Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (17): 2746-2752.doi: 10.3969/j.issn.2095-4344.1714
Previous Articles Next Articles
Association of aging with pulmonary fibrosis and chronic obstructive pulmonary disease and treatment with mesenchymal stem cells
Yang Yukun, Zhu Xiangqing, Ruan Guangping, Wang Yanying, Kan Xiaoli, Zhang Xuejuan, Yu Qianqian, Wang Mao, He Huanyu, Pan Xinghua
- Yunnan Stem Cell Engineering Laboratory, Yunnan Provincial Key Laboratory of Cell Therapy and Translational Medicine, National and Local Joint Engineering Laboratory of Stem Cell and Immune Cell Biomedical Technology, 920th Hospital of Joint Logistics Support Force of PLA, Kunming 652230, Yunnan Province, China
-
Revised:2019-01-23Online:2019-06-18Published:2019-06-18 -
Contact:Pan Xing-hua, MD, Chief physician, Professor, Doctoral supervisor, Yunnan Stem Cell Engineering Laboratory, Yunnan Provincial Key Laboratory of Cell Therapy and Translational Medicine, National and Local Joint Engineering Laboratory of Stem Cell and Immune Cell Biomedical Technology, 920th Hospital of Joint Logistics Support Force of PLA, Kunming 652230, Yunnan Province, China -
About author:Yang Yukun, Master candidate, Yunnan Stem Cell Engineering Laboratory, Yunnan Provincial Key Laboratory of Cell Therapy and Translational Medicine, National and Local Joint Engineering Laboratory of Stem Cell and Immune Cell Biomedical Technology, 920th Hospital of Joint Logistics Support Force of PLA, Kunming 652230, Yunnan Province, China -
Supported by:the Major Science and Technology Special Plan of Yunnan Province, No. 2018ZF007 (to PXH); the Science and Technology Plan Project of Yunnan Province, No. 2017FB042 (to ZXQ)
CLC Number:
Cite this article
Yang Yukun, Zhu Xiangqing, Ruan Guangping, Wang Yanying, Kan Xiaoli, Zhang Xuejuan, Yu Qianqian, Wang Mao, He Huanyu,Pan Xinghua. Association of aging with pulmonary fibrosis and chronic obstructive pulmonary disease and treatment with mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(17): 2746-2752.
share this article
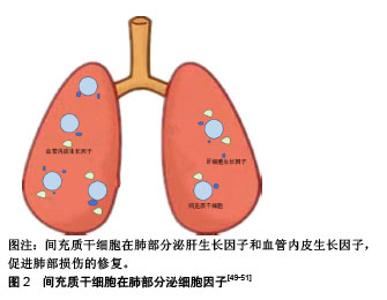
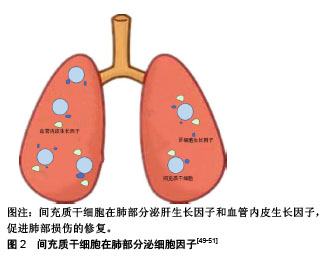
