Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (26): 4101-4109.doi: 10.3969/j.issn.2095-4344.0932
Magnesium phosphate/collagen peptide composite cements: the setting retardation effect of collagen peptides
Cao Xiao-feng1, 2, Wang Yi-hu1, 2, 3, Lu Hao-jun1, 2, Ma Ming1, 2, Guo Yan-chuan1, 2, 3, Mao Ke-ya4, Li Jiang-tao5
- 1Key Laboratory of Photochemical Conversion and Optoelectronic Material, 5Key Laboratory of Cryogenics, Technical Institute of Physics and Chemistry, Chinese Academy of Sciences, Beijing 100190, China; 2Biomaterials Research Center, Hangzhou Branch of Technical Institute of Physics and Chemistry, Chinese Academy of Sciences, Hangzhou 310018, Zhejiang Province, China; 3University of Chinese Academy of Sciences, Beijing 100049, China; 4Department of Orthopaedics, Chinese PLA General Hospital, Beijing 100853, China
-
Received:2018-01-16 -
Contact:Guo Yan-chuan, Ph.D., Professor, Doctoral supervisor, Key Laboratory of Photochemical Conversion and Optoelectronic Material, Technical Institute of Physics and Chemistry, Chinese Academy of Sciences, Beijing 100190, China -
About author:Cao Xiao-feng, Ph.D., Assistant professor -
Supported by:the National Natural Science Foundation of China, No. 51372276, 21506236; Science and Technology Service Network Initiative of the Chinese Academy of Sciences, No. KFJ-STS-ZDTP-016
CLC Number:
Cite this article
Cao Xiao-feng, Wang Yi-hu, Lu Hao-jun, Ma Ming, Guo Yan-chuan, Mao Ke-ya, Li Jiang-tao. Magnesium phosphate/collagen peptide composite cements: the setting retardation effect of collagen peptides[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(26): 4101-4109.
share this article
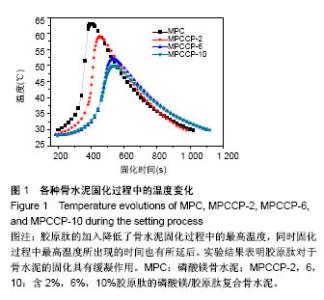
2.1 骨水泥的固化时间 不同组分磷酸镁复合骨水泥的初凝时间与终凝时间见表1。由表1可知,纯的磷酸镁骨水泥的初凝和终凝时间约为5.9 min和6.4 min。将胶原肽加入磷酸镁骨水泥中,延长了骨水泥的初凝和终凝时间。当胶原肽的含量为2%时,即MPCCP-2样品的初凝和终凝时间约为6.5 min和7.1 min。增加胶原肽的含量至6%,即MPCCP-6样品,其初凝和终凝时间分别约为7.2 min和 8.1 min。进一步增加胶原肽的含量至10%,MPCCP-10样品的初凝和终凝时间分别延长到约7.6 min和8.5 min。实验结果表明胶原肽加入的含量越高,样品的初凝和终凝时间就越长。同时,初凝和终凝时间的间隔也随着胶原肽加入量的提高而有微小的延长。 2.2 骨水泥固化过程中温度的变化 图1是磷酸镁骨水泥,MPCCP-2,MPCCP-6和MPCCP-10在室温条件下固化过程中温度变化的曲线。由图1可知,磷酸镁骨水泥固化过程中的最高温度为63 ℃。胶原肽的加入降低了骨水泥固化过程中的最高温度,随着胶原肽加入量的增加,最高温度降至低于50 ℃。同时,固化过程中最高温度所出现的时间也有所延后。实验结果表明胶原肽对于骨水泥的固化具有缓凝作用。"
