Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (10): 1625-1633.doi: 10.3969/j.issn.2095-4344.0726
Previous Articles Next Articles
Polyethylene glycol: an expert of cellular camouflage confusing the immune system
- 1The Third Affiliated Hospital, Guangzhou Medical University, Guangzhou 510150, Guangdong Province, China; 2Shaanxi Provincial Eye Clinical Research Center, Shaanxi Provincial Key Laboratory of Ophthalmology, Shaanxi Institute of Ophthalmology, The First Hospital of Xi’an, Xi’an 710002, Shaanxi Province, China
-
Received:2018-01-21Online:2018-04-08Published:2018-04-08 -
Contact:Wang Shuang-yong, M.D., Attending physician, the Third Affiliated Hospital, Guangzhou Medical University, Guangzhou 510150, Guangdong Province, China; Shaanxi Provincial Eye Clinical Research Center, Shaanxi Provincial Key Laboratory of Ophthalmology, Shaanxi Institute of Ophthalmology, The First Hospital of Xi’an, Xi’an 710002, Shaanxi Province, China -
About author:Tian Ying, Master, Attending inspector, the Third Affiliated Hospital, Guangzhou Medical University, Guangzhou 510150, Guangdong Province, China -
Supported by:the Natural Science Foundation of Shaanxi Province, No. 2016JM8017
CLC Number:
Cite this article
Tian Ying, Wu Jie, Wang Shuang-yong. Polyethylene glycol: an expert of cellular camouflage confusing the immune system[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(10): 1625-1633.
share this article
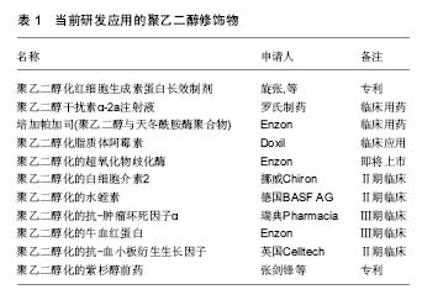
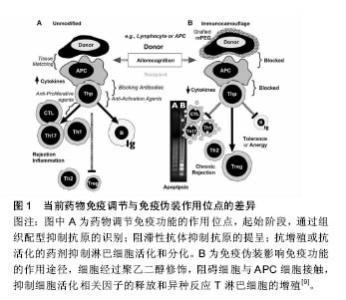
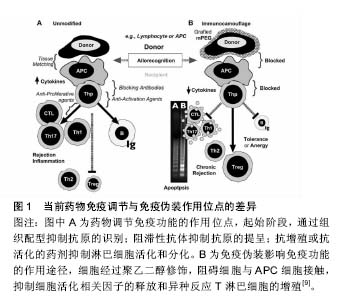
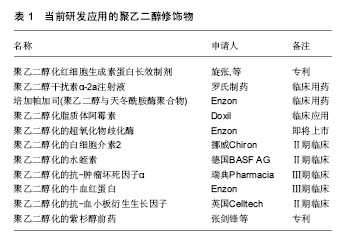
2.1 聚乙二醇修饰位点选择 不同的聚乙二醇衍生物能够选择性修饰蛋白质氨基、巯基、羧基等位点。由于蛋白质分子表面的氨基有较高的亲核反应活性,且一般不处于活性中心部位,因而是蛋白质化学修饰中最常被修饰的基团。由于蛋白质只有一个末端氨基,对氨基的定点修饰通常发生在N末端氨基。但是,一些蛋白质N端是其活性区域,聚乙二醇修饰会导致活性下降,甚至丧失,因此并不适合N端氨基修饰。氨基修饰最常用的修饰剂包括琥珀酰亚胺琥珀酸酯聚乙二醇、琥珀酰亚胺丙酸酯聚乙二醇、琥珀酰亚胺二聚乙二醇、醛类聚乙二醇及琥珀酰亚胺甲酸酯聚乙二醇。巯基在蛋白质组成中通常含量不高,但位置确定,因而聚乙二醇衍生物可针对那些对活性影响不大、呈游离状态的巯基进行定量、定点修饰。然而,活化半胱氨酸的巯基大多位于蛋白质的活性中心部位,因此,聚乙二醇修饰可能造成蛋白活性的丧失。最常用的巯基修饰剂为马来酰亚胺聚乙二醇。双烷基化聚乙二醇能够烷基化二硫键的巯基,分别形成三碳桥。聚乙二醇通过三碳桥与蛋白质共价偶联,三碳桥能够替代二硫键,起到稳定修饰产物空间结构的作用,保持聚乙二醇修饰后蛋白质的生物活性稳定。对于半胱氨酸含量较少的天然蛋白,可通过基因工程技术在合适位置定向引入巯基来实现定点修饰。羧基的修饰位点位于天冬氨酸、谷氨酸和末端羧基。但羧基修饰同时易产生其他的交联反应,故目前使用较少。此外,特殊合成的聚乙二醇衍生物也被广泛应用。由2个线型聚乙二醇连接而成的伞状聚乙二醇用于蛋白质修饰,保护蛋白质抵抗蛋白水解酶。带有氨基酸手臂的聚乙二醇是指在聚乙二醇和活性基团之间存在一个氨基酸或多肽,不同的氨基酸或多肽起不同的作用,如:氚标记的甘氨酸(Gly)可用于药代动力学研究,色氨酸可用于荧光标记等。Inui等[4]合成聚乙二醇-脂质和聚乙烯醇烷基(PVA-烷基)聚合物,将其加入细胞混悬液,该聚合物依靠疏水作用锚靠于细胞表面,达到修饰作用,而不影响细胞功能。 2.2 聚乙二醇抗原伪装作用 免疫伪装是指细胞表面生物物理及生理学的遮盖,以防止与其他细胞、大分子或病毒的相互作用[5-6](表1)。聚乙二醇免疫伪装作用机制在于(图1):第一,聚乙二醇共价结合于修饰物表面,而每个环氧乙烷分子可结合3分子水,从而在修饰物表面形成水分子层外壳,遮蔽抗原位点,阻碍免疫蛋白受体与配体的结合;第二,聚乙二醇形成的水分子外壳呈电中性,遮盖修饰物表面电荷,阻碍大分子的接近和结 合[7]。细胞膜表面的聚乙二醇修饰,可抑制免疫细胞或分子的黏附、识别及参与异种识别刺激通路,从而可能影响免疫反应的诸多方面[8]。 2.2.1 聚乙二醇抗原伪装修饰血细胞 Jeongh和Byun[10]首次报道了将聚乙二醇用于修饰红细胞,以遮蔽红细胞表面血型抗原。聚乙二醇及其衍生物可与红细胞表面的抗原决定簇共价结合,遮蔽血型抗原,降低血型抗原的免疫原性和保护其免受免疫攻击[11-12]。利用甲氧基聚乙二醇包被在人、鼠、羊红细胞表面,体内外实验观察这些细胞的结构、功能、抗原性和存活率发现:甲氧基聚乙二醇修饰可阻断抗-A、抗-B对A型或B型的人红细胞A抗原或B抗原的血清学凝集反应;甲氧基聚乙二醇修饰的羊红细胞可阻断人外周血单核细胞的吞噬,而红细胞形态、结构和功能正常;甲氧基聚乙二醇修饰的羊红细胞输注到鼠体内可存活72 h[13]。Western检测RH+甲氧基聚乙二醇-红细胞发现,红细胞表面D抗原缺失,贮存30 d后,甲氧基聚乙二醇-红细胞依然保持抗原沉默[14]。Murad与Bradley等[15-16]发现甲氧基聚乙二醇修饰的红细胞,可阻断针对红细胞抗原的抗原-抗体反应及经典和旁路补体活化途径,而该红细胞的变形性和血红蛋白携氧能力均正常,也不影响红细胞膜蛋白的功能,建议用甲氧基聚乙二醇包被红细胞制备通用型红细胞,用于需反复输血、稀有血型、特殊患者的急需用血。