2.1 衰老与肺纤维化、慢性阻塞性肺疾病的关系 衰老是一个自然老化的过程,其表现为进行性功能障碍和对环境刺激及损伤作出适当反应的能力降低[6]。像任何其他器官一样,肺也会衰老。生理性肺衰老包括组织学变化(肺泡增大、肺泡壁破坏、气道交换表面积减少,气道阻塞或闭塞减少、肺血管减少)和功能改变(弹性反冲减少、残余气体增加以及气体交换障碍)导致的肺通气与换气功能减退[7]。另一方面,根据流行病学报道,衰老与多种慢性疾病的发病率有关,如动脉粥样硬化、骨质疏松症、2型糖尿病、癌症等[2]。肺也不例外,调查发现许多慢性肺部疾病的发病率均随年龄的增加而增加,如肺气肿、肺纤维化等[8]。 当前认为,端粒长度调节、细胞和免疫衰老、氧化应激以及许多抗衰老分子和细胞外基质变化是衰老的关键[9],且在肺纤维化、慢性阻塞性肺疾病的发生发展中起重要作用[10]。 2.1.1 端粒长度的调节 端粒是染色体末端的区域,含有1-5 kb(TTAGGG)一段重复序列,可使DNA免于降解和重组,保护染色体的稳定性[11]。在大多数体细胞中,该区域DNA聚合酶难以合成DNA,端粒会随着细胞周期的增加而缩短,而过短的端粒会导致细胞周期永久性停滞,使细胞进入衰老状态。同时,在体内所有循环的体细胞迟早都会永久停止分裂,进入衰老状态,有研究表明衰老细胞的积累是造成衰老和与衰老有关疾病的重要原因[12]。因此,迄今为止端粒长度已成为描述老化的重要标志之一,已有报道称端粒长度的异常存在于某些肺疾病中。Moela等[13]发现慢性阻塞性肺疾病患者的端粒长度和正常人相比明显缩短。另有研究表明,这种缩短在有肺气肿改变的患者中更加严重[14]。同时,端粒长度的调节与肺纤维化也密切相关,据报道几乎全部的特发性肺纤维化患者均有端粒的异常缩短,并且在家族性特发性肺纤维化中有20%发生了端粒基因的突变[15-17]。 2.1.2 氧化应激 氧化应激意味着氧化剂和抗氧化剂之间的不平衡且有利于氧化。人类的有氧生活增加了细胞和组织成分过度氧化的危险,这可能会损害细胞功能和生存能力。氧化产物的形成和积累在各种病理和正常衰老过程中都有观察到,并被认为在衰老过程中起关键作用[18]。慢性阻塞性肺疾病发生和发展的一个重要原因是肺部加速衰老,这个进程可能与高水平的氧化应激对肺部功能和结构的影响有关。研究发现,在慢性阻塞性肺疾病患者的肺部、血液以及骨骼肌中均发现有高水平氧化应激的证据,这可能是线粒体功能紊乱产生过多的活性氧和线粒体氧化损伤导致的结果[19]。另外,Malli等[20]发现在肺纤维化患者的肺泡灌洗液中氧化应激因子8-异前列烷水平显著升高,这表明在肺纤维化患者的肺部存在着较高的氧化应激水平,而衰老的进程与氧化应激水平呈正相关。 2.1.3 细胞衰老和干细胞耗竭 细胞衰老是一种细胞周期停滞、趋向凋亡的细胞状态。正常情况下,衰老细胞可被免疫系统清除,但随着年龄的增加,衰老细胞数量在组织中逐渐增加。衰老细胞能分泌多种炎症因子,而且由于其增殖能力和正常生理功能丧失而干扰正常组织的稳态和修复[21]。根据以往研究来看,细胞衰老可通过至少两种非互斥的机制促成慢性阻塞性肺疾病。首先,在慢性阻塞性肺疾病中上皮细胞和内皮细胞凋亡增多,这造成了肺泡壁中细胞的损失,此时应该发生涉及细胞增殖的补偿机制,以消除肺泡细胞的丢失[22]。然而,当细胞衰老发生时,其增殖能力丧失,平衡倾向于细胞凋亡并由此形成慢性阻塞性肺疾病;其次,有证据表明细胞衰老与炎症之间存在密切关系。衰老细胞可表现出NF-κB的活化,以及释放各种炎性细胞因子,导致炎症增强[23]。这些与衰老相关的促炎机制在人肺组织中得到了证实,例如有研究发现在p16阳性的Ⅱ型肺泡上皮中,肿瘤坏死因子α表达量与对照组相比明显增加,并且这种p16阳性细胞的衰老程度与慢性阻塞性肺疾病炎症严重程度之间也存在着正相关性[24]。另外,细胞衰老也被证明与特发性肺纤维化的发病密切相关。Hecker等[25]发现,老年肺纤维化小鼠模型的肺部存在着异于正常数量的衰老和抗凋亡的肌成纤维细胞,并且还证明了这些细胞在持续肺纤维化中起关键作用。还有研究表明,支气管上皮衰老也是特发性肺纤维化进程的关键因素,其与患者支气管上皮的再上皮化密切相关,并且这种再上皮化可被SIRT6拮抗[26]。衰老过程中也伴随着干细胞的衰老和耗竭,而这种干细胞耗竭现象对组织和器官的修复存在着严重不利影响,这也使得老年动物肺部在内外刺激的作用下更容易发生纤维化[27]。 2.1.4 衰老和抗衰老分子的改变 衰老过程中,伴随着许多分子的变化,已有多项报道证实这些衰老分子的变化和肺疾病关系密切。已知衰老会伴随着DNA和RNA变化,年龄相关的DNA甲基化改变被称为“表观遗传时钟”。众所周知,香烟是慢性阻塞性肺疾病的主要危险因素,在香烟所致的慢性阻塞性肺疾病模型中,存在DNA的过度甲基化,并且这类改变与肺功能和疾病的严重程度相关[10]。另外,肺纤维化的发生与DNA的甲基化也密切相关,Dakhlallah等[28]发现在衰老过程中miR-17-92的减少可致使DNA甲基转移酶增加,导致肺纤维化的发生;再者,组蛋白去乙酰化酶可以减少组蛋白的乙酰化,其活性也与衰老密切相关。组蛋白去乙酰化酶参与了慢性阻塞性肺疾病的发病机制,导致炎性基因的表达增强。患有慢性阻塞性肺疾病人群的肺部巨噬细胞和支气管组织中均能观察到组蛋白去乙酰化酶活性的降低,并且这种降低与慢性阻塞性肺疾病严重程度呈正相关。同样,Sirtuins家族NAD+依赖的Ⅲ类蛋白质脱乙酰酶是一类抗衰老酶,其中具有抗衰老活性的SIRT1、SIRT6水平在慢性阻塞性肺疾病患者的外周血和肺组织中明显减低[29]。另外,Baker等[30]还发现Sirtuins的表达受microRNA MiR-34a调节,在慢性阻塞性肺疾病患者中高度表达的microRNA MiR-34a导致Sirtuins表达显著减少。 2.1.5 免疫失调 衰老可引起先天性和适应性免疫改变,这些改变使细胞对病原体和环境损害的防御机制下调,如衰老可使Toll样受体识别细菌抗原的先天免疫应答被钝化、巨噬细胞吞噬能力降低,从而使肺部更容易受外界病原体的感染[31]。同时,衰老可以导致炎症调控的异常,使某些肺疾病的发展进一步加剧。John-Schuster等[32]发现,暴露于香烟烟雾的老年小鼠比年轻小鼠具有更高的炎症水平,并且更容易患肺气肿。这种老年动物肺部炎症细胞和炎症递质的较高水平,可能导致更低水平的修复,从而更容易患肺气肿。