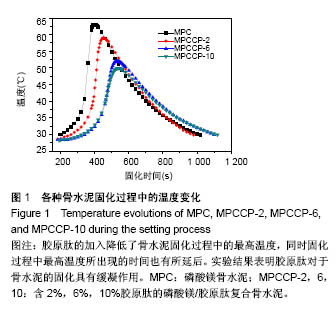
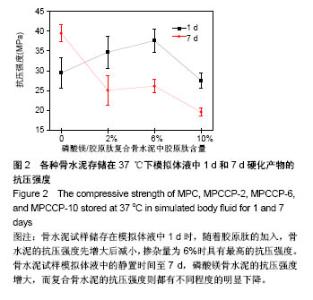
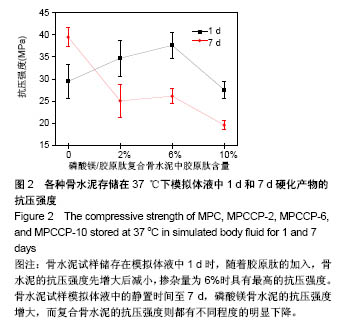
2.3 骨水泥的抗压强度 图2是4种骨水泥试样储存在模拟体液中37 ℃下1和7 d的抗压强度。当骨水泥试样储存在模拟体液中1 d时,随着胶原肽的加入,骨水泥的抗压强度增大,当掺杂量为6%时,即MPCCP-6具有最高的抗压强度。进一步增加胶原肽的含量至10%,MPCCP-10的抗压强度则明显下降。统计结果显示MPCCP-6的抗压强度比磷酸镁骨水泥和MPCCP-10显著提高(P < 0.05)。延长骨水泥试样模拟体液中的静置时间至7 d,磷酸镁骨水泥试样的抗压强度增加到(39.5±2.2) MPa,而磷酸镁/胶原肽复合骨水泥试样相对于各自1 d的抗压强度则都有不同程度的明显下降(P < 0.05)。结果显示,磷酸镁骨水泥试样的抗压强度明显高于MPCCP复合骨水泥试样(P < 0.05)。与模拟体液中静置1 d的抗压强度结果相一致,在MPCCP复合骨水泥中,MPCCP-6试样仍然具有最高的抗压强度。"
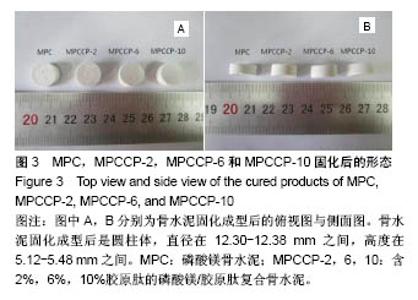
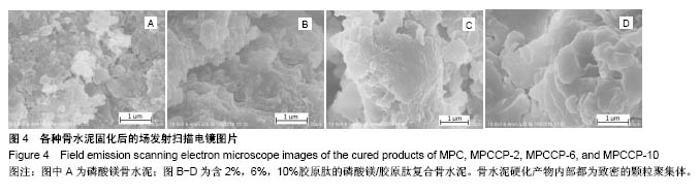
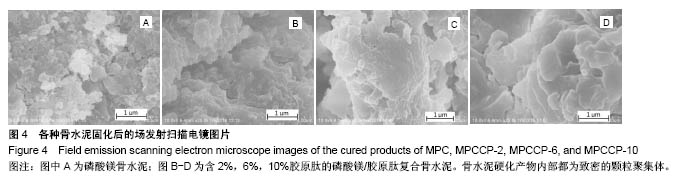
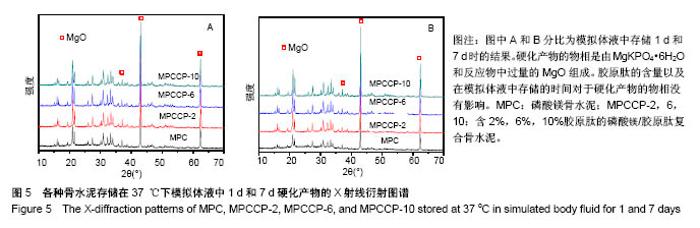
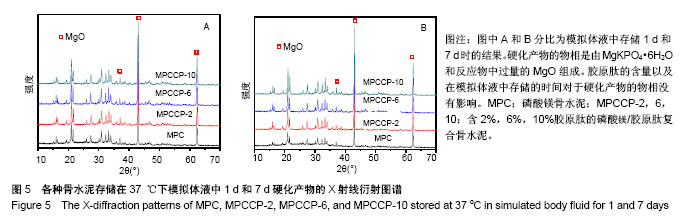
2.4 骨水泥的形貌与物相表征 图3A和B分别是磷酸镁/胶原肽复合骨水泥固化成型后的俯视图和侧面图。由图3可知成型后的骨水泥都呈圆柱体结构,截面直径为12.30-12.38 mm,高度为5.12-5.48 mm。图4是骨水泥固化后内部的场发射扫描电镜图片。由图可知硬化产物内部无规则的形貌,都为致密的颗粒聚集体。 图5A和B分别是骨水泥在37 ℃下模拟体液中静置1和7 d后的硬化产物的粉末X射线衍射谱图。通过与国际粉末衍射标准联合委员会JCPDS标准卡片35-0812 (MgKPO4•6H2O)以及89-4248(MgO)比较,硬化产物的物相是由MgKPO4•6H2O和反应物中过量的MgO组成。原料中的KH2PO4和Na2B4O7•10H2O则没有对应的衍射峰。实验结果表明胶原肽的含量以及在模拟体液中存储的时间对于硬化产物的物相没有影响。"
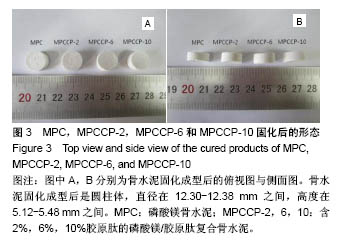
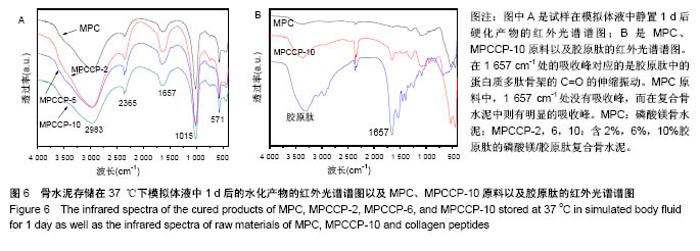
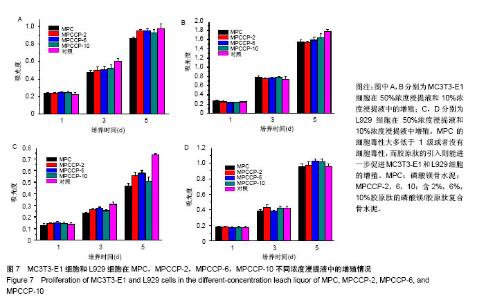
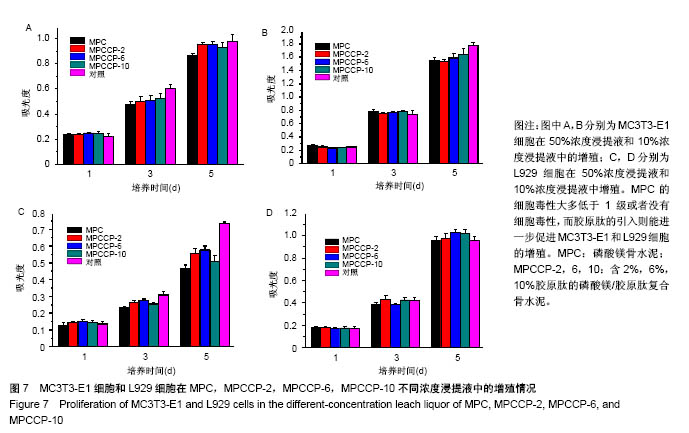
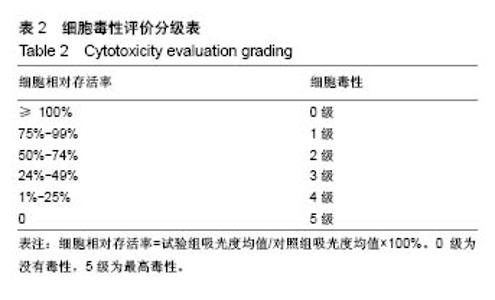
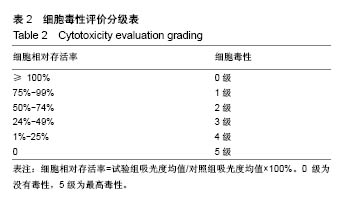
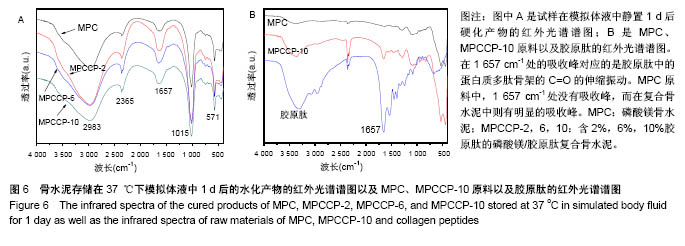
图6A是4种骨水泥试样在37 ℃下模拟体液中静置1 d后硬化产物的红外光谱谱图。由图可知,各产物在 1 008 cm-1处都有一个强吸收峰,在571 cm-1处有1个较弱的吸收峰,2个峰都归属于PO43-基团[40]。在磷酸盐的特征吸收区域(900-1 200 cm-1),尖锐的强吸收峰表明骨水泥硬化产物具有良好的结晶性 质[29],这与图5中的X射线衍射光谱中强的衍射峰相一致。在2 983 cm-1处的宽的吸收峰以及2 365 cm-1处的吸收峰归属于产物中的OH基团或水分子的吸收峰[40-41]。所有试样的谱图中,在1 600-1 700 cm-1范围内都有一中等强度的宽的吸收峰。在磷酸镁骨水泥试样谱图中,以1 620 cm-1处为中心的吸收峰,归属于水分子中H-O-H的弯曲振动[42]。在MPCCP-2,MPCCP-6和 MPCCP-10试样的红外谱图中,以1 657 cm-1处为中心的吸收峰,对应于胶原肽中的蛋白质多肽骨架的C=O的伸缩振动[43]。通过图6B中,磷酸镁骨水泥,MPCCP-10原料以及胶原肽的红外光谱可以清楚看出在1 657 cm-1处,磷酸镁骨水泥原料没有吸收峰,而MPCCP-10原料和胶原肽却有一明显的吸收峰。骨水泥试样在37 ℃下模拟体液中静置7 d后硬化产物的红外光谱谱图与图6A相同(图片未显示)。 