Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (32): 5228-5233.doi: 10.3969/j.issn.2095-4344.0557
Previous Articles Next Articles
Effect and mechanism of TRPC1 channel regulating function of vascular smooth muscle cells
Gao Chao1, Jia Xiao-ling1, Rong Li-jun2
- 1School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; 2Clinic Outpatient, Training and Administration Department of the Central Military Commission, Beijing 100852, China
-
Received:2018-08-07Online:2018-11-18Published:2018-11-18 -
Contact:Rong Li-jun, Master, Associate chief physician, Clinic Outpatient, Training and Administration Department of the Central Military Commission, Beijing 100852, China -
About author:Gao Chao, Master, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China -
Supported by:the National Natural Science Foundation of China, No. 11372030
CLC Number:
Cite this article
Gao Chao1, Jia Xiao-ling1, Rong Li-jun2 . Effect and mechanism of TRPC1 channel regulating function of vascular smooth muscle cells [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(32): 5228-5233.
share this article
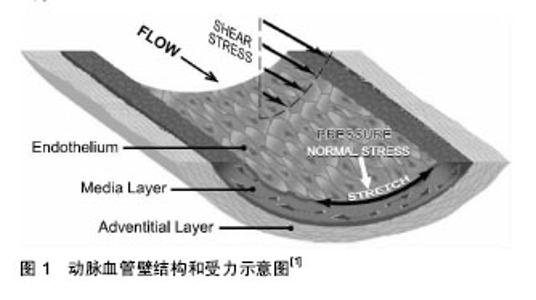
2.1 TRPC1通道简介 离子通道是广泛表达在各种细胞上的特殊的亲水性蛋白质孔道,是多种细胞生命活动的分子基础。近年来,随多种细胞分子生物学研究手段的发展,人们对多种离子通道的结构特性和生物学功能有了更加深入的了解,发现了离子通道的异常会对各种细胞的功能产生显著的影响,严重的会致使细胞功能异常,进而导致多种疾病的发生。因此,研究细胞上的离子通道,对治疗各种由离子通道结构和功能异常引起的疾病有重要的指导意义。 TRP通道是一个非选择性阳离子通道的超家族,作为细胞的传感器和效应器,调控多种细胞功能[1]。第一个TRP通道于1969年由Cosens和Manning在一种果蝇突变体的光感受器细胞上发现。在持续的光刺激下,野生型的果蝇光感受细胞会产生持续的细胞内钙浓度的升高,而突变体的果蝇光感受器细胞只能产生短暂的细胞内钙浓度升高,因此这种突变的基因被命名为TRP通道[2]。随后,在哺乳动物中,大量的TRP通道被克隆出来[3]。到目前为止,哺乳动物TRP通道超家族共有28名成员,基于结构的相似性,它们被划分为6个亚家族:TRPC(Canonical),TRPM(Melastatin),TRPML (Mucolipins),TRPV(Vanilloid),TRPP(Policystin)和TRPA(Ankyrin-rich protein)[4],其中TRPC通道因其在结构上与最早在果蝇中发现的TRP通道类似而得名[5]。 哺乳动物的TRPC通道目前共有7种,根据结构的同源性,功能的相似性和已知的相互作用,TRPC家族又可以划分为4类:TRPC1,TRPC2,TRPC3/6/7和TRPC4/5 (TRPC1有时包括在TRPC4/5中)[5-7]。TRPC1是第一个被发现的哺乳动物TRP通道蛋白[8],目前已有大量文献报道揭示其结构和特性,以及其在各种细胞中的功能。 2.1.1 TRPC1通道基因简介 1995年,人类TRPC1通道的DNA和对应的氨基酸序列以及表达模式被公布出来。编码人类TRPC1通道的基因位于3号染色体上[8]。目前发现TRPC1通道的mRNA共有5种剪接变体,分别是:TRPC1α,TRPC1β,TRPC1γ,TRPC1δ和TRPC1ε,但似乎只有3个(TRPC1α,TRPC1β和TRPC1ε)能被翻译成为有功能的通道蛋白。其中TRPC1β在第3个锚蛋白重复序列中缺少34个氨基酸残基,TRPCε则是在第一个跨膜螺旋结构域的下游有7个氨基酸残基的缺失[9-10](锚蛋白重复序列和跨膜螺旋结构域将在下一部分进行介绍)。由公布的DNA序列推测的TRPC1α和TRPC1β通道蛋白的相对分子质量应该分别是91 200和87 600,然而,在正常组织中,TRPC1通道蛋白的实际相对分子质量要大于这个大小。这个差异最初被认为是由通道蛋白的第5个跨膜结构域和孔通道区域的糖基化所导致的[11](跨膜螺旋结构域和孔通道区域将在下一部分进行介绍)。然而,有研究发现在公布的起始密码子AUG的上游,存在另一个起始密码子CUG,从该起始密码子处开始翻译,会导致得到的TRPC1通道蛋白的氨基末端延伸出78个氨基酸残基,这段延伸出的序列含有一簇带正电的氨基酸残基[10],此现象的生理功能尚不明确。有人推测,这是一种TRPC1通道功能的自我调控机制,即当TRPC1通道蛋白发生构象转变时,这个带正电荷的区域可能会阻断其带负电荷的孔通道区,使TRPC1通道关闭[12]。另外,TRPC1ε还存在一个在第5外显子上带有34个核苷酸残基缺失的有功能的剪接变体[10]。 2.1.2 TRPC1通道蛋白简介 TRPC1通道蛋白由6个跨膜结构片段(S1-S6)以及在胞质内的N端和C端组成。在第5和第6个跨膜结构片段之间有个胞外环,这个区域主要是由带负电荷的谷氨酸和天冬氨酸构成的α螺旋结构,是TRPC1通道的离子通透区。