Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (21): 3328-3334.doi: 10.3969/j.issn.2095-4344.0898
Previous Articles Next Articles
Mechanism by which UM171 sustains expansion of umbilical cord blood hematopoietic stem cells in vitro
Xu Shi-qi, Ding Ya-hui, Zhang Yu, Ji Qing, Yang Ming, Li Ying-hui, Gao Ying-dai
- Key Laboratory of Experimental Hematology, Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Tianjin 300020, China
-
Revised:2018-05-21Online:2018-07-28Published:2018-07-28 -
Contact:Gao Ying-dai, M.D., Researcher, Key Laboratory of Experimental Hematology, Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Tianjin 300020, China; Li Ying-hui, M.D., Assistant researcher, Key Laboratory of Experimental Hematology, Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Tianjin 300020, China -
About author:Xu Shi-qi, Master, Key Laboratory of Experimental Hematology, Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Tianjin 300020, China -
Supported by:the National Key Research and Development Projects of China, No. 2016YFA0100602 and 2017YFA0104903; the National Natural Science Foundation of China, No. 81430004, 81421002, 81570100, 81500086, and 81600085; CAMS Innovation Fund for Medical Sciences, No. 2016-12M-3-008 and 2016-12M-1-017
CLC Number:
Cite this article
Xu Shi-qi, Ding Ya-hui, Zhang Yu, Ji Qing, Yang Ming, Li Ying-hui, Gao Ying-dai. Mechanism by which UM171 sustains expansion of umbilical cord blood hematopoietic stem cells in vitro[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(21): 3328-3334.
share this article
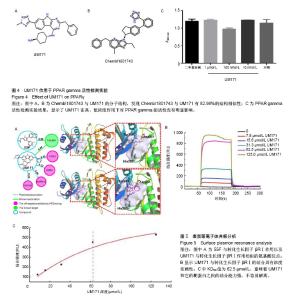
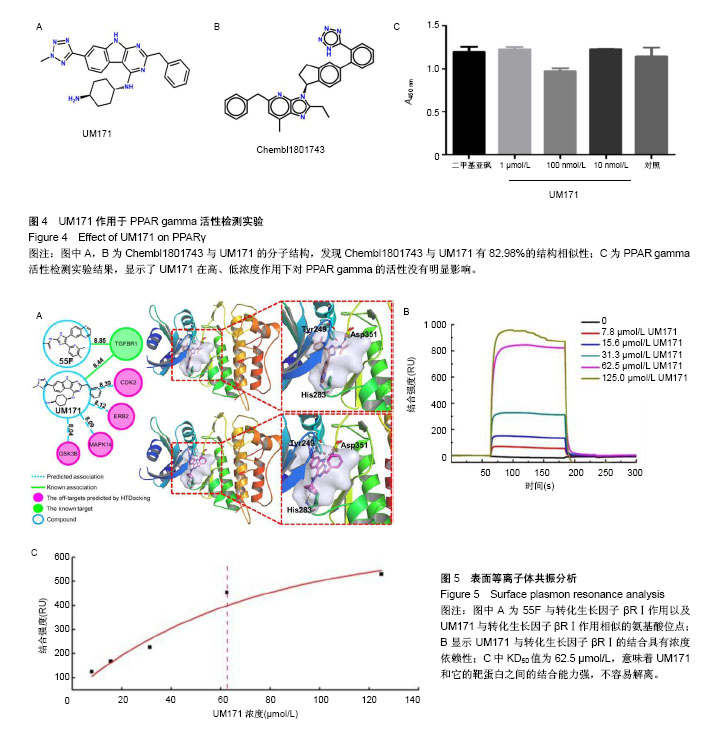
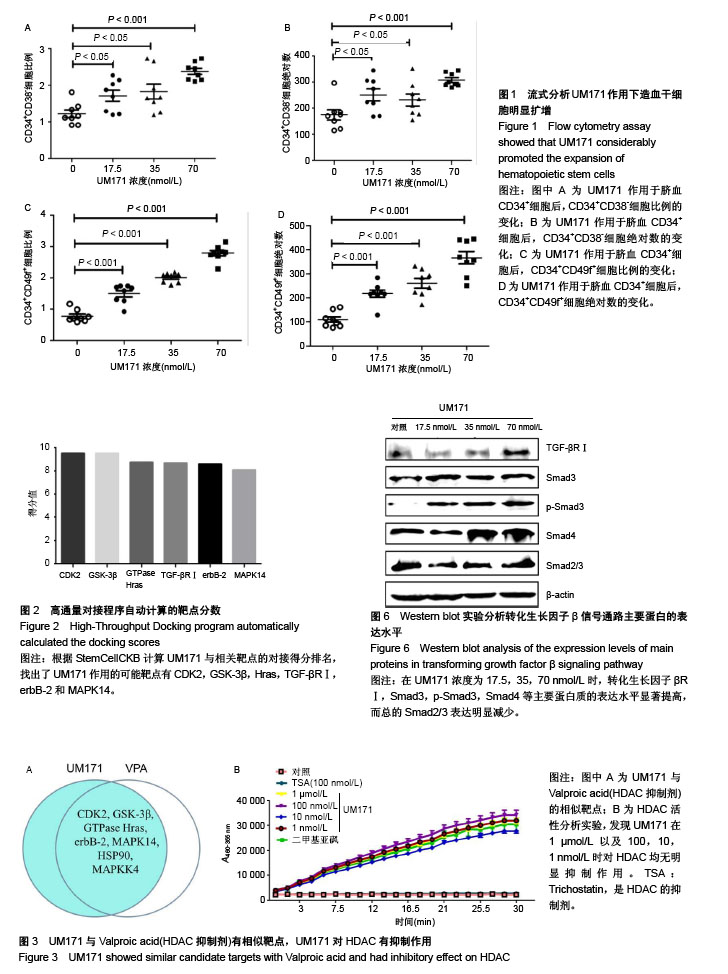
2.1 UM171作用于造血干细胞的流式检测结果 实验中使用CD34、CD38和CD49f作为造血干细胞的富集标记来验证造血干细胞扩增效果。从人脐血中收集分离的CD34+细胞进行剂量反应分析。结果显示,UM171作用 7 d后,造血干祖细胞(CD34+CD38-)的比例和绝对数量均显著增加(图1A,B)。UM171处理后相对更原始的造血干祖细胞(CD34+CD49f+)百分比增加1倍(图1C),同时CD34+CD49f+细胞的绝对数量也显著增加(图1D)。 2.2 StemCellCKB高通量对接(HTDocking)在线预测结果 首先根据HTDocking在线预测打分结果,发现UM171的可能主要蛋白靶点为CDK2,GSK-3β,Hras,TGF-βRⅠ,erbB-2和MAPK14(图2)。 2.3 高通量对接(HTDocking)提示UM171可能抑制HDAC 在蛋白靶点预测的过程中采用与UM171具有相同生物学功能的Valproic acid(HDAC抑制剂)进行对照,根据预测发现Valproic acid(HDAC抑制剂)的主要作用靶点为CDK2,GSK-3β,Hras,erbB-2和MAPK14。通过对比分析,发现Valproic acid和UM171的预测作用靶点有90%的相似度(3A),因此推测UM171也为一个HDAC抑制剂。然而通过酶活水平的验证结果可知,UM171对HDAC酶活的影响较小,因此UM171不是特异的HDAC抑制剂(图3B)。UM171与Valproic acid(HDAC抑制剂)预测结果中的差异蛋白质转化生长因子βRⅠ成为下一步研究的重点。 