Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (27): 4408-4413.doi: 10.3969/j.issn.2095-4344.1394
Previous Articles Next Articles
Denosumab-related osteonecrosis of the jaw: thinking from the direction of osteoclast targeted therapy
Wang Le, Zhang Xiaoming, Liu Tongbin
- (Department of Prosthodontics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China)
-
Received:2019-04-19Online:2019-09-28Published:2019-09-28 -
Contact:Zhang Xiaoming, Master, Associate professor, Master’s supervisor, Department of Prosthodontics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China -
About author:Wang Le, Master candidate, Department of Prosthodontics, Binzhou Medical University Hospital, Binzhou 256600, Shandong Province, China -
Supported by:the Medical and Health Technological Development Program of Shandong Province, No. 2016WS0121 (to LTB)
CLC Number:
Cite this article
Wang Le, Zhang Xiaoming, Liu Tongbin. Denosumab-related osteonecrosis of the jaw: thinking from the direction of osteoclast targeted therapy[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(27): 4408-4413.
share this article
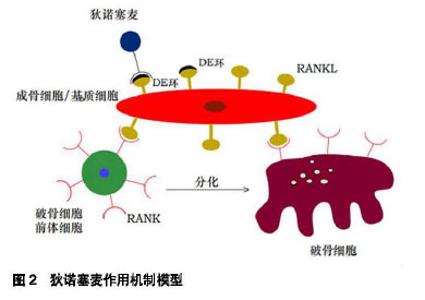
2.1 狄诺塞麦作用机制 狄诺塞麦通过作用于骨代谢核心通路RANKL/RANK来发挥作用[7]。核因子κB受体活化因子配体(receptor activator for nuclear factor-κB ligand,RANKL)是一种Ⅱ型跨膜蛋白,是人核因子κB 受体活化因子配体,主要由成骨细胞及T细胞分泌,是破骨细胞发育的主要且必须的调节剂;核因子κB受体活化因子(receptor activator for nuclear factor-κB,RANK)是位于破骨细胞(及其前体细胞)表面的I型跨膜蛋白,在树突状细胞、淋巴细胞及成熟的细胞上也有表达,影响破骨细胞(及其前体细胞)的分化与成熟,是破骨细胞维持结构、发挥功能及存活所必需的可溶性蛋白[8]。RANKL与其受体RANK相结合后,通过促进破骨细胞分化与成熟来发挥作用,调节体内成骨细胞与破骨细胞的动态平衡[9-10]。狄诺塞麦和RANKL有很高的亲和力,其竞争性结合RANKL的作用机制与成骨细胞分泌的护骨素类似,但是其亲和力更强,产生作用的有效浓度更低[11]。当狄诺塞麦与RANKL上的DE环结构相结合时,RANK信号通路传导就无法激活,破骨细胞前体细胞不能分化,导致破骨细胞功能受损甚至凋亡,骨吸收就会受到抑制。如图2所示。 "

2.2 狄诺塞麦并发症之一颌骨坏死 狄诺塞麦作为生物治疗制剂有靶向性和高效性的优点,但其在临床应用上仍有并发症发生,如颌骨坏死、低钙血症、非典型股骨骨折和免疫受损相关感染等[12]。2016年8月,Prolia®产品说明的警告和注意事项中添加了颌骨坏死,并且欧洲药品管理局以及美国食品药品管理局批准该药后都要求严格颌骨坏死的监测和预防。颌骨坏死可由放射线治疗、炎症、创伤、药物等引起,患者发生颌骨坏死后,局部血运丧失、软组织水肿破裂、颌骨骨面暴露、牙齿松动,常常伴发继发感染、骨折及神经症状等。病变部位疼痛明显,影响患者咀嚼、说话及口腔环境卫生的维护,严重损害患者的生活质量[13-14]。 药物性颌骨坏死病例报道最早出现于2003年[15],美国口腔颌面外科医师协会对药物性颌骨坏死非常关注,其定义自发现以来发生了很大变化,2014年美国口腔颌面外科医师协会完善了药物性颌骨坏死的定义与内涵[16-17]:药物性颌骨坏死多发生于下颌骨,与药物种类、给药途径及剂量、治疗持续时间、宿主机体情况、宿主口腔卫生环境情况等多种因素有关。见表1。 "

| [1]René R.Postmenopausal Osteoporosis: assessment and management. Best Pract Res Clin Endocrinol Metab. 2018; 32(5):739-757.[2]及金宝, 郑淑萍.绝经后骨质疏松症的治疗进展[J]. 世界临床药物, 2018,39(9):647-650.[3]Michael LE. Treatment of osteoporosis with denosumab. Maturitas.2010;66(2):182-186.[4]Coskun Benlidayi I. Denosumab in the treatment of glucocorticoid-induced osteoporosis. Rheumatol Int. 2018; 38(11):1975-1984.[5]Altay MA, Radu AD, Pack SE, et al.Medication-related osteonecrosis of the jaw: An institution's experience.Cranio. 2018;9:1-9.[6]Diz P, López-Cedrún JL, Arenaz J, et al.Denosumab-related osteonecrosis of the jaw. JADA.2012;143(9):981-984.[7]Kostenuik PJ,Nguyen HQ, McCabe J,et al. Denosumab, a fully human monoclonal antibody to RANKL, inhibits bone resorption and increases BMD in knock-in mice that express chimeric (murine/human) RANKL.J Bone Miner Res.2009; 24(2):182-195.[8]Fontalis A, Kenanidis E, Prousali E, et al. Safety and efcacy of denosumab in osteoporotic patients previously treated with other medications: a systematic review and meta-analysis. Expert Opin Drug Saf. 2018,17(4):413-428.[9]Lewiecki EM.New and emerging concepts in the use of denosumab for the treatment of osteoporosis. Ther Adv Musculoskelet Dis. 2018;10(11):209-223..[10]Cao X.RANKL-RANK signaling regulates osteoblast differentiation and bone formation.Bone Res.2018;6(35): 426-427.