2015年的两项研究表明,由于氧化应激、炎症和细胞凋亡,Klotho这种具有抗炎特性的抗衰老蛋白在肺泡巨噬细胞和慢性阻塞性肺疾病患者气道上皮细胞中显著减少[33-34]。根据上述案例,作者可以分析,衰老可使肺部对内外刺激反应发生异常,产生较高的炎症水平,进而加速肺部衰老,形成恶性循环;同时,衰老涉及的免疫失调还可以在没有抗原攻击的情况下通过非特异性炎症形式出现,这通常与循环细胞因子如肿瘤坏死因子α、白细胞介素β和白细胞介素6的增加有关,并且被描述为炎症衰老。炎症衰老可能代表衰老相关表型谱的一部分,对邻近细胞产生深远影响[35]。在慢性阻塞性肺疾病的进程中,也发现了肺部在没有外界因素的刺激下,产生了与炎症衰老相似的无菌性非特异性炎症[36]。作者推断,衰老与慢性阻塞性肺疾病的进展可能是相辅相成的关系。另外,慢性阻塞性肺疾病中miR-125a和miR-125b水平的增加导致NF-κB活化和病毒清除率的降低也与衰老相关炎症和免疫应答受损密不可分[37]。 2.1.6 细胞外基质的变化 已知衰老涉及几乎所有器官中细胞外基质的变化。研究表明,大多数慢性肺病,特别是慢性阻塞性肺疾病和特发性肺纤维化,与细胞外基质的表达、周转、沉积、降解的改变有关。现在认为细胞外基质的变化发生在肺损伤早期,并且能抑制修复,促进疾病的发展[38]。老化肺的特征在于较高水平的胶原沉积与弹性蛋白表达减少,老化啮齿动物肺组织中胶原蛋白、纤连蛋白和基质金属蛋白酶的表达均发生了变化[39]。同样,在慢性阻塞性肺疾病和肺纤维化患者中也观察到了类似的变化,如在肺纤维化患者肺部发现有过多胶原蛋白的累积,在慢性阻塞性肺疾病患者的肺部观察到核心蛋白聚糖和双糖链蛋白多糖的减少[40]。 综上所述,衰老与肺纤维化、慢性阻塞性肺疾病的发生发展密切相关,但当前的各种治疗手段各有弊端,并不能很好地治疗和预防上述与衰老有关的肺疾病[3,41]。从衰老与肺纤维化、慢性阻塞性肺疾病关联的机制角度出发,探究肺纤维化、慢性阻塞性肺疾病的治疗方法,或许可能成为治疗这类疾病的新道路。 2.2 间充质干细胞治疗肺纤维化、慢性阻塞性肺疾病的疗效与机制 间充质干细胞,也被称为多能基质细胞或间充质基质细胞,其最早于骨髓中被发现,富含CD105,CD73和CD90,而缺乏CD45,CD34,CD14或CD11b,CD79a或CD19和HLA-DR表面分子,并具有自我更新能力和多项分化潜能。迄今为止,间充质干细胞已能从多种不同的结缔组织中(如脂肪组织、肌肉、胎盘、脐带基质、血液、肝脏和牙髓等)分离获得。目前认为,间充质干细胞不仅具有自我更新和分化能力,并且能够在衰老损伤部位分泌多种具有生物活性的因子(如神经生长因子、血管内皮生长因子、血管内皮生长因子受体2、碱性成纤维生长因子等)、与其他细胞相接触传导信号、将自身具有良好功能的结构转移到受损细胞(如线粒体的转移)等方式,起到免疫调节、抗凋亡、血管生成、促进局部干/祖细胞的生长和分化等作用。因此间充质干细胞可能是治疗衰老相关慢性阻塞性肺疾病、肺纤维化的有效手段[5]。 从目前已有的研究来看,间充质干细胞主要从5个方面对肺纤维化、慢性阻塞性肺疾病起作用。 2.2.1 端粒的调节 衰老与端粒的改变关系密切,某些肺疾病伴随着端粒的改变。研究表明,特发性肺纤维化患者通过药物治疗可减少其端粒的损耗,并改善患者的病情[42]。间充质干细胞可以改善受损老化细胞中端粒的磨损[43]。由此推断,间充质干细胞可能通过减少衰老肺泡上皮细胞、成纤维细胞等端粒的磨损,来延缓肺部疾病的进展,改善肺功能。 2.2.2 减少氧化应激 衰老过程中逐渐增强的氧化应激是导致肺疾病的重要因素。减轻氧化应激,可以改善组织的衰老、减少肺疾病的发生、延缓慢性肺病的进展。研究发现,间充质干细胞对组织的过度氧化应激有明显的改善作用。活性氧是细胞活动中消耗氧气而产生的物质,其与氧化应激密切相关。Liu等[44]发现骨髓间充质干细胞可有效减少肺损伤大鼠肺组织中的活性氧,减轻肺部损伤。同时,超氧化物歧化酶是广泛分布于原核和真核细胞中的金属酶,其作为氧自由基的天敌,能对抗和阻断氧自由基对细胞造成的损害,并及时修复受损细胞,对人体不断补充超氧化物歧化酶具有抗衰老的特殊效果[45]。Huang等[46]研究发现骨髓间充质干细胞植入的肺纤维化小鼠模型肺组织中超氧化物歧化酶活性和总抗氧化活力显著增加,表明间充质干细胞可减轻肺组织氧化应激。另外,丙二醛和血红素氧合酶1是两种相拮抗的物质,前者具有增强氧化应激的作用,而后者则具有抗氧化应激和保护细胞的功能。Fredenblibgh等[47]发现间充质干细胞可在降低肺部丙二醛水平的同时增加血红素氧合酶1的合成。 2.2.3 补充耗竭的干细胞,调节衰老相关分子,改善细胞凋亡和衰老,修复肺组织 间充质干细胞可作为已耗竭干细胞的一种补充,通过抗衰老和凋亡的作用,治疗肺纤维化、慢性阻塞性肺疾病。目前,已有报道称骨髓间充质干细胞在体内外都可分化为肺泡上皮细胞,且能在肺部定植,例如Huang等[46]通过静脉植入骨髓间充质干细胞能减轻肺纤维化大鼠肺纤维化的程度,并且能定植在模型大鼠肺部,分化为Ⅱ型肺泡上皮。Liu等[48]发现,植入小鼠肺损伤模型的人脐带间充质干细胞能发挥抑制细胞衰老和凋亡的作用,肺组织中有间充质干细胞定植。另外,间充质干细胞还可在特定部位分泌各种因子来抑制衰老,修复肺组织,见图2。肝细胞生长因子是一种能刺激肝细胞增殖的物质,现在认为其有诱导肺上皮细胞增殖、迁移的作用,并具有抑制肺上皮细胞凋亡及抗纤维化的特性。研究发现,大鼠骨髓间充质干细胞可以分泌肝细胞生长因子来修复肺泡上皮细胞损伤,同时减少博来霉素诱导的大鼠肺纤维化[49]。同样,血管内皮生长因子是最有效的促血管生长因子,其能促进肺泡上皮增殖和肺损伤后毛细血管的形成[50]。Guan等[51]报道脐带间充质干细胞可通过上调血管内皮生长因子减少细胞凋亡,改善慢性阻塞性肺疾病大鼠肺部损害。