Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (32): 5139-5144.doi: 10.3969/j.issn.2095-4344.0931
Previous Articles Next Articles
Interactions of Notch signaling pathway and miR-21 in peripheral nerve repair
Yang Kai-yun, Sun Pei-pei, Wu Wen-liang, Liu Hai-chun
- Department of Orthopedics, Shandong University Qilu Hospital, Jinan 250012, Shandong Province, China
-
Received:2018-03-05Online:2018-11-18Published:2018-11-18 -
Contact:Liu Hai-chun, M.D., Associate chief physician, Department of Orthopedics, Shandong University Qilu Hospital, Jinan 250012, Shandong Province, China -
About author:Yang Kai-yun, Master, Research assistant, Department of Orthopedics, Shandong University Qilu Hospital, Jinan 250012, Shandong Province, China -
Supported by:the Key Research and Development Program of Shandong Province, No. 2017GSF218047
CLC Number:
Cite this article
Yang Kai-yun, Sun Pei-pei, Wu Wen-liang, Liu Hai-chun. Interactions of Notch signaling pathway and miR-21 in peripheral nerve repair[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(32): 5139-5144.
share this article
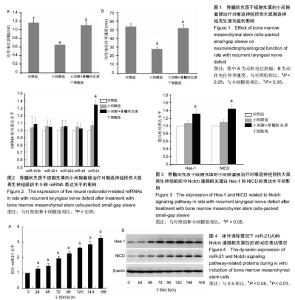
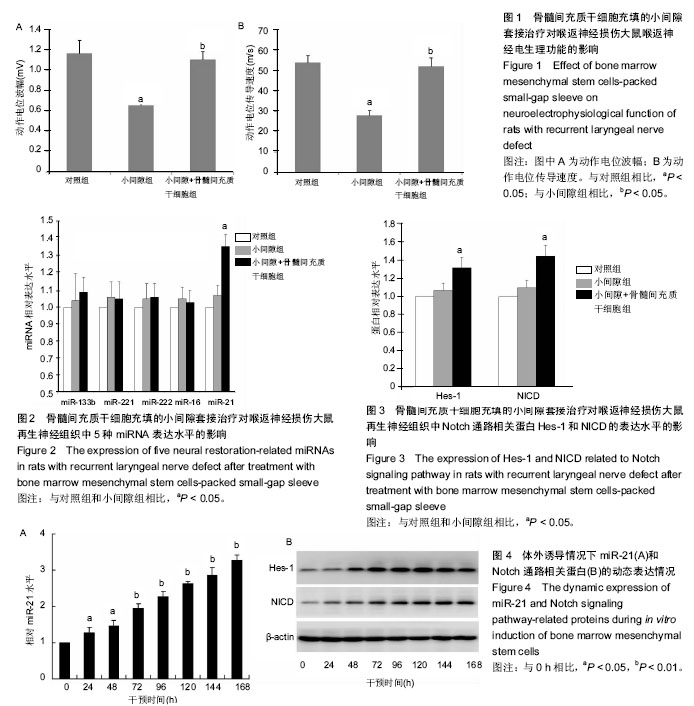
2.1 实验动物数量分析 填充骨髓间充质干细胞的小间隙套管对体内周围神经损伤治疗作用实验中小间隙组和小间隙+骨髓间充质干细胞组各有1只大鼠在实验终点前不明原因死亡,分别给予补充实验补齐。评估抑制miR-21对骨髓间充质干细胞治疗作用影响的实验中30只大鼠全部成活至实验终点。 2.2 填充骨髓间充质干细胞的小间隙套管对大鼠喉返神经损伤治疗作用 2.2.1 神经电生理结果 如图1所示,损伤后4周小间隙组神经传导速度和动作电位幅度均显著低于对照组,而小间隙+骨髓间充质干细胞组神经传导速度和动作电位幅度均显著高于小间隙组,接近对照组水平,说明小间隙+骨髓间充质干细胞组的治疗效果优于小间隙组。 2.2.2 实时PCR结果 实验选取miR-21等5种微小RNA家族成员作为检测靶点,检测了微小RNA在大鼠离体后神经组织中的异位表达情况。结果显示各组miR-221、miR-222、miR-133b和miR-16表达水平接近,对照组和小间隙组miR-21表达水平接近,而小间隙+骨髓间充质干细胞组中miR-21表达水平明显高于其他2组(图2)。 2.2.3 Western Blot检测 实验选取Hes-1和NICD作为指标以观察Notch信号通路在大鼠离体后神经组织中的表达情况。如图3所示,其中小间隙组两种蛋白的表达量稍高于对照组,差异无显著性意义;小间隙+骨髓间充质干细胞组两种蛋白的表达量明显高于其他2组。 2.3 miR-21和Notch信号通路的表达变化观察 2.3.1 miR-21和Notch信号通路表达动态测定 在体外条件下将骨髓间充质干细胞加入小间隙套管中进行神经元方向诱导分化,连续测量结果可见miR-21的表达量随时间逐"
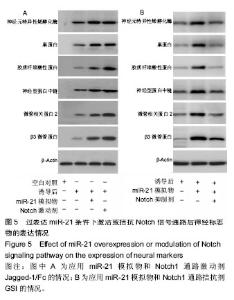
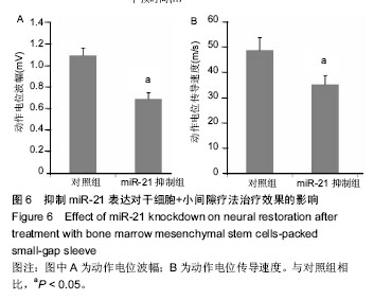
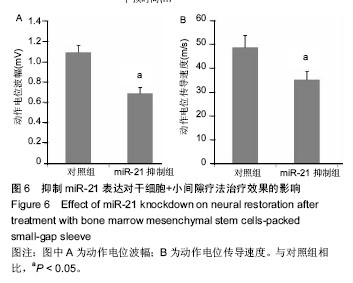
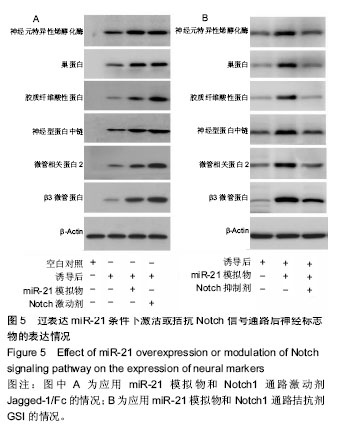
渐增加(图4A);Notch信号通路代表物Hes-1和NICD的表达亦随时间逐渐增强(图4B),其中Hes-1的表达峰值出现于120 h时间点,NICD的表达峰值出现于144 h时间点。 2.3.2 过表达miR-21条件下干预Notch信号通路的改变观察 实验首先证实在骨髓间充质干细胞中加入神经分化诱导剂后,其表达6种神经相关蛋白的能力明显增强。随后,在此体系中分别应用预先用miR-21模拟物转染的骨髓间充质干细胞使其过表达miR-21,或加入Notch通路激动剂使Notch信号通路被激活,2种条件下骨髓间充质干细胞表达6种蛋白的能力均得到明显上调(图5A)。如果在miR-21转染后的骨髓间充质干细胞体系中继续加入Notch通路抑制剂,则对6种蛋白的表达能力产生了明显的中和或抑制作用(图5B),说明Notch通路具有凌驾于miR-21之上的否决作用。"
