Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (28): 4562-4567.doi: 10.3969/j.issn.2095-4344.0369
Previous Articles Next Articles
Biomarkers for lumbar disc degeneration: how to solve the influences of specificity, sensitivity and covariates
Guo Mei-yu, Li Zhong-hai
- Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116600, Liaoning Province, China
-
Received:2018-06-26Online:2018-10-08Published:2018-10-08 -
Contact:Li Zhong-hai, MD, Associate professor, Master’s supervisor, Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116600, Liaoning Province, China -
About author:Guo Mei-yu, Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116600, Liaoning Province, China -
Supported by:the Guidance Plan of Natural Science Foundation of Liaoning Province, No. 20170540294; the Basic Scientific Research Project of Liaoning Provincial Universities, No. LQ2017022
CLC Number:
Cite this article
Guo Mei-yu, Li Zhong-hai. Biomarkers for lumbar disc degeneration: how to solve the influences of specificity, sensitivity and covariates [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(28): 4562-4567.
share this article
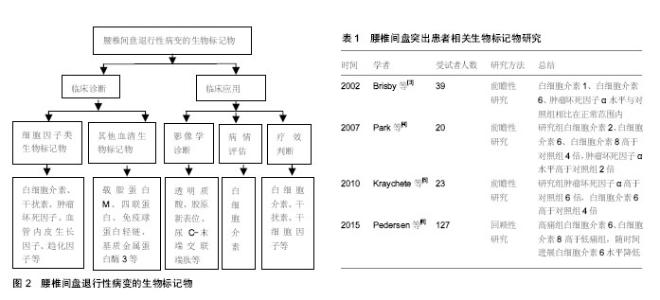
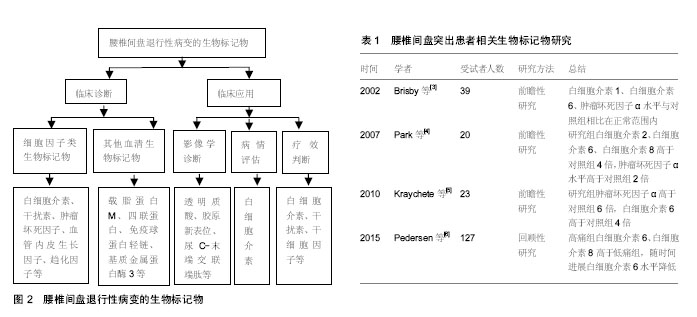
2.1 腰椎间盘退行性病变的生物标记物 2.1.1 细胞因子类生物标记物 细胞因子是由免疫细胞(如单核、巨噬细胞、T细胞、B细胞、NK细胞等)和某些非免疫细胞(内皮细胞、表皮细胞、纤维母细胞等)经刺激而合成、分泌的一类具有广泛生物学活性的小分子蛋白质。细胞因子被划分为白细胞介素、干扰素、趋化因子、生长因子等。近年来已经发现多种细胞因子与椎间盘退行性病变有关。 已有研究指出腰椎间盘突出症患者手术时的白细胞介素1、白细胞介素6、干扰素γ和肿瘤坏死因子α水平均在正常范围内[3]。Brisby等[3]研究也证实椎间盘突出患者的血清肿瘤坏死因子α水平与健康对照组相比并无显著差异。然而,Park等[4]进一步研究发现腰椎间盘突出症患者血清的白细胞介素2、白细胞介素6、白细胞介素8、肿瘤坏死因子α、可溶性活化诱导型TNFR家族受体和可溶性活化诱导型TNFR家族受体配体的平均浓度均显著高于对照组,其中白细胞介素2、白细胞介素6、白细胞介素8是对照组的4倍,肿瘤坏死因子α水平是对照组2倍。Kraychete等[5]以少量受试者为样本,并对其中由椎间盘突出引起慢性疼痛患者的血液和脑脊液中白细胞介素8、白细胞介素1、肿瘤坏死因子α、白细胞介素6和可溶性肿瘤坏死因子受体水平进行研究,结果显示椎间盘突出患者的肿瘤坏死因子α和白细胞介素6水平显著升高,但白细胞介素1或可溶性肿瘤坏死因子受体水平并未得到类似的结果。Pedersen等[6]检测了因椎间盘突出引起腰椎神经根性疼痛患者第6周和第12个月时的血清白细胞介素6、白细胞介素8水平,并比较了白细胞介素6、白细胞介素8的水平与目测类比评分之间的关系,结果证实慢性根性疼痛可能与椎间盘突出后血清促炎性细胞因子白细胞介素6和白细胞介素8持续增加有关,提示这些细胞因子可能促进腰椎间盘突出患者的慢性疼痛发展。Moon等[7]将微血管内皮细胞培养在来自椎间盘退化的纤维环细胞细胞培养物的条件培养基中,通过ELISA测定该细胞培养基中的血管内皮生长因子、白细胞介素8和神经生长因子,结果发现白细胞介素8和血管内皮生长因子可能是来自纤维环细胞的分泌因子,并通过促血管生成调节椎间盘退变发展。因此,作者认为白细胞介素8和血管内皮生长因子有望成为腰椎间盘退行性病变的生物标记物。腰椎间盘退行性病变的生物标记物见图2;腰椎间盘突出患者相关生物标记物研究见表1。 Deng等[8]选取了8项病例-对照研究,总计392例受试者,其中263例为患有椎间盘退变患者,129例患者为健康对照,证实血清白细胞介素6蛋白表达水平可能与椎间盘退变有关。进一步研究显示椎间盘突出症患者的血清白细胞介素6蛋白的表达水平明显增加,与对照组相比,差异具有统计学意义[8]。此外,大量的研究表明椎间盘退行性疾病和椎管狭窄患者的白细胞介素6水平显著高于椎间盘突出患者,椎管狭窄和椎间盘退行性疾病患者多种促炎因子和生长因子水平都高于椎间盘突出患者,包括白细胞介素2、白细胞介素3、白细胞介素8、肝细胞生长因子、干扰素2、白血病抑制因子、单核细胞趋化蛋白3和肿瘤坏死因子β[8]。因此,这些细胞因子都有可能成为椎间盘退行性疾病潜在的生物标记物。陈岩等[9]应用斑点杂交和原位杂交技术检测转化生长因子β对椎间盘原代培养及传代培养的纤维环细胞、髓核细胞中Ⅰ型胶原mRNA的调节作用时,发现转化生长因子β可以按照剂量依赖方式正向调节椎间盘Ⅰ胶原基因表达,表明转化生长因子β与椎间盘退变过程中不断发展的纤维化过程密切相关,提示转化生长因子β可作为腰椎间盘退行性病变的生物标记物。 椎间盘是人体最大的无血管组织,具有自身免疫原性,但在健康状况下为免疫耐受组织。当椎间盘突出或发生破裂时,免疫耐受组织和细胞暴露于系统环境中可诱发自身免疫应答反应。白细胞介素17是辅助性T细胞17的主要效应细胞因子,可诱导炎性细胞因子的产生。