然而,血管内皮生长因子的表达有很多不良反应,例如提高肝癌发病率等,为了避免与血管内皮生长因子表达相关的并发症,研究人员通过用c-白藜芦醇下调间充质干细胞的热休克蛋白70启动子,实现了血管内皮生长因子的条件表达,而且这种经过诱导的间充质干细胞在香烟提取物攻击中,表现出比对照组更高的存活率,与弹性蛋白酶诱导的肺气肿模拟对照相比,条件表达血管内皮生长因子的间充质干细胞具有更好的治疗效果[52]。除了以上机制外,间充质干细胞还可调节一些与凋亡、衰老相关的分子来修复肺组织。前文中已经概述了衰老与DNA的甲基化密切相关,有研究表明,间充质干细胞可通过miR-29b介导的Dnmt1下调来减低DNA的甲基化,改善细胞衰老[53];Yuan等[54]发现间充质干细胞还能上调抗衰老相关分子SIRT1来调节线粒体的功能,从而改善氧化应激。另外在脐带间充质干细胞治疗衰老小鼠肺部也发现了衰老相关蛋白(p16、p53)表达减少[55]。"
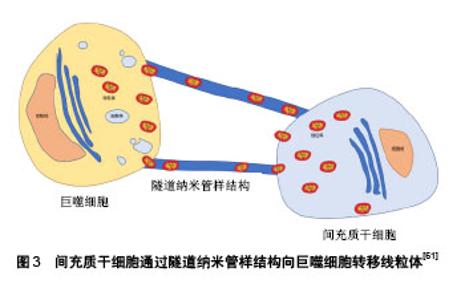
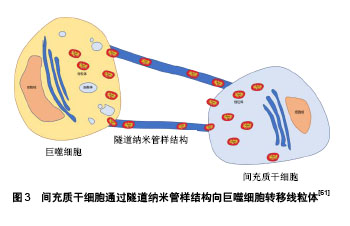
2.2.4 调节肺部免疫 衰老使机体免疫力下降、免疫调节失衡,肺疾病与这种免疫功能的变化密切相关。间充质干细胞的低免疫原性和免疫调节作用已经得到公认[56]。Gupta等[57]证明间充质干细胞移植能降低慢性阻塞性肺疾病小鼠肺泡灌洗液炎症因子肿瘤坏死因子α水平,上调抗炎因子白细胞介素10水平,并且可有效改善内毒素造成的肺损伤。同样,Li等[58]也报道间充质干细胞对肺纤维化小鼠肺部炎症有明显的抑制作用,并且可以改善其纤维化程度。间充质干细胞还可分泌一些因子来调节肺部免疫,Liu等[59]发现小鼠骨髓间充质干细胞可通过分泌TSG-6促进巨噬细胞进入抗炎表型,抑制NF-κB信号通路,在香烟烟雾和副流感嗜血杆菌诱导的慢性阻塞性肺疾病模型中调节抗炎和促炎因子的平衡,减轻肺气肿病变。间充质干细胞通过细胞间相互作用对免疫调节也不可忽视,经环氧合酶2/前列腺素E2途径促进肺泡巨噬细胞重编程,从而减轻过度的炎症。该研究还发现,间充质干细胞对巨噬细胞中环氧合酶2表达的影响仅通过p38MAPK和ERK通路介导,而不涉及JNK途径[60]。另外,间充质干细胞不仅有抗炎的作用,同时也能增强机体对病原体的清除能力。Jackson等[61]发现间充质干细胞可通过隧道纳米管样结构将自身的线粒体转移到巨噬细胞内,从而调节巨噬细胞的功能,使吞噬细菌的能力增强,见图3。Liu等[59]也发现向香烟诱导的慢性阻塞性肺疾病小鼠模型体内移植间充质干细胞,可使巨噬细胞的吞噬功能增强,经气管滴入的流感嗜血杆菌的清除率增加。"
| [1] 陈艳玫,刘子锋,李贤德,等. 2015-2050年中国人口老龄化趋势与老年人口预测[J]. 中国社会医学杂志, 2018, 35(5): 480-483.[2] Childs BG, Durik M, Baker DJ, et al. Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med. 2015; 21(12):1424-1435.[3] Shioya T, Sato S, Iwakura M, et al. Improvement of physical activity in chronic obstructive pulmonary disease by pulmonary rehabilitation and pharmacological treatment. Respir Investig. 2018;56(4):292-306.[4] Sgalla G, Iovene B, Calvello M, et al. Idiopathic pulmonary fibrosis: pathogenesis and management. Respir Res. 2018;19(1):32.[5] Xi J, Yan X, Zhou J, et al. Mesenchymal stem cells in tissue repairing and regeneration: Progress and future. Burns Trauma. 2013;1(1): 13-20.[6] Kirkwood TB. Understanding the odd science of aging. Cell. 2005; 120(4):437-447.[7] Tran D, Rajwani K, Berlin DA. Pulmonary effects of aging. Curr Opin Anaesthesiol. 2018;31(1):19-23.[8] Braman SS, Skloot GS. Pulmonary Disease in the Aging Patient. Clin Geriatr Med. 2017;33(4):xi-xii.[9] Guarente L. Sirtuins, aging, and metabolism. Cold Spring Harb Symp Quant Biol. 2011;76:81-90.[10] Ascher K, Elliot SJ, Rubio GA, et al. Lung Diseases of the Elderly: Cellular Mechanisms. Clin Geriatr Med. 2017;33(4):473-490.