2.5 骨水泥的细胞毒性 图7是MC3T3-E1和L929两种细胞在4种骨水泥2种浓度浸提液中1,3,5 d的生存率曲线。骨水泥细胞毒性的评级是基于表2进行[44]。在细胞培养1 d的结果中,各试样的50%浓度浸提液和10%浓度浸提液对MC3T3-E1和L929两种细胞都没有细胞毒性,与对照组相比,细胞存活率均在90%以上。在细胞培养3 d的结果中,各试样的50%浓度浸提液和10%浓度浸提液对两种细胞均为1级细胞毒性或者没有细胞毒性,与对照组相比,细胞存活率均在75%以上。对于同一细胞,50%浓度浸提液组的细胞存活率都稍低于10%浓度浸提液组。其中,L929细胞50%浓度浸提液组中,MPCCP组的细胞存活率明显高于磷酸镁骨水泥组(P < 0.05)。在细胞培养5 d的结果中,除个别结果外,试样的50%浓度浸提液和10%浓度浸提液对2种细胞均为1级细胞毒性或者没有细胞毒性,与对照组相比,细胞存活率均在75%以上,只在L929细胞50%浓度浸提液组,磷酸镁骨水泥和MPCCP-10试样具有2级细胞毒性,与对照组相比,细胞存活率分别为63.7%和69.0%。在结果统计中,MC3T3-E1细胞50%浓度浸提液组,MPCCP复合骨水泥试样的细胞存活率明显高于磷酸镁骨水泥试样(P < 0.05)。对L929细胞,50%浓度浸提液组,MPCCP-2、MPCCP-6复合骨水泥的试样的细胞存活率高于磷酸镁骨水泥试样(P < 0.05),而10%浓度浸提液组,MPCCP-6、MPCCP-10复合骨水泥的试样的5 d细胞存活率高于磷酸镁骨水泥试样(P < 0.05)。结果说明,磷酸镁骨水泥的细胞毒性大多低于1级或者没有细胞毒性,而胶原肽的引入则能进一步促进MC3T3-E1和L929细胞的增殖。"
| [1] 刘昌胜.硬组织修复材料与技术[M].北京:科学出版社,2014.[2] 王成健.多孔SrO/HA生物复合陶瓷骨修复材料的制备及体外生物相容性研究[D].昆明:昆明理工大学硕士学位论文,2016.[3] Van der Stok J, Van Lieshout EM, El-Massoudi Y, et al. Bone substitutes in the Netherlands - a systematic literature review. Acta Biomater. 2011;7(2):739-750. [4] Kolk A, Handschel J, Drescher W, et al. Current trends and future perspectives of bone substitute materials - from space holders to innovative biomaterials. J Craniomaxillofac Surg. 2012;40(8):706-718. [5] Bouler JM, Pilet P, Gauthier O, et al. Biphasic calcium phosphate ceramics for bone reconstruction: A review of biological response. Acta Biomater. 2017;53:1-12. [6] Li J, Baker BA, Mou X, et al. Biopolymer/Calcium phosphate scaffolds for bone tissue engineering. Adv Healthc Mater. 2014;3(4):469-484.[7] Ben-Nissan B. Advances in Calcium Phosphate Biomaterials. Springer. 2014.[8] Ginebra MP, Espanol M, Montufar EB, et al. New processing approaches in calcium phosphate cements and their applications in regenerative medicine. Acta Biomater. 2010;6(8):2863-2873. [9] Krüger R, Groll J. Fiber reinforced calcium phosphate cements -- on the way to degradable load bearing bone substitutes? Biomaterials. 2012;33(25):5887-5900.[10] Nezafati N, Moztarzadeh F, Hesaraki S, et al. Biological response of a recently developed nanocomposite based on calcium phosphate cement and sol-gel derived bioactive glass fibers as substitution of bone tissues. Ceram Int 2013;39(2):289-297.