| [1] Lundborg G. A 25-year perspective of peripheral nerve surgery: evolving neuroscientific concepts and clinical significance. J Hand Surg Am. 2000; 25(3):391-414.[2] Zujovic V, Bachelin C, Baron-Van Evercooren A. Remyelination of the central nervous system: a valuable contribution from the periphery. Neuroscientist. 2007;13(4):383-391.[3] Gerth DJ, Tashiro J, Thaller SR. Clinical outcomes for Conduits and Scaffolds in peripheral nerve repair. World J Clin Cases. 2015;3(2): 141-147.[4] Pabari A, Lloyd-Hughes H, Seifalian AM, et al. Nerve conduits for peripheral nerve surgery. Plast Reconstr Surg. 2014;133(6):1420-1430.[5] Kehoe S, Zhang XF, Boyd D. FDA approved guidance conduits and wraps for peripheral nerve injury: a review of materials and efficacy. Injury. 2012;43(5):553-572. [6] Wislet-Gendebien S, Laudet E, Neirinckx V, et al. Mesenchymal stem cells and neural crest stem cells from adult bone marrow: characterization of their surprising similarities and differences. Cell Mol Life Sci. 2012;69(15):2593-2608. [7] Croft AP, Przyborski SA. Generation of neuroprogenitor-like cells from adult mammalian bone marrow stromal cells in vitro. Stem Cells Dev. 2004;13(4):409-420.[8] Lopez-Verrilli MA, Caviedes A, Cabrera A, et al. Mesenchymal stem cell-derived exosomes from different sources selectively promote neuritic outgrowth. Neuroscience. 2016;320:129-139. [9] Woodhoo A, Alonso MB, Droggiti A, et al. Notch controls embryonic Schwann cell differentiation, postnatal myelination and adult plasticity. Nat Neurosci. 2009;12(7):839-847. [10] Yanjie J, Jiping S, Yan Z, et al. Effects of Notch-1 signalling pathway on differentiation of marrow mesenchymal stem cells into neurons in vitro. Neuroreport. 2007;18(14):1443-1447.[11] Zou D, Chen Y, Han Y, et al. Overexpression of microRNA-124 promotes the neuronal differentiation of bone marrow-derived mesenchymal stem cells. Neural Regen Res. 2014;9(12):1241-1248.[12] Zhao Y, Jiang H, Liu XW, et al. MiR-124 promotes bone marrow mesenchymal stem cells differentiation into neurogenic cells for accelerating recovery in the spinal cord injury. Tissue Cell. 2015;47(2): 140-146.[13] Zhang P, Xue F, Kou Y, et al. The experimental study of absorbable chitin conduit for bridging peripheral nerve defect with nerve fasciculu in rats. Artif Cells Blood Substit Immobil Biotechnol. 2008;36(4):360-371. [14] Yu K, Zhang C, Wang Y, et al. The protective effects of small gap sleeve in bridging peripheral nerve mutilation. Artif Cells Blood Substit Immobil Biotechnol. 2009;37(6):257-264.[15] Wang J, Zhang P, Wang Y, et al. The observation of phenotypic changes of Schwann cells after rat sciatic nerve injury. Artif Cells Blood Substit Immobil Biotechnol. 2010;38(1):24-28.[16] Wang J, Ren KY, Wang YH, et al. Effect of active Notch signaling system on the early repair of rat sciatic nerve injury. Artif Cells Nanomed Biotechnol. 