Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (10): 1593-1598.doi: 10.3969/j.issn.2095-4344.0721
Previous Articles Next Articles
Application prospects of compound gelatin-methacryloyl hydrogel in bone tissue engineering
- 1School of Graduate, Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China; 2Department of Burn and Plastic Surgery, the 253rd Hospital of Chinese PLA, Hohhot 010030, Inner Mongolia Autonomous Region, China
-
Received:2017-11-28Online:2018-04-08Published:2018-04-08 -
Contact:Chen Xiang-jun, Chief physician, Master’s supervisor, School of Graduate, Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China -
About author:Liu Zhao-xing, Master candidate, Physician, School of Graduate, Inner Mongolia Medical University, Hohhot 010030, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Liu Zhao-xing, Wu Xing, Lin Huan-huan, Liu Sha, Chen Xiang-jun. Application prospects of compound gelatin-methacryloyl hydrogel in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(10): 1593-1598.
share this article
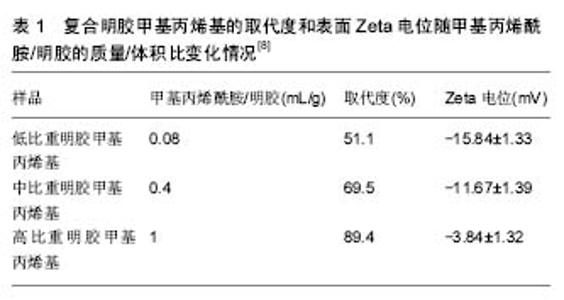
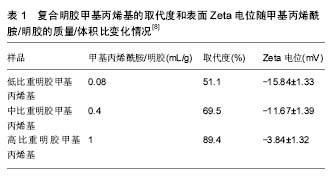
2.1 组织工程骨支架的制备及特性 制备仿生组织工程骨-复合GelMA水凝胶支架,其中GelMA水凝胶的合成都是以Van提出的方法为基础的,由于GelMA水凝胶力学强度、弹性模量较低及骨传导性能较差,遂将GelMA水凝胶与某一种或多种特定的无机物按特定的供给比混合,置于一定反应条件下,制备出具有骨相关特性及适合种子细胞增殖、迁移的仿生骨支架。因骨结构主要组成部分是干质量30%的有机基质(骨胶原纤维、骨钙素、骨唾液蛋白、骨桥蛋白、骨连接蛋白、黏多糖,Ⅰ型胶原占骨中有机物质量的93%)和70%的无机质羟基磷灰石,在有机质和无机质的作用下使骨具有韧、弹性、硬度及脆性。从仿生角度看,构建羟基磷灰石与GelMA水凝胶双相复合材料是构建组织工程骨的一种方案。Tan等[7]制备出改性明胶-羟基磷灰石水凝胶及钛表面矿化明胶水凝胶复合涂层支架材料,对其进行表征分析发现,矿化后水凝胶的表面和内部都存在碳酸型羟基磷灰石纳米晶体的生成,表明GelMA水凝胶能诱导羟基磷灰石矿物在其表面沉积,并且获得的羟基磷灰石成分和结构接近自然骨;通过力学压缩实验发现,矿化后复合水凝胶表现出更高的压缩模量、压缩强度及压缩韧性;但随着GelMA水凝胶取代度的增高,水凝胶和成核矿物之间的界面能也在提高,导致高取代度水凝胶在矿化过程中获得较低含量和结晶度的磷灰石,因此确立GelMA水凝胶取代度的最适比例尤为重要[8]。与传统GelMA水凝胶相对比,GelMA/羟基磷灰石复合水凝胶拥有较低的溶胀比、更高的机械强度及更好的生物相容性。 模块化组织工程在再生天然复合组织中拥有巨大潜力,其通过预先设计的具有几何学和生物学特征的三维模型支架来完成模块化设计。最常用的模块组织形成方法:利用微型模具控制细胞扩增形成细胞球、细胞层技术构建模块组织、细胞-水凝胶混合物构建模块组织;组装模块组织的方法有封闭模具随机填充法、组织打印技术、细胞层技术[9]。在模块化组织构建中,支架拥有利于组装制造的机械刚度和细胞存活的生物相容性,是生物构建成功的关键。Zuo等[10-11]通过模块化方法首先制备出改性明胶-羟基磷灰石水凝胶,通过调节凝胶的机械刚度和细胞功能表达方式来确定适合骨再生的复合GelMA水凝胶浓度;此外,他们利用支架材料的光敏特性,通过光刻技术成功构建了一种具有骨样同心圆双层结构的仿生骨组织,其中内环封装人脐带血管内皮细胞,而外环封装人类成骨细胞MG63。在体外三维培养期间,人脐带血管内皮细胞和MG63不仅表现出令人满意的生长状态,而且表现出与成骨相关性及血管再生相关性的基因表达。构建预血管化组织工程骨是保证植入骨成活的前提。下面简述GelMA水凝胶及其复合凝胶的合成过程。 2.1.1 GelMA水凝胶的制备及改良方法 GelMA是通过明胶和甲基丙烯酸在50 ℃下PBS(pH=7.5)中直接反应合成的,该反应方式为甲基丙烯酸醛基与氨基酸残端的胺基和羟基进行反应。通过调节反应物中的甲基丙烯酸含量,可实现不同程度的甲基丙烯酰基取代,继而产生具有不同物理性质的GelMA。