Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (13): 2390-2398.doi: 10.3969/j.issn.2095-4344.2013.13.016
Previous Articles Next Articles
Dabigatran and rivaroxaban versus enoxaparin for anticoagulant therapy after total knee or hip arthroplasty
Ruan Jia-li, Lü Tao, Miao Jing-jing, Chen Ming-yi, Han Zhong-yu, Tian Jing
- Zhujiang Hospital of Southern Medical University, Guangzhou 510282, Guangdong Province, China
-
Received:2012-09-10Revised:2012-10-15Online:2013-03-26Published:2013-03-26 -
Contact:Tian Jing, Professor, Associate chief physician, Master’s supervisor, Zhujiang Hospital of Southern Medical University, Guangzhou 510282, Guangdong Province, China tian_jing6723@yahoo.com.cn -
About author:Ruan Jia-li, Zhujiang Hospital of Southern Medical University, Guangzhou 510282, Guangdong Province, China weriamo@qq.com
CLC Number:
Cite this article
Ruan Jia-li, Lü Tao, Miao Jing-jing, Chen Ming-yi, Han Zhong-yu, Tian Jing. Dabigatran and rivaroxaban versus enoxaparin for anticoagulant therapy after total knee or hip arthroplasty[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(13): 2390-2398.
share this article
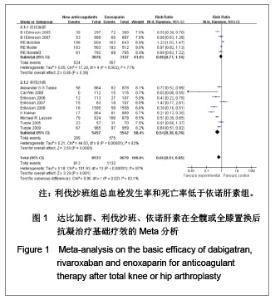
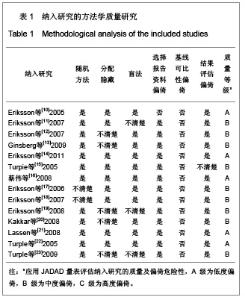
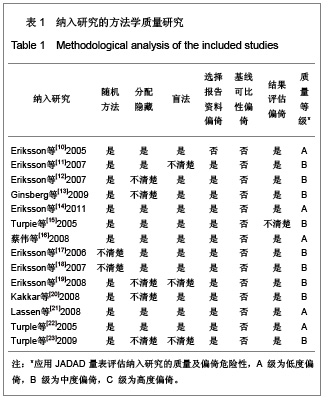
2.2.1 随机分配方法 除2个研究未明确提到具体的随机分配方法[17-18],具有选择性偏倚的中度可能性外,其余12个研究均提到了随机分配的具体方式[10-16,19-23],发生选择性偏倚的可能性低。 2.2.2 分配隐藏 有5个研究未明确提及运用了分配隐藏方案[12-13,19-20,23],有发生选择性偏倚的中度可能性。其他9个研究均采用分配隐藏方案[10-11,14-18,20-22],发生选择性偏倚的可能性低。 2.2.3 盲法 除3个研究未明确提及采用双盲法或单盲法实施试验[11,19,23],有发生实施偏倚或测量偏倚的中度可能性外,其余11个研究均采用双盲法实施[10,12-18,20-21],发生实施偏倚或测量偏倚的可能性低。 2.2.4 损耗性偏倚的评估 除1个研究未明确提及试验后随访及失访率/剔除率/退出率情况[10],有发生损耗性偏倚的中、高度可能性外,其余13个研究的试验后随访均较完善[11-23],失访率/剔除率/退出率均低于10%,发生耗损性偏倚的可能性低。 2.2.5 质量等级 根据以上质量评估,纳入的文献中质量为A级的有5篇[10,14,16,21-22],质量为B级的有9 篇[11-13,15,17-20,23],没有质量为C级的文献。 2.3 依诺肝素、达比加群、利伐沙班疗效的Meta分析 2.3.1 基础疗效的Meta分析 见图1。"
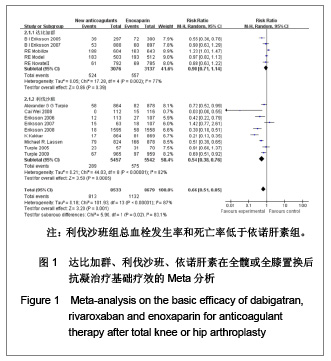
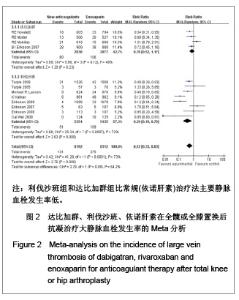
如图1所示,关于新型抗凝药达比加群和利伐沙班与依诺肝素对于全髋或全膝置换后抗凝疗效的评估方面,纳入并分析了14篇文献共17 212例患者,各研究之间有明显的异质性(χ2=101.93,P < 0.000 01,I2=87%),结果表明新药组明显优于依诺肝素组[RR=0.66,95%CI(0.51-0.85)]。 达比加群的总血栓发生率和死亡率:5篇文献共纳入 6 213例患者;各研究之间差异有显著性意义(χ2= 17.28,P=0.002,I2=77%),结果表明达比加群(220 mg, 1次/d)与依诺肝素(40 mg,1次/d)疗效差异无显著性意义[RR=0.90,95%CI(0.71-1.14)]。 利伐沙班的总血栓发生率和死亡率:9篇文献共纳入 10 999例患者;各研究之间差异具有显著性意义 (χ2=44.03,P < 0.000 01,I2=82%),结果表明利伐沙班(10 mg,1次/d)与依诺肝素(40 mg,1次/d)疗效差异有显著性意义[RR=0.54,95%CI(0.38-0.76)]。 2.3.2 主要静脉血栓形成率的Meta分析 见图2。"
