Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (13): 2127-2132.doi: 10.3969/j.issn.2095-4344.1684
Treating femoral head necrosis by directed differentiation of bone marrow mesenchymal stem cells: application and evaluation
Zhao Jun1, Zhang Xiaofeng2, Xu Xilin2, Wang Zhengchun1, Fang Xiangchun1, He Xuefeng1, Xie Xinsheng1, Zhang Cheng1
- 1School of Graduates, Heilongjiang University of Chinese Medicine, Harbin 150040, Heilongjiang Province, China; 2the Second Hospital Affiliated to Heilongjiang University of Chinese Medicine, Harbin 150001, Heilongjiang Province, China
-
Revised:2019-01-14Online:2019-05-08Published:2019-05-08 -
Contact:Zhang Xiaofeng, Doctoral supervisor, Professor, the Second Hospital Affiliated to Heilongjiang University of Chinese Medicine, Harbin 150001, Heilongjiang Province, China -
About author:Zhao Jun, Doctorate candidate, School of Graduates, Heilongjiang University of Chinese Medicine, Harbin 150040, Heilongjiang Province, China -
Supported by:the National Natural Science Foundation of China, No. 81774343 (to ZXF)
CLC Number:
Cite this article
Zhao Jun, Zhang Xiaofeng, Xu Xilin, Wang Zhengchun, Fang Xiangchun, He Xuefeng, Xie Xinsheng, Zhang Cheng. Treating femoral head necrosis by directed differentiation of bone marrow mesenchymal stem cells: application and evaluation[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(13): 2127-2132.
share this article
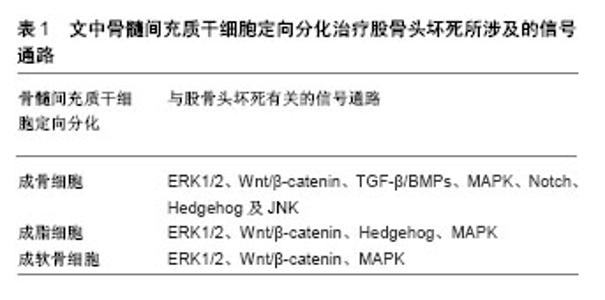
2.1 股骨头坏死的病因病理 创伤与非创伤性为股骨头坏死的主要病因,其中非创伤性因素主要为激素和酒精。ARCO对两种类型股骨头坏死的判定标准已形成共识,激素相关[6]:①3个月内有糖皮质激素使用史,> 2 g泼尼松龙或其等效物;②使用后2年内诊断骨坏死;③除糖皮质激素外,无其他危险因素。酒精相关[7]:①饮酒量>400 mL/周(320 g/周,任何类型的酒精饮料),纯乙醇超过6个月;②该剂量饮酒后1年内确诊股骨头坏死;③无其他风险因素。目前关于股骨头坏死的发病机制尚未完全清楚,主要有骨细胞脂肪变性学说、脂肪栓塞学说、骨内压增高静脉淤滞学说、微血管损伤学说、激素细胞毒性作用学说以及骨质疏松学说等。近年来,骨髓间充质干细胞成骨与成脂分化学说研究越来越多,股骨头坏死的发生和发展与骨髓间充质干细胞的成骨和成脂形成之间的不平衡分化密切相关[8]。大剂量的酒精摄入通过上调过氧化酶基因的表达激活PPARγ,使得骨髓间充质干细胞成脂分化能力显著增强而成骨分化能力减弱,脂肪细胞增多造成骨内压增高血流减少,而骨细胞的减少造成骨质疏松及骨脆性增加,动态平衡遭到破坏,最终出现股骨头坏死与塌陷[9]。杨广杰等[10]通过鉴定大鼠股骨头坏死模型的骨髓间充质干细胞成骨与成脂能力均降低。 2.2 骨髓间充质干细胞成骨分化与股骨头坏死 有研究通过microCT扫描、病理学观察和评估股骨头坏死样本不同区域骨结构,结果显示股骨头不同区域的成骨细胞活性降低、骨小梁厚度数量减少及骨质疏松可能是股骨头塌陷的原因[11]。因此,应尽早促进成骨细胞活性以防止坏死股骨头塌陷。成骨细胞的主要来源为骨髓腔内的骨髓间充质干细胞分化。Wen等[12]用肝细胞生长因子基因转染骨髓间充质干细胞联合髓芯减压治疗激素诱导的兔早期股骨头坏死,发现肝细胞生长因子有利于骨髓间充质干细胞向成骨分化,且坏死股骨头区域观察到小梁的规则排列和明显的骨再生,并推断可能是通过激活细胞外信号调节激酶1/2(ERK1/2)通路起作用,随着肝细胞生长因子的减少,通过激活成骨微环境中的Akt信号传导途径促进骨髓间充质干细胞分化,有利于骨组织的重建。