通过提高反应过程中的pH值,可增强胺基和羟基的反应性,生成更高的取代度。取代度可用Habeeb方法测定[12]。反应中通过稀释PBS中反应物浓度(稀释5倍)来终止置换反应,将稀释后的溶液置于透析袋(12-14 ku)中,在50 ℃双蒸水中透析 5-7 d,可将潜在具有细胞毒性的低分子量杂质(包括未反应的甲基丙烯酸和甲基丙烯酸副产物等)完全去除。将透析后的溶液离心、冻干后,形成白色泡沫物质(即GelMA),可储存在-4 ℃冷藏下。将合成的GelMA溶于40 ℃水中,加入光引发剂Irgacure2959,并置于365 nm紫外光(10 mW/cm2)下照射一定时间,便可交联形成具备可调节力学性能的GelMA水凝胶[4]。 Lee等[13-14]对传统方法进行了改良,通过相关实验并最终确定反应所需的最适温度、pH及时间,可用极低浓度的甲基丙烯酸,即可制备出接近99%取代度的GelMA水凝胶,此方法效率高、并可对GelMA取代度进行精准调节,具体方法如下:碳酸盐碳酸氢盐缓冲液(CB缓冲液)制备:将5.86 g NaHCO3和3.18 g Na2CO3溶于1 L蒸馏水中,浓度为0.1 mol/L。 将10 g明胶和1 mL甲基丙烯酸溶于100 mL碳酸盐碳酸氢盐缓冲液(0.25 mol/L)中,pH=9.0,反应温度为50 ℃,反应时间为3 h,3 h后将溶液pH值调至7.0,终止反应,将终止反应后的溶液置于透析袋(12-14 ku)中在50 ℃双蒸水中透析5-7 d,将透析后的溶液离心、冻干,即可获得取代度为97%的GelMA水凝胶,见图1所示。 2.1.2 制备GelMA/羟基磷灰石复合水凝胶 GelMA水凝胶制备同上。①仿生矿化液的配置:按照以下顺序,将药品加入700 mL去离子水中,磁力搅拌溶解,用 1 mol/L HCl调节pH值为7.4,定容至1 000 mL,NaCl 8.035 g,NaHCO3 0.533 g,KCl 0.225 g,K2HPO4•3H2O 0.347 g,MgCl2•6H2O 0.347 g,1.0 mol/L HCl 58.5 mL,CaCl2 0.438 g,Na2SO4 0.072 g,NH2C(CH2OH)3 9.177 g,1.0 mol/L HCl;②水凝胶的矿化:将冻干的水凝胶样品放置塑料离心管中,加入适量的仿生矿化液,放入真空箱中抽真空10 min,以排出样品中的气泡,然后置于37 ℃下的烘箱中静置12 d,每24 h换1次液。 12 d取矿化的样,使用去离子水小心清洗,冷冻干燥后便可获得GelMA/羟基磷灰石复合水凝胶[7]。GelMA/羟基磷灰石复合水凝胶的矿化程度取决于GelMA自身Zeta电位,并发现随着取代度的增加,Zeta电位也在增加,见表1所示。"

2.1.3 模块化仿生骨的制备 GelMA水凝胶制备同上。 GelMA/羟基磷灰石前体聚合溶液的制备:溶液由GelMA、CaCl2和Na2HPO4组成,将Ca和P前体物质分别溶解在等体积蒸馏水中(Ca/P=1.67),包括0.5%(质量体积比)的Irgacure2959。在每种溶液中加入等量的GelMA(10%),在37 ℃下搅拌直至溶解。然后将GelMA-P溶液逐滴加入到GelMA-Ca中,用NaOH调节混合溶液pH至8。最后将GelMA-羟基磷灰石溶液在 37 ℃下搅拌24 h,在室温下储存。另外,通过改变Ca和P的含量(保持相同Ca和P比例:1.67)可形成不同含量羟基磷灰石基团。 类骨组织结构的制备:①“圆和十字”方法:首先在玻璃板两侧放置3个载玻片(厚度为150 μm),以控制GelMA水凝胶的高度,然后吸取180 μL聚乙二醇二甲基丙烯酸酯预聚物溶液,置于玻璃板中间,溶液上方放置盖玻片。随后,将具有环形和十字型结构放入光掩模放置在盖玻片上,将预聚物在紫外光(360-480 nm, 7.9 mW/cm2)下曝光27 s,以诱导形成具有环形和十字形结构的微凝胶。经第1次交联后,使用注射器针头直接将玻璃板上的环形和十字形结构微凝胶剥落至含有25 mL矿物油的培养皿中,以组装微凝胶阵列。第2次交联7 s后,便可形成稳定的由圆环形微凝胶组件和十字形微凝胶组件组成的最终结构;②“逐层组建”方法:以GelMA为原料,将紫外光曝光时间调为20 s,按“圆和十字”方法来制备圆环形的微凝胶,在微凝胶的玻璃板上放置另一种浓度的预聚物溶液,置于可遮挡圆环结构的光掩模下紫外光下照射20 s,即可得到稳定的单层水凝胶阵列,重复上述方法,最终以紫外光下照射5 s,便可获得具有多层的结构的水凝胶阵列[10]。 GelMA/羟基磷灰石微凝胶的制备和组装:将150 μL GelMA/羟基磷灰石前体溶液用移液枪滴到载玻片上,其两侧放置厚度为150 μm载玻片,以控制微凝胶高度,溶液上方放置盖玻片,然后将具有环形结构的光掩模放置在其顶部,并通过紫外光照射(波长360-480 nm,光照强度7.9 mW/cm2)25 s,便可形成内环微凝胶。用PBS冲洗微凝胶玻璃板上的残余反应物,置入另一浓度的预聚物溶液,在第二种光掩模下紫外光照25 s,便可形成外环的水凝胶;手动将双环微凝胶从玻璃板上剥离,紫外光照5 s,便可得到双环微凝胶单元的稳定结构[11]。 2.2 种子细胞与三维支架 2.2.1 间充质干细胞 间充质干细胞是中胚层的一类多能干细胞,是成骨细胞的前体细胞,可直接转化为成骨细胞,促进骨组织再生,CD44、CD90、CD105是其特异性表面标志[15]。目前可用于制备组织工程骨的间充质干细胞主要包括骨髓间充质干细胞、脂肪间充质干细胞及脐血间充质干细胞。