[11] Xu J, Gonzalez ET, Iyer SS, et al. Use of senescence- accelerated mouse model in bleomycin-induced lung injury suggests that bone marrow-derived cells can alter the outcome of lung injury in aged mice. J Gerontol A Biol Sci Med Sci. 2009; 64(7):731-739.[12] Vidacek NŠ, Nanic L, Ravlic S, et al. Telomeres, Nutrition, and Longevity: Can We Really Navigate Our Aging. J Gerontol A Biol Sci Med Sci. 2017;73(1):39-47.[13] Morlá M, Busquets X, Pons J, et al. Telomere shortening in smokers with and without COPD. Eur Respir J. 2006;27(3):525-528.[14] Müller KC, Welker L, Paasch K, et al. Lung fibroblasts from patients with emphysema show markers of senescence in vitro. Respir Res. 2006;7:32.[15] Armanios MY, Chen JJ, Cogan JD, et al. Telomerase mutations in families with idiopathic pulmonary fibrosis. N Engl J Med. 2007;56(13): 1317-1326.[16] Tsakiri KD, Cronkhite JT, Kuan PJ, et al. Adult-onset pulmonary fibrosis caused by mutations in telomerase. Proc Natl Acad Sci U S A. 2007; 104(18):7552-7557.[17] Lawson WE, Loyd JE, Degryse AL. Genetics in pulmonary fibrosis- familial cases provide clues to the pathogenesis of idiopathic pulmonary fibrosis. Am J Med Sci. 2011;341(6):439-443.[18] Harman D. Free radical theory of aging: an update: increasing the functional life span Ann N Y Acad Sci. 2006;1067:10-21.[19] MacNee W, Tuder RM. New paradigms in the pathogenesis of chronic obstructive pulmonary disease I. Proc Am Thorac Soc. 2009;6(6): 527-531.[20] Malli F, Bardaka F, Tsilioni I, et al. 8-isoprostane levels in serum and bronchoalveolar lavage in idiopathic pulmonary fibrosis and sarcoidosis. Food Chem Toxicol. 2013;61:160-163.[21] Burton DG, Krizhanovsky V. Physiological and pathological consequences of cellular senescence. Cell Mol Life Sci. 2014; 71(22): 4373-4386.[22] Tuder RM, Yoshida T, Fijalkowka I, et al. Role of lung maintenance program in the heterogeneity of lung destruction in emphysema. Proc Am Thorac Soc. 2006;3(8):673-679.[23] von Kobbe C. Cellular senescence: a view throughout organismal life. Cell Mol Life Sci. 2018;75(19):3553-3567.[24] Tsuji T, Aoshiba K, Nagai A. Alveolar cell senescence in patients with pulmonary emphysema. Am J Respir Crit Care Med. 2006;174(8): 886-893.[25] Hecker L, Logsdon NJ, Kurundkar D, et al. Reversal of persistent fibrosis in aging by targeting Nox4-Nrf2 redox imbalance. Sci Transl Med. 2014;6(231):231ra47.[26] Minagawa S, Araya J, Numata T, et al. Accelerated epithelial cell senescence in IPF and the inhibitory role of SIRT6 in TGF-β-induced senescence of human bronchial epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2011;300(3):L391-401.