将甲氧基聚乙二醇修饰的红细胞输给一些需要长期输血且对某些次要血型敏感的患者,可减少输血反应。就异种输血而言,甲氧基聚乙二醇猪红细胞的血凝素效价降低,与人血清共育,人免疫球蛋白的结合下降61%,临床交叉配血实验显示猪红细胞与人血清有更高的相容性,猪红细胞的形态、结构、携氧度正常[17]。Gundersen等[3]探索使用琥珀酰亚胺丙酸酯-甲氧基聚乙二醇修饰的牛红细胞作为一种可能的血液替代品可行性。实验结果证实:琥珀酰亚胺丙酸酯-甲氧基聚乙二醇修饰的牛红细胞与人红细胞相比,有相似的氧结合度、氧运输和氧流量等特性,与人血清共育并不引起红细胞凝集增加。将聚乙二醇修饰的人红细胞输注于鼠,可明显减少异种红细胞的免疫原性[15],鼠体内针对异种红细胞抗原的抗体明显减少[18]。 2.2.2 聚乙二醇抗原伪装修饰淋巴细胞 Huang等[19]研究发现使用15 g/L的聚乙二醇-琥珀酰亚胺丙酸酯修饰,可遮盖淋巴细胞表面88%的CD3抗原,输注聚乙二醇-琥珀酰亚胺丙酸酯修饰的T淋巴细胞明显减轻移植物抗宿主病的临床表现及病理程度。而且,聚乙二醇对表面抗原的屏蔽作用并不随培养时间而改变。流式细胞学检测显示,甲氧基聚乙二醇修饰的T细胞及抗原递呈细胞表面黏附分子(CD2,CDlla)、信号分子(CD3,TCR)、共刺激分子(CD28,CD80)含量明显降低。因此,淋巴细胞的聚乙二醇修饰旨在切断其与抗原递呈细胞之间相互识别和黏附,使淋巴细胞处于特异性无应答 状[20-21]。既不影响氧气、二氧化碳、糖、氨基酸、无机盐、维生素等小分子物质交换,又不损伤淋巴细胞的形态、结构及功能,使受者拥有完全而有活力的免疫系 统[14]。混合淋巴细胞反应实验结果显示,甲氧基聚乙二醇修饰遮蔽外周血单个核细胞细胞表面分化抗原,阻止了记忆细胞免疫应答,抑制免疫活化,Treg细胞升高,Th17细胞居于基线水平;而对照组白细胞介素2、肿瘤坏死因子α、干扰素γ等促炎因子明显升高,Treg细胞减少,Th17细胞则明显升高。从而证明,异体淋巴细胞的聚乙二醇修饰可诱导免疫耐受或免疫静止状态[9]。 2.2.3 聚乙二醇抗原伪装修饰细胞治疗的移植细胞 当前,在细胞移植治疗研究中,聚乙二醇修饰也备受关注。聚乙二醇结合于胰岛细胞囊膜面,抑制免疫细胞的激活[6,22-23]。聚乙二醇修饰的胰岛细胞与淋巴细胞共育,淋巴细胞白细胞介素2等因子分泌明显减少,提示聚乙二醇修饰产生有效的免疫伪装作用[24]。移植聚乙二醇修饰的胰岛细胞给糖尿病模型动物,可降低免疫排斥反应,明显延长胰岛细胞的存活时间[25-27],同时,显著减少移植后免疫抑制剂的使用[25,28]。而且,聚乙二醇修饰并不影响胰岛细胞对血糖变化的反应和胰岛素分泌,也不影响细胞生存能力[29]。 2.2.4 聚乙二醇抗原伪装修饰移植器官 众多研究显示,聚乙二醇改进保存的移植器官的移植存活。Collins和Wicomb[30]首次使用相对分子质量为20 000的聚乙二醇(50 g/L)代替UW器官保存液中的羟乙基淀粉,人心脏移植后排斥反应的发生较使用其他保存液明显减少(1年内排斥反应的发生率分别为50%和78%),提示聚乙二醇免疫伪装抑制移植免疫系统的活化。随后的研究证实,聚乙二醇对移植前保存的供体肝脏[31]、肾脏[32]、肺脏[33]、小肠[34]、胰脏具有保护作用及免疫伪装作用[35],改善移植后器官功能的恢复及存活。在器官保存液加入聚乙二醇,可改善移植后器官功能的恢复及存活,其机制包括:①免疫伪装器官抗原,减少移植排斥反应的发生。不像羟乙基淀粉,含15 g/L聚乙二醇化的器官保存液并不影响移植后器官血管网顺利循环,适合腹部器官如肝肾的保存。含相对分子质量20 000的聚乙二醇15-30 g/L的SCOT保存液,可改善移植器官的存活,减少免疫移植排斥的损伤[36];②维持胶体渗透压,防止细胞水肿加重。聚乙二醇分子相对比较稳定,能够较长时间维持器官保存液稳定的胶体渗透压。在UW器官保存液加入3%聚乙二醇,可减少内皮细胞的肿胀,增加移植后功能恢复[37];③稳定细胞膜磷脂,增强对供体细胞组织器官的免疫保护作用[38]。聚乙二醇的抗氧化特性,可有效保存器官细胞活性[39];④聚乙二醇保持器官保存液中ATP含量;⑤诱导细胞死亡由细胞坏死转向凋亡,减少免疫反应。急性坏死细胞可通过Toll-like受体激活局部树突状细胞,触发急性排斥反应,是生理性凋亡引起的细胞死亡,不会引起明显排斥。 2.2.5 聚乙二醇抗原伪装修饰蛋白质及大分子物质 蛋白质及其他大分子类药物通过将活化的聚乙二醇化学偶联到药物达到聚乙二醇修饰的目的,即聚乙二醇化。一般来说,在聚乙二醇修饰过程中,聚乙二醇与蛋白质及大分子的共价结合并不改变蛋白质的二级、三级结构[40]。由于天然或重组的未经任何修饰蛋白质或其他大分子类物质易引起机体的免疫反应,同时在体内的半衰期较短,从而显著减弱了其临床效果,并限制了它们广泛的临床使用。聚乙二醇修饰能够改进蛋白质及大分子物质诸多特性[41-42]:①降低蛋白质及大分子物质的免疫原性,减少体内免疫反应等不良反应的发生。蛋白质类制剂大多来源于异种或异体,具有较强的免疫原性。聚乙二醇修饰免疫伪装蛋白质表面的抗原位点,降低蛋白质的免疫原性,减少蛋白凝集反应的发生;②延长蛋白质制剂体内血浆半衰期。由于聚乙二醇每分子可配位结合6-7水分子,聚乙二醇流体力学体积大于同体积的蛋白分子,聚乙二醇修饰后的蛋白质制剂分子量增加,超出肾脏过滤的分子量范围,从而减少此类制剂的清除率,有效延长药物的半衰期;③增强蛋白质及大分子制剂的抗酶降解能力。聚乙二醇在蛋白质表面起到屏蔽和位阻效应,使得修饰后的蛋白质抵抗胃蛋白酶及胰蛋白酶等蛋白酶降解,提高蛋白质的稳定性,同时延长体内血浆半衰期;④增强蛋白质及大分子制剂对酸碱、热的耐受性。聚乙二醇作为一种修饰剂,在蛋白质及大分子的外围形成了一个保护膜,保护稳定蛋白质及大分子的分子构象次级链,增强了对热、 酸、碱的耐受性,提高其稳定性;⑤提高蛋白质及大分子制剂的的溶解性。聚乙二醇为呈现水溶性及脂溶性的两性分子,其修饰能够改善蛋白质及大分子制剂溶解性等理化性质,从而提高了作用部位的蛋白质制剂血浆浓度,增强其体内的生物利用度;⑥降低蛋白质及大分子制剂体内最大血药浓度,减少血药浓度波动,从而降低此类制剂的毒副作用。 Abuchowsk于1977年首次使用聚乙二醇修饰牛血清蛋白,发现可有效改变其免疫原性[43]。以血红蛋白为基础的血液替代品,具有与天然红细胞类似的S形氧解离曲线。但无基质血红蛋白氧亲和力过高,在循环系统中的存留时间过短。Ajisaka等[44]制备出聚乙二醇和磷酸吡哆醛共修饰的血红蛋白,明显改善了其血浆半衰期和氧亲和力。聚乙二醇化修饰看改善凝血酶样蛋白酶的溶解度、底物结合力及半衰期,提高其生物药理学活性[42]。聚乙二醇修饰的链激酶免疫原性明显降低,蛋白稳定性升高,体内的半衰期延长2-20倍[45]。 大肠杆菌(E .Coli)来源的相对分子质量为136 000的天冬酰胺酶,主要用于急性淋巴性白血病和淋巴肉瘤的治疗。然而,天然天冬酰胺酶的血浆半衰期短,异种蛋白抗原性可引起程度不一的不良反应。Ashihara等[46]用相对分子质量为5 000的甲氧基聚乙二醇修饰天冬酰胺酶,当其表面总92氨基群中的73个被修饰时,来自兔血清样本中抗-天冬酰胺酶抗体对其的结合力完全丧失,而7%的酶活性依然被保留,同时甲氧基聚乙二醇-天冬酰胺酶抵御胰蛋白酶的降解能力增强。用琥珀酸酐及琥珀酰亚胺两步活化的单甲氧基聚乙二醇修饰天冬酰胺酶,酶活性保持在40%以上,天冬酰胺酶的抗原性显著下降,抗热、抗胰蛋白酶水解能力明显提高[47]。