2001年,Zuk等[16-17]首次通过从脂肪抽吸物组织悬液中提取出脂肪干细胞,并发现其具有多向分化潜能,可跨胚层分化形成脂肪细胞、成骨细胞、软骨细胞、骨细胞及血管内皮细胞等多种细胞类型,脂肪组织具有易获得,获取过程损伤小的特点;同体积的脂肪组织所含干细胞数量是同体积骨髓的 1 000倍[18-19]。Dong等[20]将GelMA/纳米金颗粒水凝胶支架与脂肪间充质干细胞于体外三维培养,经相关生物学特性检测(CCK-8、碱性磷酸酶活性、LIVE/DEAD检测)证实,GelMA/纳米金颗粒水凝胶支架可促进人脂肪间充质干细胞碱性磷酸酶的分泌,并明显增强成骨相关基因的表达(整合素结合涎蛋白、骨钙素、核心结合蛋白因子2),说明该支架可促进脂肪干细胞增殖、分化为成骨细胞;将载有脂肪间充质干细胞的纳米金GelMA水凝胶植入兔颅骨缺损模型中,8周后经Micro-CT分析发现植入物有明显钙化,这更进一步证实纳米金GelMA水凝胶能够实现骨组织再生。为了增强支架中细胞的增殖与迁徙力,Mahadik等[21]将一定的干细胞因子与GelMA水凝胶支架以共价反应结合,发现可明显提高体外3D培养基中干细胞的活性。胡甜甜[22-23]通过冷冻成胶的方法制备出微米羟基磷灰石GelMA冷冻水凝胶,将该支架与骨间充质干细胞进行3D培养,对其进行表征和体外生物特性检测,体外实验证实该支架具有良好的组织相容性并有骨相关基因的表达,将组织工程骨植入预先设计的大鼠颅骨缺损模型中,经Micro-CT扫描及组织切片苏木精-伊红染色分析,证实2.5%微米羟基磷灰石GelMA水凝胶组修复面积最大,新骨生成量最大、最厚,效果最好。Xavier等[24]添加纳米硅到GelMA水凝胶,证明在没有促分化因子的情况下可促进NIHMC3T3细胞的成骨分化。 方幸幸[25]将仿生GelMA水凝胶与脂肪干细胞进行三维培养,将其种植于裸鼠皮下行异位成骨研究,4周后取出裸鼠体内植入物并行苏木精-伊红、Masson’s染色及免疫荧光分析,发现在含有脂肪干细胞支架上发现持续大量的成骨细胞形成及高水平成骨相关蛋白表达。 2.2.2 血管内皮祖细胞 血管内皮祖细胞是血管内皮细胞的前体细胞,具有促进缺血组织血管新生的能力[26]。内皮祖细胞参与胚胎发育过程中及出生后的血管形成,可在体外形成内皮样克隆,依靠其旁分泌释放的促血管生成因子来促进体内血管生成,更早实现骨内血管循环系统的建立。通过免疫荧光鉴定内皮祖细胞的表面抗原为CD34、CD133。 2.2.3 多细胞共培养 多细胞共培养将2种或2种以上的细胞同时或先后接种于组织工程骨中三维培养,加快3D支架中的血管化进程和成骨效应。目前常用的细胞共培养体系有内皮祖细胞/间充质干细胞、内皮细 胞/成骨细胞等[27]。内皮祖细胞与间充质干细胞共培养后,能显著增强成骨相关性和血管化相关性标志物的表达,包括碱性磷酸酶、骨形态发生蛋白2、血管内皮生长因子、Ⅷ因子等。同时,可早期实现骨缺损区的桥接,促进骨力学性能的恢复[28]。Kavemzadehnarbut等[29]将人脐带血管内皮细胞与人类骨间充质干细胞共同培养后,置于经光刻技术处理过的GelMA水凝胶支架中,发现在支架中有稳定的血管形成,同时有高度的骨相关性基因表达。 在内皮细胞与成骨细胞的共培养体系中,通过介导p38 MAPK(P38丝裂原活化蛋白激酶)信号通路的转录,内皮细胞可支持成骨分化和维持成骨细胞功能,加快成骨进程[30];同时,成骨细胞通过自分泌或旁分泌相关的促血管生长因子,如血管内皮生长因子等,促进内皮细胞血管化。 2.3 微环境 微环境由细胞间质及其中的体液成分构成,作为细胞生存的环境,稳定的内环境是细胞增殖、分化、代谢的基础。由于干细胞具有多项分化潜能,在特定内环境和相关信号分子的诱导下即可获得相关性细胞[31]。富血小板血浆是自体全血经离心后得到的血小板浓缩物,其中含有大量生长因子:如血小板衍生生长因子、转化生长因子β、血管内皮生长因子等,可促进血管形成和骨的修复[32-33]。富血小板血浆可通过激活P13K-AKT-NFKB信号通路[34-35],促进骨髓间充质干细胞、血管内皮生长因子和血小板衍生生长因子的分泌,降低骨髓间充质干细胞在三维培养过程中的凋亡。 力学微环境:骨在人体中起到保护、支持和运动的作用,骨组织一直都处在一个活跃的力学微环境中。相关研究发现,干细胞的增殖、分化、黏附、迁徙等行为会因受到外力刺激的影响而发生改变,因此研究力学刺激对组织工程骨的构建有着重要的意义。De Araujo等[36]通过重物加力方法对包裹在Ⅰ型胶原凝胶中的牙周膜细胞施以静态压力,通过DNA micro-array方法比较了在不同应力及不同加力时间对牙周膜细胞基因表达的影响。结果表明在30 000个检测基因位点中,有108个基因表达发生了变化(PEGZ及COX-2等基因表达量上升;白细胞介素1β、白细胞介素6、TSC-22、TSG-6、BiP、RhoE, DSCR1等基因表达量下调)。Chen等[37]将 GelMA/纳米羟基磷灰石复合水凝胶与人牙周膜干细胞进行体外三维培养,并置于自制的三维力学装置环境下,结果发现,三维力学环境可显著增加Ge1MA微凝胶的杨氏模量并提高细胞活性,促进细胞增殖能力。经MTT和BrdU方法检测并证实单体浓度为10%(w/v)的GelMA/纳米羟基磷灰石复合支架具有较为良好的生物相容性,当纳米羟基磷灰石含量小于5%时,纳米羟基磷灰石的掺入并不会影响细胞的增殖能力;而当纳米羟基磷灰石含量增加至10%时,增殖细胞量显著下降[38],将载有人牙周膜干细胞的GelMA/纳米羟基磷灰石复合凝胶支架植入裸鼠皮下,饲养8周后,将支架取出进行组织学形态观察,发现含5%纳米羟基磷灰石的水凝胶中细胞、胶原分布均匀,其间可见明显毛细血管穿行,并能观察到类似骨隐窝的存在。 "

