Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (35): 5611-5618.doi: 10.3969/j.issn.2095-4344.1432
Previous Articles Next Articles
Effect of reduction of mechanical loading on subchondral bone and articular cartilage of early osteoarthritis in mice
Shao Yijie1, Jiang Huaye1, Gao Chao1, Luo Zongping2, Yang Huilin1
- (1the First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China; 2Institute of Orthopedics at Soochow University, Suzhou 215000, Jiangsu Province, China)
-
Received:2019-05-16Online:2019-12-18Published:2019-12-18 -
Contact:Luo Zongping, Professor, Institute of Orthopedics at Soochow University, Suzhou 215000, Jiangsu Province, China Corresponding author: Yang Huilin, Chief physician, Doctoral supervisor, the First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
About author:Shao Yijie, Master candidate, the First Affiliated Hospital of Soochow University, Suzhou 215000, Jiangsu Province, China -
Supported by:the National Natural Science Foundation of China, No. 81320108018, 31570943, and 31270995 (all to LZP)
CLC Number:
Cite this article
Shao Yijie1, Jiang Huaye1, Gao Chao1, Luo Zongping2, Yang Huilin1. Effect of reduction of mechanical loading on subchondral bone and articular cartilage of early osteoarthritis in mice [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(35): 5611-5618.
share this article
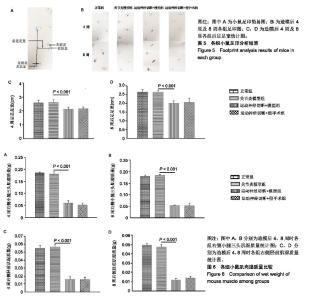
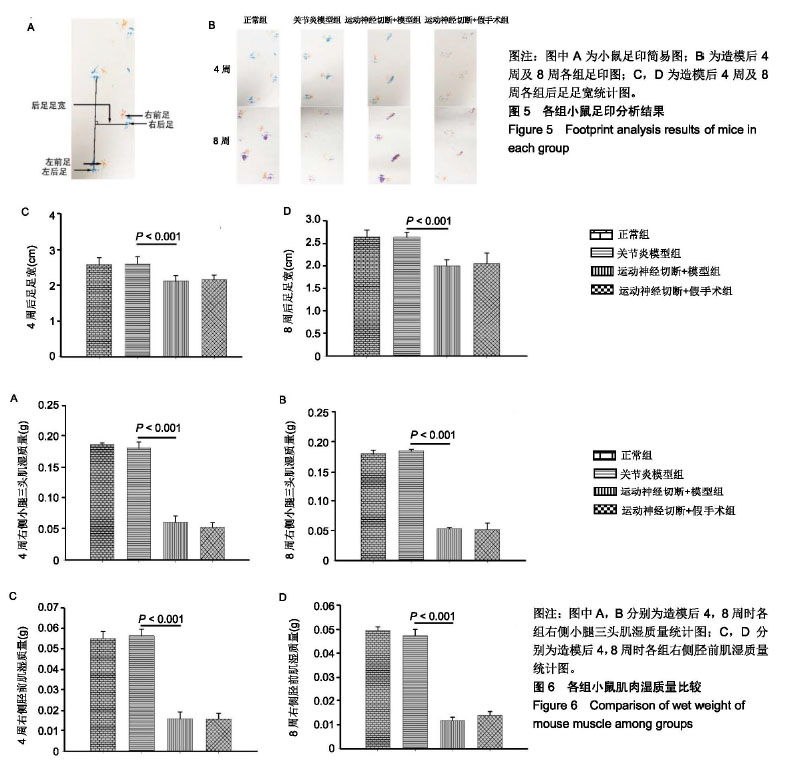
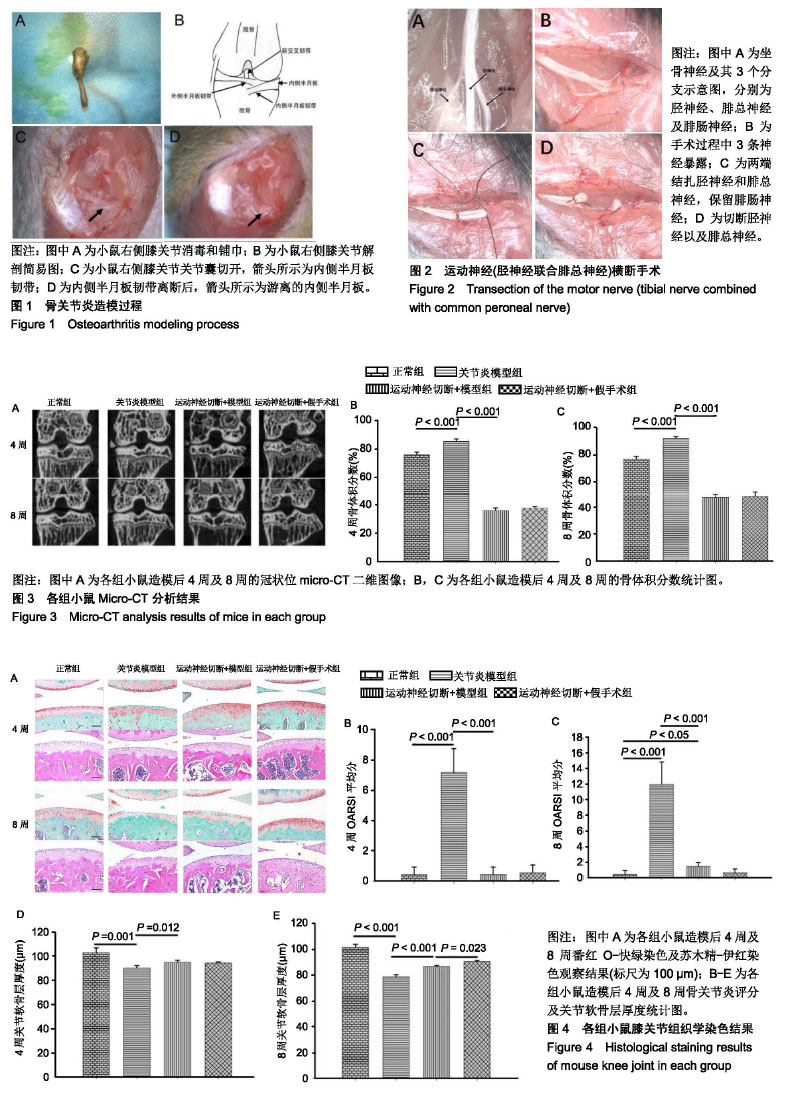
2.1 实验动物数量分析 所有48只小鼠耐受手术良好,无小鼠出现意外死亡,未出现感染迹象。 2.2 Micro-CT分析结果 造模后4周及8周,典型的Micro-CT冠状面图像见图3A,无论4周还是8周,关节炎模型组的内侧胫骨软骨下骨的骨体积分数增加,表现为骨硬化。相反,运动神经(胫神经联合腓总神经)切断后,小鼠膝关节内侧胫骨软骨下骨的骨体积分数急剧减少,表现为骨丢失。与关节炎模型组比较,造模后4周及8周运动神经切断+模型组中骨体积分数降低(P < 0.001),见图3B,C。运动神经切断+模型组与运动神经切断+假手术组之间差异无显著性意义。另外,运动神经切断+模型组8周时骨体积分数较4周有所增加(P < 0.05)。 2.3 组织学染色结果 如图4A所示,造模后4周,关节炎模型组软骨表面蛋白聚糖丢失(局部番红O失染),软骨表面粗糙,软骨细胞死亡。运动神经切断+模型组软骨表面轻微蛋白聚糖丢失,软骨表面较为光滑。造模后8周,骨关节炎模型组软骨表面磨损较为严重,软骨细胞大量死亡。运动神经切断+模型组软骨表面蛋白聚糖丢失范围较4周大。运动神经切断+假手术组4周及8周并未见到明显的蛋白聚糖丢失。图4B为OARSI半定量评分,随着时间的进展,关节炎模型组及运动神经切断+模型组软骨退变逐渐加重(P < 0.001),与关节炎模型组比较,造模后4周及8周运动神经切断+模型组软骨退变减轻(P < 0.001)。运动神经切断+假手术组造模后4周及8周相比于正常组软骨OARSI评分无明显差异。图4C,D示,造模后4周及8周时运动神经切断+假手术组软骨厚度较正常组降低,且8周时降低更为明显。 2.4 足印分析结果 运动神经(胫神经联合腓总神经)切断后,小鼠右足行五趾并拢、足轻度外翻。图5A为小鼠足印示意图,图5B为各组小鼠的足印图,从中可以发现运动神经切断组造模后4周及8周右前足和右后足足印重叠或接近。造模后4周及8周时,相比于关节炎模型组,运动神经切断+模型组后足足宽降低(P < 0.001),见图5C,D。此外,运动神经切断+模型组和运动神经切断+假手术组后足足宽在4周时与在8周时无明显差别。 2.5 肌肉湿质量 造模后4周及8周时,相比于关节炎模型组,运动神经切断+模型组右侧小腿三头肌肌肉湿质量明显降低(P < 0.001),见图6A,B。运动神经切断+模型组右侧小腿三头肌肌肉湿质量在造模后8周时要比4周稍降低,但差异无显著性意义。造模后4周及8周时,相比于关节炎模型组,运动神经切断+模型组右侧胫前肌肌肉湿质量明显降低(P < 0.001),见图6C,D。运动神经切断+模型组右侧胫前肌肌肉湿质量在造模后4周与8周无明显差异。 "
