Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (28): 4566-4573.doi: 10.3969/j.issn.2095-4344.0266
Previous Articles Next Articles
Biological role of myonectin and its correlation with exercise
Li Lin, Yu Liang
- Sport Science College, Beijing Sport University, Beijing 100084, China
-
Received:2018-01-25Online:2018-10-08Published:2018-10-08 -
Contact:Yu Liang, PhD, Associate professor, Sport Science College, Beijing Sport University, Beijing 100084, China -
About author:Li Lin, Master candidate, Sport Science College, Beijing Sport University, Beijing 100084, China -
Supported by:the National Natural Science Foundation of China, No. 31500964; the Fok Ying Tung Education Foundation, No. 151095; the Nationwide Body-Building Project of General Administration of Sport of China, No. 2015B046; the Self-Dependent Research Project of Beijing Sport University, No. 2016YB047 and 2017YB028
CLC Number:
Cite this article
Li Lin, Yu Liang. Biological role of myonectin and its correlation with exercise[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(28): 4566-4573.
share this article

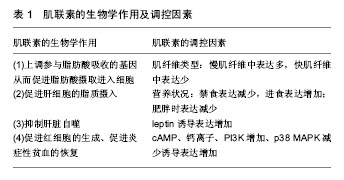
2.1 Myonectin概况 Myonectin是新发现的肌肉因子,是CTRP家族的第15位成员,即CTRP15。CTRP家族是通过使用脂联素的cDNA序列询问小鼠及人类表达序列标签(expressed sequence tags,ESTs)和基因组数据库所得到的与脂联素(adiponectin)同源的高度保守的蛋白家族,该家族成员都具有与脂联素相似的模块化组织,并且包含4个不同的结构域:N末端的信号肽、短的可变区、胶原结构域和与补体蛋白C1q同源的C-末端球状结构域,由于球形的补体 C1q样结构域的晶体结构与肿瘤坏死因子( tumor necrosis factor,TNF)非常类似,因而将该家族命名为补体 C1q /肿瘤坏死因子相关蛋白(C1q /tumor necrosis factor-related protein,CTRP)[3]。 Myonectin由位于人染色体2q37.3上的基因编码,全长9 883 bp,由8个外显子和7个内含子组成,编码2 911 bp的mRNA;小鼠myonectin的基因位于第1号染色体上,全长8 057 bp,由9个外显子和8个内含子组成,编码2 835 bp的mRNA。利用免疫印迹的方法对小鼠骨骼肌组织myonectin的蛋白表达进行测定,发现在相对分子质量 40 000-50 000分布着多个被不同程度糖基化的myonectin特异性条带。利用同样的方法对12周龄的小鼠血清中myonectin水平进行测定发现特异性条带出现在45 000左右[4]。其蛋白包括5个结构域:用于分泌的信号肽(signal peptide,SP)结构域、N-末端结构域-1(N-terminal domain 1,NTD1)、具有6个Gly-X-Y重复的胶原结构域、N-末端结构域-2(NTD2)和补体C1q/TNF样结构域,其中C1q/TNF样结构域是CTRP 15发挥生物学作用的主要功能结构域[4]。 2.2 Myonectin的生物学作用 Seldin等[4]对小鼠myonectin的组织分布及表达进行了测定,发现myonectin主要存在于血液循环中,主要表达于骨骼肌组织中,其次极少量表达于肺、眼、平滑肌、心脏和脑、肾、胎盘、肝脏、胃及胸腺等,并且其在骨骼肌中的表达量是肺、眼、平滑肌等组织的5倍,是心肌、脑组织的10倍左右,除此以外的其余组织几乎没有myonectin mRNA的表达。此外,myonectin在小鼠分化的C2C12肌管细胞中表达显著升高,而在未分化的成肌细胞中其几乎不表达[4],不是由卫星细胞分泌,所以,骨骼肌而非骨骼肌卫星细胞是myonectin特异性表达的组织,因此认定myonectin为典型的 “肌肉因子”,Myonectin以内分泌或旁分泌的方式释放入血,主要是作用于脂肪组织和肝脏,也有学者发现myonectin是红细胞生成过程中调节铁代谢的重要因子(见表1)。"
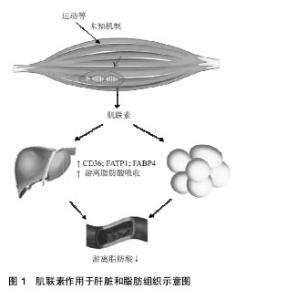
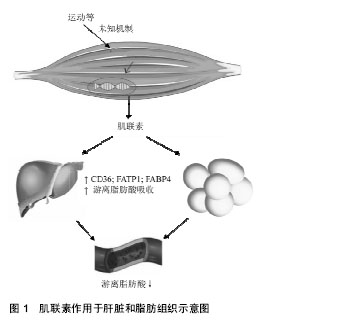
