal cord injury in rats by transplantation routes. J Korean Med Sci. 2012; 27(6):586-593. [32] Zeng X, Zeng YS, Ma YH, et al. Bone marrow mesenchymal stem cells in a three-dimensional gelatin sponge scaffold attenuate inflammation, promote angiogenesis, and reduce cavity formation in experimental spinal cord injury. Cell Transplant. 2011;20(11-12):1881-1899. [33] Wu W, Zhao H, Xie B, et al. Implanted spike wave electric stimulation promotes survival of the bone marrow mesenchymal stem cells and functional recovery in the spinal cord injured rats. Neurosci Lett. 2011;491(1):73-78. [34] Alexanian AR, Kwok WM, Pravdic D, et al. Survival of neurally induced mesenchymal cells may determine degree of motor recovery in injured spinal cord rats. Restor Neurol Neurosci. 2010;28(6):761-767. [35] Ding Y, Yan Q, Ruan JW, et al. Bone marrow mesenchymal stem cells and electroacupuncture downregulate the inhibitor molecules and promote the axonal regeneration in the transected spinal cord of rats. Cell Transplant. 2011;20(4): 475-491.[36] Cizkova D, Novotna I, Slovinska L, et al. Repetitive intrathecal catheter delivery of bone marrow mesenchymal stromal cells improves functional recovery in a rat model of contusive spinal cord injury. J Neurotrauma. 2011;28(9):1951-1961. [37] Björklund M, Koivunen E. Gelatinase-mediated migration and invasion of cancer cells. Biochim Biophys Acta. 2005;1755 (1):37-69. [38] Rosenberg GA, Dencoff JE, Correa N Jr, et al. Effect of steroids on CSF matrix metalloproteinases in multiple sclerosis: relation to blood-brain barrier injury. Neurology. 1996;46(6):1626-1632.[39] Yong VW. Metalloproteinases: mediators of pathology and regeneration in the CNS. Nat Rev Neurosci. 2005;6(12): 931-944.[40] Goussev S, Hsu JY, Lin Y, et al. Differential temporal expression of matrix metalloproteinases after spinal cord injury: relationship to revascularization and wound healing. J Neurosurg. 2003;99(2 Suppl):188-197.[41] Hsu JY, McKeon R, Goussev S, et al. Matrix metalloproteinase-2 facilitates wound healing events that promote functional recovery after spinal cord injury. J Neurosci. 2006;26(39):9841-9850.[42] Veeravalli KK, Dasari VR, Tsung AJ, et al. Human umbilical cord blood stem cells upregulate matrix metalloproteinase-2 in rats after spinal cord injury. Neurobiol Dis. 2009;36(1): 200-212. [43] Bi XB, Deng YB, Gan DH, et al. Salvianolic acid B promotes survival of transplanted mesenchymal stem cells in spinal cord-injured rats. Acta Pharmacol Sin. 2008;29(2):169-176.[44] Li X, Zhang M, Bai L, et al. Effects of 50 Hz pulsed electromagnetic fields on the growth and cell cycle arrest of mesenchymal stem cells: an in vitro study. Electromagn Biol Med. 2012;31(4):356-364. [45] Sun LY, Hsieh DK, Yu TC, et al. Effect of pulsed electromagnetic field on the proliferation and differentiation potential of human bone marrow mesenchymal stem cells. Bioelectromagnetics. 2009;30(4):251-260.[46] Park JE, Seo YK, Yoon HH, et al. Electromagnetic fields induce neural differentiation of human bone marrow derived mesenchymal stem cells via ROS mediated EGFR activation. Neurochem Int. 2013;62(4):418-424. [47] Bai WF, Xu WC, Feng Y, et al. Fifty-Hertz electromagnetic fields facilitate the induction of rat bone mesenchymal stromal cells to differentiate into functional neurons. Cytotherapy. 2013. in press. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Jiang Hongying, Zhu Liang, Yu Xi, Huang Jing, Xiang Xiaona, Lan Zhengyan, He Hongchen. Effect of platelet-rich plasma on pressure ulcers after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1149-1153. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [5] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [6] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [7] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [8] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [9] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [10] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [11] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [12] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [13] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [14] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [15] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||