| [1]Hootman JM, Helmick CG. Projections of US prevalence of arthritis and associated activity limitations. Arthritis Rheum. 2006;54(1):226-229.[2]Loeser RF, Goldring SR, Scanzello CR, et al. Osteoarthritis: a disease of the joint as an organ. Arthritis Rheum. 2012;64(6): 1697-1707.[3]Felson DT. Epidemiology of hip and knee osteoarthritis. Epidemiol Rev. 1988;10:1-28.[4]Moyer RF, Ratneswaran A, Beier F, et al. Osteoarthritis year in review 2014: mechanics--basic and clinical studies in osteoarthritis. Osteoarthritis Cartilage. 2014;22(12):1989-2002.[5]Nomura M, Sakitani N, Iwasawa H, et al. Thinning of articular cartilage after joint unloading or immobilization. An experimental investigation of the pathogenesis in mice. Osteoarthritis Cartilage. 2017;25(5):727-736.[6]Cucchiarini M, de Girolamo L, Filardo G, et al. Basic science of osteoarthritis. J Exp Orthop. 2016;3(1):22.[7]Madry H, Luyten FP, Facchini A. Biological aspects of early osteoarthritis. Knee Surg Sports Traumatol Arthrosc. 2012; 20(3):407-422.[8]Bleuel J, Zaucke F, Brüggemann GP, et al. Effects of cyclic tensile strain on chondrocyte metabolism: a systematic review. PLoS One. 2015;10(3):e0119816.[9]Silverwood V, Blagojevic-Bucknall M, Jinks C, et al. Current evidence on risk factors for knee osteoarthritis in older adults: a systematic review and meta-analysis. Osteoarthritis Cartilage. 2015;23(4):507-515.[10]Bader DL, Salter DM, Chowdhury TT. Biomechanical influence of cartilage homeostasis in health and disease. Arthritis. 2011; 2011:979032.[11]Jurvelin J, Kiviranta I, Tammi M, et al. Softening of canine articular cartilage after immobilization of the knee joint. Clin Orthop Relat Res. 1986;(207):246-252.[12]Hinterwimmer S, Krammer M, Krötz M, et al. Cartilage atrophy in the knees of patients after seven weeks of partial load bearing. Arthritis Rheum. 2004;50(8):2516-2520.[13]Kim H, Iwasaki K, Miyake T, et al. Changes in bone turnover markers during 14-day 6 degrees head-down bed rest. J Bone Miner Metab. 2003;21(5):311-315.[14]Lang TF, Leblanc AD, Evans HJ, et al. Adaptation of the proximal femur to skeletal reloading after long-duration spaceflight. J Bone Miner Res. 2006;21(8):1224-1230.[15]Bloomfield SA, Allen MR, Hogan HA, et al. Site- and compartment-specific changes in bone with hindlimb unloading in mature adult rats. Bone. 2002;31(1):149-157.[16]Stevens HY, Meays DR, Frangos JA. Pressure gradients and transport in the murine femur upon hindlimb suspension. Bone. 2006;39(3):565-572.[17]Hanson AM, Ferguson VL, Simske SJ, et al. Comparison of tail-suspension and sciatic nerve crush on the musculoskeletal system in young-adult mice. Biomed Sci Instrum. 