2.2.1 Myonectin在脂肪组织中的作用 许多CTRP家族成员是由脂肪组织分泌并以自分泌、旁分泌和内分泌方式作用于附近或远处器官,所以被称为“脂肪因子”,它们可以调节脂肪组织的功能,进而调节全身胰岛素敏感性、能量代谢及心血管疾病[5-6],例如CTRP 3可以抑制小鼠病理性心脏的重塑[7]、CTRP 5可以通过促进AMPK信号传导增加葡萄糖的摄取,改善肥胖小鼠的葡萄糖代谢[8]。Myonectin是CTRP家族成员之一,但不同的是它是由骨骼肌分泌并以内分泌方式调节体内脂质系统的平衡,并且实现骨骼肌和其他代谢组织(主要脂肪组织和肝脏)之间的“对话”[4]。 为了探究myonectin的作用,用myonectin重组蛋白作用于小鼠后发现FFA(游离脂肪酸或非酯化脂肪酸)水平降低,而脂肪细胞脂解能力几乎没有改变,提示myonectin在脂肪细胞对脂肪酸的摄取中起到了一定的作用[4,9]。非酯化脂肪酸是血浆的成分之一,在正常情况下,脂肪组织的脂肪水解释放非酯化脂肪酸进入血液,被骨骼肌、肝脏等多种外周组织摄取利用,进行氧化供能。猜测Myonectin降低循环非酯化脂肪酸水平通过2种可能的机制:抑制脂肪组织脂肪分解或促进脂肪酸摄取进入细胞,于是用myonectin分别处理3T3-L1脂肪细胞和原代睾周脂肪组织,发现myonectin对常态下的脂解及肾上腺素诱导的脂解均没有抑制作用[4],因此排除了第1种可能的机制;随后继续对myonectin是否促进了脂肪酸的摄取进行观察,用饱和剂量的胰岛素(50 nmol/L)和5 mg/L的myonectin处理脂肪细胞均能大幅度增加脂肪酸摄取,而同时用胰岛素和myonectin蛋白刺激脂肪细胞不会导致脂肪酸摄取的进一步增加,因此推测myonectin和胰岛素增强脂质摄取所激活的途径类似[4]。 在脂肪酸摄入细胞中起重要作用的包括脂肪酸转运蛋白CD36、小窝蛋白1 ( caveolin-1,Cav1)、脂肪酸转运蛋白(fatty acid transport protein 1,FATP1)、脂肪酸结合蛋白(fatty acid bindin protein 4,FABP4)等蛋白,实验中利用RT-PCR分析显示在脂肪细胞中重组myonectin可以不同程度地上调这些基因的表达,在脂代谢过程中,FAT/CD36是骨骼肌主要的脂肪酸转运体之一,在敲除FAT/CD36基因的小鼠中[10],脂肪酸摄入明显减少,而当FAT/CD36基因过表达时[11],骨骼肌对脂肪酸的利用显著增加,提示FAT/CD36在骨骼肌脂肪酸摄入中起到关键作用,所以myonectin可以通过转录上调参与脂肪酸吸收的基因促进脂肪细胞的脂质摄入,通过刺激FAT/CD36的表达来促进脂肪酸的摄入[4],而又有学者发现大鼠进行跑台训练下调了myonectin的水平,而同时非酯化脂肪酸水平降低,似乎myonectin不参与运动对血清非酯化脂肪酸的调节作用,结果相悖则需要进一步的实验探究。 2.2.2 Myonectin在肝脏中的作用 为了证实myonectin在肝脏中的作用,采用RT-PCR技术分析小鼠H4IIE肝细胞发现,饥饿或禁食可分别引起H4IIE肝细胞和小鼠肝脏中自噬相关基因Atg7和Atg12的表达增加,而重组myonectin可以增加Cav1和Fabp1的表达,促进脂肪酸摄取的增加,所以myonectin可以通过转录上调参与脂肪酸吸收的基因促进肝细胞的脂质摄入[4]。另外,Seldin等[12]发现myonectin可激活细胞自噬抑制因子mTOR(雷帕霉素受体),激活PI3K/Akt/mTOR途径从而抑制肝脏自噬。而自噬调节细胞物质的合成、降解和重新利用之间的代谢平衡起很大的作用,所以myonectin水平的异常也会影响到能量代谢的平衡,但是其机制还需要进一步的实验证明。为了研究myonectin和葡萄糖之间的关系,将重组myonectin蛋白注射到小鼠中发现没有降低葡萄糖的作用,所以myonectin主要与脂质代谢关系密切,根据Seldin等[4]的报道,在体外重组myonectin通过增加在脂肪酸吸收中起重要作用的CD36,FATP1水平、增强Fabp4 mRNA表达,促进小鼠脂肪细胞和大鼠肝细胞中的脂肪酸摄取(见图1)。此外,重组myonectin对脂肪组织脂解没有影响。因此可以推测myonectin蛋白血清长期保持高水平可以增加脂肪组织和肝脏中的脂肪酸摄取,并改善脂肪代谢和脂质处理,但是机制尚不清楚。"
| [1] Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol.2012;8(8): 457-465.[2] Giudice J,Taylor JM.Muscle as a paracrine and endocrine organ.Curr Opin Pharmacol.2017;34:49-55.[3] Wong GW,Wang J,Hug C,et al. A family of Acrp30 / adiponectin structural and functional paralogs[J]. Proc Natl Acad Sci USA.2004; 101:10302-10307.[4] Seldin MM,Peterson JM,Byerly MS,et al.Myonectin(CTRP15),a novel myokine that links skeletal muscle to systemic lipid homeostasis.J Biol Chem.2012;287(15):11968-11980.[5] Noriyuki Ouchi and Kenneth Walsh.Cardiovascular and Metabolic Regulation by the Adiponectin/CTRP Family of Proteins.Circulation. 