TRPC1通道蛋白的N 端有1个螺旋区(CC)和4个锚蛋白重复区(AAAA),人们推测它们可能参与了TRPC1通道蛋白在细胞上的锚定,其中的锚蛋白重复区还可能参与调控通道蛋白的同源多聚化。与所有其他所有的TRPC通道蛋白的结构类似,TRPC1通道蛋白的C端有一个功能未知的TRP盒结构(氨基酸残基序列为EWKFAR)以及两个钙调蛋白(Calmodulin,CaM)结合位点,后者参与调控钙离子诱导的TRPC1通道电流的失活。此外,TRPC1通道蛋白C端的2个带负电荷的天冬氨酸残基(Asp639和Asp640)能与钙感受器-基质相互作用分子1(Stromal interacting molecule 1,STIM1)上的2个带正电荷的赖氨酸残基(Lys684和Lys685)相互作用,调控TRPC1通道的开放与关闭[13]。 2.2 TRPC1通道对血管平滑肌细胞功能的影响 2.2.1 血管平滑肌细胞简介 血管平滑肌细胞位于血管壁的中层,是血管壁的重要细胞组分之一。在胚胎发生期,血管平滑肌细胞由三胚层中的中胚层发育而来,发育成熟的血管平滑肌细胞所具有的特征表型为收缩表型(contractile phenotype),这种表型的血管平滑肌细胞主要执行收缩功能,其收缩与舒张能维持血管结构的完整性,调节血压和血流分布[14]。 在正常情况下,成人血管系统内的平滑肌细胞的特征是极低的增殖速率,非常低的合成活性和表达一系列独特的离子通道、收缩蛋白和信号分子,这对维持其功能尤为重要[14-16]。不同解剖学位置的血管平滑肌细胞表达不同类型的离子通道,这些特定的通道表达模式在明确平滑肌细胞的表型时非常重要[17-18]。不同于终末分化的心肌细胞和骨骼肌细胞,血管平滑肌细胞仍保留着一定的表型可塑性,表现为它们可以对体液,环境和病理生理指标的刺激做出应答,在某些因素,如生长因子(growth factor),炎症反应(inflammatory response)和力学因素(mechanical factors)等的刺激下,收缩表型的血管平滑肌细胞可能会暂时去分化,重新转变为未分化的更接近胚胎期的表型,即合成表型(synthetic phenotype)[14]。动脉血管壁结构和受力示意图见图1。此种表型的血管平滑肌细胞表现出显著的增殖和迁移的特性,并且在其增殖和迁移的同时会合成和分泌细胞外基质和多种细胞因子等[15,17],这也是导致许多血管疾病,如高血压(hypertension)、动脉粥样硬化(atherosclerosis)、血管成形术后再狭窄(restenosis)等发生的重要原因[14]。因此,研究血管平滑肌细胞的功能改变及其机制,对许多血管疾病的治疗有着重要的意义。"

| [1] House SJ, Potier M, Bisaillon J, et al. The non-excitable smooth muscle: calcium signaling and phenotypic switching during vascular disease. Pflugers Arch. 2008;456(5): 769-790.[2] Nilius B, Szallasi A. Transient receptor potential channels as drug targets: from the science of basic research to the art of medicine. Pharmacol Rev. 2014;66:676-682. [3] Cosens DJ, Manning A. Abnormal electroretinogram from a drosophila mutant. Nature. 1969;224(5216):285-293. [4] Petersen CC, Berridge MJ, Borgese MF, et al. Putative capacitative calcium entry channels: expression of Drosophila trp and evidence for the existence of vertebrate homologues. Biochem J. 1995;311(Pt1):41-53. [5] Alonsocarbajo L, Kecskes M, Jacobs G, et al. Muscling in on TRP channels in vascular smooth muscle cells and cardiomyocytes. Cell Calcium. 2017;66:48-61. [6] Vazquez G, Wedel B J, Aziz O, et al. The mammalian TRPC cation channels. Biochim Biophys Acta. 2004;1742(3):21-36. [7] Trebak M, Lemonnier L, Smyth J T, et al. Phospholipase C-coupled receptors and activation of TRPC channels. Handb Exp Pharmacol. 2007;179(179):593-614. [8] Trebak M, Vazquez G, Bird GS, et al. The TRPC3/6/7 subfamily of cation channels. Cell Calcium. 2003;33(5-6): 451-466. [9] Clapham DE, Julius D, Montell C, et al. International union of pharmacology. XLIX. nomenclature and structure-function relationships of transient receptor potential channels. Pharmacol Rev. 2005;57(4):427-450. [10] Sakura H, Ashcroft FM. Identification of four trp1 gene variants murine pancreatic beta-cells. Diabetologia. 