2005;41:92-96.[18]Malagelada F, Vega J, Guelfi M, et al. Anatomic lectures on structures at risk prior to cadaveric courses reduce injury to the superficial peroneal nerve, the commonest complication in ankle arthroscopy. Knee Surg Sports Traumatol Arthrosc. 2019 Feb 7. doi: 10.1007/s00167-019-05373-x. [Epub ahead of print][19]Guled U, Gopinathan NR, Goni VG, et al. Proximal tibial and fibular physeal fracture causing popliteal artery injury and peroneal nerve injury: A case report and review of literature. Chin J Traumatol. 2015;18(4):238-240.[20]Geyik S, Geyik M, Yigiter R, et al. Preventing Sciatic Nerve Injury due to Intramuscular Injection: Ten-Year Single-Center Experience and Literature Review. Turk Neurosurg. 2017;27(4): 636-640.[21]Zhang Q, Chen H, Liu G, et al. Comparison of healing results between tibial nerve and common peroneal nerve after sciatic nerve injury repair in rhesus monkey. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2016;30(5):608-611.[22]Glasson SS, Blanchet TJ, Morris EA. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthritis Cartilage. 2007;15(9):1061-1069.[23]Cichon J, Sun L, Yang G. Spared Nerve Injury Model of Neuropathic Pain in Mice. Bio Protoc. 2018;8(6): e2777.[24]Nakasa T, Ishikawa M, Takada T, et al. Attenuation of cartilage degeneration by calcitonin gene-related paptide receptor antagonist via inhibition of subchondral bone sclerosis in osteoarthritis mice. J Orthop Res. 2016;34(7):1177-1184.[25]Iijima H, Aoyama T, Ito A, et al. Exercise intervention increases expression of bone morphogenetic proteins and prevents the progression of cartilage-subchondral bone lesions in a post-traumatic rat knee model. Osteoarthritis Cartilage. 2016; 24(6):1092-1102.[26]Ma M, Basso DM, Walters P, et al. Behavioral and histological outcomes following graded spinal cord contusion injury in the C57Bl/6 mouse. Exp Neurol. 2001;169(2):239-254.[27]Anderson MJ, Diko S, Baehr LM, et al. Contribution of mechanical unloading to trabecular bone loss following non-invasive knee injury in mice. J Orthop Res. 2016;34(10): 1680-1687.[28]Poulet B. Non-invasive Loading Model of Murine Osteoarthritis. Curr Rheumatol Rep. 2016;18(7):40.[29]Kuyinu EL, Narayanan G, Nair LS, et al. Animal models of osteoarthritis: classification, update, and measurement of outcomes. J Orthop Surg Res. 2016;11:19.[30]Little CB, Hunter DJ. Post-traumatic osteoarthritis: from mouse models to clinical trials. Nat Rev Rheumatol. 2013;9(8): 485-497.[31]Thysen S, Luyten FP, Lories RJ. Targets, models and challenges in osteoarthritis research. Dis Model Mech. 2015;8(1):17-30.