Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (18): 2813-2819.doi: 10.3969/j.issn.2095-4344.0868
Previous Articles Next Articles
Reconstruction of mandibular defects with tissue-engineered bone using 3D bionic printing technology
Du Guo-qing, Sun Jian, Li Ya-li, Chen Li-qiang, Chen Chen, Deng Nan, Wu Yu-tong, Li Li, Wang Zhi-hao
- Department of Oral and Maxillofacial Surgery, the Affiliated Hospital of Qingdao University, Laboratory of Oral Digital Medicine and 3D Printing Engineering, Qingdao University, Qingdao 266003, Shandong Province, China
-
Received:2018-01-15Online:2018-06-28Published:2018-06-28 -
Contact:Sun Jian, M.D., Professor, Chief physician, Master’s supervisor, Department of Oral and Maxillofacial Surgery, the Affiliated Hospital of Qingdao University, Laboratory of Oral Digital Medicine and 3D Printing Engineering, Qingdao University, Qingdao 266003, Shandong Province, China -
About author:Du Guo-qing, Master candidate, Department of Oral and Maxillofacial Surgery, the Affiliated Hospital of Qingdao University, Laboratory of Oral Digital Medicine and 3D Printing Engineering, Qingdao University, Qingdao 266003, Shandong Province, China -
Supported by:the Science and Technology Development Plan of Shandong Province, No. 2014GSF118108
CLC Number:
Cite this article
Du Guo-qing, Sun Jian, Li Ya-li, Chen Li-qiang, Chen Chen, Deng Nan, Wu Yu-tong, Li Li, Wang Zhi-hao . Reconstruction of mandibular defects with tissue-engineered bone using 3D bionic printing technology[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(18): 2813-2819.
share this article
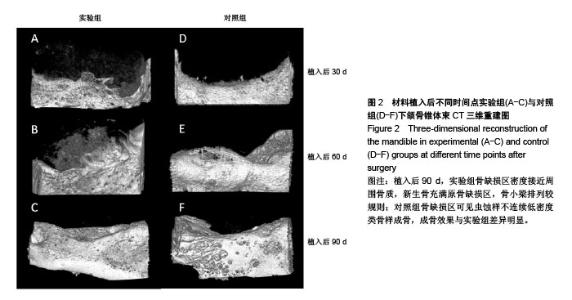
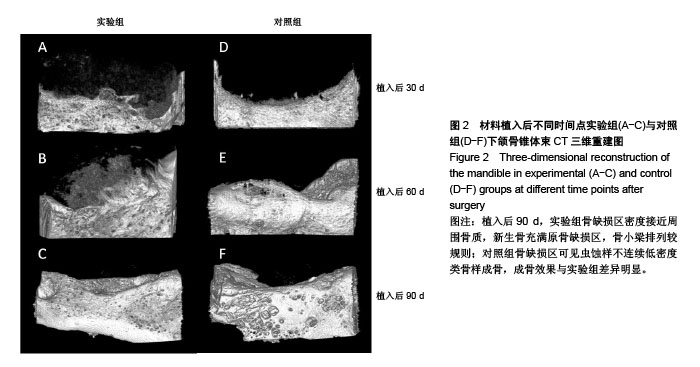
2.1 实验动物数量分析 27只新西兰大白兔均进入结果分析。 2.2 一般情况观察 术后4 d,实验兔术区肿胀,皮温稍高,未见明显渗血渗液,术后7 d切口一期愈合。 2.3 下颌骨锥体束CT三维重建结果 见图2。 实验组:植入后30 d,骨缺损区呈稀薄云雾状,未见明显钙化新骨生成;植入后60 d,骨缺损区密度较前增高,能观察到不同密度的钙化新骨生成;植入后90 d,骨缺损区密度接近周围骨质,新生骨充满原骨缺损区,骨小梁排列较规则。 对照组:植入后30 d,骨缺损区呈现低密度透光区;植入后60 d,缺损区边缘见少量不规则新骨生成,骨缺损中央区未见骨质形成;植入后90 d,骨缺损区可见虫蚀样不连续低密度类骨样成骨,其成骨效果与实验组比差异明显。"
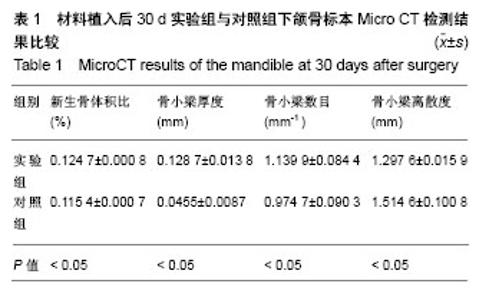
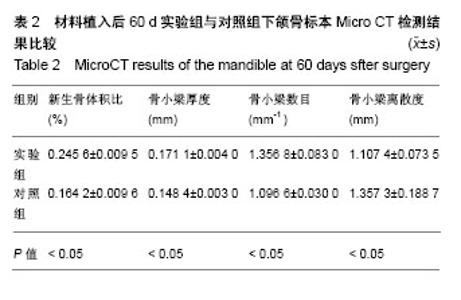
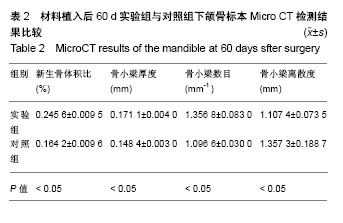
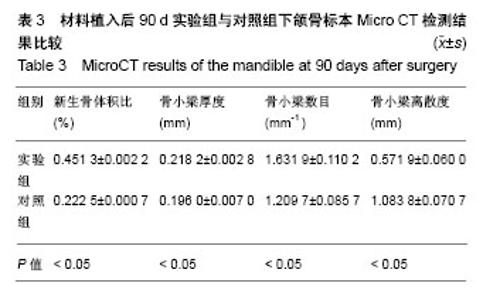
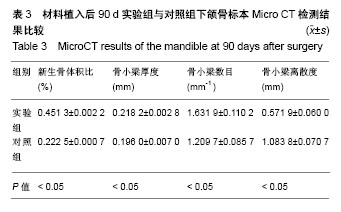