应用氰尿酰氯活化分枝状甲氧基聚乙二醇衍生物,酶分子上的51个氨基被修饰时,天冬酰胺酶的抗原性完全消除,而酶活性仍保持了原来的30%以上。新的梳状聚乙二醇衍生物分子PM13(相对分子质量约13 000)和PM100(相对分子质量约100 000)也被用于天冬酰胺酶的修饰。与线状聚乙二醇和分枝状聚乙二醇相比,PM修饰的天冬酰胺酶在较低修饰程度下的免疫原性明显降低,同时保留了较高的酶活性。将PM13-天冬酰胺酶注射给实验鼠,抗-天冬酰胺酶抗体的产生被完全抑制。鼠单次静脉注射PM100-天冬酰胺酶和未经修饰的天冬酰胺酶,两者的血浆半衰期分别是50 h和1.5 h,单次给药11 d后仍能检测到血清中PM100-天冬酰胺酶的活 性[48]。对于某些对E .Coli和欧文氏胡萝卜软腐菌来源的天冬酰胺酶有临床过敏反应的患者来说,聚乙二醇修饰的天冬酰胺酶是一种安全而有效的天冬酰胺酶替代物。临床报道显示,与天然天冬酰胺酶相比,聚乙二醇修饰的天冬酰胺酶拥有相同的安全性和有效性,同时又具有较低的免疫原性和较长的半衰期,可以替代天然天冬酰胺酶来作为急性淋巴细胞白血病的一线用药[49-50]。国内的多中心研究报道同样提示,聚乙二醇修饰的天冬酰胺酶可维持2周左右的半衰期,单次剂量应用相当于天然天冬酰胺酶多次应用,且具有较低的免疫原性[51]。 合成的腺苷脱氨酶作为替代疗法,治疗由于腺苷脱氨酶的缺乏导致的严重综合性免疫缺陷症。Hershfield[52]利用聚乙二醇修饰制备一种长循环形式的腺苷脱氨酶,用于临床治疗腺苷脱氨酶缺乏的患者。在近24年的长期临床观察中,聚乙二醇-腺苷脱氨酶的半衰期显著延长,用聚乙二醇-腺苷脱氨酶治疗几乎可完全纠正代谢异常,并可恢复不同程度的免疫功能。在大多数情况下,虽然免疫功能并未达到正常水平,但足以防止机会感染和危及生命的其他感染。同时,聚乙二 醇-腺苷脱氨酶也可作为细胞基因治疗腺苷脱氨酶缺乏症的重要辅助物。聚乙二醇-腺苷脱氨酶临床使用中,患者耐受性良好,未见有过敏反应发生。 聚乙二醇修饰的干扰素已被应用于临床治疗。吴影新等[53]采用相对分子质量为20 000的单甲氧基聚乙二醇丙醛修饰重组人干扰素α-2b,在 pH=6.5、20 mmol/L的磷酸氢二钠-柠檬酸缓冲溶液中,聚乙二醇与干扰素α-2b的摩尔比为8∶1,4 ℃时反应20 h,单修饰聚乙二醇-干扰素α-2b的转化率达到55%。分离纯化单修饰产品纯度达到97%,体外活性保留达到未修饰干扰素α-2b的13.4%,其在SD大鼠体内的循环半衰期得到了较大延长,且具有较好的水溶液稳定性。使用相对分子质量为40 000的分枝状聚乙二醇分子来修饰干扰素α-2a,修饰后聚乙二醇-干扰素α-2a治疗慢性乙型肝炎的功效明显增强。Foster等[54]对比聚乙二醇-干扰素α-2a和干扰素α-2a在531例慢性乙肝患者临床疗效,结果显示:每周一次皮下注射180 mg聚乙二醇-干扰素α-2a比每周3次(600万U/次)皮下注射干扰素α-2a更有效。Heathcote 等[55]对271例慢性乙肝病毒相关纤维硬化患者进行的临床治疗,从患者慢性乙肝病毒mRNA和丙氨酸氨基转移酶检测结果分析来看,每周一次皮下注射180 mg聚乙二醇-干扰素α-2a的疗效要显著优于每周3次(300万U/次)皮下注射干扰素α-2a的疗效。当下,关于聚乙二醇-干扰素β-1a治疗多发性硬化的临床研究也正在进行中[56]。 2.2.6 聚乙二醇抗原伪装修饰基因治疗的载体 尽管腺病毒载体具有高转导效率,并被广泛应用于基因治疗研究的动物模型中,但临床使用非常有限。一个主要原因是系统应用腺病毒载体导致先天和抗原特异性免疫的激活,引起T和B细胞特异性免疫应答。此外,近80%的人本身拥有抗-腺病毒载体抗体。Eto等[57]尝试用丙酸琥珀酰亚胺酯活化的甲氧基聚乙二醇修饰腺病毒载体,并评估了聚乙二醇化腺病毒载体逃避中和抗体能力。结果表明,与未修饰的腺病毒载体相比,聚乙二醇-腺病毒载体降低与抗腺病毒抗体黏附结合,增强腺病毒载体抵抗其抗体攻击的能力。考虑到许多人携带抗腺病毒载体抗中和抗体,而且慢性疾病治疗需要反复多次腺病毒载体载体应用,聚乙二醇修饰可作为改良载体或开发新载体的重要策略。使用活化的单甲氧基聚乙二醇修饰腺病毒载体衣壳蛋白,静脉注射此种聚乙二醇修饰腺病毒载体,其肝内表达明显增加,提示聚乙二醇聚合物化学修饰并不影响体内载体转导效率。而且,聚乙二醇化腺病毒载体降低了针对天然病毒抗原的1型辅助性T细胞亚群(Th1细胞)和细胞毒性T淋巴细胞的活化;同时针对天然病毒抗原的中和抗体也减少[58]。在卵巢癌的腹膜内模型中,与未修饰的腺病毒载体相比,聚乙二醇-腺病毒载体-FGF2在肿瘤组织中基因表达明显增加。Choi等[59]使用荧光标记的腺病毒载体研究发现,聚乙二醇修饰的腺病毒载体靶基因转染率增加,靶基因表达升高,荧光维持时间延长,腺病毒载体的免疫原性降低。聚乙二醇修饰减少腺病毒载体的聚合物定位到非目标组织,体内输注引起的Th1和Th2T淋巴细胞免疫应答显著降 低[60-61],这进一步肯定了聚乙二醇修饰对病毒抗原的免疫伪装效应。Gao等[62]用不同聚乙二醇修饰比例制备聚乙二醇-腺病毒载体,发现聚乙二醇-腺病毒载体的血浆半衰期长于未修饰的腺病毒载体,聚乙二醇-腺病毒载体体内表达随修饰比例的增加而增高。将聚乙二醇-腺病毒载体-肿瘤坏死因子α(携带编码肿瘤坏死因子基因序列)注射给携带肿瘤的小鼠,聚乙二醇-腺病毒载体-肿瘤坏死因子α表现出较强的肿瘤抑制活性,而且有较少的肝毒性副作用。因此,聚乙二醇-腺病毒载体对于实现肿瘤靶向具有很大的潜力。 此外,聚乙二醇修饰在组织工程材料研究方面也备受关注。聚乙二醇用以增加组织工程植入物的生物相容性,减少免疫排斥反应的发生。聚乙二醇修饰可改善纳米材料纳米微粒载体的生物利用度。利用聚乙二醇- Herceptin®结合形成的纳米载药微粒,降低吞噬细胞的摄取及Herceptin®本身的免疫原性[63]。与壳聚糖包裹的脂质纳米载体相比,聚乙二醇修饰的脂质纳米载体呈现“惰性”,并未引起免疫细胞的活化[64]。 2.3 影响聚乙二醇修饰的因素 2.3.1 聚乙二醇相对分子质量及浓度 聚乙二醇修饰浓度越大,修饰效率和修饰产物多态性越高。而增加聚乙二醇相对分子质量可延长被修饰物的循环半衰期。Bradley等[16]利用微粒电泳及液体双相聚合物分离技术观察发现,聚乙二醇的浓度、种类和相对分子质量均可影响红细胞聚乙二醇修饰。当其相对分子质量为 20 000、浓度达2 mmol/L时,可产生免疫伪装作用,维持聚乙二醇修饰红细胞在体内的正常存活,而不影响细胞活性。而相对分子质量为5 000的聚乙二醇,在同样浓度条件下并未产生免疫伪装作用。相对分子质量为 20 000的聚乙二醇,可伪装遮蔽细胞表面纵向距离为 13.8 nm的抗原簇,而相对分子质量5 000者只能遮蔽细胞表面纵向距离为6 nm的抗原簇。小分子量的聚乙二醇并不适合移植物免疫伪装修饰,相比较于相对分子质量8 000者,相对分子质量20 000的聚乙二醇可产生免疫伪装效果,明显提高异体小肠移植后的存活[34]。Chen等[5]认为聚合物长度是免疫屏蔽细胞最关键因素,他们通过实验证明相对分子质量为20 000的甲氧基聚乙二醇-苯并三唑碳酸酯包被细胞最有效。在1.5 mmol/L相同浓度下,相对分子质量为20 000的聚乙二醇修饰胰岛细胞较相对分子质量8 000更能保护异种移植后胰岛细胞的存活及其功能的恢复[65]。聚乙二醇修饰后,红细胞表面补体、抗体及其他免疫复合体的结合明显减少,并呈现剂量依赖模式[66]。