| [1]Gouron R,Petit L,Boudot C,et al.Osteoclasts and their precursors are present in the induced-membrane during bone reconstruction using the Masquelet technique.J Tissue Eng Regen Med.2017;11(2):382-389.[2]Bauwens K,Stengel D,Hopfner JI,et al.Reconstruction of large defects of the long bones with free vascularized bone grafts: functional results after minimum 5-year follow-up. Orthopedics.2008;31(4):369.[3]Aronson J.Limb-lengthening,skeletal reconstruction, and bone transport with the Ilizarov method. J Bone Joint Surg Am.1997;79(8):1243-1258.[4]Van Den Bulcke AI,Bogdanov B,De Rooze N,et al. Structural and rheological properties of methacrylamide modified gelatin hydrogels.Biomacromolecules. 2000;1(1): 31-38.[5]Alge DL,Anseth KS.Bioactive hydrogels: lighting the way.Nat Mater.2013;12(11):950-952.[6]Yue K,Trujillo-de Santiago G,Alvarez MM,et al.Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels.Biomaterials. 2015;73: 254-271. [7]Tan G,Zhou L,Ning C,et al.Biomimetically-mineralized composite coatings on titanium functionalized with gelatin methacrylate hydrogels.Appl Surf Sci.2013;279(9):293-299. [8]Zhou L,Tan G,Tan Y,et al.Biomimetic mineralization of anionic gelatin hydrogels: effect of degree of methacrylation.RSC Adv. 2014;4:21997-22008.[9]寇德鹏.利用BMP-胶原丝和BMSC-明胶微球构建大尺寸生物仿生骨组织[D].大连医科大学,2015.[10]Zuo Y,Xiao W,Chen X,et al.Bottom-up approach to build osteon-like structure by cell-laden photocrosslinkable hydrogel.Chem Commun(Camb).2012;48(26):3170-3172. [11]Zuo Y,Liu X,Wei D,et al.Photo-cross-linkable methacrylated gelatin and hydroxyapatite hybrid hydrogel for modularly engineering biomimetic osteon.ACS Appl Mater Interfaces. 2015;7(19):10386-10394.[12]Habeeb AF.Determination of free amino groups in Proteins by trinitrobenzeneslfonic acid.Anal Biochem. 1966;14(3): 328-336.[13]Lee BH,Shirahama H,Cho NJ,et al.Efficient and Controllable Synthesis of Highly Substituted Gelatin Methacrylamide for Mechanically Stiff Hydrogels.Rsc Adv. 2015;5(128): 106094-106097.[14]Shirahama H,Lee BH,Tan LP,et al.Precise Tuning of Facile One-Pot Gelatin Methacryloyl (GelMA) Synthesis.Sci Rep. 2016;6:31036.[15]Luang S,Leung V,Peng S,et al.Developmental definition of MSCs: new insights into pending questions. Cell Reprogram. 2011;13(6):465-472.