Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (1): 148-153.doi: 10.3969/j.issn.2095-4344.1855
Previous Articles Next Articles
Research progress of mesenchymal stem cells in treating lung injury
Zhang Yunqiang, Ke Xixian, Zhang Lingtao, Liang Guiyou
- Department of Cardiovascular Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2019-06-03Revised:2019-06-11Accepted:2019-07-05Online:2020-01-08Published:2019-12-13 -
Contact:Liang Guiyou, MD, Professor, Doctoral supervisor, Department of Cardiovascular Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Zhang Yunqiang, Master candidate, Department of Cardiovascular Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the Science and Technology Support Program of Guizhou Province, No. [2017] 2969; High-level Talents Project of Guizhou Province, No. [2016]4034; the Science and Technology Plan Project of Zunyi, No. [2018] 69; the Project of Zunyi Medical University, No. [2017] 5733-023
CLC Number:
Cite this article
Zhang Yunqiang, Ke Xixian, Zhang Lingtao, Liang Guiyou. Research progress of mesenchymal stem cells in treating lung injury[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(1): 148-153.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
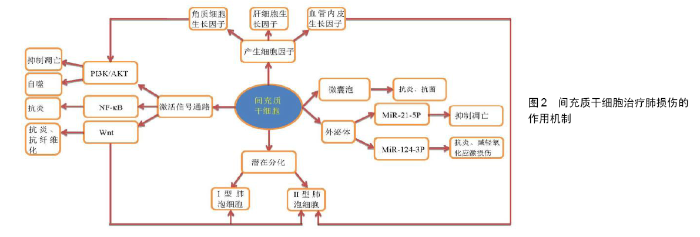
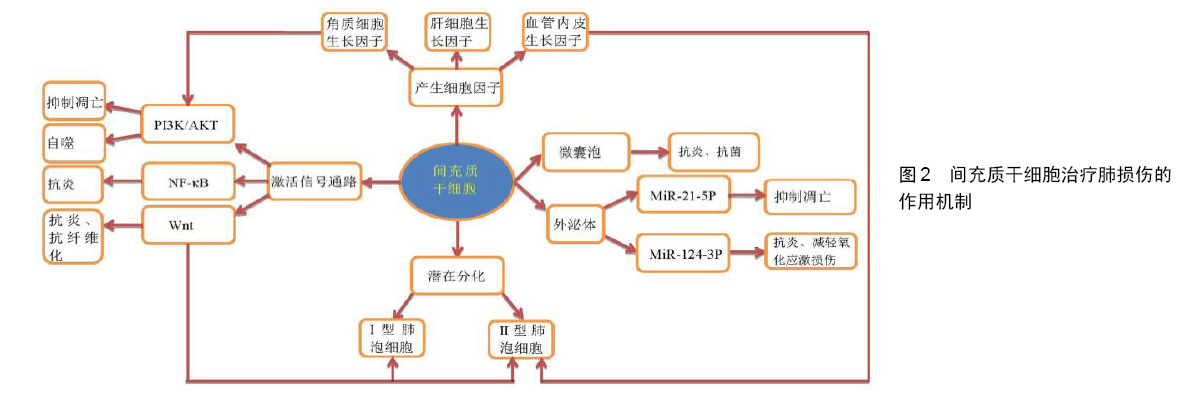
2.1 间充质干细胞 2.1.1 间充质干细胞的生物学特性 间充质干细胞属于多能干细胞,具有多向分化潜能、造血支持和促进干细胞植入、免疫调控以及自我复制、旁分泌等特点,来源于多种组织类型。在临床试验中,间充质干细胞主要来源于成人骨髓,其次是脂肪组织,还可来源于脐带组织和胎盘组织的产褥丢弃物[2-4]。国际细胞疗法协会的间充质和组织干细胞委员会定义了关于人类间充质干细胞的基本标准:首先,在标准条件下培养时,细胞必须是贴壁生长;其次,间充质干细胞表面表达CD105、CD73和CD90,不表达CD45、CD34、CD14或CD11b、CD79a或CD19及HLA-DR;同时,间充质干细胞在体外可分化为成骨细胞、脂肪细胞和成软骨细胞[5]。 2.1.2 间充质干细胞治疗肺损伤的优势 在细胞治疗的背景下,间充质干细胞与其他候选细胞类型(如胚胎干细胞或诱导多能干细胞)相比具有易获得、被自如使用、无需复杂体外操作以及通常具有低致瘤性风险等几个优点[5]。而且,间充质干细胞经静脉注射后首先归巢于肺中[6-7],这利于更好地发挥治疗肺损伤的作用。 2.2 间充质干细胞治疗肺损伤的机制 目前,就间充质干细胞治疗肺损伤的研究机制很多,如PI3K/AKT信号通路、Wnt信号通路、NF-κB信号通路、产生的细胞因子、外泌体以及潜在分化趋势机制等,见图2,它们在治疗肺损伤方面有很大优势。 2.2.1 信号通路 (1)磷脂酰肌醇3-激酶/蛋白激酶B(PI3K/AKT)信号通路:PI3K是胞内脂质激酶家族成员,可以使磷脂酰肌醇和磷酸肌醇的3'-羟基基团磷酸化,具有调节细胞代谢、存活和极性以及调控囊泡运输的功能[8-9]。PI3Ks是胰岛素和生长因子反应的关键成分,它调节新陈代谢和细胞生长[8-11],活化第二信使,参与细胞分化、增殖、凋亡以及迁徙等[13-14]。CHU等[10]报道通过激活PI3K/AKT信号通路来改善黑炭造成的肺损伤。在缺血再灌注损伤诱导的小鼠模型和氧葡萄糖剥夺/复氧诱导的人肺微血管内皮细胞模型中,骨髓间充质干细胞诱导自噬激活与PI3K/AKT信号通路有关,骨髓间充质干细胞移植可上调自噬诱导因子(包括PI3KⅢ类和LC3-Ⅱ)以及下调负调节因子(PI3KⅠ类)的表达,通过PI3K/AKT信号通路改善自噬水平,减轻肺损伤[11]。