相对分子质量为5 000的聚乙二醇衍生物修饰的胰岛细胞,当质量浓度为22 g/L时与淋巴细胞共育培养,白细胞介素2分泌下降70%;当质量浓度变为12.5 g/L时,白细胞介素2的分泌可降低约50%;当质量浓度小于7 g/L时,无免疫伪装作用[67]。此外,药物或基因载体的单层脂质体及乳胶微粒的聚乙二醇修饰同样呈现剂量和分子长度依赖性[68]。某些情况下,分子量大小不同产生不同的聚乙二醇修饰效果,小分子量聚乙二醇修饰促进细胞-蛋白分子间的吸附,大分子量聚乙二醇修饰诱导细胞-蛋白间的排斥。 2.3.2 修饰反应的时间、温度及pH值 在不同的pH值条件下,聚乙二醇衍生物的反应活性和修饰位点存在明显差异。随着pH的变化,聚乙二醇与修饰物形成的共价结合键可能不同。比如,亲电性的琥珀酰亚胺基碳酸 酯-聚乙二醇和苯并三唑碳酸酯-聚乙二醇,在pH=8.0时与赖氨酸残基反应形成氨基甲酸酯键;而处于酸性环境时,可与组氨酸残基反应,形成咪唑氨基甲酸酯键[69]。从修饰效率考虑,大多数亲电性聚乙二醇衍生物采用碱性条件进行修饰反应。Murad等[15]研究显示,聚乙二醇修饰作用的实际化学条件可变,在人红细胞甲氧基聚乙二醇修饰过程中,使用pH=9.2(4 ℃、30 min)或pH=8.0 (25 ℃、30 min)的抗原伪装效果相当。Richter等[70]指出,聚乙二醇修饰的最佳pH=8.7,温度14 ℃,反应时间为30 min。不同的聚乙二醇衍生物修饰不同的蛋白集团所需要的最佳反应时间并不完全不相同。当前,研究报道中,常用的聚乙二醇修饰反应化学条件为:pH=8.0,温度25-37 ℃,反应时间30-60 min。此外,某些种类的聚乙二醇修饰反应需要加入其他辅助试剂,比如,醛类聚乙二醇需加入硼氢氰化钠,操作时也应考虑所加试剂的反应条件。 此外,修饰细胞或组织抗原密度及抗原的拓扑形态(抗原距细胞膜表面的高度)等也会影响聚乙二醇抗原伪装的效果[16]。最近研究报道,聚乙二醇修饰干扰素呈现不同的体内药代学特点,雌性小鼠体内药物清除率高于雄性小鼠[71]。那么,性别因素是否影响修饰物分子中聚乙二醇在体内的代谢,仍需进一步的探究。 "
| [1]Roberts MJ,Bentley MD,Harris JM.Chemistry for peptide and protein PEGylation.Adv Drug Deliv Rev. 2002;54(4):459-476.[2]Fruijtier-Polloth C.Safety assessment on polyethylene glycols (PEGs) and their derivatives as used in cosmetic products. Toxicology.2005;214(1-2):1-38.[3]Gundersen SI,Palmer AF.Conjugation of methoxypolyethylene glycol to the surface of bovine red blood cells.Biotechnol Bioeng. 2007;96(6):1199-1210.[4]Inui O,Teramura Y,Iwata H.Retention dynamics of amphiphilic polymers PEG-lipids and PVA-Alkyl on the cell surface.ACS Appl Mater Interfaces.2010;2(5):1514-1520.[5]Chen AM,Scott MD.Comparative analysis of polymer and linker chemistries on the efficacy of immunocamouflage of murine leukocytes.Artif Cells Blood Substit Immobil Biotechnol. 2006;34(3):305-322.[6]Teramura Y,Asif S,Ekdahl KN,et al.Cell Surface Engineering for Regulation of Immune Reactions in Cell Therapy.Adv Exp Med Biol.2015;865:189-209.[7]Sutton TC,Scott MD.The effect of grafted methoxypoly (ethylene glycol) chain length on the inhibition of respiratory syncytial virus(RSV)infection and proliferation.Biomaterials. 2010;31(14):4223-4230.[8]Scott MD,Chen AM.Beyond the red cell: pegylation of other blood cells and tissues. Transfus Clin Biol.2004;11(1):40-46.[9]Wang D,Toyofuku WM,Chen AM,et al.Induction of immunotolerance via mPEG grafting to allogeneic leukocytes. Biomaterials.2011;32(35):9494-9503.[10]Jeong ST,Byun SM.Decreased agglutinability of methoxy-polyethylene glycol attached red blood cells: significance as a blood substitute.Artif Cells Blood Substit Immobil Biotechnol.1996;24(5):503-511.[11]Le Y, Toyofuku WM, Scott MD. Immunogenicity of murine mPEG-red blood cells and the risk of anti-PEG antibodies in human blood donors. Exp Hematol 2017;47:36-47.[12]Li L,Noumsi GT,Kwok YY,et al.Inhibition of phagocytic recognition of anti-D opsonized Rh D+ RBC by polymer-mediated immunocamouflage.Am J Hematol. 2015;90(12):1165-1170.[13]Armstrong JK,Meiselman HJ,Fisher TC.Covalent binding of poly(ethylene glycol) (PEG) to the surface of red blood cells inhibits aggregation and reduces low shear blood viscosity. Am J Hematol. 1997;56(1):26-28.[14]Tan Y,Qiu Y,Xu H,et al.Decreased immunorejection in unmatched blood transfusions by attachment of methoxypolyethylene glycol on human red blood cells and the effect on D antigen.Transfusion. 