[27] Bustos ML, Huleihel L, Kapetanaki MG, et al. Aging mesenchymal stem cells fail to protect because of impaired migration and antiinflammatory response. Am J Respir Crit Care Med. 2014; 189(7):787-798.[28] Dakhlallah D, Batte K, Wang Y, et al. Epigenetic regulation of miR-17~92 contributes to the pathogenesis of pulmonary fibrosis. Am J Respir Crit Care Med. 2013;187(4):397-405.[29] Ito K, Ito M, Elliott WM, et al. Decreased histone deacetylase activity in chronic obstructive pulmonary disease. N Engl J Med. 2005;352(19): 1967-1976.[30] Baker JR, Vuppusetty C, Colley T, et al. Oxidative stress dependent microRNA-34a activation via PI3Kα reduces the expression of sirtuin-1 and sirtuin-6 in epithelial cells. Sci Rep. 2016;6:35871.[31] Nikolich-Zugich J. The twilight of immunity: emerging concepts in aging of the immune system. Nat Immunol. 2018;19(1):10-19.[32] John-Schuster G, Günter S, Hager K, et al. Inflammaging increases susceptibility to cigarette smoke-induced COPD. Oncotarget. 2016; 7(21):30068-30083.[33] Li L, Wang Y, Gao W, et al. Klotho Reduction in Alveolar Macrophages Contributes to Cigarette Smoke Extract-induced Inflammation in Chronic Obstructive Pulmonary Disease. J Biol Chem. 2015;290(46): 27890-27900.[34] Gao W, Yuan C, Zhang J, et al. Klotho expression is reduced in COPD airway epithelial cells: effects on inflammation and oxidant injury. Clin Sci (Lond). 2015;129(12):1011-1023.[35] Acosta JC, Banito A, Wuestefeld T, et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat Cell Biol. 2013;15(8):978-990.[36] Brandsma CA, de Vries M, Costa R, et al. Lung ageing and COPD: is there a role for ageing in abnormal tissue repair. Eur Respir Rev. 2017;26(146): 170073.[37] Hsu AC, Dua K, Starkey MR, et al. MicroRNA-125a and -b inhibit A20 and MAVS to promote inflammation and impair antiviral response in COPD. JCI Insight. 2017;2(7):e90443.[38] Thannickal VJ, Henke CA, Horowitz JC, et al. Matrix biology of idiopathic pulmonary fibrosis: a workshop report of the national heart, lung, and blood institute. Am J Pathol. 2014;184(6): 1643-1651.[39] Booth AJ, Hadley R, Cornett AM, et al. Acellular normal and fibrotic human lung matrices as a culture system for in vitro investigation. Am J Respir Crit Care Med. 2012;186(9): 866-876.