同时,还有学者报道自噬在调节间充质干细胞对氧化应激的反应中起着重要的作用[12]。在骨髓间充质干细胞减少炎性细胞因子对Ⅱ型肺泡细胞(alveolar type II epithelial cell,AT-Ⅱcell)损伤的机制研究中,可通过角质细胞生长因子(keratinocyte growth factor,KGF)依赖的PI3K/AKT/ MTOR信号通路改善受损Ⅱ型肺泡细胞的炎症反应来治疗肺损伤[13]。LIN等[14]在诱导大鼠哮喘模型中发现间充质干细胞移植组的气道炎症程度和气道重塑程度明显受到抑制,且间充质干细胞移植后抑制了肺组织中磷酸化AKT(p-AKT)的表达,这表明间充质干细胞移植可通过PI3K/AKT信号通路在大鼠哮喘模型中抑制肺部炎症和气道重塑。在香烟烟雾诱导的肺损伤研究中,使用间充质干细胞条件培养基处理后可通过PI3K/AKT途径抑制细胞凋亡和促进肺成纤维细胞增殖来实现肺损伤修复[15]。以上研究说明了PI3K/AKT信号通路在肺损伤中起重要作用,间充质干细胞可以通过激活该途径进而发挥保护肺损伤的作用。 (2)Wnt信号通路:Wnt信号通路在肺泡细胞的发育生长以及损伤修复中起到了重要的作用,在Wnt/β-连环蛋白(Wnt/β-catenin)信号通路中,Wnt表达增加会抑制β-连环蛋白的产生[6,16-18]。ZHANG等[6]在光气诱导的鼠肺损伤模型中,转化生长因子β1和Wnt5a的表达与Wnt3a/β-连环蛋白信号通路的抑制相关,外源性间充质干细胞归巢于受损的肺组织,通过阻断转化生长因子β1和wnt5介导的Wnt3/β-连环蛋白信号传导的抑制而降低上皮细胞通透性,这表明Wnt/β-连环蛋白信号传导途径在肺修复中起重要作用。在大鼠骨髓间充质干细胞与脂多糖诱导巨噬细胞凋亡的体外细胞共培养实验研究中发现,骨髓间充质干细胞减少了巨噬细胞以适当比例共培养时的细胞凋亡,骨髓间充质干细胞通过抑制Wnt/β-连环蛋白途径部分减弱巨噬细胞凋亡,降低了caspase-3和促凋亡蛋白Bax的表达,增加抗凋亡蛋白Bcl-2的表达,延长了巨噬细胞的体外寿命,共培养系统中骨髓间充质干细胞可逆转巨噬细胞中Wnt/β-连环蛋白信号通路的激活[16]。在小鼠间充质干细胞与小鼠肺上皮-12细胞的小气道生长培养基共培养实验中,间充质干细胞分化为Ⅱ型肺泡上皮细胞与Wnt信号通路有关,间充质干细胞的增殖性、氧化应激诱导下细胞凋亡的降低以及诱导其向受损的肺组织迁移均与Wnt信号通路相关,同时Wnt信号通路也参与间充质干细胞分化为Ⅱ型肺泡上皮细胞[17]。在SD大鼠急性肺损伤模型中,Wnt信号通路在骨髓间充质干细胞分化为Ⅰ型肺泡上皮细胞中起重要作用,Wnt-1和Wnt-3α信号通路在骨髓间充质干细胞分化为Ⅰ型肺泡上皮细胞过程中被激活[18]。小鼠骨髓间充质干细胞也可通过过表达β-连环蛋白而激活经典Wnt/β-连环蛋白信号通路,进一步提高小鼠骨髓间充质干细胞对上皮损伤的保护,从而对急性呼吸窘迫综合征小鼠有治疗作用,该途径增加了小鼠骨髓间充质干细胞在肺中的驻留,促进了小鼠骨髓间充质干细胞向Ⅱ型肺泡上皮细胞的分化,改善了肺上皮通透性,减轻急性肺部炎症,抑制肺纤维化[19]。研究还发现Wnt/β-连环蛋白信号传导途径在肺纤维化的病理过程中起重要作用,通过特异性抑制剂来抑制该传导通路可抑制肺纤维化的发展[20]。间充质干细胞培养过程中加入白细胞介素1β可以刺激间充质干细胞的血管重塑能力,这是由miR-433的过表达导致β-连环蛋白表达增多,从而活化Wnt信号通路实现的[21]。 (3)NF-κB信号通路:NF-κB由一系列转录因子组成,这些因子在炎症、免疫、细胞增殖、分化和存活中起关键作用,NF-кB家族包含的转录因子控制大量靶基因的表达以适应环境的变化,有助于协调炎症和免疫应答[22]。间充质干细胞可通过分泌TSG-6,抑制NF-кB信号通路,减少炎症反应,促进抗炎细胞因子的表达,将巨噬细胞转化为抗炎表型[23]。在脂多糖诱导的急性肺损伤模型中,间充质干细胞可减少炎症损伤,通过调节Toll样受体2和Toll样受体4介导的NF-кB信号通路从而对急性肺损伤产生保护作用[24]。间充质干细胞还下调了促炎因子肿瘤坏死因子α、白细胞介素6和白细胞介素1β的表达,上调了抗炎因子白细胞介素10的表达[2,4,24]。另外,Toll样受体4和蛋白酶激活受体1在间充质干细胞的存活以及治疗肺损伤中发挥重要作用[25]。SU等[26]报道间充质干细胞条件培养基通过诱导中性粒细胞凋亡从而减弱脂多糖诱导的急性肺损伤也与NF-κB途径的抑制有关。间充质干细胞还可通过可溶性肿瘤坏死因子α受体1来抑制NF-кB p65的信号传导,从而抑制心脏和肺部的炎症反应[27]。 2.2.2 旁分泌 (1)分泌细胞因子:间充质干细胞可产生多种细胞因子[28-29],其可分泌具有细胞保护和修复特性的生长因子、角质细胞生长因子、血管内皮生长因子和肝细胞生长因子等。 角质细胞生长因子又称成纤维细胞生长因子7,是成纤维细胞生长因子家族中的成员之一,它介导多种组织中表皮或上皮细胞增殖和分化。角质细胞生长因子可以与成纤维细胞生长因子受体结合,激活PI3K途径,从而对细胞功能发生调控作用[30-31],其可通过PI3K/AKT/ MTOR信号通路来改善受损Ⅱ型肺泡上皮细胞的炎症以改善肺损伤[13]。角质细胞生长因子也可通过多种机制发挥作用,包括恢复肺内皮和上皮的通透性,并改善肺泡上皮细胞清除肺泡水肿的能力[28]。