[11]Chitre M,Shechter D,Grauer A.Denosumab for treatment of postmenopausal osteoporosis.Am J Health Syst Pharm. 2011;68(15):1409-1418.[12]Pittman K, Antill Y C, Goldrick A, et al. Denosumab: Prevention and management of hypocalcemia, osteonecrosis of the jaw and atypical fractures. Asia Pac J Clin Oncol.2017; 13(4):266-276.[13]Otto S, Pautke C, Van den Wyngaert T, et al.Medication- related osteonecrosis of the jaw: Prevention, diagnosis and management in patients with cancer and bone metastases. Cancer Treat Rev. 2018;69:177-187.[14]Khan AA,Morrison A,Hanley DA,et al.Diagnosis and Management of Osteonecrosis of the Jaw: A Systematic Review and International Consensus. J Bone Miner Res. 2015;30(1):3-23. [15]王杞章. 药物性颌骨坏死的研究进展[J].华西口腔医学杂志, 2018,36(5):568-572.[16]许伟建. 药物相关性颌骨骨坏死:文献回顾与6例病例报告[D]. 杭州:浙江大学, 2017.[17]Ruggiero SL,Dodson TB,Fantasia J.American Association of Oral and Maxillofacial Surgeons Position Paper on Medication-Related Osteonecrosis of the Jaw—2014 Update. J Oral Maxillofac Surg. 2014,72(10):1938-1956.[18]Wat WZM. Current Controversies on the Pathogenesis of Medication-Related Osteonecrosis of the Jaw.Dent J (Basel). 2016 Oct 28;4(4).[19]Anastasilakis AD,Polyzos SA,Makras P.THERAPY OF ENDOCRINE DISEASE: Denosumab vs bisphosphonates for the treatment of postmenopausal osteoporosis. Eur J Endocrinol. 2018 ;179(1):R31-R45. [20]Yang H,Pan H,Yu F,et al.A novel model of bisphosphonate- related osteonecrosis of the jaw in rats.Int J Clin Exp Pathol. 2015;8(5):5161-5167.[21]Saad F, Brown J E, Van Poznak C, et al.Incidence, risk factors, and outcomes of osteonecrosis of the jaw: integrated analysis from three blinded active-controlled phase III trials in cancer patients with bone metastases. Ann Oncol.2012;23(5): 1341-1347.[22]Reid IR, Miller PD, Brown JP. Effects of denosumab on bone histomorphometry: the FREEDOM and STAND studies.J Bone Miner Res.2010;25(10):2256-2265.[23]Fizazi K,Carducci M,Smith M,et al.Denosumab versus zoledronic acid for treatment of bone metastases in men with castration-resistant prostate cancer: a randomised, double-blind study. Lancet.2011;377(9768):813-822.[24]谢冰洁,冯捷,韩向龙.破骨细胞生物学特征的研究与进展[J].中国组织工程研究, 2017,21(11):1770-1775.[25]Brown JP,Prince RL,Deal C,et al.Comparison of the effect of denosumab and alendronate on BMD and biochemical markers of bone turnover in postmenopausal women with low bone mass: a randomized, blinded, phase 3 trial.J Bone Miner Res.2009;24(1):153-161.[26]Higuchi T,Soga Y,Muro M,et al.Replacing zoledronic acid with denosumab is a risk factor for developing osteonecrosis of the jaw. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125(6): 547-551.[27]Rahim I, Salt S, Heliotis M.Successful long-term mandibular reconstruction and rehabilitation using non-vascularised autologous bone graft and recombinant human BMP-7 with subsequent endosseous implant in a patient with bisphosphonate-related osteonecrosis of the jaw. Br J Oral Maxillofac Surg. 2015;53(9):870-874.[28]Stresing V, Fournier P G, Bellahcene A, et al. Nitrogen-containing bisphosphonates can inhibit angiogenesis in vivo without the involvement of farnesyl pyrophosphate synthase.Bone. 2011;48(2):259-266.[29]Pabst AM,Ziebart T,Ackermann M.Bisphos-phonates' ?antiangiogenic?potency?in?the?development?of? bisphosphonate-associated? osteonecrosis? of? the?jaws:?influence?on?microvessel?sprouting?in?an in vivo 3D? Matrigel? assay.Clin? Oral? Invest.2014;18(3):1015-1022.[30]李彦博.双膦酸盐和下颌骨坏死[J]. 现代肿瘤医学, 2012,20(5): 1065-1069.[31]Misso G,Porru M,Stoppacciaro A,et al.Evaluation of the in vitro and in vivo antiangiogenic effects of denosumab and zoledronic acid.Cancer Biol Ther.2012;13(14):1491-1500.