2006;46(12): 2122-2127.[15]Murad KL,Gosselin EJ,Eaton JW,et al.Stealth cells: prevention of major histocompatibility complex class II-mediated T-cell activation by cell surface modification. Blood.1999;94(6):2135-2141.[16]Bradley AJ,Murad KL,Regan KL,et al.Biophysical consequences of linker chemistry and polymer size on stealth erythrocytes: size does matter.Biochim Biophys Acta.2002; 1561(2):147-158.[17]Doucet J,Gao ZH,MacLaren LA,et al.Modification of xenoantigens on porcine erythrocytes for xenotransfusion. Surgery.2004;135(2):178-186.[18]Scott MD,Bradley AJ,Murad KL.Camouflaged blood cells: low-technology bioengineering for transfusion medicine? Transfus Med Rev.2000;14(1):53-63.[19]Huang Y,Feng S,Tang R,et al.Efficacy of pretreatment of allografts with methoxypolyethylene glycol-succinimidyl-propionic acid ester in combination with an anti-OX40L monoclonal antibody in relieving graft-versus-host disease in mice.Int J Hematol.2010;92(4):609-616.[20]Chen AM,Scott MD.Immunocamouflage: prevention of transfusion-induced graft-versus-host disease via polymer grafting of donor cells.J Biomed Mater Res A. 2003;67(2): 626-636.[21]Kyluik-Price DL,Scott MD.Effects of methoxypoly (Ethylene glycol) mediated immunocamouflage on leukocyte surface marker detection, cell conjugation, activation and alloproliferation. Biomaterials. 2016;74:167-177.[22]Giraldo JA,Molano RD,Rengifo HR,et al.The impact of cell surface PEGylation and short-course immunotherapy on islet graft survival in an allogeneic murine model.Acta Biomater. 2017;49:272-283.[23]Manzoli V,Villa C,Bayer AL,et al.Immunoisolation of murine islet allografts in vascularized sites through conformal coating with polyethylene glycol.Am J Transplant.2017.doi: 10.1111/ajt.14547. [Epub ahead of print][24]Aghajani-Lazarjani H,Vasheghani-Farahani E,Shojaosadati SA,et al.The effect of two different polyethylene glycol (PEG) derivatives on the immunological response of PEG grafted pancreatic islets. J Artif Organs.2010;13(4):218-224.[25]Haque MR,Kim J,Park H,et al.Xenotransplantation of layer-by-layer encapsulated non-human primate islets with a specified immunosuppressive drug protocol.J Control Release.2017;258:10-21.[26]Jeong JH,Yook S,Hwang JW,et al.Synergistic effect of surface modification with poly(ethylene glycol) and immunosuppressants on repetitive pancreatic islet transplantation into antecedently sensitized rat.Transplant Proc.2013;45(2):585-590.[27]Haque MR,Jeong JH,Byun Y.Combination strategy of multi-layered surface camouflage using hyperbranched polyethylene glycol and immunosuppressive drugs for the prevention of immune reactions against transplanted porcine islets.Biomaterials.2016;84:144-156.[28]Lee DY,Lee S,Nam JH,et al.Minimization of immunosuppressive therapy after islet transplantation: combined action of heme oxygenase-1 and PEGylation to islet.Am J Transplant.2006;6(8):1820-1828.[29]Hashemi J,Hashemi-Najafabadi S,Vasheghani-Farahani E.Synergistic effect of PEGylation and pentoxifylline addition on immunoprotection of pancreatic islets.J Biomater Sci Polym Ed. 2017;28(1):33-49.[30]Collins GM,Wicomb WN,Levin BS,et al.Heart preservation solution containing polyethyleneglycol: an immunosuppressive effect? Lancet.1991;338(8771): 890-891.