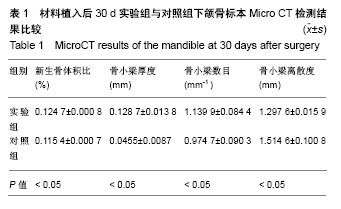
2.4 Micro CT骨组织微结构检测结果 通过图像分析系统软件对标本进行形态计量学分析得出,同一时间点实验组新生骨体积比、骨小梁厚度、骨小梁数目均大于对照组(P < 0.05),骨小梁离散度小于对照组(P < 0.05),见表1-3。 随着时间的推移,实验组新生骨体积比(F=38 584.165,P=0)、骨小梁厚度(F=83.045,P=0)、骨小梁数目(F=20.916,P=0.002)逐渐增加,骨小梁离散度(F=131.045,P=0)逐渐减小。随着时间的推移,对照组新生骨体积比(F=14384.410,P=0)、骨小梁厚度(F=183.676,P=0)、骨小梁数目(F=7.568,P=0.023)逐渐增加,骨小梁离散度(F=27.554,P=0.001)逐渐减小,差异有显著性意义(P < 0.05)。"
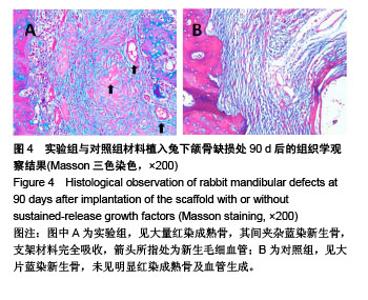
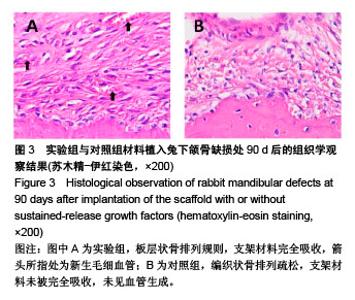
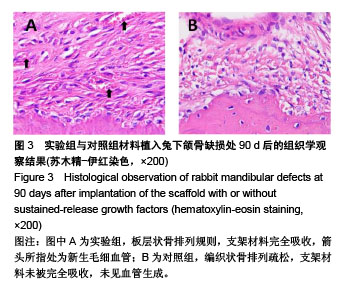
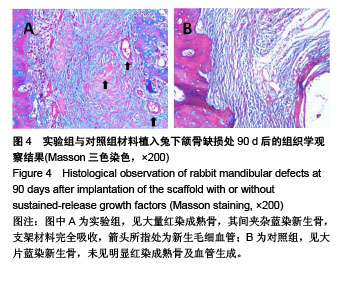
2.5.2 Masson三色染色 Masson三染在染色试剂及染色顺序一定的情况下,最终着色的颜色取决于于骨组织的成分[18-19],骨组织中主要存在Ⅰ型胶原,其成熟度直接影响Masson染色的结果,成熟骨Masson染色其胶原被染成鲜红色,在新生骨向成熟的过度中为红-蓝相间着色,由此可用Masson染色来衡量骨的成熟度,新生骨质多呈蓝染红染少,成熟骨多呈红染蓝染少[20] 。 植入后30 d,实验组成骨区截骨线边缘见少量蓝染新生骨形成,着色较浅,支架材料开始降解;对照组未见明显改变。植入后60 d,实验组蓝染新生骨增多,开始出现红染成熟骨,支架材料被大部分降解;对照组仅见少量类骨质或未成熟骨结构,未见成熟骨,支架材料开始降解。植入后90 d,实验组骨缺损区支架材料被大量红染成熟板层骨代替,其间夹杂蓝染成熟新生骨,见图4A;对照组材料大部分降解,见少量稀疏蓝染新生骨,呈不规则疏松网格状排列,未见明显红染成熟骨形成,见图4B。"
| [1] Sun J,Li YL,Chen LQ,et al.An experimental study on reconstruction of the condyle of the temporomandibular joint using free autogenous costal perichondrial grafts in rabbits. Oral Med Oral Pathol Oral Radiol Endodontol. 2010;109(5):649-796.[2] Shimada K,Tanaka H,Matsumoto T,et al.Cylindrical costal osteochondral autograft for reconstruction of large defects of the capitellum due to osteochondritis dissecans.J Bone Joint Surg Am. 2012;94(11):992-1002.[3] Mori Y,Saito T,Chang SH,et al.Identification of fibroblast growth factor-18 as a molecule to protect adult articular cartilage by gene expression profiling.J Biol Chem. 2014;289(14):10192.[4] Klammert U,Mueller TD,Hellmann TV,et al.GDF-5 can act as a context-dependent BMP-2antagonist.BMC Biol. 2015;13(1):1-18.[5] Seemann P,Mundlos S,Reissner C,et al.Bone morphogenetic protein 2 (BMP2) variants with reduced BMP antagonist sensitivity: WO,US 8828937 B2[P].2014.[6] Holly VL,Widen SA,Famulski JK,et al.Sfrp1a and Sfrp5 function as positive regulators of Wnt and BMP signaling during early retinal development.Dev Biol.2014;388(2):192.[7] Osses N,Henríquez JP.Bone morphogenetic protein signaling in vertebrate motor neurons and neuromuscular communication.Front Cell Neurosci.2015;8:453. [8] 周庆梅,孙健,李亚莉,等.