Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (1): 118-124.doi: 10.3969/j.issn.2095-4344.1532
Previous Articles Next Articles
Differentiation of human pluripotent stem cells into dopaminergic neurons: security risk for heterogeneity
Chen Li1, Hu Lan1, Peng Yanan2, Yang Liu1, Shen Hui1, Wang Tan1, Zhao Zhenqiang1
- 1Department of Neurology, the First Affiliated Hospital of Hainan Medical University, Haikou 570102, Hainan Province, China; 2the United Laboratory for Neurosciences, Hainan Medical University, Haikou 570100, Hainan Province, China
-
Revised:2018-09-07Online:2019-01-08Published:2018-11-28 -
Contact:Zhao Zhenqiang, MD, Chief physician, Master’s supervisor, the United Laboratory for Neurosciences, Hainan Medical University, Haikou 570102, Hainan Province, China -
About author:Chen Li, Master candidate, Physician, Department of Neurology, the First Affiliated Hospital of Hainan Medical University, Haikou 570102, Hainan Province, China -
Supported by:the National Natural Science Foundation of China, No. 31260231 (to ZZQ); the Major Research and Development Project of Hainan Province, No. ZDYF2018233 (to ZZQ); Hainan International Cooperation Project, No. KJHZ2015-09 (to ZZQ); Research and Cultivation Project of Hainan Medical University, No. HY2018-20 (to PYN); Research Projects of Hainan Health and Family Planning Industry, No. 18A200171 (to ZZQ)
CLC Number:
Cite this article
Chen Li, Hu Lan, Peng Yanan, Yang Liu, Shen Hui, Wang Tan, Zhao Zhenqiang. Differentiation of human pluripotent stem cells into dopaminergic neurons: security risk for heterogeneity[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(1): 118-124.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
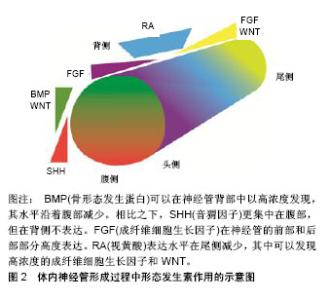
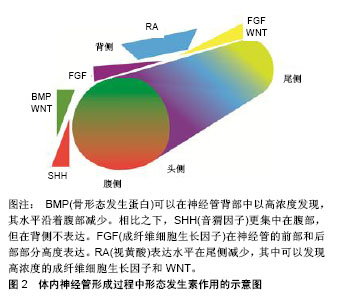
2.1 帕金森病的细胞替代疗法现状 胚胎腹侧中脑组织中含有多巴胺能神经祖细胞,其移植治疗帕金森病患者的临床研究已经开展了30多年。尽管临床结局好坏不一,但这些临床研究已证明细胞替代疗法的确是一种非常有希望的治疗策略,成为持续的研究热点[10]。然而由于伦理问题,难以得到大量的胎儿中脑组织,存在移植物质量参差不齐,难以标准化等问题,胚胎腹侧中脑细胞移植很难大规模开展。 人多能干细胞(human pluripotent stem cells,hPSCs)包括人胚胎干细胞(human embryonic stem cells,hESCs)和人诱导多能干细胞(human induced pluripotent stem cells,hiPSCs),均具有自我更新和分化成多种人类细胞类型(包括中脑多巴胺能神经元)的潜能[11]。因此,人多能干细胞为细胞替代疗法的开展提供了有价值的资源[12-13]。通常情况下,人多能干细胞在体内分化的不可控性,并不适合直接移植。此外,神经干细胞倾向于分化为中间神经元类型和星形胶质细胞,体外很难产生足够数量的多巴胺能神经元,因此从治疗的安全性和有效性综合考虑,移植已经定向的多巴胺能祖细胞更具可行性[14]。Kikuchi小组[15]的实验显示无饲养层和无血清条件下由人诱导多能干细胞分化衍生的多巴胺能神经元,在1-甲基-4-苯基-1,2,3,6-四氢吡啶(1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine,MPTP)损伤的帕金森病猴体内存活6个月。该报道支持人诱导多能干细胞对未来临床试验的治疗潜力。Kirkeby小组[16]则使用优化的方案分化人胚胎干细胞,将衍生的多巴胺能神经元移植入纹状体内,能改善6-羟基多巴胺诱导的半帕金森病大鼠运动功能。同时多个研究小组证实,通过特定的诱导方案可将人多能干细胞分化为A9多巴胺能神经元[17-19]。移植后的多巴胺能神经元可以在动物模型脑内存活、神经支配,促进功能修复[20-21],而没有产生肿瘤[22]。2017年中国科学院周琪院士主持的“人胚胎干细胞来源的神经祖细胞治疗帕金森病”的临床前期研究在郑州大学第一附属医院正式开展,表明干细胞替代疗法已经十分接近临床。 2.2 当前人多能干细胞分化为中脑多巴胺能神经元诱导方案的设计基本原理及机制 2.2.1 中脑多巴胺能神经元发育的前后模式和背腹模式 在胚胎发育过程中,发育模式和细胞命运特异性分化受到局部分泌的形态发生素和生长因子的调节。神经的形成是一个发展的过程。它从外胚层开始,神经上皮层或神经板折叠形成神经管,大脑和脊髓从该神经管发育而来。神经管沿着延喙尾轴和背腹轴生长,此发育模式在时间和空间上受沿前后模式和背腹模式轴分布的形态发生素浓度梯度的协调控制。此过程中,细胞分化由形态发生因子和邻近细胞分泌的信号分子诱导[23]。影响前后模式的形态发生素包括成纤维细胞生长因子、WNT和视黄酸,经典WNT/ β-catenin信号通路在其中扮演了关键角色。影响背腹模式的成分包括WNT、骨形态发生蛋白和音猬因子,SHH信号通路起到了决定性作用。沿前后模式和背腹模式轴分布的形态发生素浓度梯度限定了转录密码和特定区域神经前体细胞的身份,见图2。形态发生素浓度轻微的变化就可以引起细胞命运的转变[24]。这两种模式是体外方案中调控WNT信号通路和SHH信号通路可以决定中脑神经前体表型的理论依据。"