[31]Ben AH,El RZ,Steghens JP,et al.Effective pig liver preservation with an extracellular-like UW solution containing the oncotic agent polyethylene glycol: a preliminary study. Transplant Proc. 2002;34(3):762-763.[32]Faure JP,Petit I,Zhang K,et al.Protective roles of polyethylene glycol and trimetazidine against cold ischemia and reperfusion injuries of pig kidney graft.Am J Transplant.2004;4(4):495-504.[33]Jayle C,Corbi P,Eugene M,et al.Beneficial effect of polyethylene glycol in lung preservation: early evaluation by proton nuclear magnetic resonance spectroscopy.Ann Thorac Surg.2003;76(3):896-902.[34]Itasaka H,Burns W,Wicomb WN,et al.Modification of rejection by polyethylene glycol in small bowel transplantation. Transplantation.1994;57(5):645-648.[35]Weber LM,Cheung CY,Anseth KS.Multifunctional pancreatic islet encapsulation barriers achieved via multilayer PEG hydrogels.Cell Transplant.2008;16(10):1049-1057.[36]Giraud S,Bon D,Neuzillet Y,et al.Concentration and chain length of polyethylene glycol in islet isolation solution: evaluation in a pancreatic islet transplantation model.Cell Transplant. 2012;21(9):2079-2088.[37]Yandza T,Tauc M,Canioni D,et al.Effect of polyethylene glycol in pig intestinal allotransplantation without immunosuppression. J Surg Res.2012;176(2):621-628.[38]Hauet T,Eugene M.A new approach in organ preservation: potential role of new polymers.Kidney Int. 2008;74(8):998-1003.[39]Dutheil D,Rioja-Pastor I,Tallineau C,et al.Protective effect of PEG 35,000 Da on renal cells: paradoxical activation of JNK signaling pathway during cold storage.Am J Transplant. 2006; 6(7):1529-1540.[40]冯翠,王祺,张纯,等.PEG定点修饰重组人睫状神经营养因子及其生物活性评价[J].中国生物工程杂志, 2015,35(5):15-21.[41]Basu A,Yang K,Wang M,et al.Structure-function engineering of interferon-beta-1b for improving stability, solubility, potency, immunogenicity, and pharmacokinetic properties by site-selective mono-PEGylation.Bioconjug Chem. 2006; 17(3):618-630.[42]Da-Silva-Freitas D,Boldrini-Franca J,Arantes EC.PEGylation: a successful approach to improve the biopharmaceutical potential of snake venom thrombin-like serine protease. Protein Pept Lett. 2015;22(12):1133-1139.[43]Abuchowski A,van Es T,Palczuk NC,et al.Alteration of immunological properties of bovine serum albumin by covalent attachment of polyethylene glycol.J Biol Chem. 1977;252(11):3578-3581.[44]Ajisaka K,Iwashita Y.Modification of human hemoglobin with polyethylene glycol : a new candidate for blood substitute.Biochem Biophys Res Commun. 1980;97(3): 1076-1081.[45]Sawhney P,Kumar S,Maheshwari N,et al.Site-Specific Thiol-mediated PEGylation of Streptokinase Leads to Improved Properties with Clinical Potential.Curr Pharm Des. 2016;22(38):5868-5878.[46]Ashihara Y,Kono T,Yamazaki S,et al.Modification of E. coli L-asparaginase with polyethylene glycol: disappearance of binding ability to anti-asparaginase serum.Biochem Biophys Res Commun. 1978;83(2):385-391.[47]周笑艳,刘景晶.聚乙二醇对大肠杆菌L-天冬酰胺酶的化学修饰[J].中国药科大学学报,2000,31(3):230.[48]Kodera Y,Sekine T,Yasukohchi T,et al.Stabilization of L-asparaginase modified with comb-shaped poly(ethylene glycol) derivatives, in vivo and in vitro.Bioconjug Chem.1994; 5(4):283-286.