Cheng等[10]通过对破裂和未破裂腰椎间盘突出症患者和健康对照组的外周血中辅助性T细胞17水平与椎间盘突出的联系进行研究,证实椎间盘突出患者的外周血辅助性T细胞17出现频率和白细胞介素17表达水平高于健康对照组,破裂椎间盘患者的外周血辅助性T细胞17出现频率和白细胞介素17表达明显高于未破裂椎间盘突出患者,故作者认为椎间盘突出会诱发自身免疫反应,使白细胞介素17表达的增加,加速炎症的发生,从而进一步引起椎间盘退变程度的加重,椎间盘发生破裂时这种效应更为显著,这可能是导致椎间盘破裂的患者比不破裂的患者疼痛严重的原因之一。 白细胞介素21的功能为调节辅助效应T细胞的活性,并促进辅助性T细胞17和B细胞的分化。Xue等[11]发现腰椎间盘突出症患者外周血和椎间盘组织中的白细胞介素21、白细胞介素17和环氧化酶2水平明显高于健康对照组,目测类比疼痛评分与白细胞介素17、环氧化酶2、白细胞介素21的水平呈正相关,提示这些因子在腰椎间盘突出症的发病机制中占有重要地位。 趋化因子C-C基序配体5和C-X-C基序配体6由破坏的椎间盘细胞释放。Grad等[12]研究表明腰椎间盘退变患者趋化因子C-C基序配体5是非退变组的1.6倍,C-X-C基序配体6是非退变组的1.3倍,提示趋化因子C-C基序配体5和C-X-C基序配体6可作为诊断椎间盘退行性疾病的生物标记物。另外,作者还评估了这两种因子的诊断灵敏度和特异性,通过计算两种因子的曲线下面积(AUC),证实CXCL6的诊断准确性略高于趋化因子C-C基序配体5,其能做出诊断的可能性接近曲线下面积(即AUC = 0.5)[11]。 2.1.2 其他血清生物标记物 此外,相关研究还发现了其他与椎间盘炎症和变性相关的生物标记物。Goode 等[13]研究指出腰椎关节突关节发生创伤和炎症时可释放其他一些炎性递质,如5-羟色胺、缓激肽、前列腺素E等,这些炎症递质在腰椎小关节炎的发生和进展过程"
| [1] Wuertz K, Haglund L.Inflammatory mediators in intervertebral disk degeneration and discogenic pain. Global Spine.2013; 3(3):175-184.[2] Risbud MV,Shapiro IM.Role of cytokines in intervertebral disc degeneration:pain and disccontent. Nat Rev Rheumatol. 2014; 10(1):44-56.[3] Brisby H,Olmarker K,Larsson K,et al.Proinflammatory cytokines in cerebrospinal fluid and serum in patients with disc herniation and sciatica. Eur Spine J.2002;11(1):62-66.[4] Park MS,Lee HM,Hahn SB,et al.The association of the activation-inducible tumor necrosis factor receptor and ligand with lumbardisc herniation.Yonsei Med J.2007;48(5):839-846.[5] Kraychete DC,Sakata RK,Issy AM,et al.Serum cytokine levels in patients with chronic low back pain due to herniated disc: analyticalcross-sectional study.Sao Paulo Med J.2010;128(5):259-262.[6] Pedersen LM,Schistad E,Jacobsen LM,et al.Serum levels of the pro-inflammatory interleukins -6 (IL-6) and -8 (IL-8) patients with lumbarradicular pain due to disc her niation:a 12-month prospective study. Brain Behav. Immun.2015;46: 132-136.[7] Moon HJ,Yurube T,Lozito TP,et al.Effects of secreted factors in culture medium of annulus fibrosus cells on microvascular endothelial cells: elucidating the possible pathomechanisms of matrix degradation and nerve in-growth in disc degeneration. Osteoarthritis Cartilage. 2014;22(2):344-354.[8] Deng X,Zhao F,Kang B,et al.Elevated interleukin-6 expression levels are associated with intervertebral disc degeneration. Exp Ther Med.2016;11(4):1425-1432.[9] 陈岩,胡有谷.转化生长因子β与椎间盘细胞Ⅰ型胶原基因调控的关系[J].中国矫形外科杂志,2000, 7(2):151-153. [10] Cheng L,Fan W,Liu B,et al.Th17 lymphocyte levels are higher in patients with ruptured than non-ruptured lumbar discs, and are correlated with pain intensity.Injury.2013;44(12): 1805-1810. [11] Xue H,Yao Y,Wang X,et al.Interleukin-21 is associated with the pathogenesis of lumbar disc herniation. Iran J Allergy Asthma Immunol.2015;14(5):509-518.[12] Grad S,Bow C,Karppinen J,et al.Systemic blood plasma CCL5 and CXCL6:Potential biomarkers for human lumbar disc degeneration. Eur Cell Mater.2016;31:1-10.