有研究表明,间充质干细胞可依赖角质细胞生长因子的旁分泌机制促进肺损伤后的修复[32]。角质细胞生长因子是通过刺激上皮细胞增殖进行促进肺上皮修复的关键介质,间充质干细胞可减弱肺纤维化,经角质细胞生长因子预处理的间充质干细胞可预防肺纤维化,这种作用部分归因于间充质干细胞向纤维化肺的动员增强;此外,角质细胞生长因子也可激活与干细胞分化相关的SHH信号传导途径,从而间充质干细胞表达角质细胞生长因子的特异性受体,这表明角质细胞生长因子可能对间充质干细胞有促分泌作用[33]。 在使用间充质干细胞条件培养基以及加入肝细胞生长因子和血管内皮生长因子受体抑制剂的实验中,间充质干细胞条件培养基显著降低了脂多糖诱导肺损伤的内皮细胞旁通透性和跨细胞通透性,也能降低内皮细胞凋亡和诱导内皮细胞增殖,同时发现间充质干细胞分泌的肝细胞生长因子具有保护肺损伤后的内皮屏障功能,间充质干细胞分泌的血管内皮生长因子可以与肝细胞生长因子协同作用以稳定内皮细胞屏障功能[34]。在接受间充质干细胞注射的高氧诱导的肺损伤模型中,肺组织血管内皮生长因子的表达明显提高,并且损伤肺内Ⅱ型肺泡上皮细胞的功能得到了改善,潜在说明间充质干细胞的输注增加了血管内皮生长因子的产生[35]。 将间充质干细胞加入到含肺祖细胞的3D培养基中发现其增加了肺泡的分化和器官形成,间充质干细胞条件培养基不仅可以促进肺泡器官的形成[36],而且可促使中性粒细胞凋亡以减轻肺损伤[26],这表明间充质干细胞条件培养基中的一些因子发挥作用。骨髓间充质干细胞移植增加了大鼠吸入烟雾致吸入性肺损伤的微血管数量,增加了肺湿干质量比,通过骨髓间充质干细胞的旁分泌机制减轻肺损伤[37]。 (2)微囊泡和胞外囊泡:许多临床前研究表明祖细胞和干细胞群体(包括间充质干细胞)在肺纤维化动物模型损伤早期炎症阶段有治疗效果,旁分泌在抗炎中起着关键作用[29,38]。PARK等[39]进行的体外肺实验表明,间充质干细胞释放的微囊泡增加了肺泡液体清除率和降低了肺蛋白的通透性,经过聚肌胞苷酸预处理后显著减低细菌定植数量。通过Toll样受体3激动剂预处理后的间充质干细胞来源微囊泡在离体肺内的抗大肠杆菌能力明显增强,其与增强巨噬细胞吞噬细菌活性有关,间充质干细胞来源微囊泡明显增加肺泡液体清除率,减少蛋白质通透性以及降低损伤肺泡的细菌数量,这表明离体人肺泡巨噬细胞在体外与间充质干细胞来源微囊泡联合治疗增加了抗菌能力。将微囊泡的量增加1倍,其疗效并未明显增加,说明微囊泡有治疗上限[39-40]。MORRISON等[41]报道人骨髓间充质干细胞抑制急性呼吸窘迫综合征患者支气管肺泡灌洗液中巨噬细胞促炎性细胞因子的分泌,是间充质干细胞产生的胞外囊泡,通过使线粒体从间充质干细胞转导到巨噬细胞内以及增强巨噬细胞中氧化磷酸化来实现的,这提高了巨噬细胞的吞噬能力和M2巨噬细胞标志物CD206的表达。此外,肺泡巨噬细胞凋亡在急性肺损伤及其严重型急性呼吸窘迫综合征中起致病作用[16]。XU等[42]在光气诱导的急性肺损伤模型研究中,发现间充质干细胞衍生的外泌体通过调节炎症、抑制基质金属蛋白酶9合成、提高表面活性物质相关蛋白C水平,对急性肺损伤发挥有益作用。 (3)外泌体:miR-21-5p是一种抗凋亡miRNA,是间充质干细胞来源外泌体中的主要成分之一,对氧化应激诱导的细胞凋亡有保护作用,经气管内给予miR-21-5p可显著减轻肺水肿、肺功能障碍、肺泡巨噬细胞极化及白细胞介素8、白细胞介素1β、白细胞介素6、白细胞介素17、肿瘤坏死因子α的分泌,说明间充质干细胞通过miR-21-5p抑制内源性和外源性凋亡途径,改善了缺血再灌注肺损伤以及缺氧/复氧诱导的细胞凋亡[7]。间充质干细胞外泌体中另一重要成分miR-124-3p,能抑制嘌呤能受体P2X配体门控离子通道7(P2X7)的表达,改善创伤性急性肺损伤的氧化应激损伤以及炎性反应[43]。 2.2.3 分化能力 鼠源间充质干细胞可分化为鼠上皮细胞,鼠源间充质干细胞中过表达p130或E2F4可以进一步改善急性呼吸窘迫综合征小鼠肺上皮细胞损伤,降低其高渗透性,促进鼠源间充质干细胞向Ⅱ型肺泡上皮细胞分化从而改善肺通透性和肺纤维化,减少肺水肿,进一步提高鼠源间充质干细胞治疗小鼠急性呼吸窘迫综合征的效果[44]。LI等[45]移植Lats1敲低的鼠源间充质干细胞后,抑制了脂多糖诱导的急性呼吸窘迫综合征小鼠的Hippo信号通路,促进了鼠源间充质干细胞在肺组织的保留及其向Ⅱ型肺泡上皮细胞的分化。 2.2.4 与其他药物联合 间充质干细胞与其他药物联合应用可以增强保护肺损伤的作用。间充质干细胞与1-磷酸鞘氨醇类似物FTY720联合治疗脂多糖诱导的急性肺损伤/急性呼吸窘迫综合征的疗效更好[46]。间充质干细胞和血管紧张素转换酶Ⅱ共同给药可明显抑制急性肺损伤小鼠的细胞凋亡[47]。LUAN等[48]提出间充质干细胞与促红细胞生成素联合应用可减轻高氧诱导的肺损伤,减少肺纤维化,二者是通过抑制转化生长因子β1信号通路来促进肺泡发育不良损伤肺的修复。小鼠间充质干细胞与促红细胞生成素联合治疗支气管肺发育不良的实验中,血管密度和血管内皮生长因子水平、基质细胞衍生因子1及其受体CXC趋化因子受体4(CXCR4)的表达水平显著增加。促红细胞生成素显著促进了间充质干细胞的增殖、迁移以及抗凋亡能力[49]。与临床上单独使用的皮质类固醇治疗方法相比,联合应用间充质成血管细胞衍生的间充质干细胞对实验性慢性过敏性气道疾病有更好的保护作用[50]。 2.2.5 其他 间充质干细胞培养过程中加入小分子脯氨酰羟化酶抑制剂AKB-4924可以提高间充质干细胞中的缺氧诱导因子1α水平,增强体外治疗小鼠实验性细菌性肺炎及急性肺损伤的能力[51]。缺氧条件下间充质干细胞实现了较高的缺氧诱导因子1α和Akt活化水平,通过促进缺氧诱导因子1α介导的间充质干细胞增殖和提高其抗氧化能力来增强间充质干细胞对辐射诱导的肺损伤的治疗效果,其停留肺内时间延长、细胞活力和增殖潜力增加、活性氧水平降低且抗氧化能力增强[52]。经修饰的间充质干细胞还改善了急性呼吸窘迫综合征动物的肺水肿,减少了炎症反应,减轻了病理性损伤和肺组织中的纤维化[17,19,45]。 "