生长因子缓释系统复合纳米支架材料修复犬下颌骨缺损[J].中国组织工程研究, 2013,17(21):3816-3822.[9] 安世昌,孙健,李亚莉,等.复合壳聚糖纳米微球聚乳酸-羟基乙酸/纳米羟基磷灰石缓释载体系统对蛋白的缓释作用[J].中国组织工程研究与临床康复,2011,15(25):4615-4618.[10] Vishnubalaji R,Yue S,Alfayez M,et al.Bone morphogenetic protein 2(BMP2) induces growth suppression and enhances chemosensitivity of human colon cancer cells.Cancer Cell Int. 2016;16(1):77.[11] Zhou N,Li Q,Lin X,et al.BMP2 induces chondrogenic differentiation, osteogenic differentiation and endochondral ossification in stem cells. Cell Tissue Res.2016;366(1):101-111.[12] Çak?r-Özkan N,E?ri S,Bekar E,et al.The Use of Sequential VEGF- and BMP2-Releasing Biodegradable Scaffolds in Rabbit Mandibular Defects.J Oral Maxillofac Surg. 2017;75(1):221.e1-221.e14. [13] John PS,Jeffery OH.The critical size defect as perimentalmodel for craniomandibulofacia Nonunions.Clini Orthop Rel Res. 1986;20(5): 299-230.[14] Lee JC,Min HJ,Park HJ,et al.Synovial membrane-derived mesenchymal stem cells supported by platelet-rich plasma can repair osteochondral defects in a rabbit model.Arthroscopy. 2013;29(6): 1034-1046.[15] 荣小芳,吴琳,杨晓东,等.兔下颌骨临界骨缺损人工材料植入实验动物模型的建立[J].中国医科大学学报, 2008,37(1):62-64.[16] 臧晓龙,孙健,李亚莉,等.3D生物打印构建聚乳酸羟基乙酸/纳米羟基磷灰石支架骨形态发生蛋白2缓释复合体的实验研究[J].中国组织工程研究, 2016,20(16):2405-2411.[17] The Ministry of Science and Technology of the People’s Republic of China. Guidance suggestion of caring laboratory animals.2006-09-30.[18] 朱明华,杨成民.植入性高分子材料的组织病理学观察[J].北京生物医学工程,1991,10(2):10217.[19] Tan W,Krishnaraj R,Desai TA.Evaluation of nanostructuredcomposite collagen-chitosan matrices for tissue engineering.Tissue Eng. 2001; 7(2):203-210.[20] Rosenberg L.Chemical basis for the histological use ofsafranin O in the study of articular cartilage.J Bone Joint Surg Am.1971;53(1):69-82.[21] Martyna K,Katarzyna W,Ma?gorzata LS,et al.Chitosan and composite microsphere-basedscaffold for bone tissue engineering: evaluation of tricalcium phosphate contentinfluence on physical and biological properties.J Nat Gas Sci Eng.2015;26(3):1-12.[22] Adnan H,Kailash CG,Inn-Kyu K,et al.PLGA/nHA hybrid nanofiber scaffold as a nanocargo carrier of insulin for accelerating bone tissue regeneration.Nanoscale Res Lett.2014;9(1):314-316.