该课题组用同样的方法治疗创伤性兔早期股骨头坏死发现两者的效果及恢复机制相似[13],高浓度肝细胞生长因子促进ERK1/2信号传导途径的活化并抑制Akt信号传导途径的活化,Akt促进骨髓间充质干细胞增殖但抑制成骨分化;然而,低浓度肝细胞生长因子可以激活Akt信号传导途径并促进成骨分化。Chen等[14]发现乙醇可影响Akt向质膜募集,并抑制Ser473处的Akt磷酸化,进而抑制骨髓间充质干细胞中Akt/GSK3β/β-连环蛋白信号传导途径,最终下游β-连环蛋白降解,骨髓间充质干细胞成骨分化能力减弱,而Akt活化剂SC-79在体内和体外给药均可阻断乙醇对成骨分化的抑制作用,并推测SC-79是乙醇诱导股骨头坏死的潜在治疗方法。Huang等[15]发现股骨头坏死区域比正常髂骨处的骨髓间充质干细胞成骨分化明显降低,并且在Wnt信号传导途径中具有更高的GSK3β表达。在添加氯化锂(GSK3β抑制剂)之后,股骨头坏死区域的骨髓间充质干细胞成骨分化能力显著增加,推测Wnt信号通路介质(如GSK3β的抑制剂)可能是早期股骨头坏死的临床治疗靶点。基于研究发现成骨分化受Mdm2-p53信号传导调节及乙醇通过刺激氧化应激抑制Wnt信号通路传导进而抑制骨髓间充质干细胞成骨分化[16-17]。Chen等[18]发现PFTα可减弱乙醇诱导的p53活化和GSK3β去磷酸化,从而恢复细胞核和细胞质中的β-连环蛋白水平,促进骨髓间充质干细胞的成骨分化,起到对酒精性股骨头坏死的保护作用,即与p53/Wnt/β-连环蛋白信号通路有关;同样,Chen等[19]发现虫草素能调节Wnt/β-连环蛋白途径减弱乙醇抑制骨髓间充质干细胞成骨分化的作用而起到治疗酒精性股骨头坏死的效果。在激素性股骨头坏死方面,Zhun等[20]体外实验发现在Dkk-1干扰下能够激活骨髓间充质干细胞的Wnt/β-连环蛋白途径,抑制成脂分化并促进成骨分化达到防止其疾病进展的目的;另有研究发现geraniin和七厘接骨胶囊亦可能通过活化Wnt/β-连环蛋白途径增强骨髓间充质干细胞的增殖和成骨分化能力[21-22],或许为股骨头坏死的预防提供方向。因此作者认为,各种方式稳定细胞内相关β-连环蛋白通路水平可能作为促进骨髓间充质干细胞成骨分化治疗股骨头坏死的一个新方向。 除Wnt/β-连环蛋白信号通路的调控之外,TGF-β/ BMPs、MAPK、Notch、Hedgehog及JNK信号通路在骨髓间充质干细胞成骨分化中发挥了重要作用[23-24]。骨细胞Wnt的作用机制是通过调节自身Notch配体的表达而进一步激活骨髓间充质干细胞的Notch信号通路,从而调控其成骨分化[25]。与此同时,Hedgehog和BMP信号通路之间可相互作用,调节smad共同促进成骨分化[26]。各个信号通路在骨髓间充质干细胞的定向分化过程中并不是单独作用,而是形成信号通路网络,在特定的刺激因素下发挥协同调控作用。王大伟等[27]在体内实验通过兔早期酒精性股骨头坏死股骨头穿刺介入给药的方法在股骨头按0.1 mL/kg局部注射三七总皂甙(对照组为等量的生理盐水和复方骨肽),每周1次,连续治疗4周后,通过扫描电镜观察骨组织超微结构发现骨细胞可见到嵴结构,且脂滴数量明显减少,证明三七总皂甙可有效促进早期兔酒精性股骨头坏死骨细胞超微结构形态修复,认为三七总皂甙可作为预防早期股骨头坏死新的靶点。研究发现氧化应激在早期激素性股骨头坏死的发生中发挥了积极作用[28],并且三七总皂甙可通过其抗氧化作用保护兔免受早期类固醇诱导的股骨头坏死。如Wang等[29]发现沉默转化生长因子β1基因可以中断三七总皂甙的成骨作用,并认为三七总皂甙可能通过上调转化生长因子β1的基因表达,在体外促进兔骨髓间充质干细胞成骨分化。研究发现miR-708靶向通过SMAD3(转化生长因子β信号传导的重要组成部分)途径促进激素对骨髓间充质干细胞成骨分化,抑制成脂分化[30],并推测miR-708可作为预防和治疗股骨头坏死的新型治疗靶点。李学东等[31]在成骨诱导液中分别加入P38MAPK抑制剂和三七总皂甙后,检测发现P38MAPK抑制剂组细胞成骨活性明显低于三七总皂甙组,推测三七总皂甙可能活化P38MAPK信号通路促进骨髓间充质干细胞的成骨分化及增殖。以上可见一种药物或不同药物通过不同的信号通路以及相互之间的作用方式调控骨髓间充质干细胞的成骨分化达到预防股骨头坏死的作用。 另外,仍有很多药物研究发现能促进骨髓间充质干细胞的成骨分化起到降低股骨头坏死发病的风险,但未发现其有关信号通路的作用机制[32-34],需要进一步研究。 2.3 骨髓间充质干细胞成脂分化与股骨头坏死 研究表明类固醇和酒精都能促进脂肪生成[35],同时抑制骨生成和血管生成,导致骨质疏松和骨坏死,且股骨头坏死骨髓间充质干细胞可以优先分化为脂肪细胞而不是成骨细胞[36],因此抑制骨髓间充质干细胞成脂分化可能成为股骨头坏死的潜在治疗策略。Han等[37]在大鼠类固醇诱导的股骨头坏死模型体外及体内实验发现利福平可通过增强P-gp(P-糖蛋白,且糖皮质激素激素是P-gp的底物)活性抑制骨髓间充质干细胞的成脂分化,从而降低股骨头坏死发生的风险。研究发现P-gp基因三磷酸腺苷结合盒B1(ABCB1)基因多态性与中国人群激素性股骨头坏死风险的遗传相关[38],且Sun等[39]发现淫羊藿苷可通过ABCB1启动子的去甲基化诱导P-gp重新表达,抑制激素性股骨头坏死患者骨髓间充质干细胞的成脂分化增强其成骨分化。