[11] Zhang J, Liu W, Schnitzler V, et al. Calcium phosphate cements for bone substitution: chemistry, handling and mechanical properties. Acta Biomater. 2014;10(3):1035-1049. [12] Comuzzi L, Ooms E, Jansen JA. Injectable calcium phosphate cement as a filler for bone defects around oral implants: an experimental study in goats. Clin Oral Implants Res. 2002;13(3):304-311.[13] Vater C, Lode A, Bernhardt A, et al. Influence of different modifications of a calcium phosphate bone cement on adhesion, proliferation, and osteogenic differentiation of human bone marrow stromal cells. J Biomed Mater Res A. 2010;92(4):1452-1460. [14] Farrar DF. Bone adhesives for trauma surgery: A review of challenges and developments. Int J Adhes Adhes. 2012;33(3):89-97.[15] Ostrowski N, Roy A, Kumta PN. Magnesium Phosphate Cement Systems for Hard Tissue Applications: A Review. ACS Biomater Sci Eng. 2016;2(7):1067-1083.[16] Tamimi F, Le Nihouannen D, Bassett DC, et al. Biocompatibility of magnesium phosphate minerals and their stability under physiological conditions. Acta Biomater. 2011;7(6):2678-2685. [17] Pina S, Ferreira JMF. Brushite-forming Mg-, Zn- and Sr-substituted bone cements for clinical applications. Materials. 2010;3(1):519-535.[18] Ostrowski N, Lee B, Hong D, et al. Synthesis, osteoblast, and osteoclast viability of amorphous and crystalline tri-magnesium phosphate. ACS Biomater Sci Eng. 2015;1(1):52-63.[19] Driessens FCM, Boltong MG, Zapatero MI, et al. In vivo behaviour of three calcium phosphate cements and a magnesium phosphate cement. J Mater Sci Mater Med. 1995;6(5):272-278.[20] Liu C. Inorganic bone adhesion agent and its use in human hard tissue repair: US, 7094286. 2006-08-22.[21] Waselau M, Samii VF, Weisbrode SE, et al. Effects of a magnesium adhesive cement on bone stability and healing following a metatarsal osteotomy in horses. Am J Vet Res. 2007;68(4):370-378.[22] Gulotta LV, Kovacevic D, Ying L, et al. Augmentation of tendon-to-bone healing with a magnesium-based bone adhesive. Am J Sports Med. 2008;36(7):1290-1297. [23] Li Y, Chen B. Factors that affect the properties of magnesium phosphate cement. Constr Build Mater. 