| [1] VILLAR J, BLANCO J, KACMAREK RM. Current incidence and outcome of the acute respiratory distress syndrome. Curr Opin Crit Care. 2016; 22(1): 1-6. [2] 强勇,梁贵友,余丽梅,等.人羊膜间充质干细胞移植减轻体外循环再灌注肺损伤[J].中国组织工程研究,2016,20(1): 70-77. [3] GALIPEAU J, SENSÉBÉ L. Mesenchymal Stromal Cells: Clinical Challenges and Therapeutic Opportunities. Cell Stem Cell. 2018; 22(6):824-833. [4] QIANG Y, LIANG G, YU L. Human amniotic mesenchymal stem cells alleviate lung injury induced by ischemia and reperfusion after cardiopulmonary bypass in dogs. Lab Invest. 2016;96(5):537-546. [5] DOMINICI M, LE BLANC K, MUELLER I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006; 8(4):315-317. [6] ZHANG J, SHAO Y, HE D, et al. Evidence that bone marrow-derived mesenchymal stem cells reduce epithelial permeability following phosgene-induced acute lung injury via activation of wnt3a protein-induced canonical wnt/β-catenin signaling. Inhal Toxicol. 2016;28(12):572-579. [7] LI JW, WEI L, HAN Z, et al. Mesenchymal stromal cells-derived exosomes alleviate ischemia/reperfusion injury in mouse lung by transporting anti-apoptotic miR-21-5p. Eur J Pharmacol. 2019;852: 68-76. [8] ENGELMAN JA, LUO J, CANTLEY LC. The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism. Nat Rev Genet. 2006;7(8):606-619. [9] LORUSSO PM. Inhibition of the PI3K/AKT/mTOR Pathway in Solid Tumors. J Clin Oncol. 2016;34(31):3803-3815. [10] CHU H, HAO W, CHENG Z, et al. Black carbon particles and ozone-oxidized black carbon particles induced lung damage in mice through an interleukin-33 dependent pathway. Sci Total Environ. 2018; 644:217-228. [11] LI J, ZHOU J, ZHANG D, et al. Bone marrow-derived mesenchymal stem cells enhance autophagy via PI3K/AKT signalling to reduce the severity of ischaemia/reperfusion-induced lung injury. J Cell Mol Med. 2015;19(10):2341-2351. [12] GHANTA S, TSOYI K, LIU X, et al. Mesenchymal Stromal Cells Deficient in Autophagy Proteins Are Susceptible to Oxidative Injury and