[16]Zuk PA,Zhu M,Mizuno H,et al.Multilineage cells from human adipose tissue:implications for cell-based therapies.Tissue Eng.2001;7(2):211-228.[17]Murphy SV,Atala A.Organ engineering--combining stem cells, biomaterials, and bioreactors to produce bioengineered organs for transplantation.Bioessays.2013;35(3):163-172.[18]Fraser JK,Wulur I,Alfonso Z,Hedrick MH.Fat tissue: an underappreciated sourceof stem cells for biotechnology. Trends Biotechnol.2006;24(4):150-154. [19]Zuk PA.The adipose-derived stem cell: looking back and looking ahead.Mol Biol Cell. 2010;21(11):1783-1787.[20]Dong NH,Ko WK,Min SB,et al.Enhanced bone regeneration with a gold nanoparticle–hydrogel complex.J Materials Chem B.2014;2(2):1584-1593.[21]Mahadik BP,Pedron HS,Skertich LJ,et al.The use of covalently immobilized stem cell factor to selectively affect hematopoietic stem cell activity within a gelatin hydrogel. Biomaterials.2015; 67:297-307.[22]胡甜甜.微米羟基磷灰石—明胶丙烯酸甲酯冷冻水凝胶三维支架的制备及体内外诱导成骨性能研究[D].南方医科大学,2016.[23]Mishra R,Kumar A.Effect of plasma polymerization on physicochemical properties of biocomposite cryogels causing a differential behavior of human osteoblasts.J Colloid Interface Sci.2014;431:139-148.[24]Xavier JR,Teena T,Prachi D,et al.Bioactive nanoengineered hydrogels for bone tissue engineering: agrowth-factor-free approach.Acs Nano.2015;9(3):3109-3118.[25]方幸幸.仿生明胶甲基丙烯酰胺水凝胶支架的骨组织工程研究[D].南方医科大学,2016.[26]Alev C,Ii M,Asahara T.Endothelial progenitor cells: a novel tool for the therapy of ischemic diseases.Antioxid Redox Signal.2011;15(4):949-965.[27]付维力,项舟.血管化组织工程骨构建中细胞共培养体系的研究进展[J].中国修复重建外科杂志,2014,28(2):179-185.[28]赵娴.自体外周血EPCs与BMSCs联合PDPBB构建微血管化生物骨[D].昆明:昆明医科大学,2013.[29]Kazemzadehnarbat M,Rouwkema J,Annabi N,et al. Engineering Photocrosslinkable Bicomponent Hydrogel Constructs for Creating 3D Vascularized Bone.Adv Healthc Mater.2017;6(10).doi: 10.1002/adhm.201601122. Epub 2017 Feb 27.2.[30]Estrela C,AIencarA,Kitten GT,et al.Mesenchymal stem cells in the dental tissues:perspectives for tissue regeneration. BrazDent J.2011;22(2):91-98.[31]Frese L,Dijkman PE,Hoerstrup SP.Adipose Tissue-Derived Stem Cells in Regenerative Medicine.Transfus Med Hemother. 2016;43(4):268-274.[32]Badran Z,Abdallah MN,Torres J,et al.Platelet concentrates for bone regeneration: Current evidence and future challenges. Platelets.2017:1-8.