因此推测P-gp活性水平可作为抑制骨髓间充质干细胞成脂分化治疗股骨头坏死的一个监测指标。 PPARγ主要调节脂肪的生成,PPARγ的高表达可促进骨髓间充质干细胞成脂分化,降低成骨分化而引起股骨头坏死。双酚A二缩水甘油醚作为PPARγ的合成拮抗剂,可通过抑制体外和体内的骨髓间充质干细胞成脂分化达到降低激素性股骨头坏死发生的风险[40]。Jiang等[41]发现普伐他汀可以通过抑制PPARγ的表达和激活Wnt信号通路,增加类固醇诱导股骨头坏死大鼠股骨头中Wnt3a、LRP5、β-catenin和RUNX2在mRNA和蛋白水平的表达,预防类固醇诱导的股骨头坏死。Kong等[42]发现HuoGu配方可抑制PPARγ、LPL和AP2的mRNA和蛋白表达,并调节Wnt/β-连环蛋白信号通路抑制骨髓间充质干细胞的成脂分化,从而延迟股骨头坏死的进展。因此Wnt/β-连环蛋白信号通路与骨髓间充质干细胞的成脂分化在股骨头坏死形成及治疗中同样起到重要作用。鉴于骨髓间充质干细胞成骨分化和成脂分化动态平衡关系,诸多信号通路能抑制成脂分化同时促进成骨分化达到干预股骨头坏死的目的。如Hedgehog信号通路促进骨髓间充质干细胞的成骨分化,抑制其成脂分化[43],促红细胞生成素通过激活ERK和p38 MAPK抑制小鼠骨髓间充质干细胞成脂分化并促进其成骨分化[44]。 2.4 骨髓间充质干细胞成软骨分化与股骨头坏死 目前关于股骨头坏死关节软骨改变的报道较少。研究显示股骨头骨小梁坏死势必会导致软骨下区的微循环障碍,关节软骨代谢变化发生在股骨头塌陷前,并随坏死程度的加重而加重[45]。研究发现股骨头坏死关节软骨的退变开始于塌陷后的坏死区域[46],并且随着骨坏死骨缺损表面积比的增加,股骨头表面软骨坏死率也随之增高[47],可见改善早期股骨头坏死软骨代谢或许成为保髋治疗的一种新的方法。Han等[48]发现颗粒体蛋白前体在股骨头坏死患者关节软骨中的表达显著上调,并能激活ERK1/2通路改善软骨细胞合成代谢,在股骨头坏死的发病机制中发挥治疗作用。另外体外实验证实多种方法可以促进骨髓间充质干细胞成软骨分化。在股骨头软骨修复方面,杨飞[49]制备犬股骨头软骨缺损模型,然后用骨髓间充质干细胞复合Bio-gide胶原膜处理,组织学观察发现软骨损伤处有软骨陷窝形成,与周围软骨愈合良好,基本接近软骨组织;同样,李国威等[50]为探讨小儿股骨头坏死症与髋关节软骨损伤关系,对幼年大鼠髋关节软骨损伤模型行骨髓间充质干细胞复合双相磷酸钙陶瓷处理,结果发现软骨损伤程度得到明显改善,促进软骨细胞增殖以及坏死股骨头修复,并推测该方法或许为股骨头坏死的保髋治疗提供一种软骨修复的新思路和方法。 虽然在体外诱导骨髓间充质干细胞成软骨分化且有部分信号通路参与,如Wnt/β-catenin信号通路和MAPK/ERK信号通路[51-52],见表1,但其定向分化为软骨细胞的机制以及在体内作用效果仍需进一步研究。骨髓间充质干细胞定向成软骨分化修复股骨头坏死不同时期软骨病变而达到改善股骨头坏死髋关节功能甚至是保髋治疗目的的研究比较少,需要进一步骨组织工程研究。"
| [1] Kubo T, Ueshima K, Saito M, et al. Clinical and basic research on steroid-induced osteonecrosis of the femoral head in Japan. J Orthop Sci. 2016;21(4):407-413.[2] Piuzzi NS, Chahla J, Schrock JB, et al. Evidence for the Use of Cell-Based Therapy for the Treatment of Osteonecrosis of the Femoral Head: A Systematic Review of the Literature. J Arthroplasty. 2017; 32(5):1698-1708.[3] Rackwitz L, Reichert JC, Haversath M, et al. Cell-based and future therapeutic strategies for femoral head necrosis. Orthopade. 2018; 47(9):770-776.[4] Bianco P, Robey PG, Simmons PJ. Mesenchymal stem cells: revisiting history, concepts, and assays. Cell Stem Cell. 2008;2(4):313-319.[5] 李瑞琦,张国平,任立中,等.骨髓间充质干细胞治疗股骨头坏死的评价[J] .中国组织工程研究,2013,17(35):6327-6332.[6] Yoon BH, Jones LC, Chen CH, et al. Etiologic Classification Criteria of ARCO on Femoral Head Osteonecrosis Part 1: Glucocorticoid-Associated Osteonecrosis. J Arthroplasty. 