[13] Goode AP,Marshall SW,Kraus VB,et al.Association between serum and urine biomarkers and lumbar spine individual radiographicfeatures: the Johnston County Osteoarthritis Project.Osteoarthritis Cartilage. 2012;20(11):1286-1293.[14] Xie P,Liu B,Chen R,et al.Comparative analysis of serum proteomes:Identification of proteins associated with sciatica due to lumbar intervertebral disc herniation.Biomed Rep.2014; 2(5):693-698.[15] Moen A,Lind AL,Thulin M,et al.Protein Profiling of Patients with Lumbar Inflammatory Serum Radicular Pain One Year after DiscHerniation.Int J Inflam.2016;2016:3874964.[16] 贾长青,王臣,陈勇,等.基质金属蛋白酶3和白细胞介素1在突出的腰椎间盘组织中的含量及其相关性研究[J].中国修复重建外科杂志,2006,20(12):1180-1182.[17] Tertti M,Paajanen H,Laato M,et al.Disc degeneration in magnetic resonance imaging.A comparative biochemical, histologic, and radiologic study in cadaver spines. Spine. 1991;16(6):629-634.[18] Pearce RH, Thompson JP, Bebault GM, et al. Magnetic resonance imaging reflects the chemical changes of aging degeneration in the humanintervertebral disk.J Rheumatol Suppl.1991; 27:42-43.[19] Southern EP,Fye MA,Panjabi MM,et al.Disc degeneration: a human cadaveric study correlating magnetic resonance imaging and quantitative discomanometry.Spine.2000;25(17) 2171-2175.[20] Weidenbaum M,Foster RJ,Best BA,et al.Correlating magnetic resonance imaging with the biochemical content of the normal humanintervertebral disc.J Orthop Res.1992;10(4):552-561.[21] Schiebler ML,Camerino VJ,Fallon MD,et al.In vivo and ex vivo magnetic resonance imaging evaluation of early disc degeneration with histopathologic correlation. Spine.1991; 16(6):635-640.[22] Blumenkrantz G,Zuo J,Li X,et al.In vivo 3.0-tesla magnetic resonance T1rho and T2 relaxation mapping in subjects with intervertebral disc degeneration and clinical symptoms.Magn Reson Med.2010;63(5):1193-1200. [23] Taylor TK,Melrose J,Burkhardt D,et al.Spinal biomechanics and aging are major determinants of the proteoglycan metabolism of intervertebral disc cells.Spine (Phila Pa 1976). 2000;25(23):3014-3020.[24] Iatridis JC,MacLean JJ,O'Brien M,et al.Measurements of proteoglycan and water content distribution in human lumbarintervertebral discs.Spine (Phila Pa 1976). 2007;32(14): 1493-1497.[25] Filippi CG, Duncan CT, Watts R, et al.In vivo quantification of T1 in lumbar spine disk spaces at 3T using parallel transmission MRI. AJR Am J Roentgenol. 2013;201(1): W110-116.[26] Radek M,Pacholczyk-Sienicka B,Jankowski S,et al.Assessing the correlation between the degree of disc degeneration on the Pfirrmann scale and the metabolites identified in HR-MAS NMR spectroscopy. Magn Reson Imaging. 2016;34(4): 376-380.[27] Pfirrmann CW,Metzdorf A,Zanetti M,et al.Magnetic resonance classification of lumbar intervertebral disc degeneratio.Spine (Phila Pa 1976).2001;26(17):1873-1878.