[33]Liu Z,Yuan X,Fernandes G,et al.The combination of nano-calcium sulfate/platelet rich plasma gel scaffold with BMP2 gene-modified mesenchymal stem cells promotes bone regeneration in rat critical-sized calvarial defects.Stem Cell Res Ther.2017;8(1):122.[34]彭艳.富血小板血浆激活PI3K/AKT/NFKB信号通路促进骨髓间充质干细胞再生及修复功能的相关研究[D].广州:南方医科大学, 2013.[35]赵天源,孙红.骨组织工程支架材料及其血管化的研究进程[J].中国组织工程研究, 2013,17(38):6832-6838.[36]De Araujo R,Oba Y,Moriyama K.Identification of genes related to mechanical stress in human periodontal ligament cells using microarray analysis.J Periodontal Res. 2007;42(1): 15-22.[37]Chen X,Bai S,Li B,et al.Fabrication of gelatin methacrylate/nanohydroxyapatite Microgel arrays for periodontal tissue regeneration.Int J Nanomedicine. 2016;11: 4707. [38]陈晞.应用GelMA水凝胶构建牙周膜干细胞力学三维微环境及牙周组织再生的实验研究[D].第四军医大学,2016.[39]Kalyani S,Dhiman N,Laha A,et al.Three-Dimensional Bioprinting for Bone Tissue Regeneration. Curr Opin Biomed Eng.2017;2:22-28.[40]Heo DN,Castro NJ,Lee SJ,et al.Enhanced bone tissue regeneration using a 3D printed microstructure incorporated with a hybrid nano hydrogel. Nanoscale. 2017;9(16): 5055-5062. [1]Gouron R,Petit L,Boudot C,et al.Osteoclasts and their precursors are present in the induced-membrane during bone reconstruction using the Masquelet technique.J Tissue Eng Regen Med.2017;11(2):382-389.[2]Bauwens K,Stengel D,Hopfner JI,et al.Reconstruction of large defects of the long bones with free vascularized bone grafts: functional results after minimum 5-year follow-up. Orthopedics.2008;31(4):369.[3]Aronson J.Limb-lengthening,skeletal reconstruction, and bone transport with the Ilizarov method. J Bone Joint Surg Am.1997;79(8):1243-1258.[4]Van Den Bulcke AI,Bogdanov B,De Rooze N,et al. Structural and rheological properties of methacrylamide modified gelatin hydrogels.Biomacromolecules. 2000;1(1): 31-38.[5]Alge DL,Anseth KS.Bioactive hydrogels: lighting the way.Nat Mater.2013;12(11):950-952.[6]Yue K,Trujillo-de Santiago G,Alvarez MM,et al.Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels.Biomaterials. 2015;73: 254-271. [7]Tan G,Zhou L,Ning C,et al.Biomimetically-mineralized composite coatings on titanium functionalized with gelatin methacrylate hydrogels.Appl Surf Sci.2013;279(9):293-299. [8]Zhou L,Tan G,Tan Y,et al.Biomimetic mineralization of anionic gelatin hydrogels: effect of degree of methacrylation.RSC Adv. 2014;4:21997-22008.[9]寇德鹏.利用BMP-胶原丝和BMSC-明胶微球构建大尺寸生物仿生骨组织[D].大连医科大学,2015.