Mitochondrial Dysfunction. Am J Respir Cell Mol Biol. 2017;56(3): 300-309. [13] LI J, HUANG S, ZHANG J, et al. Mesenchymal stem cells ameliorate inflammatory cytokine-induced impairment of AT-II cells through a keratinocyte growth factor-dependent PI3K/Akt/mTOR signaling pathway. Mol Med Rep. 2016;13(5):3755-3762. [14] LIN HY, XU L, XIE SS, et al. Mesenchymal stem cells suppress lung inflammation and airway remodeling in chronic asthma rat model via PI3K/Akt signaling pathway. Int J Clin Exp Pathol. 2015;8(8):8958-8967. [15] KIM SY, LEE JH, KIM HJ, et al. Mesenchymal stem cell-conditioned media recovers lung fibroblasts from cigarette smoke-induced damage. Am J Physiol Lung Cell Mol Physiol.2012;302(9): L891-908. [16] LI B, ZHANG H, ZENG M, et al. Bone marrow mesenchymal stem cells protect alveolar macrophages from lipopolysaccharide-induced apoptosis partially by inhibiting the Wnt/β-catenin pathway. Cell Biol Int. 2015;39(2):192-200. [17] LIU AR, LIU L, CHEN S, et al. Activation of canonical wnt pathway promotes differentiation of mouse bone marrow-derived MSCs into type II alveolar epithelial cells, confers resistance to oxidative stress, and promotes their migration to injured lung tissue in vitro. J Cell Physiol. 2013;228(6):1270-1283. [18] GAO P, YANG J, GAO X, et al. Salvianolic acid B improves bone marrow-derived mesenchymal stem cell differentiation into alveolar epithelial cells type I via Wnt signaling. Mol Med Rep. 2015;12(2): 1971-1976. [19] CAI SX, LIU AR, CHEN S, et al. Activation of Wnt/β-catenin signalling promotes mesenchymal stem cells to repair injured alveolar epithelium induced by lipopolysaccharide in mice. Stem Cell Res Ther. 2015;6:65. [20] WANG C, ZHU H, SUN Z, et al. Inhibition of Wnt/β-catenin signaling promotes epithelial differentiation of mesenchymal stem cells and repairs bleomycin-induced lung injury. Am J Physiol Cell Physiol. 2014; 307(3):C234-244. [21] SUN J, CHEN J, CAO J, et al. IL-1β-stimulated β-catenin up-regulation promotes angiogenesis in human lung-derived mesenchymal stromal cells through a NF-κB-dependent microRNA-433 induction. Oncotarget. 2016;7(37):59429-59440. [22] OECKINGHAUS A, GHOSH S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol. 2009;1(4): a000034. [23] WANG G, CAO K, LIU K, et al. Kynurenic acid, an IDO metabolite, controls TSG-6-mediated immunosuppression of human mesenchymal stem cells. Cell Death Differ. 