2.4 TargetHunter在线预测提示UM171可能抑制PPAR gamma 根据TargetHunter在线预测的结果可知,Chembl1801743化合物与UM171有82.98%的结构相似性(图4A,B),因此推测Chembl1801743化合物和UM171具有相同的作用靶点。Chembl1801743是PPAR gamma的抑制剂,根据相同结构具有相似作用机制的设想,作者初步推测UM171也为PPAR gamma的抑制剂。采用PPAR gamma分析试剂盒进行验证表明,UM171对PPAR gamma并没有显著的抑制作用(图4C)。 2.5 表面等离子体共振分析 根据HTDocking和TargetHunter在线预测的结果进行分析和排除后,初步选定转化生长因子βRⅠ成为下一步研究的重点。通过查阅相关文献也发现,转化生长因子βRⅠ与造血干细胞自我更新和干性维持具有较强的相关性[12,22]。而且已知55F可以与转化生长因子βRⅠ结合,通过结构模拟分析UM171与转化生长因子βRⅠ相互作用的评分与55F接近,作用相似氨基酸位点(图2,图5A)。为了确定UM171和转化生长因子βRⅠ之间的相互作用,使用了表面等离子体共振分析。重组的转化生长因子βRⅠ蛋白质通过标准氨基偶联化学过程,被偶联在CM5芯片上固定了10 min,结合在芯片上的蛋白达到6 000 RU,UM171被运行缓冲液稀释后在25 ℃以10 μL/min的流速上样。结果显示UM171与转化生长因子βRⅠ的结合具有浓度依赖性(图5B)。KD50值为62.5 µmol/L(图5C),这意味着UM171和它的靶蛋白之间的结合能力强,不容易解离。 2. 6转化生长因子β信号通路激活 为了证明UM171通过转化生长因子β信号通路扩增造血干细胞的途径,研究了几种已知的调节该途径的重要蛋白质。据报道,Smad2、Smad3和Smad4羧基端磷酸化被激活,引起细胞状态特异性调节的转录[23]。首先在细胞膜形成转化生长因子βRⅠ-转化生长因子βRⅡ复合物,然后转化生长因子βRⅡ磷酸化转化生长因子βRⅠ。激活的转化生长因子βRⅠ接下来激活Smads(p-Smad2,p-Smad3是Smad2和Smad3的激活形式)[24]。激活的Smad2和Smad3可以与Smad4形成复合物[25-26],转移到细胞核,然后作为转录因子调节转化生长因子β通路的靶基因[20,27-28]。分析结果表明,转化生长因子βRⅠ,Smad3,p-Smad3,Smad4等主要蛋白质的表达水平显著提高,然而总的Smad2/3表达明显减少,提示UM171主要通过上调p-Smad3和Smad3/4来扩增造血干细胞。"
| [1] Bozda? SC, Yüksel MK, Demirer T. Adult Stem Cells and Medicine. Adv Exp Med Biol. 2018 Mar 20. doi: 10. 1007/5584_2018_184. [Epub ahead of print][2] Peccatori J, Ciceri F. Allogeneic stem cell transplantation for acute myeloid leukemia. Haematologica. 2010;95(6): 857-859. [3] Gragert L, Eapen M, Williams E, et al. HLA match likelihoods for hematopoietic stem-cell grafts in the U. S. registry. N Engl J Med. 2014;371(4):339-348. [4] Fares I, Chagraoui J, Gareau Y, et al. Cord blood expansion. Pyrimidoindole derivatives are agonists of human hematopoietic stem cell self-renewal. Science. 2014;345 (6203):1509-1512. [5] Ramírez P, Brunstein CG, Miller B, et al. Delayed platelet recovery after allogeneic transplantation: a predictor of increased treatment-related mortality and poorer survival. Bone Marrow Transplant. 2011;46(7):981-986. [6] Rocha V, Labopin M, Sanz G, et al. Transplants of umbilical-cord blood or bone marrow from unrelated donors in adults with acute leukemia. N Engl J Med. 2004;351(22): 2276-2285. [7] Miller PH, Knapp DJ, Eaves CJ. Heterogeneity in hematopoietic stem cell populations: implications for transplantation. Curr Opin Hematol. 