2019;34(1): 163-168.[7] Yoon BH, Jones LC, Chen CH, et al. Etiologic Classification Criteria of ARCO on Femoral Head Osteonecrosis Part 2: Alcohol-Associated Osteonecrosis. J Arthroplasty. 2019;34(1):169-174.[8] Wang Q, Yang Q, Chen G, et al. LncRNA expression profiling of BMSCs in osteonecrosis of the femoral head associated with increased adipogenic and decreased osteogenic differentiation. Sci Rep. 2018;8(1):9127.[9] Li J, Wang Y, Li Y, et al. The effect of combined regulation of the expression of peroxisome proliferator-activated receptor-γ and calcitonin gene-related peptide on alcohol-induced adipogenic differentiation of bone marrow mesenchymal stem cells. Mol Cell Biochem. 2014;392(1-2):39-48.[10] 杨广杰,卜一多,周炳康,等.激素性股骨头坏死模型大鼠骨髓间充质干细胞增殖及定向诱导能力均降低[J].中国组织工程研究,2015,19(41): 6579-6583.[11] Wang C, Meng H, Wang Y, et al. Analysis of early stage osteonecrosis of the human femoral head and the mechanism of femoral head collapse. Int J Biol Sci. 2018;14(2):156-164.[12] Wen Q, Ma L, Chen YP, et al. Treatment of avascular necrosis of the femoral head by hepatocyte growth factor-transgenic bone marrow stromal stem cells. Gene Ther. 2008;15(23):1523-1535.[13] Wen Q, Jin D, Zhou CY, et al. HGF-transgenic MSCs can improve the effects of tissue self-repair in a rabbit model of traumatic osteonecrosis of the femoral head. PLoS One. 2012;7(5):e37503.[14] Chen YX, Tao SC, Xu ZL, et al. Novel Akt activator SC-79 is a potential treatment for alcohol-induced osteonecrosis of the femoral head. Oncotarget. 2017;8(19):31065-31078.[15] Huang L, Wang Y, Jiang Y, et al. High levels of GSK-3β signalling reduce osteogenic differentiation of stem cells in osteonecrosis of femoral head. J Biochem. 2018;163(3):243-251.[16] Chen H, Kolman K, Lanciloti N, et al. p53 and MDM2 are involved in the regulation of osteocalcin gene expression. Exp Cell Res. 2012; 318(8):867-876.[17] Chen JR, Lazarenko OP, Shankar K, et al. A role for ethanol-induced oxidative stress in controlling lineage commitment of mesenchymal stromal cells through inhibition of Wnt/beta-catenin signaling. J Bone Miner Res. 2010;25(5):1117-1127.[18] Chen YX, Zhu DY, Yin JH, et al. The protective effect of PFTα on alcohol-induced osteonecrosis of the femoral head. Oncotarget. 2017;8(59):100691-100707.