1997; 40(5): 528-537. [11] Ong EC, Nesin V, Long CL, et al. A TRPC1 protein-dependent pathway regulates osteoclast formation and function. J Biol Chem. 2013;288(31):22219-22245. [12] Rychkov G, Barritt GJ. TRPC1 Ca2+-permeable channels in animal cells. Handb Exp Pharmacol. 2007;179(179):23-47. [13] Nilius B, Flockerzi V. Mammalian Transient Receptor Potential (TRP) Cation Channels. Springer Berlin Heidelberg. 2014:105-117. [14] 崔媛媛,史娟.在神经系统中TRPC1的研究进展[J].生理科学进展,2017,48(6):58-67.[15] Owens GK, Kumar MS, Wamhoff BR. Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol Rev. 2004;84(3):767-801. [16] Somlyo AP, Somlyo AV. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: modulated by G proteins, kinases, and myosin phosphatase. Physiol Rev. 2003;83(4): 1325-1355. [17] Yoshida T, Owens GK. Molecular determinants of vascular smooth muscle cell diversity. Circ Res. 2005;96(3):280-301.[18] Rzucidlo EM, Martin KA, Powell RJ. Regulation of vascular smooth muscle cell differentiation. J Vasc Surg. 2007;45 Suppl A(6):25-38. [19] Inoue R, Jensen LJ, Shi J, et al. Transient receptor potential channels in cardiovascular function and disease. Circ Res. 2006;99(2):119-136. [20] Xu SZ, Beech DJ. TrpC1 is a membrane-spanning subunit of store-operated Ca2+ channels in native vascular smooth muscle cells. Circ Res. 2001;88(1):84-97. [21] Beech DJ. TRPC1: store-operated channel and more. Pflügers Archiv. 2005;451(1):53-60. [22] Ahmad AA, Streiff M, Hunter C, et al. Physiological and pathophysiological role of transient receptor potential canonical channels in cardiac myocytes. Prog Biophys Mol Biol. 130:254-263. [23] Trebak M. Canonical transient receptor potential channels in disease: targets for novel drug therapy? Drug Discov Today. 2006;11(19-20):924-930. [24] Dietrich A, Chubanov V, Kalwa H, et al. Cation channels of the transient receptor potential superfamily: their role in physiological and pathophysiological processes of smooth muscle cells. Pharmacol Ther. 2006;112(3):744-760. [25] Watanabe H, Murakami M, Ohba T, et al. TRP channel and cardiovascular disease. Pharmacol Ther. 2008;118(3): 337-351. [26] Kumar B, Dreja K, Shah SS, et al. Upregulated TRPC1 channel in vascular injury in vivo and its role in human neointimal hyperplasia. Circ Res. 2006;98(4):557-580. [27] Takahashi Y, Watanabe H, Murakami M, et al. Upregulation of TRPC1 is involved in Angiotensin II-induced vascular smooth muscle cell hypertrophy. J Mol Cell Cardiol. 2006;41(6): 1057-1057. [28] Sweeney M, Yu Y, Platoshyn O, et al. Inhibition of endogenous TRP1 decreases capacitative Ca2+ entry and attenuates pulmonary artery smooth muscle cell proliferation. Am J Physiol Lung Cell Mol Physiol. 2002;283(1):144-155. [29] Li J, Mckeown L, Ojelabi O, et al. Nanomolar potency and selectivity of a Ca2+ release-activated Ca2+ channel inhibitor against store-operated Ca2+ entry and migration of vascular smooth muscle cells. Br J Pharmacol. 