[10]Zuo Y,Xiao W,Chen X,et al.Bottom-up approach to build osteon-like structure by cell-laden photocrosslinkable hydrogel.Chem Commun(Camb).2012;48(26):3170-3172. [11]Zuo Y,Liu X,Wei D,et al.Photo-cross-linkable methacrylated gelatin and hydroxyapatite hybrid hydrogel for modularly engineering biomimetic osteon.ACS Appl Mater Interfaces. 2015;7(19):10386-10394.[12]Habeeb AF.Determination of free amino groups in Proteins by trinitrobenzeneslfonic acid.Anal Biochem. 1966;14(3): 328-336.[13]Lee BH,Shirahama H,Cho NJ,et al.Efficient and Controllable Synthesis of Highly Substituted Gelatin Methacrylamide for Mechanically Stiff Hydrogels.Rsc Adv. 2015;5(128): 106094-106097.[14]Shirahama H,Lee BH,Tan LP,et al.Precise Tuning of Facile One-Pot Gelatin Methacryloyl (GelMA) Synthesis.Sci Rep. 2016;6:31036.[15]Luang S,Leung V,Peng S,et al.Developmental definition of MSCs: new insights into pending questions. Cell Reprogram. 2011;13(6):465-472.[16]Zuk PA,Zhu M,Mizuno H,et al.Multilineage cells from human adipose tissue:implications for cell-based therapies.Tissue Eng.2001;7(2):211-228.[17]Murphy SV,Atala A.Organ engineering--combining stem cells, biomaterials, and bioreactors to produce bioengineered organs for transplantation.Bioessays.2013;35(3):163-172.[18]Fraser JK,Wulur I,Alfonso Z,Hedrick MH.Fat tissue: an underappreciated sourceof stem cells for biotechnology. Trends Biotechnol.2006;24(4):150-154. [19]Zuk PA.The adipose-derived stem cell: looking back and looking ahead.Mol Biol Cell. 2010;21(11):1783-1787.[20]Dong NH,Ko WK,Min SB,et al.Enhanced bone regeneration with a gold nanoparticle–hydrogel complex.J Materials Chem B.2014;2(2):1584-1593.[21]Mahadik BP,Pedron HS,Skertich LJ,et al.The use of covalently immobilized stem cell factor to selectively affect hematopoietic stem cell activity within a gelatin hydrogel. Biomaterials.2015; 67:297-307.[22]胡甜甜.微米羟基磷灰石—明胶丙烯酸甲酯冷冻水凝胶三维支架的制备及体内外诱导成骨性能研究[D].南方医科大学,2016.[23]Mishra R,Kumar A.Effect of plasma polymerization on physicochemical properties of biocomposite cryogels causing a differential behavior of human osteoblasts.J Colloid Interface Sci.2014;431:139-148.[24]Xavier JR,Teena T,Prachi D,et al.Bioactive nanoengineered hydrogels for bone tissue engineering: agrowth-factor-free approach.Acs Nano.2015;9(3):3109-3118.[25]方幸幸.仿生明胶甲基丙烯酰胺水凝胶支架的骨组织工程研究[D].南方医科大学,2016.[26]Alev C,Ii M,Asahara T.Endothelial progenitor cells: a novel tool for the therapy of ischemic diseases.Antioxid Redox Signal.2011;15(4):949-965.