[49]Ettinger LJ,Kurtzberg J,Voǔte PA,et al.An open-label,multicenter study of polyethylene glycol-L-asparaginase for the treatment of acute lymphoblastic leukemia.Cancer-Am Cancer Soc. 2015; 75(5):1176-1181.[50]Ettinger LJ,Kurtzberg J,Voǔte PA,et al.An open-label, multicenter study of polyethylene glycol-L-asparaginase for the treatment of acute lymphoblastic leukemia.Cancer-Am Cancer Soc. 2015;75(5):1176-1181.[51]Wen Z,Ren H,Ke X,et al.PEG-asparaginase in BFM-90 regimen improves outcomes in adults with newly diagnosed lymphoblastic lymphoma.Chin J Cancer Res.2017;29(1):66-74.[52]Hershfield MS. PEG-ADA: an alternative to haploidentical bone marrow transplantation and an adjunct to gene therapy for adenosine deaminase deficiency.Hum Mutat.1995;5(2):107-112.[53]吴影新,翟艳琴,雷建都,等.干扰素α-2b的聚乙二醇修饰[J].生物工程学报,2008,24(9):1658-1663.[54]Foster GR,Serfaty LD.Triple combination treatment for chronic hepatitis C with protease inhibitors, pegylated interferon and ribavirin: 'lead-in or no lead-in'?Liver Int. 2012;32 Suppl 1:61-63.[55]Heathcote EJ,Shiffman ML,Cooksley WG,et al.Peginterferon alfa-2a in patients with chronic hepatitis C and cirrhosis.N Engl J Med.2000;343(23):1673-1680.[56]Khan UT,Tanasescu R,Constantinescu CS.PEGylated IFNβ-1a in the treatment of multiple sclerosis. Expert Opin Biol Ther.2015;15(7):1077.[57]Eto Y,Gao JQ,Sekiguchi F,et al.Neutralizing antibody evasion ability of adenovirus vector induced by the bioconjugation of methoxypolyethylene glycol succinimidyl propionate (MPEG-SPA).Biol Pharm Bull. 2004;27(6):936-938.[58]Croyle MA,Chirmule N,Zhang Y,et al.PEGylation of E1-deleted adenovirus vectors allows significant gene expression on readministration to liver.Hum Gene Ther.2002; 13(15):1887-1900.[59]Choi JW,Dayananda K,Jung SJ,et al.Enhanced anti-tumor efficacy and safety profile of tumor microenvironment-responsive oncolytic adenovirus nanocomplex by systemic administration. Acta Biomater.2015;28:86-98.[60]Lanciotti J,Song A,Doukas J,et al.Targeting adenoviral vectors using heterofunctional polyethylene glycol FGF2 conjugates.Mol Ther.2003;8(1):99-107.[61]Singarapu K,Pal I,Ramsey JD.Polyethylene glycol-grafted polyethylenimine used to enhance adenovirus gene delivery.J Biomed Mater Res A.2013;101(7):1857-1864.[62]Gao JQ,Eto Y,Yoshioka Y,et al.Effective tumor targeted gene transfer using PEGylated adenovirus vector via systemic administration.J Control Release.2007;122(1):102-110.[63]Badkas A,Frank E,Zhou Z,et al.Modulation of in vitro phagocytic uptake and immunogenicity potential of modified Herceptin((R))-conjugated PLGA-PEG nanoparticles for drug delivery.Colloids Surf B Biointerfaces.2017;162:271-278.[64]Farace C,Sanchez-Moreno P,Orecchioni M,et al.Immune cell impact of three differently coated lipid nanocapsules: pluronic, chitosan and polyethylene glycol.Sci Rep.2016;6:18423.[65]Neuzillet Y,Giraud S,Lagorce L,et al.Effects of the molecular weight of peg molecules (8, 20 and 35 KDA) on cell function and allograft survival prolongation in pancreatic islets transplantation. Transplant Proc.2006;38(7):2354-2355[66]Bradley AJ, Scott MD. Immune complex binding by immunocamouflaged [poly(ethylene glycol)-grafted] erythrocytes. Am J Hematol 2007;82(11):970-975.