[23] Yang XZ,Li AS. Electrospun Hydroxyapatite/BMP-2 Grafted PLLA Nanofibers for Guided Bone Rebuilding Scaffold.Adv Mater Res.2015; 1095:322-325.[24] 许尧祥,李亚莉,陈立强,等.壳聚糖微球/纳米羟基磷灰石/聚乳酸-羟基乙酸复合支架制备及其蛋白缓释效果:与单纯纳米羟基磷灰石/聚乳酸-羟基乙酸支架、壳聚糖微球的比较[J].中国组织工程研究与临床康复, 2010, 14(3):452-456.[25] 刘峰,孙健,李亚莉,等.骨形态发生蛋白2壳聚糖纳米缓释载体的制备及性能测定[J].上海口腔医学, 2015,24(2):147-150.[26] S Dhivya S,Ajita J,Selvamurugan N.Metallic Nanomaterials for Bone Tissue Engineering.J Biomed Nanotechnol. 2015;11(10):1675-1700.[27] Labate GF,Baino F,Brovarone CV,et al.A biomimetic approach to 3D scaffold architectural characterization for veterinarian bone tissue engineering[C]// E-MRS Spring Meeting.2015.[28] Naga SM,El-Maghraby HF,Sayed M,et al.Highly porous scaffolds made of nanosized hydroxyapatite powder synthesized from eggshells.J Ceram Sci Technol. 2015;6(3):237-243.[29] Repanas A,Mavrilas D,Glasmacher B,et al.Blend electrospinning of biodegradable chitosan/ Polycaprolactone fibers as a process to create scaffolds for cardiovascular tissue engineering[C]// Esb.2015.[30] Huang J,Fu H,Wang ZY,et al.BMSCs-laden gelatin/sodium alginate/carboxymethylchitosan hydrogel for 3D bioprinting.Rsc Adv. 016;6(110):108423-108430.[31] Maji K,Dasgupta S,Pramanik K,et al.Preparation and Evaluation of Gelatin- Chitosan-Nanobioglass 3D Porous Scaffold for Bone Tissue Engineering.Int J Biomater. 2016;(4):183-187.[32] Dan Y,Liu O,Liu Y,et al.Development of Novel Biocomposite Scaffold of Chitosan-Gelatin/Nanohydroxyapatite for Potential Bone Tissue Engineering Applications.Nanoscale Res Lett. 2016;11(1):487.[33] Rahman MS,Akhtar N,Jami HMl,et al.TGF-b/BMP signaling and other molecular events:regulation of osteoblastogenesis and bone formation. Bone Res.2015;3(1):11-30.[34] Drosse I,Volkmer E,Capanna R,et al.Tissue engineering for bone defect healing: An update on a multi-component approach.Injury. 2008;39(2):S9-S20. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Fan Jiabing, Zhang Junmei. Morphological measurement and analysis of the mandible in adult females with different vertical skeletal types [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1177-1183. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||