2013;47:977-983.[24] Mestres G, Abdolhosseini M, Bowles W, et al. Antimicrobial properties and dentin bonding strength of magnesium phosphate cements. Acta Biomater. 2013;9(9):8384-8393. [25] Yu Y, Wang J, Liu C, et al. Evaluation of inherent toxicology and biocompatibility of magnesium phosphate bone cement. Colloids Surf B Biointerfaces. 2010;76(2):496-504. [26] Wu F, Wei J, Guo H, et al. Self-setting bioactive calcium-magnesium phosphate cement with high strength and degradability for bone regeneration. Acta Biomater. 2008;4(6):1873-1884. [27] Yang G, Liu J, Li F, et al. Bioactive calcium sulfate/magnesium phosphate cement for bone substitute applications. Mater Sci Eng C Mater Biol Appl. 2014;35:70-76. [28] Mestres G, Ginebra MP. Novel magnesium phosphate cements with high early strength and antibacterial properties. Acta Biomater. 2011; 7(4):1853-1861. [29] Zhou H, Agarwal AK, Goel VK, et al. Microwave assisted preparation of magnesium phosphate cement (MPC) for orthopedic applications: a novel solution to the exothermicity problem. Mater Sci Eng C Mater Biol Appl. 2013;33(7):4288-4294. [30] Wang AJ, Zhang J, Li JM, et al. Effect of liquid-to-solid ratios on the properties of magnesium phosphate chemically bonded ceramics. Mater Sci Eng C Mater Biol Appl. 2013;33(5):2508-2512. [31] Wang AJ, Fan XJ, Li JM, et al. Curing Behavior and Structure of Magnesium Phosphate Chemically Bonded Ceramics with Different MgO to KH2PO4 Ratios. Int J Appl Ceram Technol. 2015;12(6): 1124-1130.[32] Inzana JA, Olvera D, Fuller SM, et al. 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials. 2014;35(13):4026-4034.[33] Villa MM, Wang L, Huang J, et al. Bone tissue engineering with a collagen-hydroxyapatite scaffold and culture expanded bone marrow stromal cells. J Biomed Mater Res B Appl Biomater. 2015;103(2):243-253. [34] Sarker B, Hum J, Nazhat SN, et al. Combining collagen and bioactive glasses for bone tissue engineering: a review. Adv Healthc Mater. 2015;4(2):176-194. [35] 陈俊德,易瑞灶,陈晖.