2015;43(6):383-389. [17] Yang T, Liu LY, Ma YY, et al. Notch signaling-mediated neural lineage selection facilitates intrastriatal transplantation therapy for ischemic stroke by promoting endogenous regeneration in the hippocampus. Cell Transplant. 2014;23(2):221-238. [18] Chuang JH, Tung LC, Lin Y. Neural differentiation from embryonic stem cells in vitro: An overview of the signaling pathways. World J Stem Cells. 2015;7(2):437-447. [19] Xing Y, Chen X, Cao Y, et al. Expression of Wnt and Notch signaling pathways in inflammatory bowel disease treated with mesenchymal stem cell transplantation: evaluation in a rat model. Stem Cell Res Ther. 2015; 6:101. [20] Xu H, Miki K, Ishibashi S, et al. Transplantation of neuronal cells induced from human mesenchymal stem cells improves neurological functions after stroke without cell fusion. J Neurosci Res. 2010;88(16):3598-3609.[21] Osathanon T, Manokawinchoke J, Nowwarote N, et al. Notch signaling is involved in neurogenic commitment of human periodontal ligament-derived mesenchymal stem cells. Stem Cells Dev. 2013;22(8): 1220-1231.[22] Valencia-Sanchez MA, Liu J, Hannon GJ, et al. Control of translation and mRNA degradation by miRNAs and siRNAs. Genes Dev. 2006;20(5): 515-524.[23] Guo Q, Chen Y, Guo L, et al. miR-23a/b regulates the balance between osteoblast and adipocyte differentiation in bone marrow mesenchymal stem cells. Bone Res. 2016;4:16022. [24] Foshay KM, Gallicano GI. Small RNAs, big potential: the role of MicroRNAs in stem cell function. Curr Stem Cell Res Ther. 2007;2(4): 264-271.[25] Pandey A, Singh P, Jauhari A, et al. Critical role of the miR-200 family in regulating differentiation and proliferation of neurons. J Neurochem. 2015;133(5):640-652. [26] Ge XT, Lei P, Wang HC, et al. miR-21 improves the neurological outcome after traumatic brain injury in rats. Sci Rep. 2014;4:6718. [27] Zhao Y, Jiang H, Liu XW, et al. MiR-124 promotes bone marrow mesenchymal stem cells differentiation into neurogenic cells for accelerating recovery in the spinal cord injury. Tissue Cell. 2015;47(2): 140-146. [28] Zhou S, Zhang S, Wang Y, et al. MiR-21 and miR-222 inhibit apoptosis of adult dorsal root ganglion neurons by repressing TIMP3 following sciatic nerve injury. Neurosci Lett. 2015;586:43-49. [29] Junker F, Chabloz A, Koch U, et al. Dicer1 imparts essential survival cues in Notch-driven T-ALL via miR-21-mediated tumor suppressor Pdcd4 repression. Blood. 2015;126(8):993-1004.[30] Xiong Y, Zhang YY, Wu YY, et al. Correlation of over-expressions of miR-21 and Notch-1 in human colorectal cancer with clinical stages. Life Sci. 2014;106(1-2):19-24. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||