[28] Luoma K,Vehmas T,Riihimäki,et al.Disc height and signal intensity of the nucleus pulposus on magnetic resonance imaging as indicators of lumbar disc degeneration. Spine (Phila Pa 1976). 2001;26(6):680-686.[29] Antoniou J,Pike GB,Steffen T,et al.Quantitative magnetic resonance imaging in the assessment of degenerative disc disease. Magn Reson Med.1998;40(6):900-907.[30] Kettler A,Wilke HJ.Review of existing grading systems for cervical or lumbar disc and facet joint degeneration.Eur Spine J.2006;15(6):705-718.[31] Ye S,Ju B,Wang H,et al.Bone morphogenetic protein-2 provokes interleukin-18-induced human intervertebral disc degeneration.Bone Joint Res.2016;5(9):412-418.[32] Weber KT,Alipui DO,Sison CP,et al.Serum levels of the proinflammatory cytokine interleukin-6 vary based on diagnoses in individuals with lumbar intervertebral disc diseases. Arthritis Res Ther.2016;18:3. |
| [1] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [2] | Lu Dezhi, Mei Zhao, Li Xianglei, Wang Caiping, Sun Xin, Wang Xiaowen, Wang Jinwu. Digital design and effect evaluation of three-dimensional printing scoliosis orthosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1329-1334. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Yao Rubin, Wang Shiyong, Yang Kaishun. Minimally invasive transforaminal lumbar interbody fusion for treatment of single-segment lumbar spinal stenosis improves lumbar-pelvic balance [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1387-1392. |
| [5] | Wang Haiying, Lü Bing, Li Hui, Wang Shunyi. Posterior lumbar interbody fusion for degenerative lumbar spondylolisthesis: prediction of functional prognosis of patients based on spinopelvic parameters [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1393-1397. |
| [6] | Chen Jinping, Li Kui, Chen Qian, Guo Haoran, Zhang Yingbo, Wei Peng. Meta-analysis of the efficacy and safety of tranexamic acid in open spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1458-1464. |
| [7] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [8] | Zhang Wei, Hu Jiang, Tang Liuyi, Wan Lun, Yu Yang, Lin Shu, Tang Zhi, Wang Fei. Advantages of robot assisted percutaneous biopsy in the diagnosis of spinal lesions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 844-848. |
| [9] | He Li, Tian Wei, Xu Song, Zhao Xiaoyu, Miao Jun, Jia Jian. Factors influencing the efficacy of lumbopelvic internal fixation in the treatment of traumatic spinopelvic dissociation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 884-889. |
| [10] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [11] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [12] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [13] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [14] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [15] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||