2013;20(4):257-264. [8] Morin V. Stem-cell replication to treat blood diseases. CMAJ. 2014;186(17):E648. [9] Brunstein CG, Gutman JA, Weisdorf DJ, et al. Allogeneic hematopoietic cell transplantation for hematologic malignancy: relative risks and benefits of double umbilical cord blood. Blood. 2010;116(22):4693-4699. [10] Ali S, Yu SM, Yao ZJ. Gram-Scale Laboratory Synthesis of UM171, a Potent Agonist of Human Hematopoietic Stem Cell Self-Renewal. J Org Chem. 2016;81(21):10236-10241. [11] Zhang Y, Wang L, Feng Z, et al. StemCellCKB: An Integrated Stem Cell-Specific Chemogenomics KnowledgeBase for Target Identification and Systems-Pharmacology Research. J Chem Inf Model. 2016;56(10):1995-2004. [12] Blank U, Karlsson S. TGF-β signaling in the control of hematopoietic stem cells. Blood. 2015;125(23):3542-3550. [13] Batard P, Monier MN, Fortunel N, et al. TGF-(beta)1 maintains hematopoietic immaturity by a reversible negative control of cell cycle and induces CD34 antigen up-modulation. J Cell Sci. 2000;113(Pt 3):383-390. [14] Fortunel NO, Hatzfeld A, Hatzfeld JA. Transforming growth factor-beta: pleiotropic role in the regulation of hematopoiesis. Blood. 2000;96(6):2022-2036. [15] Garbe A, Spyridonidis A, Möbest D, et al. Transforming growth factor-beta 1 delays formation of granulocyte- macrophage colony-forming cells, but spares more primitive progenitors during ex vivo expansion of CD34+ haemopoietic progenitor cells. Br J Haematol. 1997;99(4):951-958. [16] Yamazaki S, Iwama A, Takayanagi S, et al. Cytokine signals modulated via lipid rafts mimic niche signals and induce hibernation in hematopoietic stem cells. EMBO J. 2006;25(15): 3515-3523. [17] Yamazaki S, Iwama A, Takayanagi S, et al. TGF-beta as a candidate bone marrow niche signal to induce hematopoietic stem cell hibernation. Blood. 2009;113(6):1250-1256. [18] Cheshier SH, Morrison SJ, Liao X, et al. In vivo proliferation and cell cycle kinetics of long-term self-renewing hematopoietic stem cells. Proc Natl Acad Sci U S A. 1999; 96(6):3120-3125. [19] Wilson A, Laurenti E, Oser G, et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell. 2008;135(6):1118-1129. [20] Heldin CH, Landström M, Moustakas A. Mechanism of TGF-beta signaling to growth arrest, apoptosis, and epithelial-mesenchymal transition. Curr Opin Cell Biol. 2009;21(2):166-176. [21] Zuo W, Huang F, Chiang YJ, et al. c-Cbl-mediated neddylation