[40] Burgess JK, Mauad T, Tjin G, et al. The extracellular matrix - the under-recognized element in lung disease. J Pathol. 2016;240(4): 397-409.[41] Lederer DJ, Martinez FJ. Idiopathic Pulmonary Fibrosis. N Engl J Med. 2018;378(19):1811-1823.[42] Jouneau S, Kerjouan M, Ricordel C. Danazol Treatment for Telomere Diseases. N Engl J Med. 2016;375(11):1095.[43] Brooks RW, Robbins PD. Treating Age-Related Diseases with Somatic Stem Cells. Adv Exp Med Biol. 2018;1056:29-45.[44] Liu YY, Chiang CH, Hung SC, et al. Hypoxia-preconditioned mesenchymal stem cells ameliorate ischemia/reperfusion- induced lung injury. PLoS One. 2017;12(11):e0187637.[45] Johnson F, Giulivi C. Superoxide dismutases and their impact upon human health. Mol Aspects Med. 2005;26(4-5):340-352.[46] Huang K, Kang X, Wang X, et al. Conversion of bone marrow mesenchymal stem cells into type II alveolar epithelial cells reduces pulmonary fibrosis by decreasing oxidative stress in rats. Mol Med Rep. 2015;11(3):1685-1692.[47] Fredenburgh LE, Perrella MA, Mitsialis SA. The role of heme oxygenase-1 in pulmonary disease. Am J Respir Cell Mol Biol. 2007; 36(2):158-165.[48] Liu G, Lv H, An Y, et al. Tracking of transplanted human umbilical cord-derived mesenchymal stem cells labeled with fluorescent probe in a mouse model of acute lung injury. Int J Mol Med. 2018; 41(5): 2527-2534.[49] Gazdhar A, Susuri N, Hostettler K, et al. HGF Expressing Stem Cells in Usual Interstitial Pneumonia Originate from the Bone Marrow and Are Antifibrotic. PLoS One. 2013;8(6):e65453.[50] Tuder RM, Yun JH. Vascular endothelial growth factor of the lung: friend or foe. Curr Opin Pharmacol. 2008;8(3):255-260.[51] Guan XJ, Song L, Han FF, et al. Mesenchymal stem cells protect cigarette smoke-damaged lung and pulmonary function partly via VEGF-VEGF receptors. J Cell Biochem. 2013;114(2):323-335.[52] Chen YB, Lan YW, Hung TH, et al. Mesenchymal stem cell-based HSP70 promoter-driven VEGFA induction by resveratrol promotes angiogenesis in a mouse model. Cell Stress Chaperones. 2015; 20(4): 643-652.[53] Liu S, Liu D, Chen C, et al. MSC Transplantation Improves Osteopenia via Epigenetic Regulation of Notch Signaling in Lupus. Cell Metab. 2015;22(4):606-618.[54] Yuan Y, Shi M, Li L, et al. Mesenchymal stem cell-conditioned media ameliorate diabetic endothelial dysfunction by improving mitochondrial bioenergetics via the Sirt1/AMPK/PGC-1α pathway. Clin Sci (Lond). 2016;130(23):2181-2198.[55] 和法莲. 人脐带间充质干细胞对小鼠老年性器官功能退变的治疗作用及其机制研究[D]. 