2018;25(7):1209-1223. [24] LI D, PAN X, ZHAO J, et al. Bone Marrow Mesenchymal Stem Cells Suppress Acute Lung Injury Induced by Lipopolysaccharide Through Inhibiting the TLR2, 4/NF-κB Pathway in Rats with Multiple Trauma. Shock. 2016;45(6):641-646. [25] GUPTA N, SINHA R, KRASNODEMBSKAYA A, et al. The TLR4-PAR1 Axis Regulates Bone Marrow Mesenchymal Stromal Cell Survival and Therapeutic Capacity in Experimental Bacterial Pneumonia. Stem Cells. 2018;36(5):796-806. [26] SU VY, LIN CS, HUNG SC, et al. Mesenchymal Stem Cell-Conditioned Medium Induces Neutrophil Apoptosis Associated with Inhibition of the NF-κB Pathway in Endotoxin-Induced Acute Lung Injury. Int J Mol Sci. 2019;20(9): E2208. [27] MARTIRE A, BEDADA FB, UCHIDA S, et al. Mesenchymal stem cells attenuate inflammatory processes in the heart and lung via inhibition of TNF signaling. Basic Res Cardiol. 2016;111(5):54. [28] MATTHAY MA, THOMPSON BT, READ EJ, et al. Therapeutic potential of mesenchymal stem cells for severe acute lung injury. Chest. 2010; 138(4): 965-972. [29] XU S, LIU C, JI HL. Concise Review: Therapeutic Potential of the Mesenchymal Stem Cell Derived Secretome and Extracellular Vesicles for Radiation-Induced Lung Injury: Progress and Hypotheses. Stem Cells Transl Med. 2019;8(4):344-354. [30] FINCH PW, RUBIN JS. Keratinocyte growth factor expression and activity in cancer: implications for use in patients with solid tumors. J Natl Cancer Inst. 2006;98(12):812-824. [31] VADIJA R, MUSTYALA KK, NAMBIGARI N, et al. Homology modeling and virtual screening studies of FGF-7 protein-a structure-based approach to design new molecules against tumor angiogenesis. J Chem Biol. 2016;9(3):69-78. [32] CURLEY GF, HAYES M, ANSARI B, et al. Mesenchymal stem cells enhance recovery and repair following ventilator-induced lung injury in the rat. Thorax. 2012;67(6):496-501. [33] YAO L, LIU CJ, LUO Q, et al. Protection against hyperoxia-induced lung fibrosis by KGF-induced MSCs mobilization in neonatal rats. Pediatr Transplant. 