| [1] Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord. 2015;30(12): 1591-1601. [2] Chaudhuri KR, Healy DG, Schapira AH, et al. Non-motor symptoms of Parkinson’s disease: diagnosis and management. Lancet Neurol. 2006;5(3):235-245. [3] Codrich M, Bertuzzi M, Russo R, et al. Neuronal hemoglobin affects dopaminergic cells’ response to stress. Cell Death Dis. 2017;8(1):e2538. [4] 中华医学会神经病学分会帕金森病及运动障碍学组.中国帕金森病的诊断标准(2016版)[J].中华神经科杂志, 2016,49(4): 268-271.[5] Kirkeby A, Parmar M, Barker RA. Strategies for bringing stem cell-derived dopamine neurons to the clinic: A European approach (STEM-PD). Prog Brain Res. 2017;230:165-190. [6] Udd M, Lyytinen J, Eerola-Rautio J, et al. Problems related to levodopa-carbidopa intestinal gel treatment in advanced Parkinson's disease. Brain Behav. 2017;7(7):e00737. [7] Suarez-Cedeno G, Suescun J, Schiess MC. Earlier Intervention with Deep Brain Stimulation for Parkinson's Disease. Parkinsons Dis. 2017;2017:9358153. [8] Virhammar J, Nyholm D. Levodopa-carbidopa enteral suspension in advanced Parkinson's disease: clinical evidence and experience. Ther Adv Neurol Disord. 2017; 10(3):171-187. [9] Tillmann AC, Andrade A, Swarowsky A, et al. Brazilian Samba Protocol for Individuals With Parkinson's Disease: A Clinical Non-Randomized Study. JMIR Res Protoc. 2017;6(7):e129. [10] Li W, Englund E, Widner H, et al. Extensive graft-derived dopaminergic innervation is maintained 24 years after transplantation in the degenerating parkinsonian brain. Proc Natl Acad Sci U S A. 2016;113(23):6544-6549. [11] Xia N, Fang F, Zhang P, et al. A Knockin Reporter Allows Purification and Characterization of mDA Neurons from Heterogeneous Populations. Cell Rep. 2017;18(10): 2533-2546. [12] Fox IJ, Daley GQ, Goldman SA, et al. Stem cell therapy. Use of differentiated pluripotent stem cells as replacement therapy for treating disease. Science. 2014;345(6199):1247391. [13] Nguyen HN, Byers B, Cord B, et al. LRRK2 mutant iPSC-derived DA neurons demonstrate increased susceptibility to oxidative stress. Cell Stem Cell. 2011;8(3): 267-280. [14] 王昱凯,周琪,胡宝洋.人胚胎干细胞来源多巴胺祖细胞治疗帕金森病猴[J].生命科学,2016(8):902-906.[15] Kikuchi T, Morizane A, Doi D, et al. Survival of human induced pluripotent stem cell-derived midbrain dopaminergic neurons in the brain of a primate model of Parkinson's disease. J Parkinsons Dis. 2011;1(4):395-412. [16] Kirkeby A, Grealish S, Wolf DA, et al. Generation of regionally specified neural progenitors and functional neurons from human embryonic stem cells under defined conditions. Cell Rep. 2012;1(6):703-714. [17] Nolbrant S, Heuer A, Parmar M, et al. Generation of high-purity human ventral midbrain dopaminergic progenitors for in vitro maturation and intracerebral transplantation. Nat Protoc. 