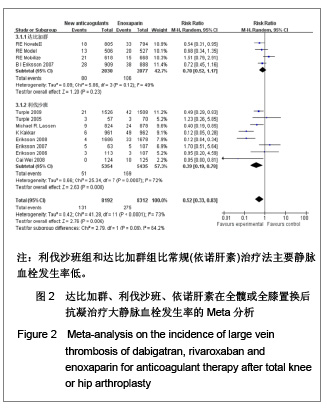
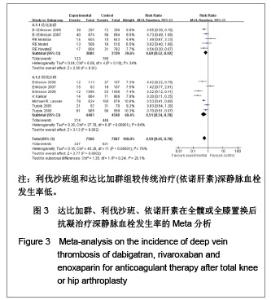
达比加群的主要静脉血栓发生率:4篇文献共纳入 5 715例患者;各研究间无明显异质性(χ2=5.86,P=0.12,I2=49%),结果表明达比加群(220 mg,1次/d)和依诺肝素(40 mg,1次/d)组主要静脉血栓发生率差异无显著性意义[RR=0.78,95%CI(0.52-1.17)]。 利伐沙班的主要静脉血栓发生率:8篇文献共纳入 10 789例患者;各研究间存在较大异质性(χ2=25.34,P=0.000 7,I2=72%),结果表明利伐沙班(40 mg, 1次/d)比依诺肝素(40 mg,1次/d)组主要静脉血栓发生率低[RR=039,95%CI(0.19-0.78)]。 新药组主要静脉血栓发生率:12篇文献共纳入16 504例患者;各研究间存在明显异质性(χ2= 41.28,P < 0.000 01,I2=73%),结果表明新药组[利伐沙班(10 mg,1次/d)和达比加群(220 mg,1次/d)]比常规(依诺肝素)治疗法主要静脉血栓发生率低[RR=0.52,95%(0.33-0.83)]。 2.3.3 深静脉血栓形成率的Meta分析 见图3。"
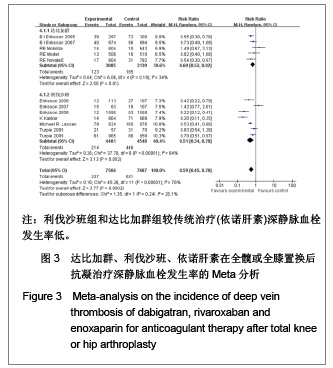
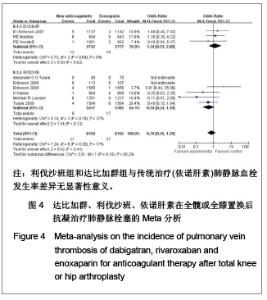
达比加群的深静脉血栓形成率:5篇文献共纳入6 224例患者;各研究间无明显异质性(χ2=6.06,P=0.19,I2=34%),结果表明达比加群(220 mg,1次/d)较依诺肝素(40 mg,1次/d)组深静脉血栓发生率低[RR=0.69,95%CI(0.52-0.92)]。 利伐沙班的深静脉血栓发生率:7篇文献共纳入9 029例患者;各研究间存在明显异质性(χ2=37.78,P < 0.000 01,I2=84%),结果表明利伐沙班(10 mg,1次/d)较依诺肝素(40 mg,1次/d)组深静脉血栓发生率低[RR=0.51,95%CI (0.34-0.78)]。 新药组深静脉血栓发生率:12篇文献共纳入15 253例患者;各研究间存在明显异质性(χ2=45.26,P < 0.000 01,I2=76%)。结果表明新药组较传统治疗(依诺肝素40 mg, 1次/d)深静脉血栓发生率低[RR=0.59,95%CI (0.45-0.78)]。 2.3.4 肺血栓栓塞形成率的Meta分析 见图4。"
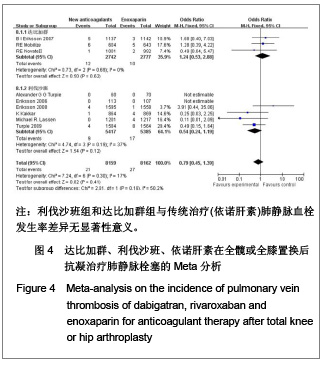

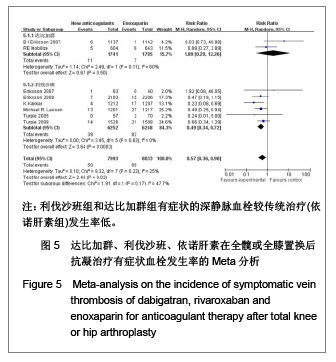
达比加群的肺血栓栓塞形成率:3篇文献共纳入5 519例患者;各研究间无明显异质性(χ2=0.73,P=0.69,I2=0%),结果表明达比加群(220 mg,1次/d)与依诺肝素(40 mg,1次/d)组肺静脉血栓发生率差异无显著性意义[RR=1.24,95%CI(0.53-2.88)]。 利伐沙班的肺血栓栓塞发生率:6篇文献共纳入10 802例患者;各研究间无明显异质性(χ2=4.74,P=0.19,I2=37%),结果表明利伐沙班(10 mg,1次/d)与依诺肝素(40 mg,1次/d)组肺静脉血栓发生率差异无显著性意义[RR=0.54,95%CI (0.24-1.19]。 新药组的肺血栓栓塞发生率:12篇文献共纳入16 321例患者;各研究间无明显异质性(χ2=7.24,P=0.30,I2=17%),结果表明新药组与传统治疗(依诺肝素40 mg, 1次/d)肺静脉血栓发生率差异无显著性意义[RR=0.79,95%CI(0.45-1.39]。 2.3.5 有症状的深静脉血栓发生率的Meta分析 见图5。 达比加群的有症状深静脉血栓形成率:2篇文献共纳入 3 526例患者;各研究间存在明显异质性(χ2=2.49,P=0.11,I2=60%),结果表明达比加群(220 mg,1次/d)和依诺肝素(40 mg,1次/d)组主要静脉血栓发生率差异无显著性意义[RR=1.89,95%CI(0.29-12.26)]。 利伐沙班的有症状深静脉血栓发生率:6篇文献共纳入12 500例患者;各研究间无明显异质性(χ2=3.45,P=0.63,I2=0%),结果表明利伐沙班(10 mg,1次/d)较依诺肝素(40 mg,1次/d)组有症状的深静脉血栓发生率低[RR=0.49,95%CI (0.34-0.72)]。 新药组的有症状深静脉血栓发生率:8篇文献共纳入 16 026例患者;各研究间无明显异质性(χ2=9.32,P=0.23,I2=25%),结果表明新药组有症状的深静脉血栓较传统治疗(依诺肝素组)发生率低[RR=0.57,95%CI (0.36-0.90)]。"
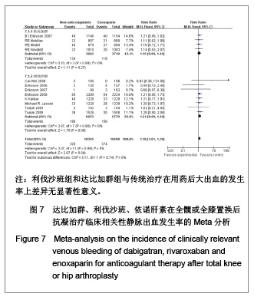
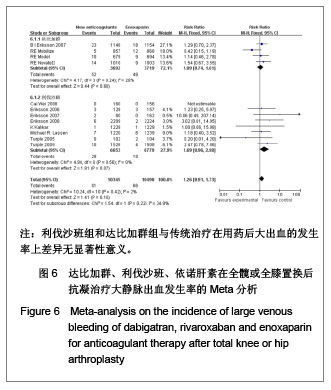