2013;17(7):676-682. [34] YANG Y, CHEN QH, Liu AR, et al. Synergism of MSC-secreted HGF and VEGF in stabilising endothelial barrier function upon lipopolysaccharide stimulation via the Rac1 pathway. Stem Cell Res Ther. 2015;6:250. [35] MEI Y, CHEN C, DONG H, et al. Treatment of Hyperoxia-Induced Lung Injury with Lung Mesenchymal Stem Cells in Mice. Stem Cells Int. 2018; 2018:5976519. [36] LEEMAN KT, PESSINA P, LEE JH, et al. Mesenchymal Stem Cells Increase Alveolar Differentiation in Lung Progenitor Organoid Cultures. Sci Rep. 2019;9(1):6479. [37] ZHU F, WANG J, QIU X, et al. Smoke inhalation injury repaired by a bone marrow-derived mesenchymal stem cell paracrine mechanism: Angiogenesis involving the Notch signaling pathway. J Trauma Acute Care Surg. 2015;78(3):565-572. [38] MCNULTY K, JANES SM. Stem cells and pulmonary fibrosis: cause or cure. Proc Am Thorac Soc. 2012;9(3):164-171. [39] PARK J, KIM S, LIM H, et al. Therapeutic effects of human mesenchymal stem cell microvesicles in an ex vivo perfused human lung injured with severe E. coli pneumonia. Thorax. 2019;74(1):43-50. [40] LIU HM, LIU YT, ZHANG J, et al. Bone marrow mesenchymal stem cells ameliorate lung injury through anti-inflammatory and antibacterial effect in COPD mice. J Huazhong Univ Sci Technolog Med Sci. 2017;37(4): 496-504. [41] MORRISON TJ, JACKSON MV, CUNNINGHAM EK, et al. Mesenchymal Stromal Cells Modulate Macrophages in Clinically Relevant Lung Injury Models by Extracellular Vesicle Mitochondrial Transfer. Am J Respir Crit Care Med. 2017;196(10):1275-1286. [42] XU N, SHAO Y, YE K, et al. Mesenchymal stem cell-derived exosomes attenuate phosgene-induced acute lung injury in rats. Inhal Toxicol. 2019; 31(2):52-60. [43] LI QC, LIANG Y, SU ZB. Prophylactic treatment with MSC-derived exosomes attenuates traumatic acute lung injury in rats. Am J Physiol Lung Cell Mol Physiol. 2019;316(6):L1107-L1117. [44] ZHANG X, CHEN J, XUE M, et al. Overexpressing p130/E2F4 in mesenchymal stem cells facilitates the repair of injured alveolar epithelial cells in LPS-induced ARDS mice. Stem Cell Res Ther. 2019;10(1):74. [45] LI L, DONG L, ZHANG J, et al. Mesenchymal stem cells with downregulated Hippo signaling attenuate lung injury in mice with lipopolysaccharide‑induced acute respiratory distress syndrome. Int J Mol Med. 2019;43(3): 1241-1252. [46] HUANG Z, LIU H, ZHANG X, et al. Transcriptomic analysis of lung tissues after hUC-MSCs and FTY720 treatment of lipopolysaccharide- induced acute lung injury in mouse models. Int Immunopharmacol. 