2017;12(9):1962-1979. [18] Zhao Z, Ma Y, Chen Z, et al. Effects of Feeder Cells on Dopaminergic Differentiation of Human Embryonic Stem Cells. Front Cell Neurosci. 2016;10:291. [19] 彭雅南,胡兰,王埮,等.人胚胎干细胞源多巴胺能神经元的功能性分化[J].中国组织工程研究,2017,21(33):5360-5368.[20] Kirkeby A, Nolbrant S, Tiklova K, et al. Predictive Markers Guide Differentiation to Improve Graft Outcome in Clinical Translation of hESC-Based Therapy for Parkinson's Disease. Cell Stem Cell. 2017;20(1):135-148. [21] Doi D, Samata B, Katsukawa M, et al. Isolation of human induced pluripotent stem cell-derived dopaminergic progenitors by cell sorting for successful transplantation. Stem Cell Reports. 2014;2(3):337-350. [22] Grealish S, Diguet E, Kirkeby A, et al. Human ESC-derived dopamine neurons show similar preclinical efficacy and potency to fetal neurons when grafted in a rat model of Parkinson's disease. Cell Stem Cell. 2014;15(5):653-665. [23] Jessell TM. Neuronal specification in the spinal cord: inductive signals and transcriptional codes. Nat Rev Genet. 2000;1(1):20-29. [24] Arenas E, Denham M, Villaescusa JC. How to make a midbrain dopaminergic neuron. Development. 2015;142(11): 1918-1936. [25] Bragdon B, Moseychuk O, Saldanha S, et al. Bone morphogenetic proteins: a critical review. Cell Signal. 2011;23(4):609-620. [26] Ji EH, Kim J. SoxD Transcription Factors: Multifaceted Players of Neural Development. Int J Stem Cells. 2016;9(1): 3-8. [27] Panman L, Papathanou M, Laguna A, et al. Sox6 and Otx2 control the specification of substantia nigra and ventral tegmental area dopamine neurons. Cell Rep. 2014;8(4): 1018-1025. [28] Ebrahimi-Fakhari D, Maas B, Haneke C, et al. Disruption of SOX6 is associated with a rapid-onset dopa-responsive movement disorder, delayed development, and dysmorphic features. Pediatr Neurol. 2015;52(1):115-118. [29] Kriks S, Shim JW, Piao J, et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson’s disease. Nature. 2011;480(7378):547-551. [30] Veenvliet JV, Dos Santos MT, Kouwenhoven WM, et al. Specification of dopaminergic subsets involves interplay of En1 and Pitx3. Development. 2013;140(16):3373-3384. [31] Memic F, Knoflach V, Morarach K, et al. Transcription and Signaling Regulators in Developing Neuronal Subtypes of Mouse and Human Enteric Nervous System. Gastroenterology. 2018;154(3):624-636. [32] Jovanovic VM, Salti A, Tilleman H, et al. BMP/SMAD Pathway Promotes Neurogenesis of Midbrain Dopaminergic Neurons In Vivo and in Human Induced Pluripotent and Neural Stem Cells. J Neurosci. 2018;38(7):1662-1676. [33] Tao Y, Zhang SC. Neural Subtype Specification from Human Pluripotent Stem Cells. Cell Stem Cell. 2016;19(5):573-586. [34] Smidt MP. Molecular Programming of Mesodiencephalic Dopaminergic Neuronal Subsets. Front Neuroanat. 