antagonizes ubiquitination and degradation of the TGF-β type II receptor. Mol Cell. 2013;49(3):499-510. [22] Xie WL, Jiang R, Shen XL, et al. Diosgenin attenuates hepatic stellate cell activation through transforming growth factor-β/Smad signaling pathway. Int J Clin Exp Med. 2015; 8(11):20323-20329. [23] Wang Z, Fei S, Suo C, et al. Antifibrotic Effects of Hepatocyte Growth Factor on Endothelial-to-Mesenchymal Transition via Transforming Growth Factor-Beta1 (TGF-β1)/Smad and Akt/mTOR/P70S6K Signaling Pathways. Ann Transplant. 2018;23:1-10. [24] You SP, Ma L, Zhao J, et al. Phenylethanol Glycosides from Cistanche tubulosa Suppress Hepatic Stellate Cell Activation and Block the Conduction of Signaling Pathways in TGF-β1/smad as Potential Anti-Hepatic Fibrosis Agents. Molecules. 2016;21(1):102. [25] Feng XH, Derynck R. Specificity and versatility in tgf-beta signaling through Smads. Annu Rev Cell Dev Biol. 2005;21: 659-693. [26] Massagué J, Seoane J, Wotton D. Smad transcription factors. Genes Dev. 2005;19(23):2783-2810. [27] Ikushima H, Miyazono K. TGFbeta signalling: a complex web in cancer progression. Nat Rev Cancer. 2010;10(6):415-424. [28] Carli C, Giroux M, Delisle JS. Roles of transforming growth factor-β in graft-versus-host and graft-versus-tumor effects. Biol Blood Marrow Transplant. 2012;18(9):1329-1340. [29] Lee MKS, Al-Sharea A, Dragoljevic D, et al. Hand of FATe: lipid metabolism in hematopoietic stem cells. Curr Opin Lipidol. 2018;29(3):240-245. [30] Sugimura R. Bioengineering Hematopoietic Stem Cell Niche toward Regenerative Medicine. Adv Drug Deliv Rev.2016; 99(Pt B):212-220. [31] Bari S, Seah KK, Poon Z, et al. Expansion and homing of umbilical cord blood hematopoietic stem and progenitor cells for clinical transplantation. Biol Blood Marrow Transplant. 2015;21(6):1008-1019. [32] Fares I, Rivest-Khan L, Cohen S, et al. Small molecule regulation of normal and leukemic stem cells. Curr Opin Hematol. 2015;22(4):309-316. [33] Sitnicka E, Ruscetti FW, Priestley GV, et al. Transforming growth factor beta 1 directly and reversibly inhibits the initial cell divisions of long-term repopulating hematopoietic stem cells. Blood. 1996;88(1):82-88. [34] Hao S, Chen C, Cheng T. Cell cycle regulation of hematopoietic stem or progenitor cells. Int J Hematol. 2016; 103(5):487-497. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||