2.4 依诺肝素,达比加群,利伐沙班安全性的Meta分析 2.4.1 用药后大出血发生率的Meta分析 见图6。 达比加群用药后大出血的发生率:4篇文献共纳入7 411例患者;各研究组无明显异质性(χ2=4.17,P=0.24,I2=28%),结果表明达比加群(220 mg,1次/d)组与依诺肝素(40 mg,1次/d)组在大出血发生率上差异无显著性意义[RR=1.09,95%CI(0.74-1.61)]。 利伐沙班用药后大出血的发生率:8篇文献共纳入 13 432例患者;各研究组间无明显异质性(χ2=4.84,P=0.56,I2=0%),结果表明利伐沙班(10 mg,1次/d)组与依诺肝素(40 mg,1次/d)组在大出血发生率上差异无显著性意义[RR=1.69,95%CI(0.96-2.98)]。 新药组用药后大出血的发生率:12篇文献共纳入20 843例患者;各研究组之间无明显异质性(χ2=10.24,P=0.16,I2=2%),结果表明新药组与传统治疗在用药后大出血的发生率上差异无显著性意义[RR=1.26,95%CI (0.91-1.73)]。"

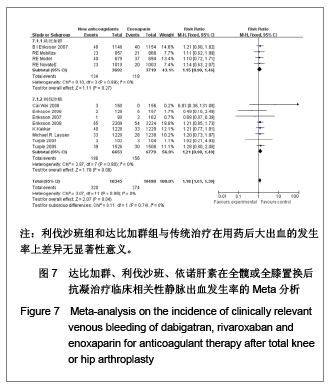
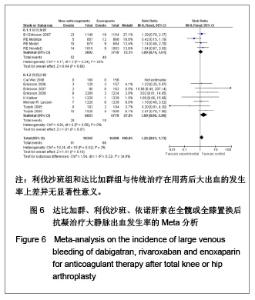
达比加群临床相关的少量出血的发生率:4篇文献共纳入7 411例患者;各研究组无明显异质性(χ2=0.10,P=0.99,I2=0%),结果表明达比加群(220 mg,1次/d)组与依诺肝素(40 mg,1次/d)组在临床相关少量出血发生率差异无显著性意义[RR=1.15,95%CI(0.90-1.46)]。 利伐沙班临床相关的少量出血的发生率:8篇文献共纳入13 432例患者;各研究组间无明显异质性(χ2=2.87,P=0.90,I2=0%),结果表明利伐沙班(10 mg,1次/d)组与依诺肝素(40 mg,1次/d)组在临床相关少量出血发生率上差异无显著性意义[RR=1.21,95%CI(0.98-1.49)]。 新药组临床相关的少量出血的发生率:12篇文献共纳入20 843例患者;各研究组之间无明显异质性(χ2=3.07,P=0.99,I2=0%),结果表明新药组与传统治疗在用药后大出血的发生率上差异无显著性意义[RR=1.18,95%CI (1.01-1.39)]。 2.5 敏感性分析 鉴于文献质量的异质性和临床实验数据的选择,可能使统计结果产生一定的偏倚。作者对该荟萃分析进行了敏感性分析,结论如下:仅纳入明确采用盲法的随机对照试验,不会使本荟萃分析的有效性和安全性产生明显差异;剔除低级质量的文献后重新进行统计学分析不影响荟萃分析的最终结果;若同时纳入30,40 mg剂量组的依诺肝素进行比较,分析所得有效性和安全性差异无显著性意义。由此,可以认为该荟萃分析纳入的临床试验之间的异质性对所需的统计结果造成的偏倚在可接受范围之内。"

| [1] Sobieraj DM, Coleman CI, Tongbram V, et al. Venous Thromboembolism Prophylaxis in Orthopedic Surgery [Internet]. Cells Tissues Organs. 2003;174(3):101-109.[2] Llau JV, Gil-Garay E, Castellet E, et al. [Thromboprophylaxis with enoxaparin for total knee replacement: An observational, retrospective and multicentre study comparing starting the treatment before and after the operation]. Rev Esp Anestesiol Reanim. 2012;59(6): 306-314.[3] Carrier M, Cushman M. Assessing the utility of venous thrombosis prophylaxis in orthopedic surgery patients. Ann Intern Med. 2012;156(10): 748-749.[4] Bosque J Jr, Coleman SI, Di Cesare P.Relationship between deep vein thrombosis and pulmonary embolism following THA and TKA. Orthopedics. 2012;35(3): 228-233; quiz 234-235.[5] Enokiya T, Muraki Y, Iwamoto T, et al. Risk factor for a residual deep vein thrombosis after fondaparinux administration in patient with postoperative replacement arthroplasty. Yakugaku Zasshi. 2012;132(5): 683-687.[6] Aaron RK, Boyan BD, Ciombor DM, et al. Stimulation of growth factor synthesis by electric and electromagnetic fields. Clin Orthop Relat Res. 2004;(419): 30-37.[7] Ufer M. Comparative efficacy and safety of the novel oral anticoagulants dabigatran, rivaroxaban and apixaban in preclinical and clinical development. Thromb Haemost. 