[32]Iijima H, Aoyama T, Ito A, et al. Effects of short-term gentle treadmill walking on subchondral bone in a rat model of instability-induced osteoarthritis. Osteoarthritis Cartilage. 2015;23(9):1563-1574.[33]Iijima H, Aoyama T, Ito A, et al. Destabilization of the medial meniscus leads to subchondral bone defects and site-specific cartilage degeneration in an experimental rat model. Osteoarthritis Cartilage. 2014;22(7):1036-1043.[34]Fang H, Huang L, Welch I, et al. Early Changes of Articular Cartilage and Subchondral Bone in The DMM Mouse Model of Osteoarthritis. Sci Rep. 2018;8(1):2855.[35]Lin C, Shao Y, Zeng C, et al. Blocking PI3K/AKT signaling inhibits bone sclerosis in subchondral bone and attenuates post-traumatic osteoarthritis. J Cell Physiol. 2018;233(8):6135-6147.[36]Khorasani MS, Diko S, Hsia AW, et al. Effect of alendronate on post-traumatic osteoarthritis induced by anterior cruciate ligament rupture in mice. Arthritis Res Ther. 2015;17:30.[37]Madry H, van Dijk CN, Mueller-Gerbl M. The basic science of the subchondral bone. Knee Surg Sports Traumatol Arthrosc. 2010;18(4):419-433.[38]Iwamoto J, Matsumoto H, Takeda T, et al. Effects of vitamin K2 on cortical and cancellous bone mass, cortical osteocyte and lacunar system, and porosity in sciatic neurectomized rats. Calcif Tissue Int. 2010;87(3):254-262.[39]Kingery WS, Offley SC, Guo TZ, et al. A substance P receptor (NK1) antagonist enhances the widespread osteoporotic effects of sciatic nerve section. Bone. 2003;33(6):927-936.[40]Rehan Youssef A, Longino D, Seerattan R, et al. Muscle weakness causes joint degeneration in rabbits. Osteoarthritis Cartilage. 2009;17(9):1228-1235.[41]Herzog W, Longino D, Clark A. The role of muscles in joint adaptation and degeneration. Langenbecks Arch Surg. 2003; 388(5):305-315.[42]Herzog W, Longino D. The role of muscles in joint degeneration and osteoarthritis. J Biomech. 2007;40 Suppl 1:S54-63.[43]Karamanidis K, Oberländer KD, Niehoff A, et al. Effect of exercise-induced enhancement of the leg-extensor muscle-tendon unit capacities on ambulatory mechanics and knee osteoarthritis markers in the elderly. PLoS One. 2014;9(6): e99330.[44]van der Poel C, Levinger P, Tonkin BA, et al. Impaired muscle function in a mouse surgical model of post-traumatic osteoarthritis. Osteoarthritis Cartilage. 2016;24(6):1047-1053.[45]Kim H, Wrann CD, Jedrychowski M, et al. Irisin Mediates Effects on Bone and Fat via αV Integrin Receptors. Cell. 2018; 175(7):1756-1768.e17.[46]Chen H, Hu B, Lv X, et al. Prostaglandin E2 mediates sensory nerve regulation of bone homeostasis. Nat Commun. 2019; 10(1):181.[47]Mapp PI, Walsh DA. Mechanisms and targets of angiogenesis and nerve growth in osteoarthritis. Nat Rev Rheumatol. 2012; 8(7):390-398. |