[67]Lazarjani HA,Vasheghani-Farahani E,Barani L,et al.Effect of polymer concentration on camouflaging of pancreatic islets with mPEG-succinimidyl carbonate.Artif Cells Blood Substit Immobil Biotechnol. 2010;38(5):250-258.[68]Le Y,Li L,Wang D,et al.Immunocamouflage of latex surfaces by grafted methoxypoly(ethylene glycol) (mPEG): proteomic analysis of plasma protein adsorption.Sci China Life Sci.2012; 55(3):191-201.[69]Miron T, Wilchek M. A simplified method for the preparation of succinimidyl carbonate polyethylene glycol for coupling to proteins.Bioconjug Chem.1993;4(6):568-569.[70]Richter AW,Akerblom E.Polyethylene glycol reactive antibodies in man: titer distribution in allergic patients treated with monomethoxy polyethylene glycol modified allergens or placebo, and in healthy blood donors.Int Arch Allergy Appl Immunol.1984;74(1):36-39.[71]Landersdorfer CB,Caliph SM,Shackleford DM,et al. PEGylated interferon displays differences in plasma clearance and bioavailability between male and female mice and between female immunocompetent C57Bl/6J and athymic nude mice.J Pharm Sci.2015;104(5):1848-1855.[72]Zhang P,Sun F,Liu S,et al.Anti-PEG antibodies in the clinic: Current issues and beyond PEGylation. J Control Release. 2016;244(Pt B):184-193.[73]Yang Q,Lai SK.Anti-PEG immunity: emergence, characteristics, and unaddressed questions. Wiley Interdiscip Rev Nanomed Nanobiotechnol.2015;7(5):655-677.[74]Ando H,Abu LA,Kawanishi M,et al.Reactivity of IgM antibodies elicited by PEGylated liposomes or PEGylated lipoplexes against auto and foreign antigens.J Control Release.2017;270:114-119.[75]Henry CE,Wang YY,Yang Q,et al.Anti-PEG antibodies alter the mobility and biodistribution of densely PEGylated nanoparticles in mucus.Acta Biomater.2016;43:61-70.[76]Ganson NJ,Kelly SJ,Scarlett E,et al.Control of hyperuricemia in subjects with refractory gout, and induction of antibody against poly(ethylene glycol) (PEG), in a phase I trial of subcutaneous PEGylated urate oxidase.Arthritis Res Ther. 2006;8(1):R12.[77]Armstrong JK,Hempel G,Koling S,et al.Antibody against poly(ethylene glycol) adversely affects PEG-asparaginase therapy in acute lymphoblastic leukemia patients.Cancer-Am Cancer Soc. 2007;110(1):103-111.[78]Poppenborg SM,Wittmann J,Walther W,et al.Impact of anti-PEG IgM antibodies on the pharmacokinetics of pegylated asparaginase preparations in mice.Eur J Pharm Sci.2016;91:122-130.[79]Elsabahy M,Li A,Zhang F,et al.Differential immunotoxicities of poly(ethylene glycol)- vs. poly(carboxybetaine)-coated nanoparticles.J Control Release.2013;172(3):641-652.[80]Verhoef JJ,Carpenter JF,Anchordoquy TJ,et al.Potential induction of anti-PEG antibodies and complement activation toward PEGylated therapeutics.Drug Discov Today. 2014; 19(12):1945-1952.[81]Ishida T,Kiwada H.Accelerated blood clearance (ABC) phenomenon upon repeated injection of PEGylated liposomes. Int J Pharm.2008;354(1-2):56-62.[82]Carter MC,Meyerhoff ME.Instability of succinyl ester linkages in O2'-monosuccinyl cyclic AMP-protein conjugates at neutral pH.J Immunol Methods.1985;81(2):245-257. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||