[19] Chen YX, Zhu DY, Xu ZL, et al. The Protective Effect of Cordycepin On Alcohol-Induced Osteonecrosis of the Femoral Head. Cell Physiol Biochem. 2017;42(6):2391-2403.[20] Zhun W, Donghai L, Zhouyuan Y, et al. Efficiency of Cell Therapy to GC-Induced ONFH: BMSCs with Dkk-1 Interference Is Not Superior to Unmodified BMSCs. Stem Cells Int. 2018;2018:1340252.[21] Mo J, Yang R, Li F, et al. Geraniin promotes osteogenic differentiation of bone marrow mesenchymal stem cells (BMSCs) via activating β-catenin: a comparative study between BMSCs from normal and osteoporotic rats. J Nat Med. 2019;73(1):262-272.[22] Song HB, Jiang Y, Liu JX, et al. Stimulation of osteogenic differentiation in bone marrow stromal cells via Wnt/β-catenin pathway by Qili Jiegu-containing serum. Biomed Pharmacother. 2018;103: 1664-1668.[23] Deng ZL, Sharff KA, Tang N, et al. Regulation of osteogenic differentiation during skeletal development. Front Biosci. 2008;13: 2001-2021.[24] Kwon HS, Johnson TV, Tomarev SI. Myocilin stimulates osteogenic differentiation of mesenchymal stem cells through mitogen-activated protein kinase signaling. J Biol Chem. 2013;288(23):16882-16894.[25] 任磊,代光明,林枭,等.骨细胞Wnt/β-Catenin通过Notch信号促进BMSCs成骨分化[J].中国骨质疏松杂志,2018,24(5):600-605.[26] 迟博婧,刘光源,邢磊,等.Hedgehog信号通路调控骨形成及BMSCs成骨分化的研究进展[J].中国修复重建外科杂志,2016,30(12):1545-1550.[27] 王大伟,卢培根,贾永龙,等.三七总皂苷干预酒精性股骨头缺血坏死模型兔的超微结构[J].中国组织工程研究, 2014,18(27):4277-4281.[28] Qiang H, Liu H, Ling M, et al. Early Steroid-Induced Osteonecrosis of Rabbit Femoral Head and Panax notoginseng Saponins: Mechanism and Protective Effects. Evid Based Complement Alternat Med. 2015; 2015:719370.[29] Wang Y, Huang X, Tang Y, et al. Effects of panax notoginseng saponins on the osteogenic differentiation of rabbit bone mesenchymal stem cells through TGF-β1 signaling pathway. BMC Complement Altern Med. 2016;16(1):319.[30] Hao C, Yang S, Xu W, et al. MiR-708 promotes steroid-induced osteonecrosis of femoral head, suppresses osteogenic differentiation by targeting SMAD3. Sci Rep. 2016;6:22599.[31] 李学东,陈斌,郑创义,等.三七总皂甙通过P38MAPK信号通路促进大鼠骨髓基质细胞向成骨细胞的增殖、分化[J].中国伤残医学, 2009,17(4):167-168.[32] Han N, Li Z, Cai Z, et al. P-glycoprotein overexpression in bone marrow-derived multipotent stromal cells decreases the risk of steroid-induced osteonecrosis in the femoral head. J Cell Mol Med. 2016;20(11):2173-2182.