[32]Leibbrandt A,Penninger JM.TNF conference 2009: beyond bones - RANKL/RANK in the immune system. Adv Exp Med Biol.2011;691:5-22.[33]Bishop KA,Coy HM,Nerenz RD.Mouse Rankl expression is regulated in T cells by c-Fos through a cluster of distal regulatory enhancers designated the T cell control region. J Biol Chem. 2011 Jun 10;286(23):20880-20891.[34]Watts NB,Roux C, Modlin J F, et al.Infections in postmenopausal women with osteoporosis treated with denosumab or placebo : Coincidence or causal association. Osteoporos Int.2012;23(1):327-337.[35]Soutome S, Hayashida S, Funahara M, et al. Factors affecting development of medication-related osteonecrosis of the jaw in cancer patients receiving high-dose bisphosphonate or denosumab therapy: Is tooth extraction a risk factor?.PLOS ONE.2018;13(7):e201343.[36]Boquete-Castro A,Gomez-Moreno G,Calvo-Guirado JL,et al.Denosumab and osteonecrosis of the jaw. A systematic analysis of events reported in clinical trials.Clin Oral Implants Res. 2016,27(3):367-375.[37]车路阳,刘长振,黄鹏.RANKL/RANK/OPG通路及其相关药物狄诺塞麦治疗骨质疏松的研究进展[J].世界最新医学信息文摘, 2017,17(34):43-46.[38]Ohga N, Sato J, Asaka T, et al.Successful conservative treatment of jaw osteonecrosis caused by denosumab in patients with multiple bone metastasis. J Oral Sci. 2018;60(1): 159-162.[39]Pichardo SE, van Merkesteyn JP. Evaluation of a surgical treatment of denosumab-related osteonecrosis of the jaws. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122(3):272-278.[40]Hoefert S,Yuan A,Munz A,et al.Clinical course and therapeutic outcomes of operatively and non-operatively managed patients with denosumab-related osteonecrosis of the jaw (DRONJ). J Craniomaxillofac Surg. 2017;45(4):570-578. [41]Voss PJ,Steybe D,Poxleitner P,et al.Osteonecrosis of the jaw in patients transitioning from bisphosphonates to denosumab treatment for osteoporosis.Odontology.2018;106(4):469-480.[42]苏程.药物相关性颌骨坏死的研究进展[J]. 国际口腔医学杂志, 2017,44(2):228-234.[43]Spanou A,Lyritis GP, Chronopoulos E, et al. Management of bisphosphonate-related osteonecrosis of the jaw: a literature review. Oral Diseases.2015;21(8):927-936.[44]郭玉兴,郭传瑸.二膦酸盐相关颌骨骨坏死临床治疗研究进展[J].中国实用口腔科杂志, 2016,9(3):178-181.[45]Emma D.Deeks. Denosumab: A Review in Postmenopausal Osteoporosis. Drugs Aging. 2018;35(2):163-173.[46]Tsourdi E,Zillikens MC.Certainties and Uncertainties About Denosumab Discontinuation. Calcif Tissue Int.2018;103(1):1-4.[47]McClung MR,Wagman RB,Miller PD,et al.Observations following discontinuation of long-term denosumab therapy. Osteoporos Int.2017;28(5):1723-1732.[48]Zanchetta MB, Boailchuk J, Massari F,et al.Significant bone loss after stopping long-term denosumab treatment: a post FREEDOM study.Osteoporos Int.2018;29(1):41-47.[49]Anastasilakis AD,Polyzos SA,Makras P,et al.Clinical Features of 24 Patients With Rebound-Associated Vertebral Fractures After Denosumab Discontinuation: Systematic Review and Additional Cases.J Bone Miner Res.2017;32(6):1291-1296.[50]Maryam I,Crystal D,Pilar M,et al.Effects of Denosumab After Treatment Discontinuation : A Review of the Literature. Consult Pharm. 2018;33(3):142-151. [51]Katsarelis H,Shah NP,Dhariwal DK,et al.Infection and medication-related osteonecrosis of the jaw.J Dent Res.2015; 94(4):534-539.[52]Owosho AA,Blanchard A,Levi L, et al. Osteonecrosis of the jaw in patients treated with denosumab for metastatic tumors to the bone: A series of thirteen patients.J Craniomaxillofac Surg. 2016;44(3):265-270.[53]Matsumoto A,Sasaki M,Schmelzeisen R,et al.Primary wound closure after tooth extraction for prevention of medication- related osteonecrosis of the jaw in patients under denosumab. Clin Oral Investig.2017;21(1):127-134.[54]Chan BH,Yee R,Puvanendran R, et al. Medication-related osteonecrosis of the jaw in osteoporotic patients: prevention and management. Singapore Med J. 2018;59(2):70-75. |