2012;5(31):1524-1539[6] Matsuzawa Y,Funahashi T,Kihara S,Shimomura I.Adiponectin and metabolic syndrome.Arterioscler Thromb Vasc Biol.2004;24:29-33.[7] Yi W,Sun Y,Lau WB,et al.C1q/TNF-related protein-3, a newly identified adipokine, is a novel anti-apoptotic, pro-angiogenic,and cardioprotective molecule in the ischemic mouse heart. Circulation. 2012;125:134-145.[8] Park SY, Choi JH, Ryu HS,et al. C1q tumor necrosis factor alpha-related protein isoform 5 is increased in mitochondrial DNA-depleted myocytes and activates AMP-activated protein kinase. J Biol Chem.2009;284:27780-27789.[9] Seldin M M,Wong G W.Regulation of tissue crosstalk by skeletal muscle-derived myonectin and other myokines.Adipocyte.2012;1(4) : 200-202[10] Febbraio M, Abumrad NA, Hajjar DP, et al. A null mutation in murine CD36 reveals an important role in fatty acid and lipoprotein metabolism.J Biol Chem.1999;274 (27):19055-19062.[11] Ibrahimi A , Bonen A , Blinn WD, et al. Muscle -specific overexpression of FAT/CD36 enhances fatty acid oxidation by contracting muscle ,reduces plasma triglycerides and fatty acids,and increases plasma glucose and insulin.J Biol Chem.1999;274(38):26761-26766.[12] Seldin MM,Lei X,Tan SY,et al.Skeletal muscle-derived myonectin activates the mammalian target of rapamycin (mTOR) pathway to suppress autophagy in liver.J Biol Chem.2013;288(50): 36073-36082.[13] Kautz L,Jung G,Valore EV,et al.Identification of erythroferrone as an erythroid regulator of iron metabolism.Nat Genet.2014;46:678-684.[14] Honda H,Kobayashi Y,Onuma S,et al.Associations among erythroferrone and biomarkers of erythropoiesis and iron metabolism, and treatment with long-term erythropoiesis-stimulating agents in patients on hemodialysis.PLOS ONE.2016;11:e151601.[15] Kautz L, Jung G, Nemeth E,et al. Erythroferrone contributes to recovery from anemia of inflammation.Blood. 2014,124:2569-2574.[16] Nai A,Rubio A,Campanella A,et al.Limiting hepatic Bmp-Smad signaling by matriptase-2 is required for erythropoietin-mediated hepcidin suppression in mice.Blood.2016;127:2327-2336[17] 张靓,董泽源,马谨,等.跑台运动对高脂饮食诱导的肥胖大鼠骨骼肌肌联素表达的影响[J].中国运动医学杂志, 2016,35(6):547-552.[18] Peterson JM, Mart R, Bond CE.Effect of obesity and exercise on the expression of the novel myokines, Myonectin and Fibronectin type III domain containing 5.PeerJ.2014;2:e605.[19] Rodríguez A, Becerril S, Méndez-Giménez L, et al.Leptin administration activates irisin-induced myogenesis via nitric oxide-dependent mechanisms, but reduces its effect on subcutaneous fat browning in mice. Int J Obes (Lond). 