2017; 11:59. [35] Mesman S, von Oerthel L, Smidt MP. Mesodiencephalic dopaminergic neuronal differentiation does not involve GLI2A-mediated SHH-signaling and is under the direct influence of canonical WNT signaling. PLoS One. 2014;9(5): e97926. [36] Sánchez-Danés A, Consiglio A, Richaud Y, et al. Efficient generation of A9 midbrain dopaminergic neurons by lentiviral delivery of LMX1A in human embryonic stem cells and induced pluripotent stem cells. Hum Gene Ther. 2012;23(1): 56-69. [37] Xi J, Liu Y, Liu H, et al. Specification of midbrain dopamine neurons from primate pluripotent stem cells. Stem Cells. 2012;30(8):1655-1663. [38] Nishimura K, Takahashi J. Therapeutic application of stem cell technology toward the treatment of Parkinson's disease. Biol Pharm Bull. 2013;36(2):171-175. [39] Samata B, Doi D, Nishimura K, et al. Purification of functional human ES and iPSC-derived midbrain dopaminergic progenitors using LRTM1. Nat Commun. 2016;7:13097. [40] Kee N, Volakakis N, Kirkeby A, et al. Single-Cell Analysis Reveals a Close Relationship between Differentiating Dopamine and Subthalamic Nucleus Neuronal Lineages. Cell Stem Cell. 2017;20(1):29-40. [41] Niclis JC, Gantner CW, Hunt CPJ, et al. A PITX3-EGFP Reporter Line Reveals Connectivity of Dopamine and Non-dopamine Neuronal Subtypes in Grafts Generated from Human Embryonic Stem Cells. Stem Cell Reports. 2017;9(3): 868-882. [42] Sundberg M, Bogetofte H, Lawson T, et al. Improved cell therapy protocols for Parkinson's disease based on differentiation efficiency and safety of hESC-, hiPSC-, and non-human primate iPSC-derived dopaminergic neurons. Stem Cells. 2013;31(8):1548-1562. [43] Politis M, Wu K, Loane C, et al. Serotonergic neurons mediate dyskinesia side effects in Parkinson's patients with neural transplants. Sci Transl Med. 2010;2(38):38ra46. [44] Fasano CA, Chambers SM, Lee G, et al. Efficient derivation of functional floor plate tissue from human embryonic stem cells. Cell Stem Cell. 2010;6(4):336-347. [45] Andersson ER, Saltó C, Villaescusa JC, et al. Wnt5a cooperates with canonical Wnts to generate midbrain dopaminergic neurons in vivo and in stem cells. Proc Natl Acad Sci U S A. 2013;110(7):E602-610. [46] Lu J, Zhong X, Liu H, et al. Generation of serotonin neurons from human pluripotent stem cells. Nat Biotechnol. 2016; 34(1):89-94. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [3] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| [4] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [5] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Wu Dandan, Liu Qi. Epigenetic mechanisms in periodontal disease, periodontal cell osteogenesis and bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5716-5722. |
| [13] | Li Yuwei, Wang Haijiao, Cui Wei, Zhou Peng, Li Cheng, Xiao Wei, Hu Bingtao, Li Fan. Percutaneous vertebroplasty with mesh-hold in the treatment of spinal metastases with extensive destruction of cortical bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5490-5494. |
| [14] | Wang Pei. Application of polycaprolactone-based biopolymer scaffolds in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(34): 5506-5510. |
| [15] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||