[33] Zhou D, Chen YX, Yin JH, et al. Valproic acid prevents glucocorticoid?induced osteonecrosis of the femoral head of rats. Int J Mol Med. 2018; 41(6):3433-3447.[34] Samy AM. Management of osteonecrosis of the femoral head: A novel technique. Indian J Orthop. 2016;50(4):359-365.[35] Seamon J, Keller T, Saleh J, et al. The pathogenesis of nontraumatic osteonecrosis. Arthritis. 2012;2012:601763.[36] Yu Z, Fan L, Li J, et al. Lithium chloride attenuates the abnormal osteogenic/adipogenic differentiation of bone marrow-derived mesenchymal stem cells obtained from rats with steroid-related osteonecrosis by activating the β-catenin pathway. Int J Mol Med. 2015;36(5):1264-1272.[37] Han N, Yan ZQ, Guo CA, et al. Effect of rifampicin on the risk of steroid-induced osteonecrosis of the femoral head. Orthop Surg. 2010;2(2):124-133.[38] Zhang Y, Kong X, Wang R, et al. Genetic association of the P-glycoprotein gene ABCB1 polymorphisms with the risk for steroid-induced osteonecrosis of the femoral head in Chinese population. Mol Biol Rep. 2014;41(5):3135-3146.[39] Sun ZB, Wang JW, Xiao H, et al. Icariin may benefit the mesenchymal stem cells of patients with steroid-associated osteonecrosis by ABCB1-promoter demethylation: a preliminary study. Osteoporos Int. 2015;26(1):187-197.[40] Yuan N, Li J, Li M, et al. BADGE, a synthetic antagonist for PPARγ, prevents steroid-related osteonecrosis in a rabbit model. BMC Musculoskelet Disord. 2018;19(1):129.[41] Jiang Y, Zhang Y, Zhang H, et al. Pravastatin prevents steroid-induced osteonecrosis in rats by suppressing PPARγ expression and activating Wnt signaling pathway. Exp Biol Med (Maywood). 2014;239(3):347-355.[42] Kong X, Li X, Zhang C, et al. Ethyl acetate fraction of Huogu formula inhibits adipogenic differentiation of bone marrow stromal cells via the BMP and Wnt signaling pathways. Int J Biol Sci. 2017;13(4):480-491.[43] 蔡加琴,邓力.Hedgehog信号通路对MSCs的调控[J].中国修复重建外科杂志,2010,24(8):993-996.[44] 刘革修,朱锦灿,陈小宇,等.促红细胞生成素通过激活ERK和p38 MAPK抑制小鼠骨髓间充质干细胞成脂分化[J].中国病理生理杂志, 2013,29(11): 2006-2010.[45] 刘朝晖,郭万首,杨东,等.软骨延迟增强核磁成像法检测Ⅱ期、Ⅲ期股骨头坏死软骨改变[J].中华医学杂志,2011,91(21):1467-1470.[46] Sonoda K, Motomura G, Kawanami S, et al. Degeneration of articular cartilage in osteonecrosis of the femoral head begins at the necrotic region after collapse: a preliminary study using T1 rho MRI. Skeletal Radiol. 