| [1] | Tang Hui, Yao Zhihao, Luo Daowen, Peng Shuanglin, Yang Shuanglin, Wang Lang, Xiao Jingang. High fat and high sugar diet combined with streptozotocin to establish a rat model of type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1207-1211. |
| [2] | Li Zhongfeng, Chen Minghai, Fan Yinuo, Wei Qiushi, He Wei, Chen Zhenqiu. Mechanism of Yougui Yin for steroid-induced femoral head necrosis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1256-1263. |
| [3] | Hou Guangyuan, Zhang Jixue, Zhang Zhijun, Meng Xianghui, Duan Wen, Gao Weilu. Bone cement pedicle screw fixation and fusion in the treatment of degenerative spinal disease with osteoporosis: one-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 878-883. |
| [4] | Li Shibin, Lai Yu, Zhou Yi, Liao Jianzhao, Zhang Xiaoyun, Zhang Xuan. Pathogenesis of hormonal osteonecrosis of the femoral head and the target effect of related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 935-941. |
| [5] | Cao Xuhan, Bai Zixing, Sun Chengyi, Yang Yanjun, Sun Weidong. Mechanism of “Ruxiang-Moyao” herbal pair in the treatment of knee osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 746-753. |
| [6] | Xiao Fangjun, Chen Shudong, Luan Jiyao, Hou Yu, He Kun, Lin Dingkun. An insight into the mechanism of Salvia miltiorrhiza intervention on osteoporosis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 772-778. |
| [7] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [8] | Zhong Yuanming, Wan Tong, Zhong Xifeng, Wu Zhuotan, He Bingkun, Wu Sixian. Meta-analysis of the efficacy and safety of percutaneous curved vertebroplasty and unilateral pedicle approach percutaneous vertebroplasty in the treatment of osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 456-462. |
| [9] | Nie Shaobo, Li Jiantao, Sun Jien, Zhao Zhe, Zhao Yanpeng, Zhang Licheng, Tang Peifu. Mechanical stability of medial support nail in treatment of severe osteoporotic intertrochanteric fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 329-333. |
| [10] | Zhu Yun, Chen Yu, Qiu Hao, Liu Dun, Jin Guorong, Chen Shimou, Weng Zheng. Finite element analysis for treatment of osteoporotic femoral fracture with far cortical locking screw [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3832-3837. |
| [11] | Yang Caihui, Liu Qicheng, Dong Ming, Wang Lina, Zuo Meina, Lu Ying, Niu Weidong. Serine/threonine protein kinases can promote bone destruction in mouse models of chronic periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3654-3659. |
| [12] | Feng Guancheng, Fang Jianming, Lü Haoran, Zhang Dongsheng, Wei Jiadong, Yu Bingbing. How does bone cement dispersion affect the early outcome of percutaneous vertebroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3450-3457. |
| [13] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [14] | Huo Hua, Cheng Yuting, Zhou Qian, Qi Yuhan, Wu Chao, Shi Qianhui, Yang Tongjing, Liao Jian, Hong Wei. Effects of drug coating on implant surface on the osseointegration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3558-3564. |
| [15] | Jiang Shengyuan, Li Dan, Jiang Jianhao, Shang-you Yang, Yang Shuye. Biological response of Co2+ to preosteoblasts during aseptic loosening of the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(21): 3292-3299. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||