2015;39(3):397-407. [20] Yang M,Wei D,Mo C,et al.Saturated fatty acid palmitate-induced insulin resistance is accompanied with myotube loss and the impaired expression of health benefit myokine genes in C2C12 myotubes.Lipids Health Dis.2013;12: 104.[21] 陈婷,李仲文,张艳艳,等.甘油诱导的骨骼肌损伤修复过程肌间脂形态学及肌分泌因子表达变化[J]. 中国生物化学与分子生物学报,2015,31(1): 64-71.[22] Gamas L, Matafome P, Seiça R.Irisin and Myonectin Regulation in the Insulin Resistant Muscle: Implications to Adipose Tissue: Muscle Crosstalk.J Diabetes Res.2015;8:121-128.[23] Chen T,Li ZW,Zhang YY,et al.Muscle-selective knockout of AMPKα2 does not exacerbate diet-induced obesity probably related to altered myokines expression.Biochem Biophysic Res Com.2015;458(12): 449-455.[24] Sharma N,Castorena CM,Cartee GD. Greater insulin sensitivity in calorie restricted rats occurswith unaltered circulating levels of several important myokines and cytokines.Nutr Metab (Lond). 2012; 9(1):90. [25] Pedersen BK.The diseasome of physical inactivity — and the role of myokines in muscle-fat cross talk.J Physiol.2009;587:5559-5568.[26] Catoire M, Kersten S.The search for exercise factors in humans. FASEB J.2015;29(5):1615-28.[27] Hawley JA, Hargreaves M, Joyner MJ et al.Integrative biology of exercise. Cell.2014; 159(4):738-749. [28] Park SY,Choi JH,Ryu HS,et al.C1q tumor necrosis factor alpha-related protein isoform 5 is increased in mitochondrial DNA-depleted myocytes and activates AMP-activated protein kinase. J Biol Chem. 2009;284(41):27780-27789.[29] Lim S,Choi SH,Koo BK,et al.Effects of aerobic exercise training on C1q tumor necrosis factor alpha-related protein isoform 5 (myonectin): association with insulin resistance and mito-chondrial DNA density in women.J Clin Endocrinol Metab.2009;97:88-93.[30] Phielix E,Meex R,Moonen-Kornips E,et al.Exercise training increases mitochondrial content and exvivo mitochondrial function similarly in patients with type 2 diabetes and in control individuals.Diabetologia. 2010;53(8):1714-1721.[31] Xu FX,Zhou X,Shen F,et al.Decreased peripheral blood mitochondrial DNA content is related to HbA1c, fasting plasmaglucose level and age of onset in type 2 diabetes mellitus.Diabet Med.2012;29(7):e47-54.[32] Jager S,Handschin C, St-Pierre J, et al.AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylalion of PGC-1 alpha.Proc Natl Acad Sci U S A.2007; 104(29) :12017-12022. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||