2011;164(2):382. [30] Ingueneau C, Huynh-Do U, Marcheix B, et al. TRPC1 is regulated by caveolin-1 and is involved in oxidized LDL-induced apoptosis of vascular smooth muscle cells. J Cell Mol Med. 2009;13(8b):1620-1631.[31] Clapham DE. Calcium signaling. Cell. 1995;80(2):259-288. [32] Petersen OH, Michalak M, Verkhratsky A. Calcium signalling: past, present and future. Cell Calcium. 2005;38(3):161-169. [33] Michael J. Berridge. Inositol trisphosphate and calcium signalling. Nature. 1993;361(6410):315-325. [34] Putney JW. Pharmacology of store-operated calcium channels. Mol Interv. 2010;10(4):209-218. [35] Potier M, Trebak M. New developments in the signaling mechanisms of the store-operated calcium entry pathway. Pflugers Arch. 2008;457(2):405-415. [36] Putney JW Jr. Capacitative calcium entry revisited. Cell calcium. 1990;11(10):611. [37] Albert AP, Large WA. Store-operated Ca2+ -permeable non-selective cation channels in smooth muscle cells. Cell Calcium. 2003;33(5-6):345-356. [38] Takahashi Y, Watanabe H, Murakami M, et al. Involvement of transient receptor potential canonical 1 (TRPC1) in angiotensin II-induced vascular smooth muscle cell hypertrophy. Atherosclerosis. 2007;195(2):287-296. [39] Brueggemann LI, Markun DR, Henderson KK, et al. Pharmacological and electrophysiological characterization of store-operated currents and capacitative Ca2+ entry in vascular smooth muscle cells. J Pharmacol Exp Ther. 2006; 317(2):488-499. [40] Guo R, Yang L, Li M, et al. Stim1- and Orai1-mediated store-operated calcium entry is critical for angiotensin II-induced vascular smooth muscle cell proliferation. Cardiovasc Res. 2012;93(2):360-370. [41] Zhang Y, Lu W, Yang K, et al. Bone morphogenetic protein 2 decreases TRPC expression, store-operated Ca2+ entry, and basal [Ca2+]i in rat distal pulmonary arterial smooth muscle cells. Am J Physiol Cell Physiol. 2013;304(9):C833-C843. [42] Wang Y, Wang Y, Li GR. TRPC1/TRPC3 channels mediate lysophosphatidylcholine-induced apoptosis in cultured human coronary artery smooth muscles cells. Oncotarget. 2016;7(32): 50937-50951. [43] Shi J, Ju M, Abramowitz J, et al. TRPC1 proteins confer PKC and phosphoinositol activation on native heteromeric TRPC1/C5 channels in vascular smooth muscle: comparative study of wild-type and TRPC1-/- mice. FASEB J. 2012;26(1): 409-419. [44] Bird GS, Dehaven WI, Smyth JT, et al. Methods for studying store-operated calcium entry. Methods. 2008;46(3):204-212. [45] Trebak M, Bird GSJ, Mckay RR, et al. Comparison of human TRPC3 channels in receptor-activated and store-operated modes differential sensitivity to channel blockers suggests fundamental differences in channel composition. J Biol Chem. 2002;277(24):21617-21623. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||