[27]付维力,项舟.血管化组织工程骨构建中细胞共培养体系的研究进展[J].中国修复重建外科杂志,2014,28(2):179-185.[28]赵娴.自体外周血EPCs与BMSCs联合PDPBB构建微血管化生物骨[D].昆明:昆明医科大学,2013.[29]Kazemzadehnarbat M,Rouwkema J,Annabi N,et al. Engineering Photocrosslinkable Bicomponent Hydrogel Constructs for Creating 3D Vascularized Bone.Adv Healthc Mater.2017;6(10).doi: 10.1002/adhm.201601122. Epub 2017 Feb 27.2.[30]Estrela C,AIencarA,Kitten GT,et al.Mesenchymal stem cells in the dental tissues:perspectives for tissue regeneration. BrazDent J.2011;22(2):91-98.[31]Frese L,Dijkman PE,Hoerstrup SP.Adipose Tissue-Derived Stem Cells in Regenerative Medicine.Transfus Med Hemother. 2016;43(4):268-274.[32]Badran Z,Abdallah MN,Torres J,et al.Platelet concentrates for bone regeneration: Current evidence and future challenges. Platelets.2017:1-8.[33]Liu Z,Yuan X,Fernandes G,et al.The combination of nano-calcium sulfate/platelet rich plasma gel scaffold with BMP2 gene-modified mesenchymal stem cells promotes bone regeneration in rat critical-sized calvarial defects.Stem Cell Res Ther.2017;8(1):122.[34]彭艳.富血小板血浆激活PI3K/AKT/NFKB信号通路促进骨髓间充质干细胞再生及修复功能的相关研究[D].广州:南方医科大学, 2013.[35]赵天源,孙红.骨组织工程支架材料及其血管化的研究进程[J].中国组织工程研究, 2013,17(38):6832-6838.[36]De Araujo R,Oba Y,Moriyama K.Identification of genes related to mechanical stress in human periodontal ligament cells using microarray analysis.J Periodontal Res. 2007;42(1): 15-22.[37]Chen X,Bai S,Li B,et al.Fabrication of gelatin methacrylate/nanohydroxyapatite Microgel arrays for periodontal tissue regeneration.Int J Nanomedicine. 2016;11: 4707. [38]陈晞.应用GelMA水凝胶构建牙周膜干细胞力学三维微环境及牙周组织再生的实验研究[D].第四军医大学,2016.[39]Kalyani S,Dhiman N,Laha A,et al.Three-Dimensional Bioprinting for Bone Tissue Regeneration. Curr Opin Biomed Eng.2017;2:22-28.[40]Heo DN,Castro NJ,Lee SJ,et al.Enhanced bone tissue regeneration using a 3D printed microstructure incorporated with a hybrid nano hydrogel. Nanoscale. 2017;9(16): 5055-5062. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [5] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Liu Fang, Shan Zhengming, Tang Yulei, Wu Xiaomin, Tian Weiqun. Effects of hemostasis and promoting wound healing of ozone sustained-release hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3445-3449. |
| [14] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [15] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||