| [1] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [2] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [3] | Tang Hui, Yao Zhihao, Luo Daowen, Peng Shuanglin, Yang Shuanglin, Wang Lang, Xiao Jingang. High fat and high sugar diet combined with streptozotocin to establish a rat model of type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1207-1211. |
| [4] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [5] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [6] | Li Yonghua, Feng Qiang, Tan Renting, Huang Shifu, Qiu Jinlong, Yin Heng. Molecular mechanism of Eucommia ulmoides active ingredients treating synovitis of knee osteoarthritis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 765-771. |
| [7] | Song Shan, Hu Fangyuan, Qiao Jun, Wang Jia, Zhang Shengxiao, Li Xiaofeng. An insight into biomarkers of osteoarthritis synovium based on bioinformatics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 785-790. |
| [8] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [9] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [10] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [11] | Lü Jiaxing, Bai Leipeng, Yang Zhaoxin, Miao Yuesong, Jin Yu, Li Zhehong, Sun Guangpu, Xu Ying, Zhang Qingzhu. Evaluation of internal fixation with proximal femoral nail antirotation in elderly knee osteoarthritis patients with femoral intertrochanteric fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 391-396. |
| [12] | Zheng Li, Li Dadi, Hu Weifan, Tang Jinlong, Zhao Fengchao. Risk assessment of contralateral knee arthroplasty after unilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 374-379. |
| [13] | Luo Anyu, Liu Hanlin, Xie Xiaofei, Huang Chen. Effect of antioxidant mixture on structural degeneration of an osteoarthritis rat model [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3625-3629. |
| [14] | Gao Kun, Chen Dayu, Zhang Yong, Liu Weidong, Sun Shufen, Lai Wenqiang, Ma Dujun, Wu Yihong, Lin Zhanpeng, Jiang Yinglu, Yu Weiji. Achyranthes bidentata alcohol extract inhibits extracellular matrix degradation of the cartilage by regulating synovial fibroblast exosomes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3636-3640. |
| [15] | Liu Jinfu, Zeng Ping, Nong Jiao, Fan Siqi, Feng Chengqin, Huang Jiaxing. Integrative analysis of biomarkers and therapeutic targets in synovium of patients with osteoarthritis by multiple microarrays [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3690-3696. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||