2017;46(4):463-467.[47] 苏敬阳,韩永台,张艳芳,等.兔股骨头软骨下骨缺损表面积比与软骨塌陷预测的相关性研究[J].临床误诊误治,2012,25(2):17-19.[48] Han Y, Si M, Zhao Y, et al. Progranulin Protects Against Osteonecrosis of the Femoral Head by Activating ERK1/2 Pathway. Inflammation. 2017;40(3):946-955.[49] 杨飞.犬骨髓间充质干细胞复合Bio-gide胶原膜修复股骨头软骨缺损的实验研究[D]. 遵义:遵义医学院,2013.[50] 李国威,郭远清,陈涛,等.骨髓间充质干细胞复合双相磷酸钙陶瓷对幼年大鼠软骨损伤修复的影响[J].中国组织工程研究,2017,21(30):4781-4786.[51] 汪建样,殷嫦嫦,武翠翠,等.淫羊藿素通过Wnt/β-catenin信号通路促进BMSCs成软骨分化[J].中国中药杂志,2016,41(4):694-699.[52] 杨海杰,李志超,王勉,等.IL-6通过MAPK/ERK信号通路调节BMSCs软骨定向分化[J].中国生物化学与分子生物学报,2015,31(2):153-160.[53] 崔操,李厚成,郭一行,等. 激素性股骨头坏死微血管变化及高压氧治疗机制的研究[J].中华航海医学与高气压医学杂志,2014,21(1):59-63.[54] Tancharoen W, Aungsuchawan S, Pothacharoen P, et al. Differentiation of mesenchymal stem cells from human amniotic fluid to vascular endothelial cells. Acta Histochem. 2017;119(2):113-121.[55] Wang C, Li Y, Yang M, et al. Efficient Differentiation of Bone Marrow Mesenchymal Stem Cells into Endothelial Cells in Vitro. Eur J Vasc Endovasc Surg. 2018;55(2):257-265.[56] Chen Y, Wang J, Zhu X, et al. The directional migration and differentiation of mesenchymal stem cells toward vascular endothelial cells stimulated by biphasic calcium phosphate ceramic. Regen Biomater. 2018;5(3):129-139.[57] Xu J, Liu X, Chen J, et al. Simvastatin enhances bone marrow stromal cell differentiation into endothelial cells via notch signaling pathway. Am J Physiol Cell Physiol. 2009; 296(3):C535-543.[58] 沈彦.右归饮联合骨髓基质干细胞两种介入法治疗早期股骨头坏死的对比实验研究[D].杭州:浙江中医药大学,2009.[59] Lee NJ, Doyle KL, Sainsbury A, et al. Critical role for Y1 receptors in mesenchymal progenitor cell differentiation and osteoblast activity. J Bone Miner Res. 2010;25(8):1736-1747.[60] Beckmann J, Knödl M, Bauser E, et al. Loss of sympathetic nerve fibers in vital intertrochanteric bone cylinders lateral to osteonecrosis of the femoral head. Joint Bone Spine. 2013;80(2):188-194.[61] 王磊,王坤正,王娜,等.骨组织神经在激素性股骨头坏死中作用的探讨[J].中国矫形外科杂志,2009,17(7):515-517.[62] 喻钧伦,唐曦,肖文,等.胶质细胞源性神经营养因子对股骨头缺血坏死的保护作用[J].中国骨质疏松杂志,2017,23(6):701-706.[63] 张战峰.激素对人骨髓间充质干细胞神经肽受体mRNA表达的影响[D]. 郑州:郑州大学,2012. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [14] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||