2018;63: 26-34. [47] ZHANG X, GAO F, LI Q, et al. MSCs with ACE II gene affect apoptosis pathway of acute lung injury induced by bleomycin. Exp Lung Res. 2015;41(1):32-43. [48] LUAN Y, ZHANG L, CHAO S, et al. Mesenchymal stem cells in combination with erythropoietin repair hyperoxia-induced alveoli dysplasia injury in neonatal mice via inhibition of TGF-β1 signaling. Oncotarget. 2016;7(30): 47082-47094. [49] SUN C, ZHANG S, WANG J, et al. EPO enhances the protective effects of MSCs in experimental hyperoxia-induced neonatal mice by promoting angiogenesis. Aging (Albany NY). 2019;11(8):2477-2487. [50] ROYCE SG, MAO W, LIM R, et al. iPSC- and mesenchymoangioblast- derived mesenchymal stem cells provide greater protection against experimental chronic allergic airways disease compared with a clinically used corticosteroid. FASEB J. 2019;33(5): 6402-6411. [51] GUPTA N, NIZET V. Stabilization of Hypoxia-Inducible Factor-1 Alpha Augments the Therapeutic Capacity of Bone Marrow-Derived Mesenchymal Stem Cells in Experimental Pneumonia. Front Med (Lausanne). 2018;5:131. [52] LI B, LI C, ZHU M, et al. Hypoxia-Induced Mesenchymal Stromal Cells Exhibit an Enhanced Therapeutic Effect on Radiation-Induced Lung Injury in Mice due to an Increased Proliferation Potential and Enhanced Antioxidant Ability. Cell Physiol Biochem. 2017;44(4):1295-1310. [53] WILSON JG, LIU KD, ZHUO H, et al. Mesenchymal stem (stromal) cells for treatment of ARDS: a phase 1 clinical trial. Lancet Respir Med. 2015;3(1):24-32.
|
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [5] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [6] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [7] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [8] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [9] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [10] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [11] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [12] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [13] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [14] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [15] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||