2010; 103(3): 572-585.[8] Anderson FA Jr, Zayaruzny M, Heit JA, et al. Estimated annual numbers of US acute-care hospital patients at risk for venous thromboembolism. Am J Hematol. 2007;82(9): 777-782.[9] Cao YB, Zhang JD, Shen H, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after total hip or knee arthroplasty: a meta-analysis of randomized controlled trials. Eur J Clin Pharmacol. 2010;66(11): 1099-1108.[10] Eriksson BI, Dahl OE, Büller HR, et al. A new oral direct thrombin inhibitor, dabigatran etexilate, compared with enoxaparin for prevention of thromboembolic events following total hip or knee replacement: the BISTRO II randomized trial. J Thromb Haemost. 2005;3(1): 103-111.[11] Eriksson BI, Dahl OE, Rosencher N, et al. Oral dabigatran etexilate vs. subcutaneous enoxaparin for the prevention of venous thromboembolism after total knee replacement: the RE-MODEL randomized trial. J Thromb Haemost. 2007;5(11): 2178-2185.[12] Eriksson BI, Dahl OE, Rosencher N, et al. Dabigatran etexilate versus enoxaparin for prevention of venous thromboembolism after total hip replacement: a randomised, double-blind, non-inferiority trial. Lancet. 2007;370(9591): 949-956.[13] Ginsberg JS, Davidson BL, RE-MOBILIZE Writing Committee, et al. Oral thrombin inhibitor dabigatran etexilate vs North American enoxaparin regimen for prevention of venous thromboembolism after knee arthroplasty surgery. J Arthroplasty. 2009;24(1): 1-9.[14] Eriksson BI, Dahl OE, Huo MH, et al. Oral dabigatran versus enoxaparin for thromboprophylaxis after primary total hip arthroplasty (RE-NOVATE II*). A randomised, double-blind, non-inferiority trial. Thromb Haemost. 2011;105(4): 721-729.[15] Turpie AG, Fisher WD, Bauer KA, et al. BAY 59-7939: an oral, direct factor Xa inhibitor for the prevention of venous thromboembolism in patients after total knee replacement. A phase II dose-ranging study. J Thromb Haemost. 2005; 3(11): 2479-2486.[16] 柴伟,王岩.利伐沙班预防成人全髋关节置换术后静脉血栓栓塞的有效性及安全性研究[D].中国人民解放军军医进修学院, 2008.[17] Eriksson BI, Borris L, Dahl OE, et al. Oral, direct Factor Xa inhibition with BAY 59-7939 for the prevention of venous thromboembolism after total hip replacement. J Thromb Haemost. 2006;4(1): 121-128.