昆明:昆明医科大学, 2017.[56] Vizoso FJ, Eiro N, Cid S, et al. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int J Mol Sci. 2017;18(9): E1852.[57] Gupta N, Su X, Popov B, et al. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J Immunol. 2007;179(3):1855-1863.[58] Li X, Wang Y, An G, et al. Bone marrow mesenchymal stem cells attenuate silica-induced pulmonary fibrosis via paracrine mechanisms. Toxicol Lett. 2017;270:96-107.[59] Liu HM, Liu YT, Zhang J, et al. Bone marrow mesenchymal stem cells ameliorate lung injury through anti-inflammatory and antibacterial effect in COPD mice. J Huazhong Univ Sci Technolog Med Sci. 2017;37(4):496-504.[60] Gu W, Song L, Li XM, et al. Mesenchymal stem cells alleviate airway inflammation and emphysema in COPD through down-regulation of cyclooxygenase-2 via p38 and ERK MAPK pathways. Sci Rep. 2015;5:8733.[61] Jackson MV, Morrison TJ, Doherty DF, et al. Mitochondrial Transfer via Tunneling Nanotubes is an Important Mechanism by Which Mesenchymal Stem Cells Enhance Macrophage Phagocytosis in the In Vitro and In Vivo Models of ARDS. Stem Cells. 2016;34(8):2210-2223.[62] Moodley Y, Atienza D, Manuelpillai U, et al. Human umbilical cord mesenchymal stem cells reduce fibrosis of bleomycin-induced lung injury. Am J Pathol. 2009;175(1):303-313.[63] Fikry EM, Safar MM, Hasan WA, et al. Bone Marrow and Adipose- Derived Mesenchymal Stem Cells Alleviate Methotrexate-Induced Pulmonary Fibrosis in Rat: Comparison with Dexamethasone. J Biochem Mol Toxicol. 2015;29(7): 321-329.[64] Jin Z, Pan X, Zhou K, et al. Biological effects and mechanisms of action of mesenchymal stem cell therapy in chronic obstructive pulmonary disease. J Int Med Res. 2015;43(3):303-310. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [4] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [5] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [6] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [7] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [8] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [9] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [10] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [11] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [12] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [13] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [14] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [15] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||