鱼胶原蛋白及其活性肽的研究进展[J].中国海洋药物杂志,2009,8(4):52-56.[36] 张兵,王毅虎,王富荣,等.浅谈胶原蛋白水解物对于人关节软骨的作用[J].明胶科学与技术,2015,35(1):44-46.[37] 蒋建新,张先龙,金东旭,等.服用胶原蛋白多肽对中老年女性膝骨关节炎患者关节疼痛和功能的临床效果研究[J].中国骨质疏松杂志, 2013,19(11): 1184-1186,1199.[38] Liu J, Zhang B, Song S, et al. Bovine collagen peptides compounds promote the proliferation and differentiation of MC3T3-E1 pre-osteoblasts. PLoS One. 2014;9(6):e99920. [39] ASTM C266-99. Standard test method for time of setting of hydraulic-cement paste by gillmore needles. PA, USA: ASTM International. 1999.[40] Lee J, Farag MM, Park EK, et al. A simultaneous process of 3D magnesium phosphate scaffold fabrication and bioactive substance loading for hard tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2014;36:252-260. [41] Wu X, Tao Y, Song C, et al. Morphological control and luminescent properties of YVO4:Eu nanocrystals. J Phys Chem B. 2006;110(32): 15791-15796.[42] 曹霄峰,张雷,马英丽,等.Zn3(OH)2V2O7•2H2O 纳米片的水热制备[J].无机化学学报,2010,26(5):787-792.[43] 杨伟,袁芳,高彦祥.鱼胶原蛋白肽与表没食子儿茶素没食子酸酯相互作用的研究[J].光谱学与光谱分析,2015,35(1):184-188.[44] 张秉文.磷酸镁骨粘合剂的生物安全性研究[D].上海:复旦大学, 2010.[45] Soudée E, Péra J. Mechanism of setting reaction in magnesia-phosphate cements. Cem Concr Res. 2000;30(2):315-321.[46] Qiao F, Chau CK, Li ZJ. Calorimetric study of magnesium potassium phosphate cement. Mater Struc. 2012;45(3):447-456.[47] Wang AJ, Yuan ZL, Zhang J, et al. Effect of raw material ratios on the compressive strength of magnesium potassium phosphate chemically bonded ceramics. Mater Sci Eng C Mater Biol Appl. 2013;33(8): 5058-5063. [48] Wang CW, Chiang TY, Chang HC, et al. Physicochemical properties and osteogenic activity of radiopaque calcium silicate-gelatin cements. J Mater Sci Mater Med. 2014;25(9):2193-2203. [49] Babaie E, Lin B, Goel VK, et al. Evaluation of amorphous magnesium phosphate (AMP) based non-exothermic orthopedic cements. Biomed Mater. 2016;11(5):055010.[50] Samali A, Holmberg CI, Sistonen L, et al. Thermotolerance and cell death are distinct cellular responses to stress: dependence on heat shock proteins. FEBS Lett. 1999;461(3):306-310. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||