[18] Eriksson BI, Borris LC, Dahl OE, et al. Dose-escalation study of rivaroxaban (BAY 59-7939)--an oral, direct Factor Xa inhibitor--for the prevention of venous thromboembolism in patients undergoing total hip replacement. Thromb Res. 2007; 120(5): 685-693.[19] Eriksson BI, Borris LC, Friedman RJ, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N Engl J Med. 2008;358(26): 2765-2775.[20] Kakkar AK, Brenner B, Dahl OE, et al., Extended duration rivaroxaban versus short-term enoxaparin for the prevention of venous thromboembolism after total hip arthroplasty: a double-blind, randomised controlled trial. Lancet. 2008;372 (9632): 31-39.[21] Lassen MR, Ageno W, Borris LC, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty. N Engl J Med. 2008;358(26): 2776-2786.[22] Turpie AG, Fisher WD, Bauer KA, et al. BAY 59-7939: an oral, direct factor Xa inhibitor for the prevention of venous thromboembolism in patients after total knee replacement. A phase II dose-ranging study. J Thromb Haemost. 2005;3(11): 2479-2486.[23] Turpie AG, Lassen MR, Davidson BL, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty (RECORD4): a randomised trial. Lancet. 2009; 373(9676): 1673-1680.[24] Rahman S. Deep vein thrombosis prophylaxis: friend or foe. Am J Ther. 2009;16(4): 300-303.[25] Anderson FA Jr, Zayaruzny M, Heit JA, et al. Estimated annual numbers of US acute-care hospital patients at risk for venous thromboembolism. Am J Hematol. 2007;82(9): 777-782.[26] Galson SK. Prevent deep vein thrombosis and pulmonary embolism with a healthful diet. J Am Diet Assoc. 2009;109(4): 592.[27] Goldhaber SZ, Fanikos J. Cardiology patient pages. Prevention of deep vein thrombosis and pulmonary embolism. Circulation. 2004;110(16): e445-447.[28] Leizorovicz A, Cohen AT, Turpie AG, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110(7): 874-849.[29] Turun S, Banghua L, Yuan Y, et al. A systematic review of rivaroxaban versus enoxaparin in the prevention of venous thromboembolism after hip or knee replacement. Thromb Res. 2011;127(6): 525-534.[30] Ansell J, Hirsh J, Poller L, et al. The pharmacology and management of the vitamin K antagonists: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest.2004;126(3 Suppl): 204S-233S. |
| [1] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [2] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| [3] | Chen Junming, Yue Chen, He Peilin, Zhang Juntao, Sun Moyuan, Liu Youwen. Hip arthroplasty versus proximal femoral nail antirotation for intertrochanteric fractures in older adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1452-1457. |
| [4] | Chen Jinping, Li Kui, Chen Qian, Guo Haoran, Zhang Yingbo, Wei Peng. Meta-analysis of the efficacy and safety of tranexamic acid in open spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1458-1464. |
| [5] | Hu Kai, Qiao Xiaohong, Zhang Yonghong, Wang Dong, Qin Sihe. Treatment of displaced intra-articular calcaneal fractures with cannulated screws and plates: a meta-analysis of 15 randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1465-1470. |
| [6] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [7] | Wang Yongsheng, Wu Yang, Li Yanchun. Effect of acute high-intensity exercise on appetite hormones in adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1305-1312. |
| [8] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [9] | Zhao Zhongyi, Li Yongzhen, Chen Feng, Ji Aiyu. Comparison of total knee arthroplasty and unicompartmental knee arthroplasty in treatment of traumatic osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 854-859. |
| [10] | Yuan Jun, Yang Jiafu. Hemostatic effect of topical tranexamic acid infiltration in cementless total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 873-877. |
| [11] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Intravenous, topical tranexamic acid alone or their combination in total knee arthroplasty: a meta-analysis of randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 948-956. |
| [12] | Li Yan, Wang Pei, Deng Donghuan, Yan Wei, Li Lei, Jiang Hongjiang. Electroacupuncture for pain control after total knee arthroplasty: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 957-963. |
| [13] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [14] | Hua Haotian, Zhao Wenyu, Zhang Lei, Bai Wenbo, Wang Xinwei. Meta-analysis of clinical efficacy and safety of antibiotic artificial bone in the treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 970-976. |
| [15] | Zhan Fangbiao, Cheng Jun, Zou Xinsen, Long Jie, Xie Lizhong, Deng Qianrong. Intraoperative intravenous application of tranexamic acid reduces perioperative bleeding in multilevel posterior spinal surgery: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 977-984. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||